Biochemistry 1 Biochemistry I The cell is a
Biochemistry 1

Biochemistry I. The cell is a complex "chemical factory". - Cells are composed of elements - There are 108 elements known today - Some elements found in cells are: C - Carbon H - Hydrogen 0 - Oxygen N - Nitrogen The most abundant elements in living organisms 2

S - Sulfur P - Phosphorus Mg - Magnesium Fe - Iron I - Iodine Ca - Calcium Na - Sodium Cl - Chlorine K – Potassium The atoms of elements bond together to form compounds. Organisms consist of Inorganic and Organic compounds. 3

C 6 H 12 O 6 H 2 O CO 2 CH 4 Na. Cl C 12 H 22 O 11 C 30 H 45 O 16 N 3 Organic - contains both carbon and hydrogen. Inorganic - lacks carbon and/or hydrogen. II. Types of Organic Compounds 1. Carbohydrates 2. Proteins 3. Lipids 4. Nucleic Acids 4

1. Carbohydrates - sugars and starches A. Composed of C, H, 0 B. ratio of Hydrogen : Oxygen is 2: 1 C. Ends in ose D. Sugars - source of immediate energy E. surplus sugars are stored as starches 5
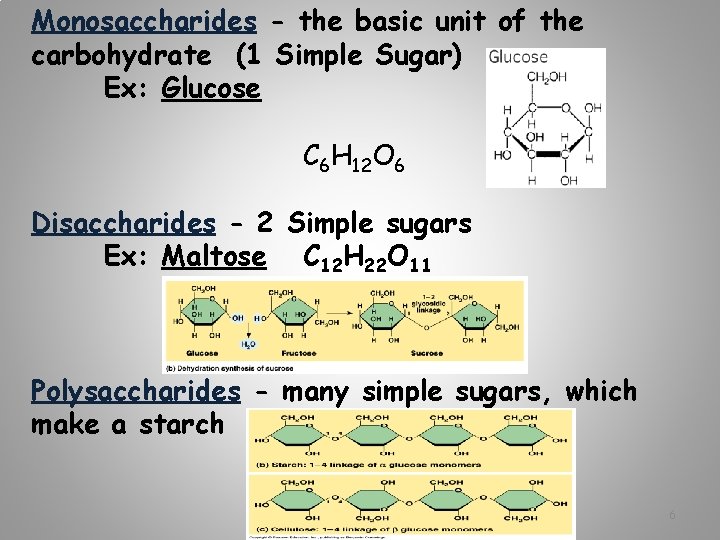
Monosaccharides - the basic unit of the carbohydrate (1 Simple Sugar) Ex: Glucose C 6 H 12 O 6 Disaccharides - 2 Simple sugars Ex: Maltose C 12 H 22 O 11 Polysaccharides - many simple sugars, which make a starch 6
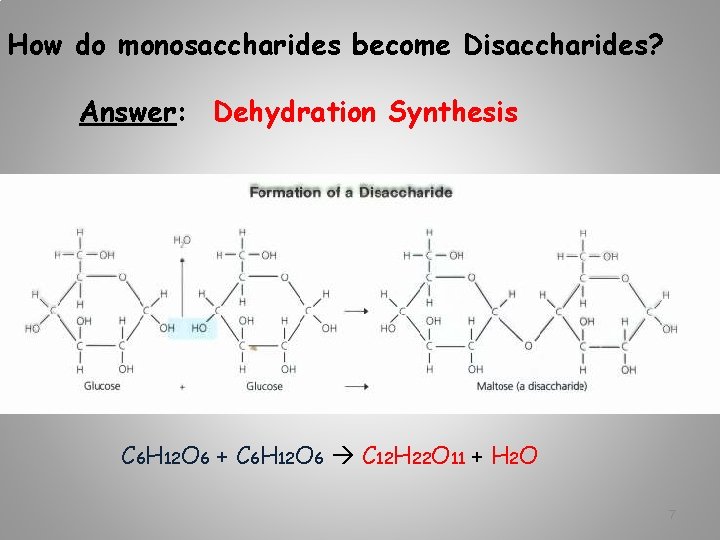
How do monosaccharides become Disaccharides? Answer: Dehydration Synthesis C 6 H 12 O 6 + C 6 H 12 O 6 C 12 H 22 O 11 + H 2 O 7

Dehydration Synthesis - 2 simple molecules bond to form a more complex molecule with the release of H 2 O How do polysaccharides or Disaccharides get broken down into smaller saccharides? Answer: Put the water back in! 8
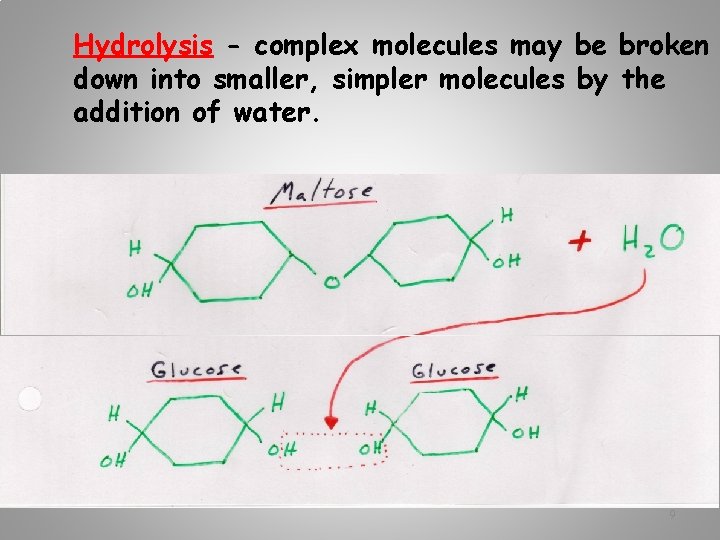
Hydrolysis - complex molecules may be broken down into smaller, simpler molecules by the addition of water. 9

2. Proteins A. Composed of C, H, O, N and sometimes S (sulfur) B. Building blocks are Amino acids C. There are 20 different amino acids D. All enzymes are proteins 10
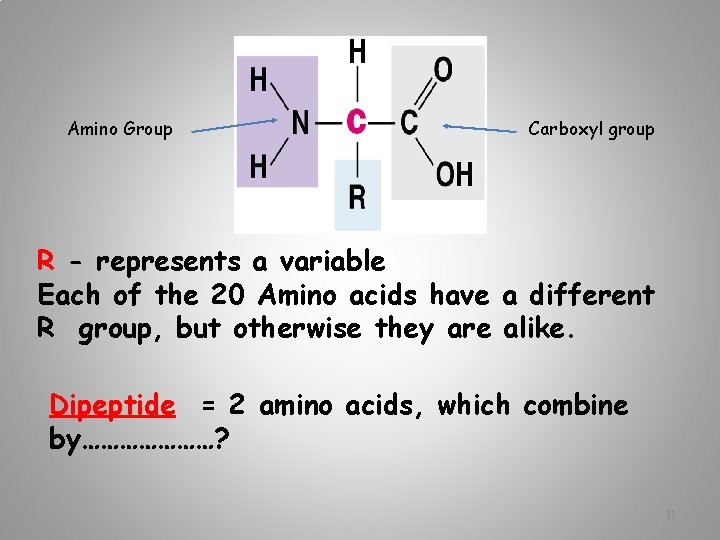
Amino Group Carboxyl group R - represents a variable Each of the 20 Amino acids have a different R group, but otherwise they are alike. Dipeptide = 2 amino acids, which combine by…………………? 11
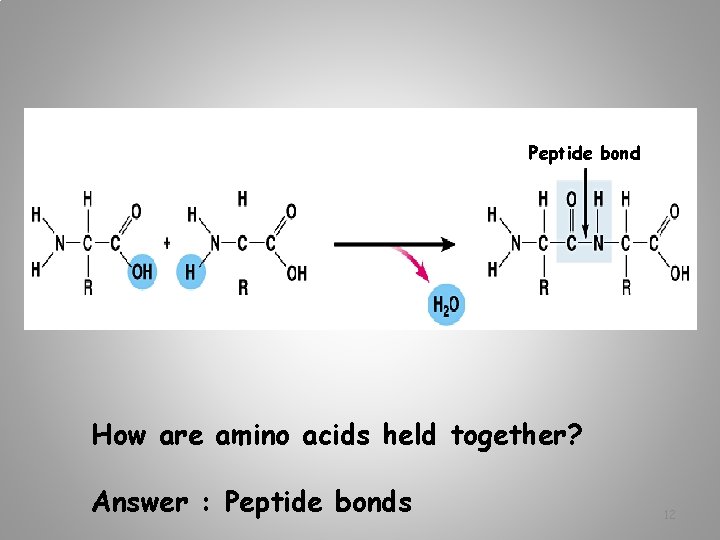
Peptide bond How are amino acids held together? Answer : Peptide bonds 12
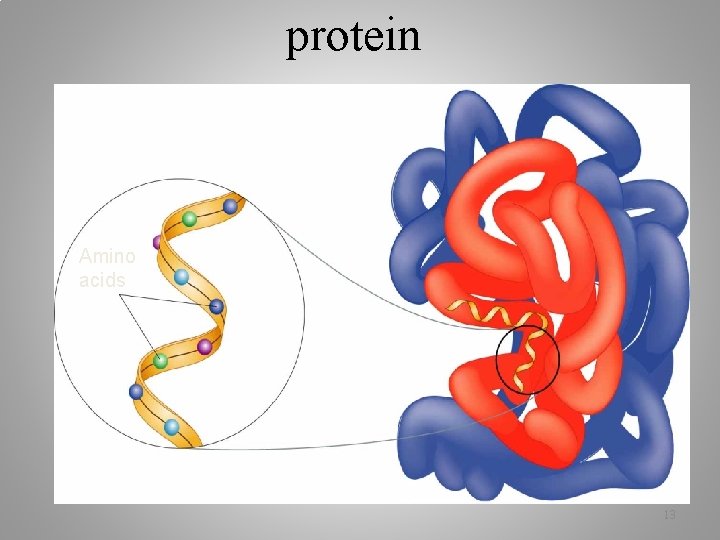
protein Amino acids 13

Polypeptide - many amino acids linked together, which forms a protein Proteins include: 1. All enzymes 2. Cell membrane 3. Hemoglobin 4. Hormones Ex: insulin 5. Skin, eyes, etc 14
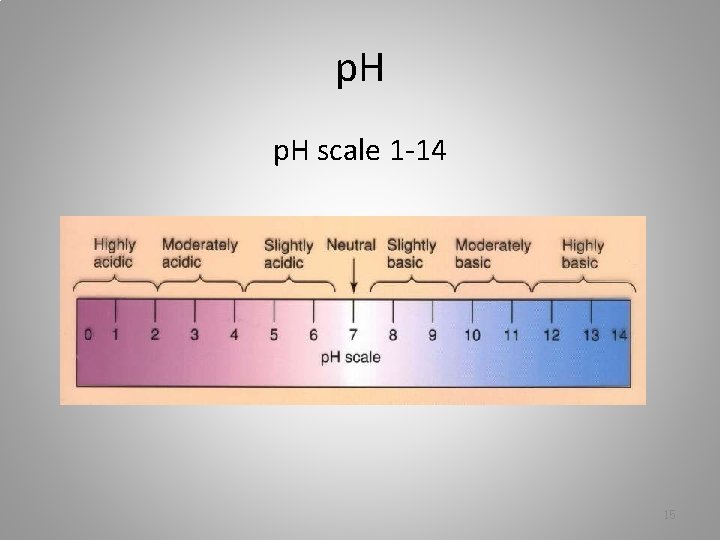
p. H scale 1 -14 15

Acids – p. H between 1 and 7 – release hydrogen ions when mixed with water – citric acid (lemon juice) – acetic acid (vinegar) – hydrochloric acid 16

Bases – p. H between 7 and 14 – when mixed with water, release ions that bond with hydrogen ions – baking soda – ammonia – antacids • p. H 7 is neutral (example: pure water) 17

3. Lipids – fats, oils and waxes A. Contains C, H, O B. No constant Hydrogen to Oxygen ratio C. Source of stored energy, and insulation D. Part of cell membrane E. Saturated fats are linked to cardiovascular diseases (polyunsaturated fats are safer to eat). 18
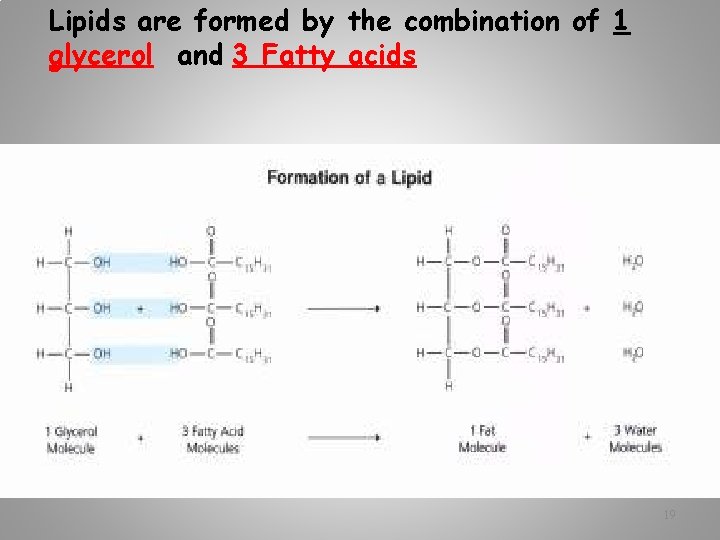
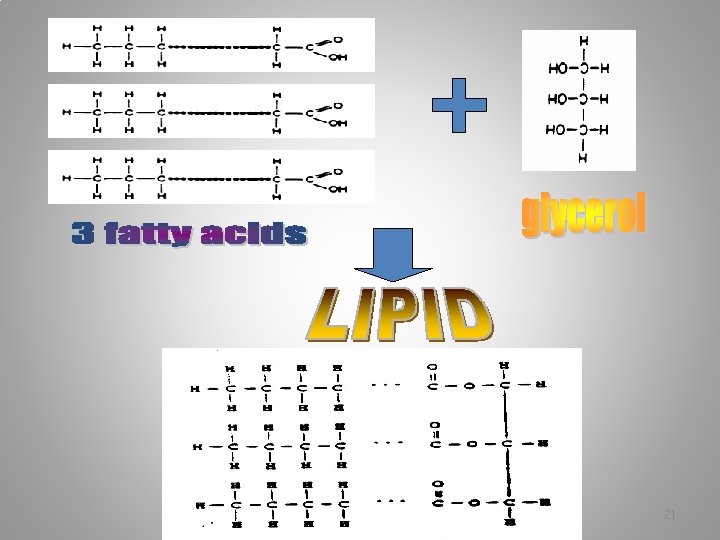
Lipids are formed by the combination of 1 glycerol and 3 Fatty acids 19

How do 3 fatty acids combine with 1 Glycerol to form a lipid? Answer: Dehydration synthesis What else is formed besides the lipid? Answer: 3 H 20 molecules 20
21
Diagrams - 1 22
Diagrams - 2 23

OTHER LIPIDS • triglycerides – used for reserve energy, insulation and cushioning • phospholipids – used for cell membranes glycerol + 2 fatty acids + phosphate group = phospholipid • saturated fats – carbon chains that have all single bonds contributes to fat deposits in arteries • unsaturated fats – contain carbon chains that have one or more double or triple bonds 24
- Slides: 24