BIOCHEMICAL CYCLES Living cycles Earth Biosphere Carbon cycle
BIOCHEMICAL CYCLES Living cycles Earth
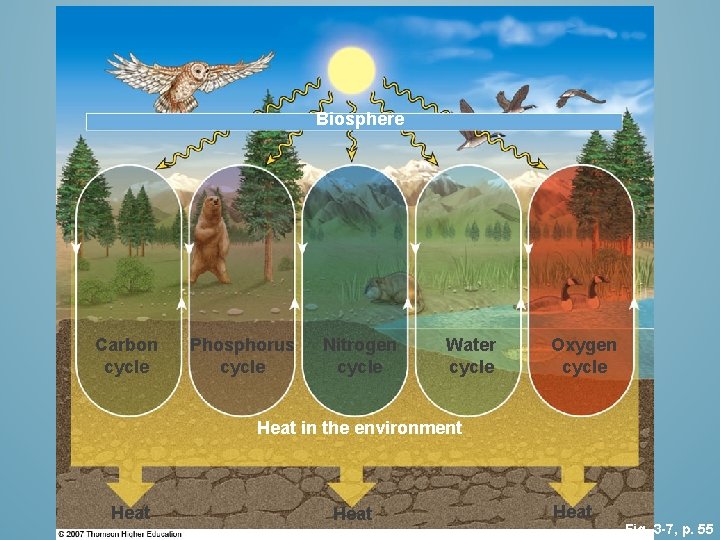
Biosphere Carbon cycle Phosphorus cycle Nitrogen cycle Water cycle Oxygen cycle Heat in the environment Heat Fig. 3 -7, p. 55
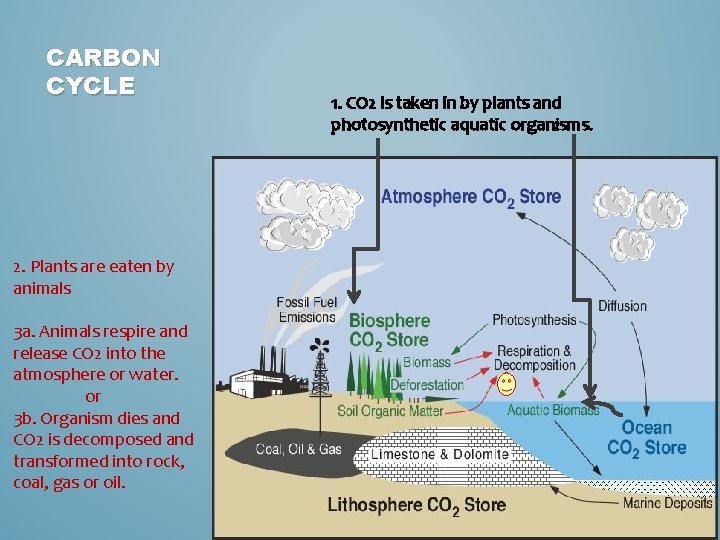
CARBON CYCLE 2. Plants are eaten by animals 3 a. Animals respire and release CO 2 into the atmosphere or water. or 3 b. Organism dies and CO 2 is decomposed and transformed into rock, coal, gas or oil. 1. CO 2 is taken in by plants and photosynthetic aquatic organisms.
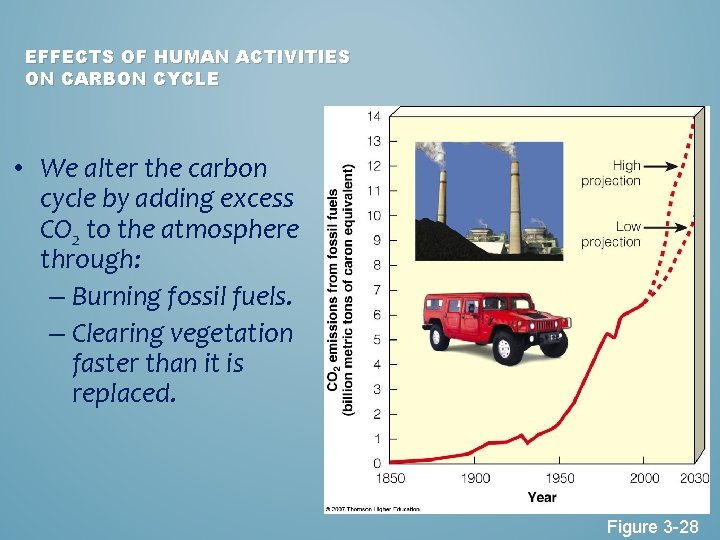
EFFECTS OF HUMAN ACTIVITIES ON CARBON CYCLE • We alter the carbon cycle by adding excess CO 2 to the atmosphere through: – Burning fossil fuels. – Clearing vegetation faster than it is replaced. Figure 3 -28
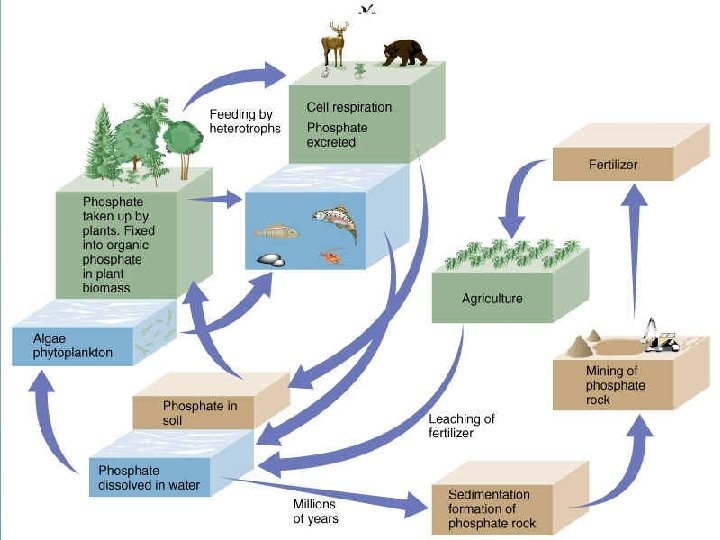
Phosphorous Cycle

FYI: PHOSPHORUS • Bacteria are not as important in the phosphorus cycle as in the nitrogen cycle. • Phosphorus is not usually found in the atmosphere or in a gas state only as dust. • The phosphorus cycle is slow and phosphorus is usually found in rock formations and ocean sediments. • Phosphorus is found in fertilizers because most soil is deficient in it and plants need it. • Phosphorus is usually insoluble in water and is not found in most aquatic environments.

EFFECTS OF HUMAN ACTIVITIES ON THE PHOSPHOROUS CYCLE • We remove large amounts of phosphate from the earth to make fertilizer. • We reduce phosphorous in tropical soils by clearing forests. • We add excess phosphates to aquatic systems from runoff of animal wastes and fertilizers.
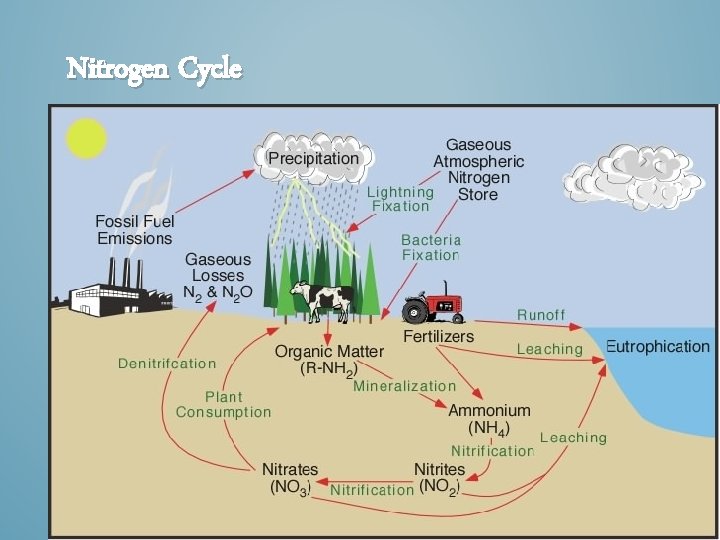
Nitrogen Cycle

STEP 1: NITROGEN FIXATION • This is the first step of the nitrogen cycle where specialized bacteria convert gaseous nitrogen to ammonia(NH 4 + ) that can be used by plants. This is done by cyanobacteria or bacteria (Rhizobium) living in the nodules on the root of various plants.
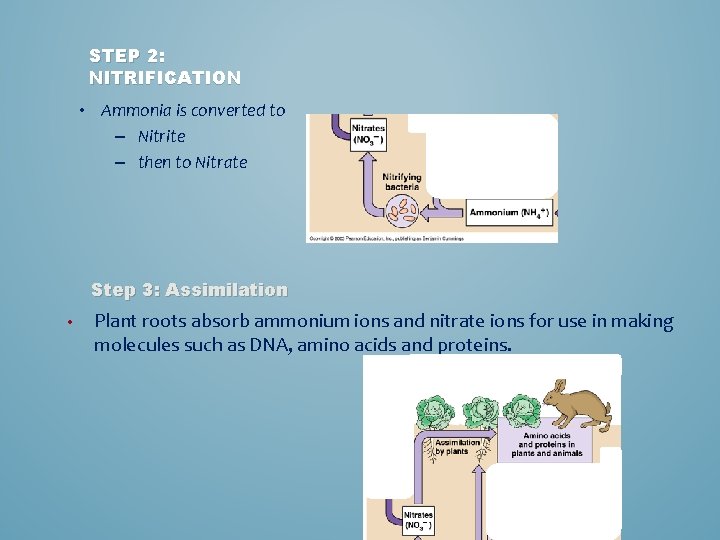
STEP 2: NITRIFICATION • Ammonia is converted to – Nitrite – then to Nitrate Step 3: Assimilation • Plant roots absorb ammonium ions and nitrate ions for use in making molecules such as DNA, amino acids and proteins.
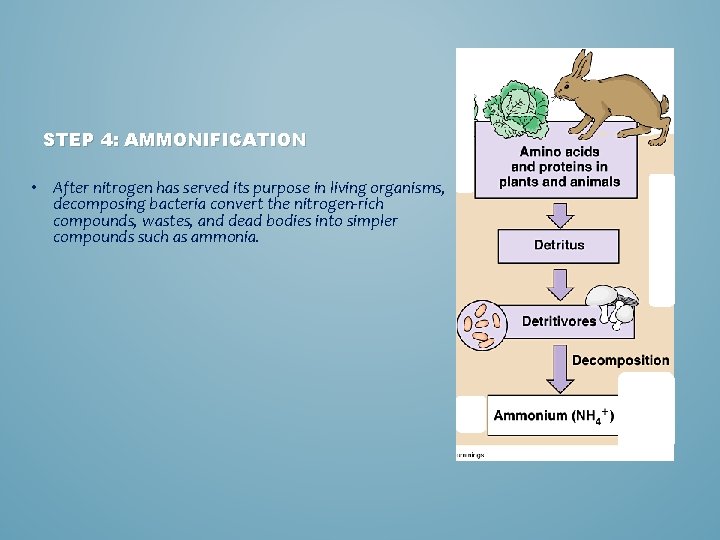
STEP 4: AMMONIFICATION • After nitrogen has served its purpose in living organisms, decomposing bacteria convert the nitrogen-rich compounds, wastes, and dead bodies into simpler compounds such as ammonia.
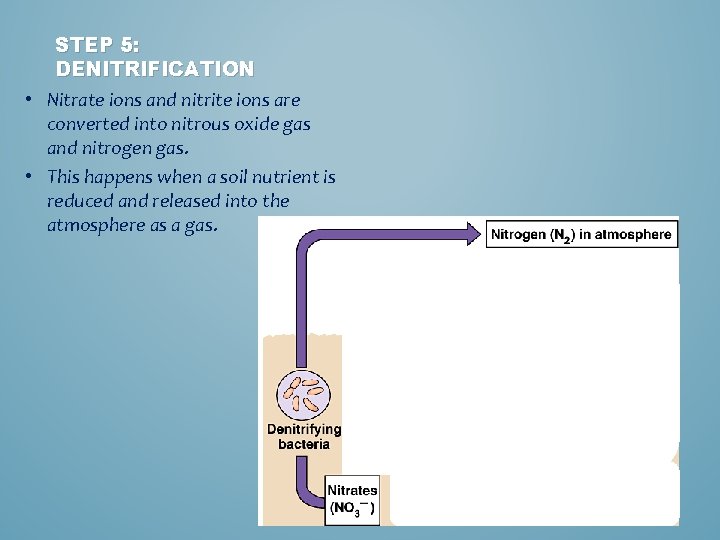
STEP 5: DENITRIFICATION • Nitrate ions and nitrite ions are converted into nitrous oxide gas and nitrogen gas. • This happens when a soil nutrient is reduced and released into the atmosphere as a gas.
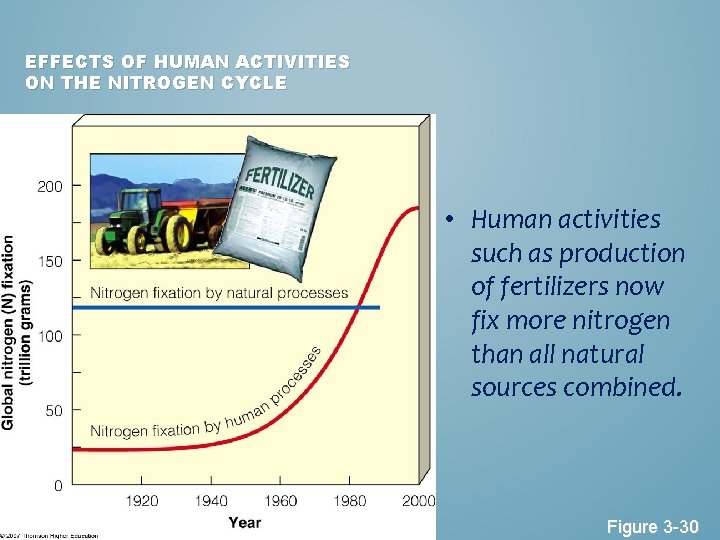
EFFECTS OF HUMAN ACTIVITIES ON THE NITROGEN CYCLE • Human activities such as production of fertilizers now fix more nitrogen than all natural sources combined. Figure 3 -30

EFFECTS OF HUMAN ACTIVITIES ON THE NITROGEN CYCLE • We alter the nitrogen cycle by: – Adding gases that contribute to acid rain. – Adding nitrous oxide to the atmosphere through farming practices which can warm the atmosphere and deplete ozone. – Contaminating ground water from nitrate ions in inorganic fertilizers. – Releasing nitrogen into the troposphere through deforestation.
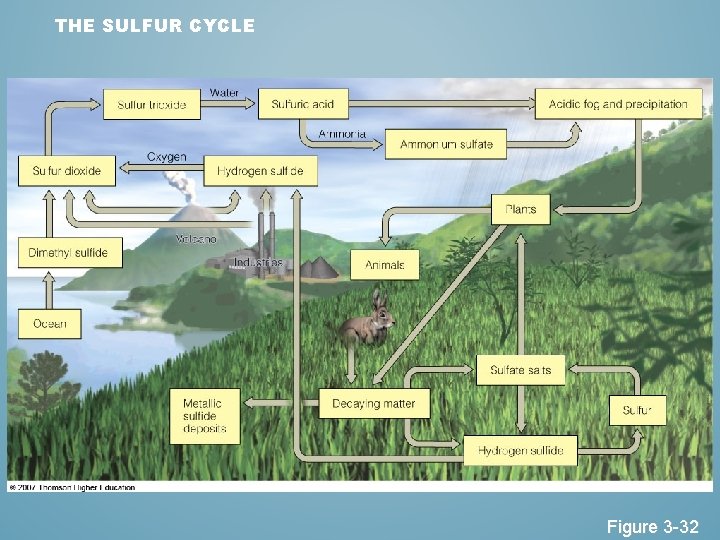
THE SULFUR CYCLE Figure 3 -32

EFFECTS OF HUMAN ACTIVITIES ON THE SULFUR CYCLE • We add sulfur dioxide to the atmosphere by: – Burning coal and oil – Refining sulfur containing petroleum. – Convert sulfur-containing metallic ores into free metals such as copper, lead, and zinc releasing sulfur dioxide into the environment.
- Slides: 16