BIOCERAMICS Prof Dr SEVL YCEL 1 Ceramics ceramicos
BIOCERAMICS Prof. Dr. SEVİL YÜCEL 1

Ceramics ceramicos (burned object) Generally ceramics are the materials that are chemically inorganic, non-metallic solid and can withstand very high temperatures(such as temperatures that range from 1, 000 °C to 1, 600 °C). Ceramic materials may have a crystalline or partly crystalline structure, or may be amorphous (e. g. , a glass) Ceramics are materials which include metal oxides (Al 2 O 3 Mg. O, Si. O 2 and Zr. O 2), carbides, silicate different hybrides, sulfite and selenoid. Ceramics have more corrosion resistance than metals and plastics. Ceramics generally does not react with liquids, gases, acids and alkaline substances. • They are good insulators, some of them have magnetic, piezoelectric, and semiconductive properties • Lightweight (40% lighter than metals) • Fragility 2
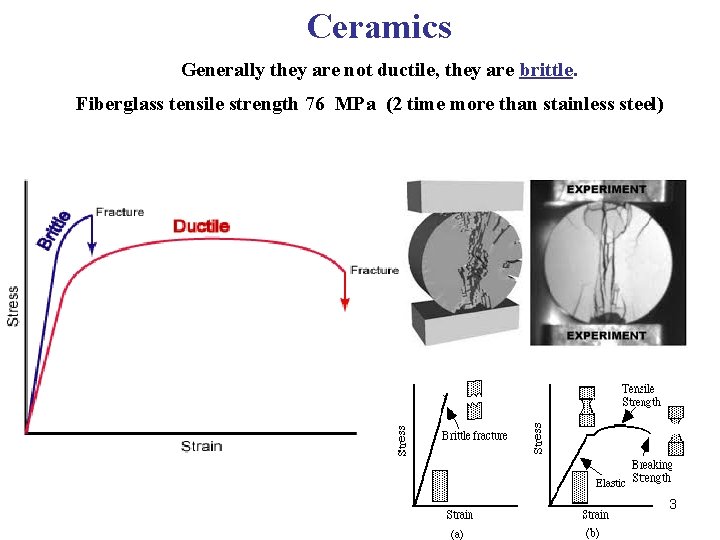
Ceramics Generally they are not ductile, they are brittle. Fiberglass tensile strength 76 MPa (2 time more than stainless steel) 3
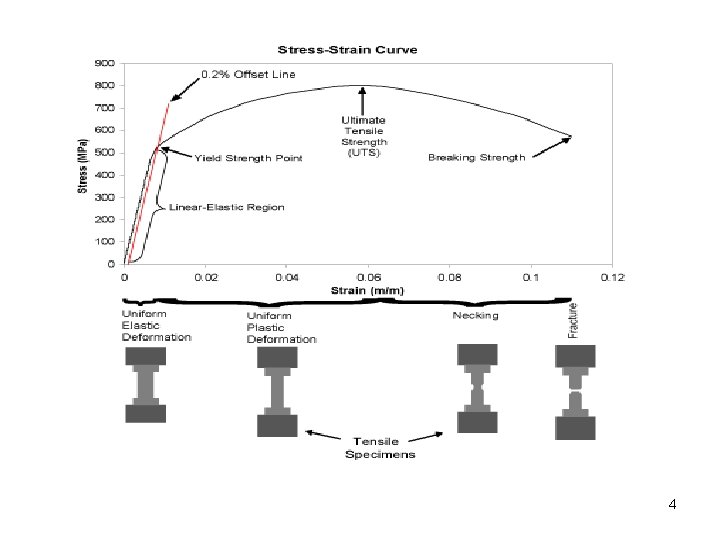
4
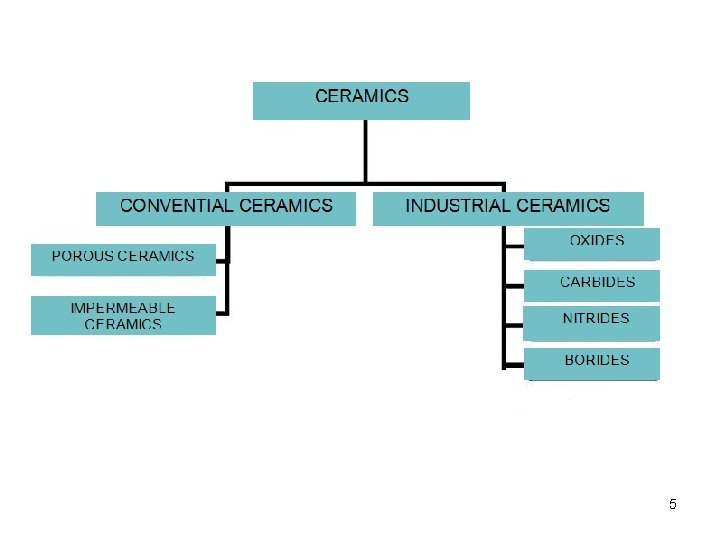
5

Ceramics • The other great revolution of ceramics created in human life is repearing the damaged or losted body parts functions, restructuring or replacing by the development and use of ceramics which were specially designed over the past 40 years. • Ceramics used for this "Bioceramics". purpose is called 6

Bioceramics • Can withstand very high temperatures • Enduring polycrystally compound • Are composed of ions which are found in human body (Ca, K, Mg, Na, etc. ) and ions that has low toxic property (Zr and Ti). • Have used in human body after 1960 s. Firstly high corrosion resistance and biocompatible high purity inert oxide materials- reactive ceramic materials • Are designed and produced to use in the body’s sickly and damaged parts. Example: Hip and dental implants. • Are used as covering to surface of metal implants (Porous ceramics) 7
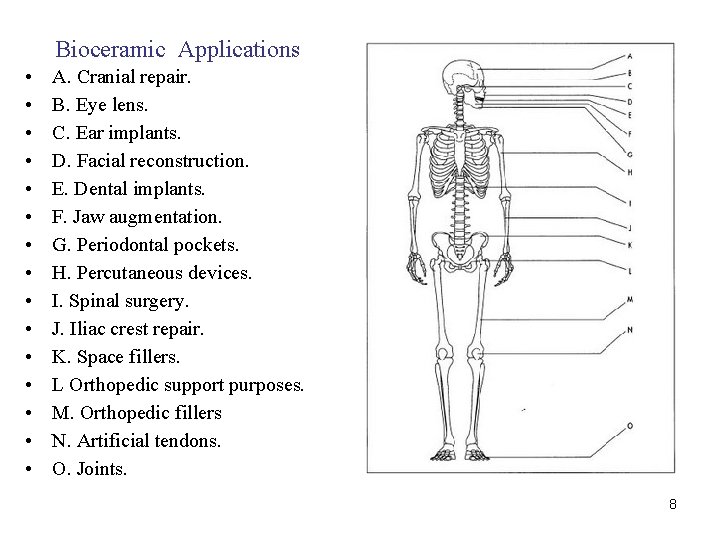
Bioceramic Applications • • • • A. Cranial repair. B. Eye lens. C. Ear implants. D. Facial reconstruction. E. Dental implants. F. Jaw augmentation. G. Periodontal pockets. H. Percutaneous devices. I. Spinal surgery. J. Iliac crest repair. K. Space fillers. L Orthopedic support purposes. M. Orthopedic fillers N. Artificial tendons. O. Joints. 8

Bioceramics • Resistance: High melting point, high corrosion resistance , high chemical stability, high compressive strength, high Young module, tough • Belongs to Mohr scale Diamond 10 Al 2 O 3 9 Kuvartz 8 Hydroxiapatite 5 • Generally inorganic • Inert • Biocompatible 9

Bioceramics • • APPLICATONS Orthopaedic load bearing coatings Dental implants Bone graft substitues Bone cements Advantages Biocompatible High wear resistance Light weight Disadvantages Low tensile strength Difficult to fabricate Low toughness Not resillient 10

CLASSIFICATION OF BIOCERAMICS • • Alumina Zirconia Pyrolytic Carbon Calcium phosphates Hydroxyapatite (Ca 10(PO 4)6(OH)2) Tricalciumphosphates • Bioactive glasses and glass ceramics 11

Bioceramics must have these properties : • • • Non- toxic Non-allergic Non-cancerogenic effect Must not inflame Must be biofunctional for long life Biocompatible Classification of Ceramics 1. Bioinert (There is not any chemical change. If there is a change it is controlled by human body’s natural regulator mechanism. ) Alumina , Zirkonium, Silicone nitride, carbon 2. Bioactive (surface reactive ceramics) Glass ceramics, dense hydroxyapatite 3. Biodegredable or resorbable Calcium phosphate, calcium aluminate 12

Classification of Bioceramics According to Tissue Response Implant Type Tissue Response Example Non-Porous intense and inert ceramics Very thin fibrous tissue forming Alumina, zirkonium (morphologic fixation) Porous inert ceramics Tissue growing inside pores Hydroksiapatite, HA (biological fixation) Metals covered with HA, bioactive ceramics Tissue implant interface binding Bioactive glasses (bioactive fixation) Glass ceramics HA Resorbable ceramics phosphate Absorbed Tricalcium Bioactive glasses This classification has no distinct borders. Hydroxyapatite is a bioactive ceramic but porous forms of it shows biodegradable feature. 13

Tissue response to Implant material : 1. If the material toxic, surrounding tissue is dying. 2. If the material is not toxic but is bioinert, fibrous (filamantous capsule) tissue is formed in different thickness. 3. If the material is not toxic but is bioactive, there is a bound between tissue-implant interface. (bioactive surface) 4. If the material is not toxic but is resorbable , surrounding tissue replace implant. 14

Morphological fixation: • Filamontous tissue • Thin tissue at optimum conditions, thick interface layer for metals • Metal, polymer and ceramics • Also compatibility, and mobility at the interface affects the thickness of the layer. • Al, Zr : Mechanical suitability and clinical success 15
Biological fixation • Porous implant : Pores must has 100 -150 µm radious for tissue liveliness and wellness. • Increased surface area due to proliferation between implant and tissue, develop a resistance against implant’s movement. • Surface area determined by growth tissue in pores. So it is called biological fixatıon. • Porous metal state: metal loss and corrosion. • High porosity reduce material resistance. • For interface stability metal alloys covered by porous ceramics. 16

Resorbable implants after definite usage period graded disintegration İnterface is very thin or there is not any interface. Important Points at Improvement of Resorbable ceramics: 1. During disintegration, interface stability and resistance must be protected 2. It must be suitable for tissue type, tissue regeneratıon speed due to life and health situatıon, absorptıon speed. 3. Material must consist of substances that are only adopted metabolic. Otherwise chronic pyogenesis and pain starts. 4. Chemically broken down by the body ad degrade 5. Resorbed material is replaced by endogenous tissue. TCP: Jaw and head regeneration of bioactive glass bone 17

Bioactive material: An other solution for interface problems is usage of bioactive material. It perform bone formation between material and tissues by specific biological reaction at interface. At this approach, many bioactive material produced which has different binding time, resistance and mechanism. Example bioglass; bioactive glass ceramics(cerevital, A-W glass ceramic; Dense HA(dupotit)) 18
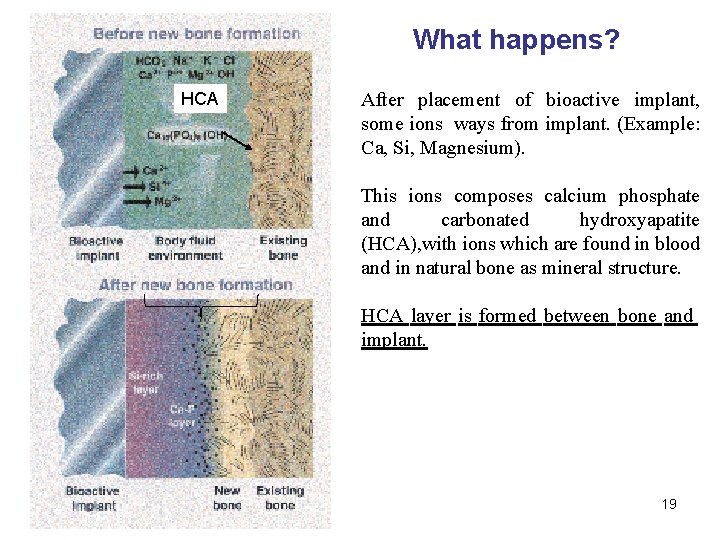
What happens? HCA After placement of bioactive implant, some ions ways from implant. (Example: Ca, Si, Magnesium). This ions composes calcium phosphate and carbonated hydroxyapatite (HCA), with ions which are found in blood and in natural bone as mineral structure. HCA layer is formed between bone and implant. 19
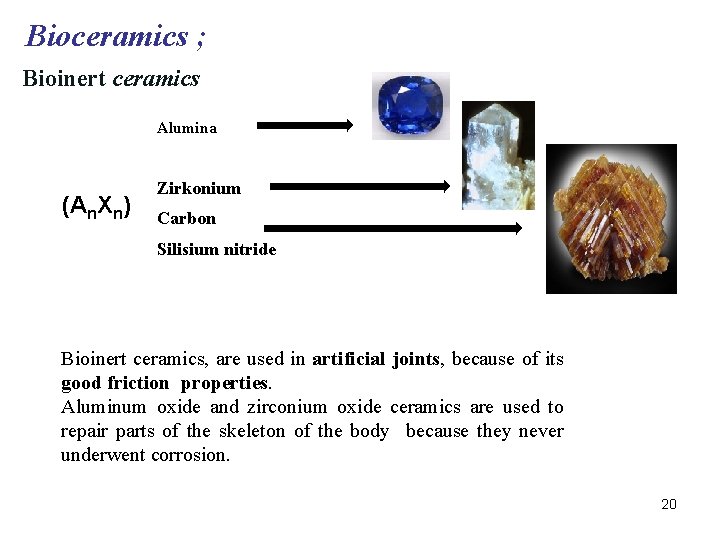
Bioceramics ; Bioinert ceramics Alumina (An. Xn) Zirkonium Carbon Silisium nitride Bioinert ceramics, are used in artificial joints, because of its good friction properties. Aluminum oxide and zirconium oxide ceramics are used to repair parts of the skeleton of the body because they never underwent corrosion. 20

Alumina (Al 2 O 3): • Natural • Polycrystalline Bauxite and natural corundum is the most common naturally occurring crystalline form of aluminium oxide BAYER PROCESS: In the Bayer process, bauxite is digested by washing with a hot solution of sodium hydroxide, Na. OH, at 175 °C. This converts the aluminium oxide in the ore to sodium aluminate. The other components of bauxite do not dissolve. The solution is clarified by filtering off the solid impurities. The mixture of solid impurities is called red mud, and presents a disposal problem. Next, the alkaline solution is cooled, and aluminium hydroxide precipitates. Then, when heated to 980°C (calcined), the aluminium hydroxide decomposes to aluminium oxide. Approximately 93% of alumina is metallurgical alumina that is used to produce aluminum. 21

Alumina (Al 2 O 3): • The implant use: purity property (ASTM), includes 99. 6% alumina, 0. 12 % Si. O 2, 0. 03% Fe 2 O 3 and 0. 04 %Na 20 (weight base) -Flame fusion method (Verneuil Process): • An ultra-thin alumina powders that are filtrated from electric arc or flame oxihidrogen are slowly fed to one crystal. • Growth up to 10 -cm diameter can be made with this method. -Drawing method (Czochralski Process) • Crystal grain • Mold 22

Alumina (Al 2 O 3): • Most widely used alumina can be prepared with calsinatıon of alfa alumina trihidrate. Include (Weight ratio): %99. 6 alumina, %0. 12 Si. O 2, %0. 03 Fe 2 O 3 and %0. 04 Na 2 O. • Aluminium pieces are obtained by mixing aluminium powder with organic binder and than sintering. • Highly stable oxide-very chemically inert • Low fracture toughness and tensile strength • Alumina which are used at applications mostly has good grain structure, are obtained by milling and sintering polycristal alfa Al 2 O 3 at 1600 -1700 °C. • Very Low friction and wear, being inert in vivo permits the use of it in joints. Its high hardness allows the use of it as a carrier element at active points. This high hardness, make advantageous use of alumina at joints despite brittleness. 2000 kg/mm 2(19. 6 GPa) -3000 kg/mm 2 (29. 4 GPa) • • 23
Alumina (Al 2 O 3): • 350 day in animals there is not any non acceptance and toxic properties. Used as: Hip prosthesis Al 2 O 3+UHMWPE; • chemically inert, • high-strength, • high resistance to abrasion, • low friction coefficient, • excellent corrosion resistance • low formation of the wound • Dental implants. 24
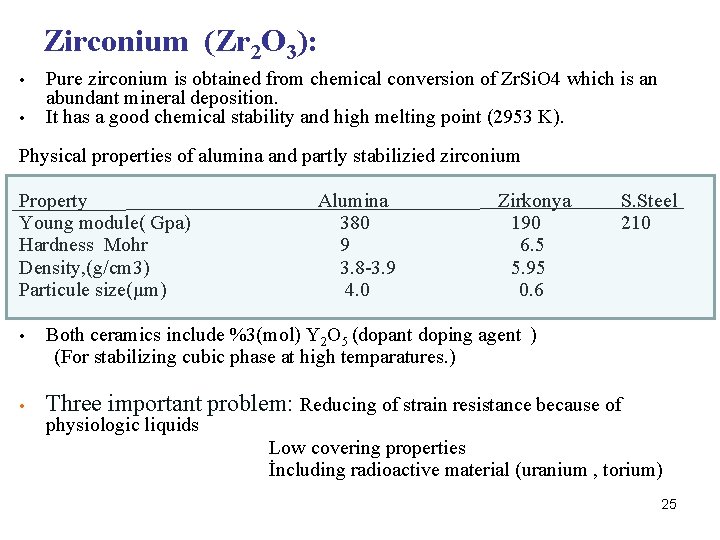
Zirconium (Zr 2 O 3): • • Pure zirconium is obtained from chemical conversion of Zr. Si. O 4 which is an abundant mineral deposition. It has a good chemical stability and high melting point (2953 K). Physical properties of alumina and partly stabilizied zirconium Property Alumina Zirkonya S. Steel Young module( Gpa) 380 190 210 Hardness Mohr 9 6. 5 Density, (g/cm 3) 3. 8 -3. 9 5. 95 Particule size(µm) 4. 0 0. 6 • Both ceramics include %3(mol) Y 2 O 5 (dopant doping agent ) (For stabilizing cubic phase at high temparatures. ) • Three important problem: Reducing of strain resistance because of physiologic liquids Low covering properties İncluding radioactive material (uranium , torium) 25
Zirconium (Zr 2 O 3): Advantage of zirconium according to other ceramics, has much more high cracking and distortion resistance. • Zirconium used as femur bone prosthesis. • It is used as globe material at hip joint prosthesis because of its mechanical properties. • At animals perfect biocompatibility in bone, 350 days non-reactive • With UHMWPE perfect biocompatibility , abrasıon and friction resistance • Has already widely use to replace alumina and metals. 26
Zirconium (Zr 2 O 3): • Both of the ceramics include 3% (moles) Y 2 O 5 (dopant doping agent) (used to stabilize high temperature the cubic phase). • The advantage of zirconia to other ceramics, having a much higher resistance to cracking and warpage, so zirconia used in the femoral prosthesis. • It is used as sphere material of hip joint prosthesis system by its mechanical properties. • It showed excellent biocompatibility in animals bone, a non-reactive state in 350 days. • Along with UHMWPE excellent biocompatibility, wear and abrasion resistant • Three major problems: Reduction in tensile strength due to physiological fluids Being poor coating properties Contain radioactive materials (uranium, thorium)

Carbon • It is mostly found element at bioorganic compounds. Because of chemical and mechanical properties they are used partly or completely, for regeneratıon of damaged biological structures as bone, cartilage, tendon, bond and blood vessel • Carbon baed materials can be used as hard tissue implant materials because of their strength. • Carbons do not form a chemical bond with living tissue, but are not considered completely inert. • There are 3 types of carbon using in biomedical cycles: 1. Pyrolitic carbon 2. Vitrified carbon 3. Vapour collected carbon • • Other carbon forms treated for living tissues, too. Knit carbon fiber systems for regenaratıon of tendon and bonds, carbon-carbon composites are used as load carrier element at connectıon points instead of bone. Researchs are going on for using semi carbon forms to fine down blood and as adsorbent. The most ımportant advantage of it according to other materials is being perfect biocompability. Valve(Abrasıon resistance- strength’s not important) 28

Resorbable Ceramics Biodegradable=Bioabsorble a. Calcium phosphates ( β TCP, α TCP, Hydroksiapatite, Aluminium calcium phosphate ceramics (ALCAP) ) b. Coral c. Calcium sulphates (granule hydroksi apatite+calcium sulphate (Plaster of Paris ) bone filling material d. Fe-Ca P oxides e. Zn-Ca-P oxides, Zn-sulphate-Ca-P oxides Resorbable Ceramic Bone Graft Material 29
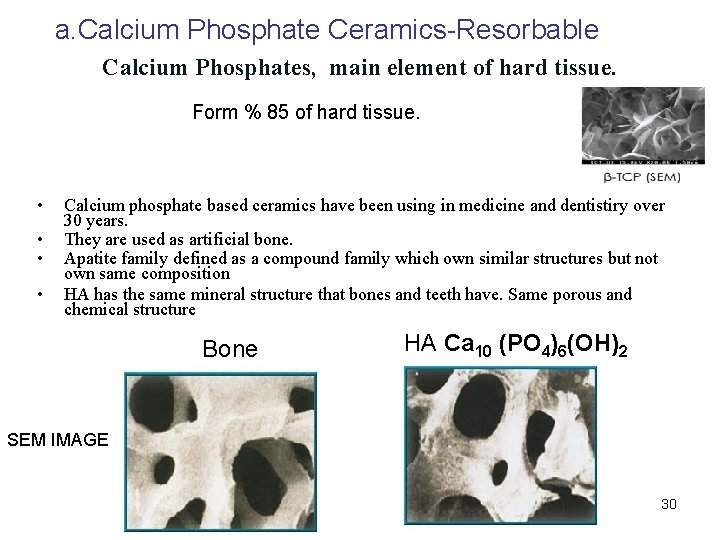
a. Calcium Phosphate Ceramics-Resorbable Calcium Phosphates, main element of hard tissue. Form % 85 of hard tissue. • • Calcium phosphate based ceramics have been using in medicine and dentistiry over 30 years. They are used as artificial bone. Apatite family defined as a compound family which own similar structures but not own same composition HA has the same mineral structure that bones and teeth have. Same porous and chemical structure Bone HA Ca 10 (PO 4)6(OH)2 SEM IMAGE 30
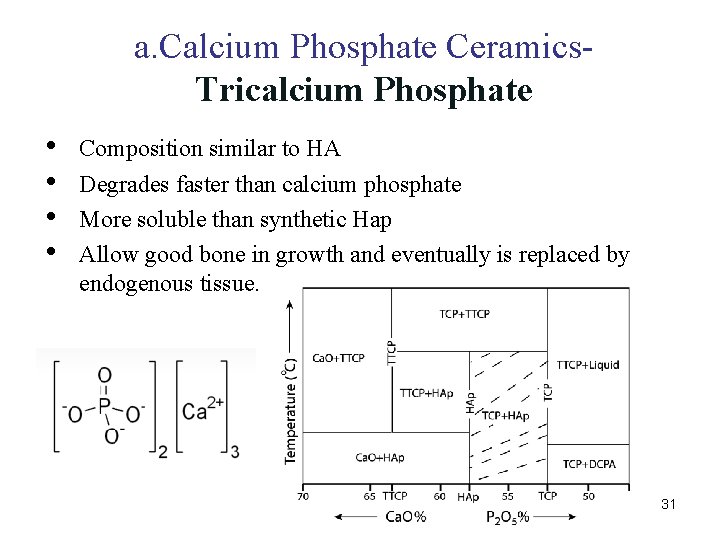
a. Calcium Phosphate Ceramics. Tricalcium Phosphate • • Composition similar to HA Degrades faster than calcium phosphate More soluble than synthetic Hap Allow good bone in growth and eventually is replaced by endogenous tissue. 31

Ca/P=1. 5 Tricalcium Phosphate • Tricalcium phosphate (TCP) is getting attraction as an another family of calcium phosphate for medical applications. • • Chemical formula is Ca 3(PO 4)2. It has 4 polymorph as α, β, ɣ, and super α. ɣ polymorph is high-pressure phase and super α polymorph is obtained at temperatures above approximately 1500 °C. α and β polymorph is the most commonly used TCP in bioceramics. TCP is a biodegradable ceramic and dissolution rates are as follows: HA< β TCP < α TCP • • • β-tricalcium phosphate α-tricalsium phosphate >1200º C β-whitlockite • • Osteoconductive Biodegradable (better than HA) 32

a. Calcium Phosphate Ceramics-Resorbable • • • The bones, dentin, tooth enamel, HA, protein, and other organic substances are natural composites containing water. Strength of bone tissue, is greater than the strength of all the other components. HA’s perfect biocompability , Osteoconductive properties( porous and bioresorbable) makes it mostly ued calcium phospate in medical area. Parts of human’s cortical and trabecular bone matrix is composed of two phases of HA and tricalcium phosphate. The main phase of natural calcium HA, shall not be less than 50% of cortical bone in a porous structure. Trabecular bones have a cage with 75% porosity. Hydroksiapatit (HA) 33

a. Calcium Phosphate Ceramics- Hydroxyapatite • HA is brittle as other ceramics. Implant material must be stronger than surrounding bone stucture. • Only HA is not suitable as implant material. • Dense (produced less than %5 porous and macroporous structure) • (500µ. pore sizes) • Dense HA powders are pressed as pellet and heat treatment applied. (Sintering) • Sintering treatment increase strength of last product’s structure. Small particle size increase strength but reduce biocompability. • Porous HA comparing to dense materials is more biocompatible so is used as bone filling material at non carried load regions. It forms a surface for bone growing and mechanically grabbing. • Disadvantage of porosity : Reduce mechanical propeties of compact material and , increase amount of corrossion and dissolutıon part due to enlarged surface area. 34
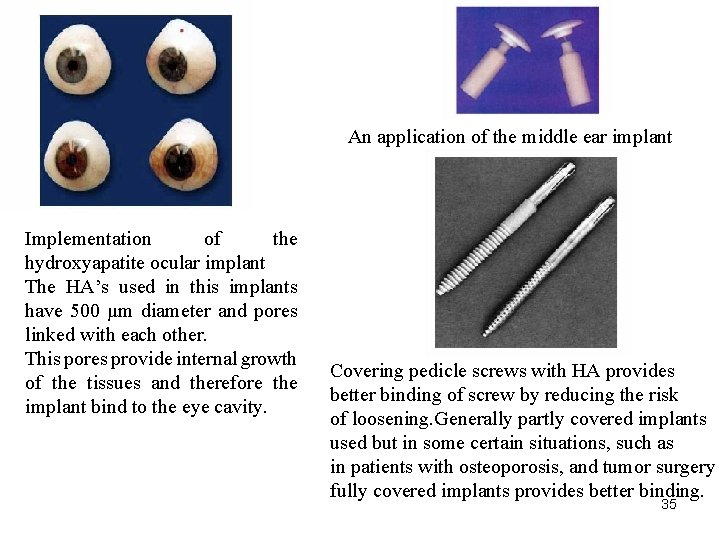
An application of the middle ear implant Implementation of the hydroxyapatite ocular implant The HA’s used in this implants have 500 μm diameter and pores linked with each other. This pores provide internal growth of the tissues and therefore the implant bind to the eye cavity. Covering pedicle screws with HA provides better binding of screw by reducing the risk of loosening. Generally partly covered implants used but in some certain situations, such as in patients with osteoporosis, and tumor surgery fully covered implants provides better binding. 35

a. Calcium Phosphate Ceramics. Aluminium Calcium Phosphate Ceramics (ALCAP) • Non-conducting, do not have magnetic or piezoelectric properties. • ALCAP are prepared from aluminum oxide, Ca oxide, and phosphorus penta-oxide powders. • Weight ratio: Al 2 O 3: Cao: P 2 O 5 50: 34: 16 • At this weight ratio • Calcination to 1350 °C for 12 hours • Milling • Elimination • Powders pressed in the form of solid blocks or empty cylinder, and 36 h sintering at 1400 °C 36
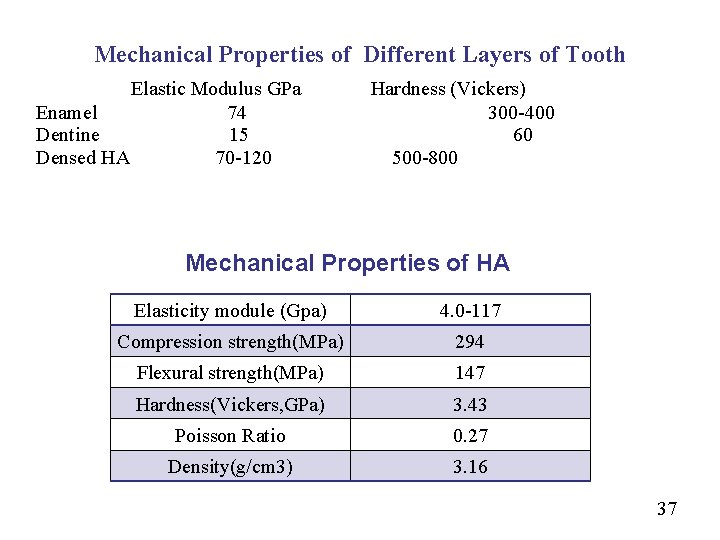
Mechanical Properties of Different Layers of Tooth Elastic Modulus GPa Hardness (Vickers) Enamel 74 300 -400 Dentine 15 60 Densed HA 70 -120 500 -800 Mechanical Properties of HA Elasticity module (Gpa) 4. 0 -117 Compression strength(MPa) 294 Flexural strength(MPa) 147 Hardness(Vickers, GPa) 3. 43 Poisson Ratio 0. 27 Density(g/cm 3) 3. 16 37

Calcium Phosphate Ceramics. USES • • Drug delivery systems Bone defects Gaps in the bone tumor Face and jaw surgery Ocular implant Dental Fillings Coatings for metal implants, eg. Heart valves to inhibit clotting, femur implant heads • Repair of herniated disks 38

39
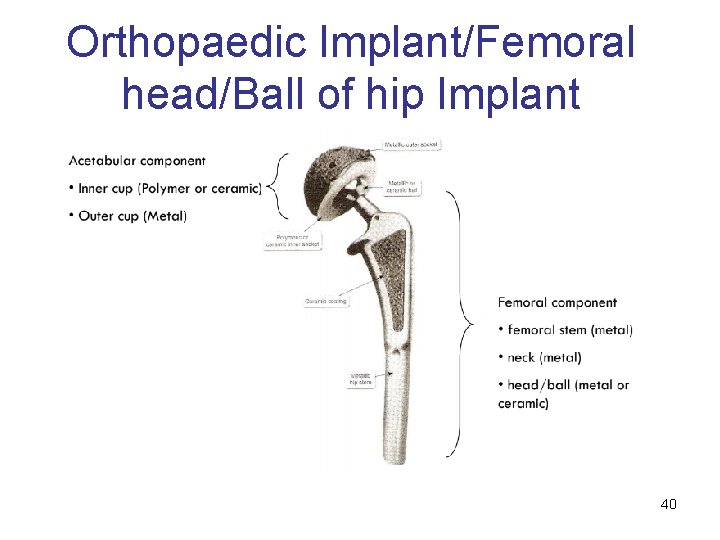
Orthopaedic Implant/Femoral head/Ball of hip Implant 40
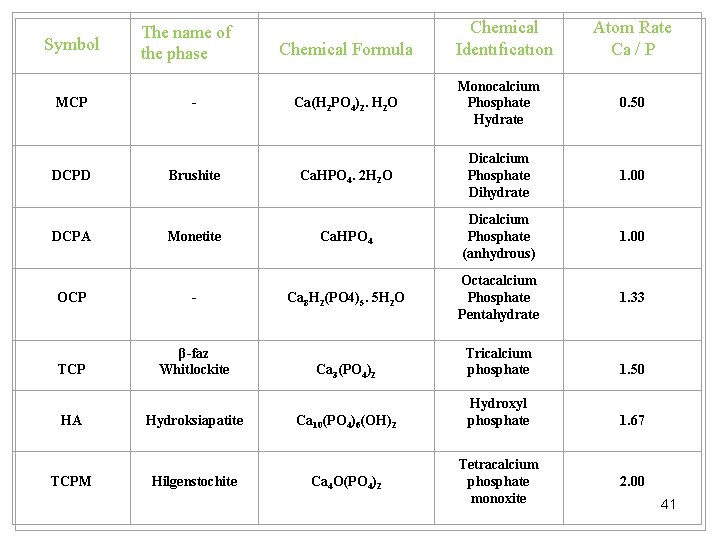
Symbol The name of the phase Chemical Formula Chemical Identıfıcatıon Atom Rate Ca / P MCP - Ca(H 2 PO 4)2. H 2 O Monocalcium Phosphate Hydrate 0. 50 DCPD Brushite Ca. HPO 4. 2 H 2 O Dicalcium Phosphate Dihydrate 1. 00 DCPA Monetite Ca. HPO 4 Dicalcium Phosphate (anhydrous) 1. 00 OCP - Ca 8 H 2(PO 4)5. 5 H 2 O Octacalcium Phosphate Pentahydrate 1. 33 TCP β-faz Whitlockite Ca 3(PO 4)2 Tricalcium phosphate 1. 50 HA Hydroksiapatite Ca 10(PO 4)6(OH)2 Hydroxyl phosphate 1. 67 TCPM Hilgenstochite Ca 4 O(PO 4)2 Tetracalcium phosphate monoxite 2. 00 41

Synthetic calcium phosphates Manufacturing Methods 1. Chemical Precipitation 2. Sol - Gel 3. Hydrothermal Techniques 4. Hydrolysis of Calcium Phosphates 42

1. CHEMICAL PRECIPITATION • For Chemical Precipitation as a calcium and phosphate source; Ca(OH)2 and H 3 PO 4 Ca(NO 3)2 and (NH 4)2 PO 4 Ca. Cl 2 and Na. H 2 PO 4 Ca/P 10: 6 Hydroxiapatite, Ca 10(PO 4)6(OH)2 3: 2 β-tricalcium phosphate Ca 3(PO 4)2 • Washing and drying • Compression to speed up crystallization • Cooking 43

PRODUCTION Precipitation from Ca(NO 3)2 and (NH 4)2 PO 4 containing aqueous solutions Calcination at 1150 °C for 1 hour Grinding and sieving (bone additive and ceramic matrix drug delivery system) Pressing and sintering at 1150 -1200 ° C. 44

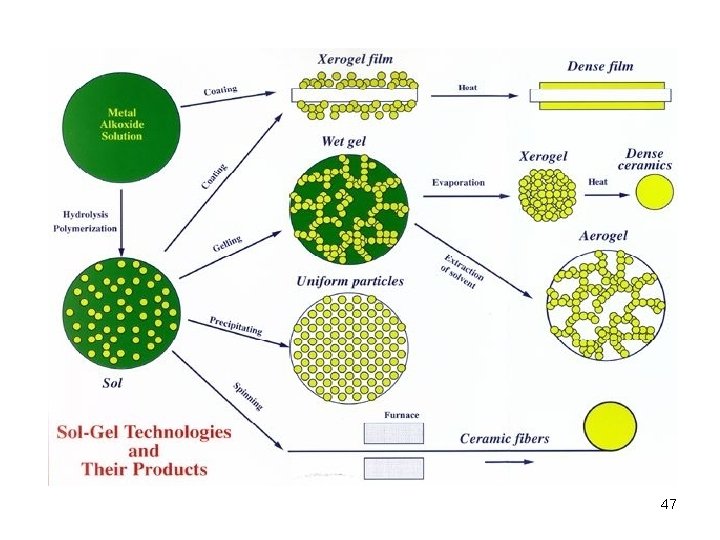
2. SOL-GEL METHOD • Sol-gel method is most commonly used method for making homogeneous and high purity materials, also that allows features such as homogeneity of particle size distribution and chemical structure, purity. • Sol; a liquid phase (continuous phase) containing dispersed solid particles, • Gel, a structure between the solid phase and liquid phase (continuous phase is the phase of solid) 45

The principle of the sol-gel method; • To prepare homogeneous solutions of the very pure starting materials. • To add required substances to the solution to obtain sol. • To ensure condensation reactions on sol to obtain gel from mixture. • To do appropriate processing of the gel (heat) to achieve designed material. 46
47

b. Coral (Coralline) • The porous structure of the coral is a rare structure among marine in vertebrates. It is used in bone implants. Calcium carbonate, the main component, is absorbed in the body gradually. Porous structure is suitable for applications in bone tissue by resembling a human bone. Coral and coralline HA are used as orbital implant and bone tissue since 1980. Because porous structure allows the growth of blood vessels in the bone. • Coral with a hydrothermal process can be converted to hydroxyapatite. Interpore 200 is a coral-hydroxy apatite. Similar to the spongy bone. This coralline HA is used to repair injured bone. • Due to pore structure compressive stress 26 -395 MPa (50% porous-dense) and modulus of elasticity (8100 GPa) changes. 48

c. Calcium Sulphates • Calcium sulfate has been used for bone substitutes for long time. Peltier et al. (1957) mixed a plaster of paris (POP), calcium sulfate hemihydrates, and water allowing to set in the mold to make column- and tablet-form calcium sulfate dihydrate. The set plaster was used to fill defects in dog bone. They observed bone regeneration as the calcium sulfate was 49 absorbed

d. Ferric calcium phosphate oxide ceramics (FECAP) • • • They are prepared from Fe III oxide, Ca. O, P 2 O 5 powders. Powders are mixed in various weight ratios with blender. Pressed and calcined at 1100 °C for 1 h. Calcined ceramic blocks are crushed and grinded in a ball mill. The desired particle size obtained. It is used for artificial bone formation and to eliminate bone defects on people that have anemia, and other similar diseases. 50

e. Zinc calcium phosphate ceramics (ZCAP) • • Zn is used to repair wound healing, bone defects and carrier systems. Nonconducting, don’t have magnetic or piezoelectric properties. Zinc oxide, are produced by mixing the calcium oxide, and phosphorus pentaoxide powders in a hot environment. Oxide powders are mixed in a ball mill, and is calcined at 1800 ° C for 24 hours. Calcined ceramic are eliminated and milled to the desired particle size. e. Zinc sulfate calcium phosphate ceramics (ZSCAP) • • • Zinc sulfate, is obtained by the processing of calcium phosphate ceramics, zinc sulfate, zinc oxide, calcium oxide and phosphorus penta-oxide (15. 30: 25). 24 -hour calcination at 650 ° C Grinding and sieving Hardening by adding water The insertion of it into the implantation made bone, eliminates errors that can be found in bone. 51
General Usage • Powder, Granules • Plate • Implant Coating • Gel 52

HAPEX • • • Bioceramic-polymer composite Composed of organic matrix that is reinforced by cortical bone mineral component, HA containing 50% by volume high-density polyethylene matrix, has a high hardness and a rigidity similar to cortical bone, Osteoblasts proliferate on Hydroksiapatite in the surface of HAPEX. • In vivo tests showed that a strong and stable interface between the material • and bones was developed. The middle ear implant (HA heads, HAPEX shafts) 53
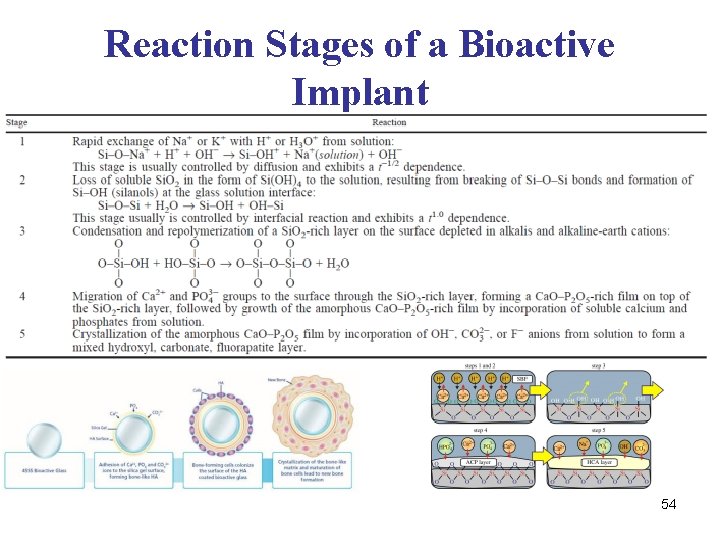
Reaction Stages of a Bioactive Implant 54
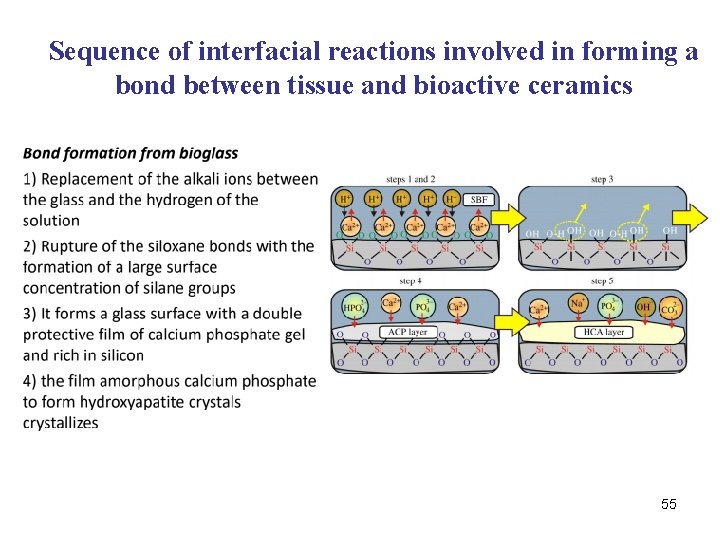
Sequence of interfacial reactions involved in forming a bond between tissue and bioactive ceramics 55
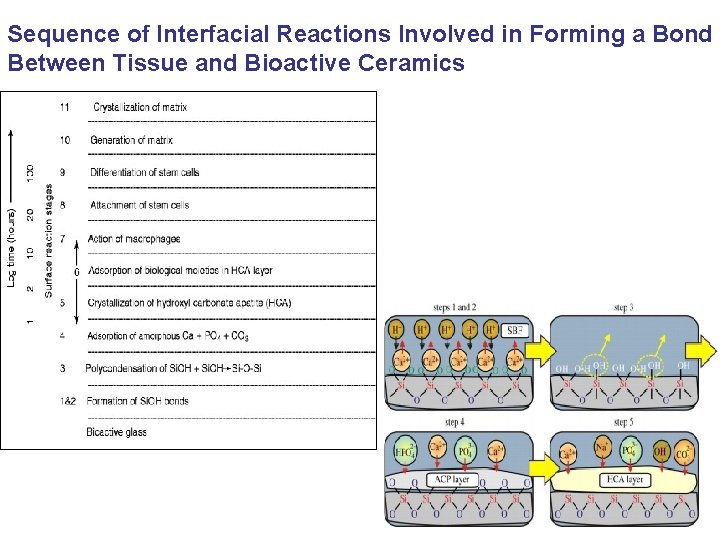
Sequence of Interfacial Reactions Involved in Forming a Bond Between Tissue and Bioactive Ceramics 56
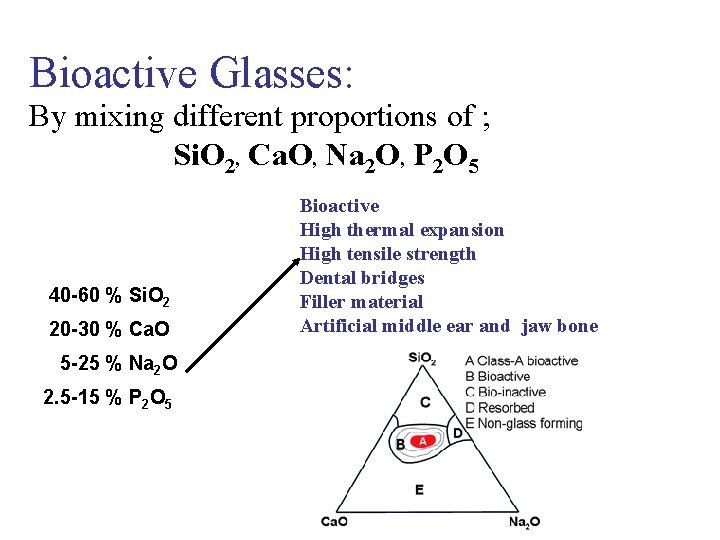
Bioactive Glasses: By mixing different proportions of ; Si. O 2, Ca. O, Na 2 O, P 2 O 5 40 -60 % Si. O 2 20 -30 % Ca. O 5 -25 % Na 2 O 2. 5 -15 % P 2 O 5 Bioactive High thermal expansion High tensile strength Dental bridges Filler material Artificial middle ear and jaw bone

Bioactive Glasses: Three key feature of components creates bioactivity; 1. % Si. O 2 (moles) content of less than 60 2. High Ca. O and Na 2 O content 3. High Ca. O/P 2 O 5 45 S 5 Bioglass (patent) %45 Si. O 2 %6 P 205 %24. 5 Cao %24. 5 Na 2 O 5: 1 Cao-P 205 EN: 1300 ° C Binding to the bone does not exist in lower rates of Ca. O-P 205. In the early 1970 s, Larry Hench (U of Florida), showed that some glass compositions is connected to the bone. 58

Bioactive Glasses • Bone tend to create a chemical bond with the bioglass. In the body fluid, before the surface of bioglass the alkali part soluble, calcium, phosphorus and sodium oscillation occurs. The released material is mixed with body fluid and creates an intermediate layer on the surface of the implant. This layer has a structure that is ideal for the growth of bone. After implantation calcium phosphate crystals forms by precipitation of bone minerals on the surface of the implant and by this interfacial layer of crystals chemically linked through the glass surface. • Therefore, these materials are considered as bioactive materials. 59

Glass Ceramics • Silica based ceramics. • • Glass ceramics are multi-crystalline materials that are obtained by crystallization of glasses by heat treatment. All of the glass phase does not often crystallized due to process. Crystalline phase or phases formed and their percentages changes due to heat treatment program during the production(nucleation and crystal growth temperature and the retention times at these temperatures). One of the most important factor affecting the properties of glass ceramics contained crystalline phase (residual glass phase) ratio. The properties such as material strength, fracture toughness, modulus of elasticity, machinability vary with the amount of the formed crystalline phase. High resistance to chemical stability, electrical resistance, thermal shock resistance and lower thermal expansion coefficient. To create glass ceramic from 45 S 5 glass, 2 type of composition are important: Magnesium alumina silicates and Li alumina silicates, Attachment to bone depends on the formation of a film on the surface that is rich by calcium phosphate silicon oxide. Glass ceramics leads to bone growth faster than HA (hydroxy apatite). It is found that mineralized layers on the surface of glass ceramics have a direct connection to the osteoblasts. 60

A Special Bioglass Oven 61

Glass Ceramics • Another example of bioactive ceramics is glass ceramics used in dental bridge. • The purpose of using glass ceramics as dental bridge giving the appearance of aesthetic tooth and reducing tooth plaque formation. • Glass ceramics also is used to change the vertebrae. • The use of glass ceramics for this purpose is because of their durability and high connectivity to bone. 62

Glass Ceramics In addition, glass ceramics are used in; glasses, diagnostic devices, thermometers, tissue culture dishes, fiber optics in endoscopy 63

Glass Ceramics Itriya Aluminosilicate glass microspheres are used in the treatment of cancer. The microspheres provides transmission radiation in local regions of cancer cells of body. 17 Y 2 O 3 -19 Al 2 O 3 -64 Si. O 2 (mole%) glass Microspheres are 20 -30 µm in diameter. After the injection of it into the hepatic vein, SEM image of Y O microspheres 2 3 it prevent the flow of blood entering the blood vessels in the region of cancer and emit β radiation (64 -hour semi- life). 64
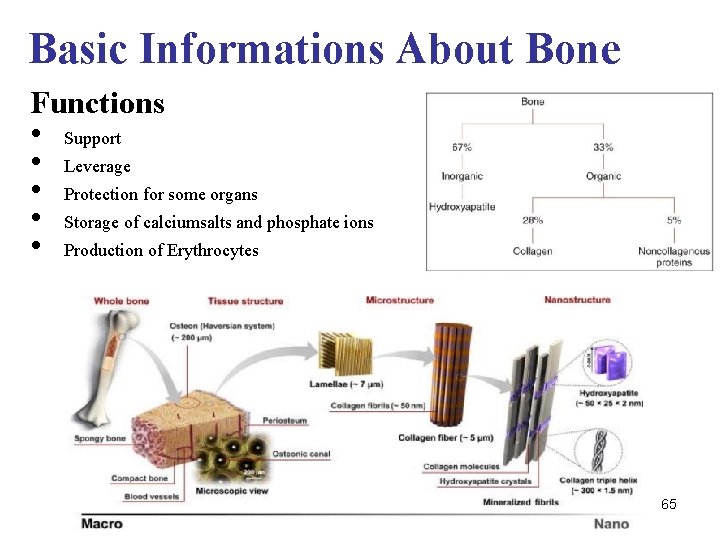
Basic Informations About Bone Functions • Support • Leverage • Protection for some organs • Storage of calciumsalts and phosphate ions • Production of Erythrocytes 65
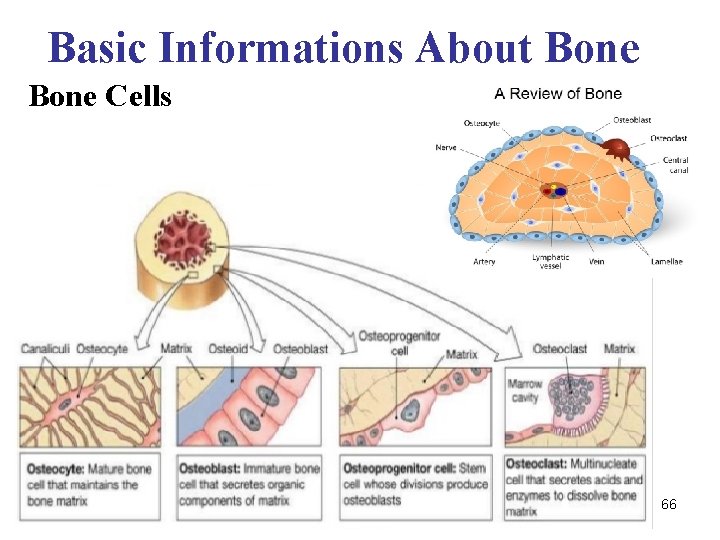
Basic Informations About Bone Cells 66
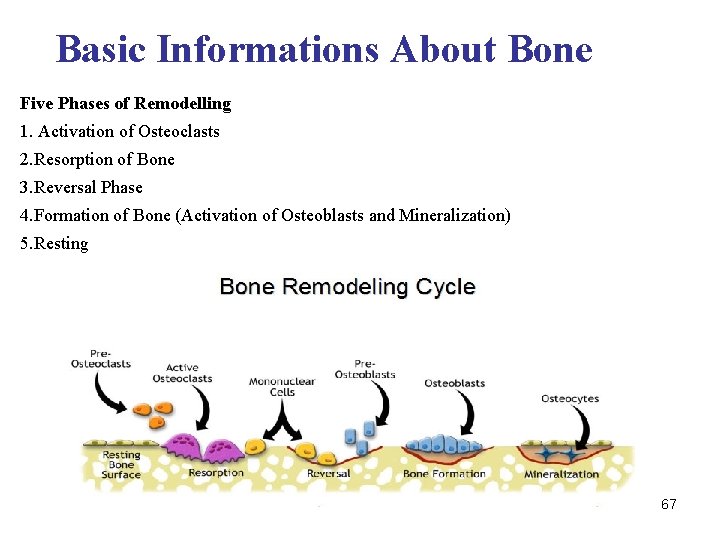
Basic Informations About Bone Five Phases of Remodelling 1. Activation of Osteoclasts 2. Resorption of Bone 3. Reversal Phase 4. Formation of Bone (Activation of Osteoblasts and Mineralization) 5. Resting 67
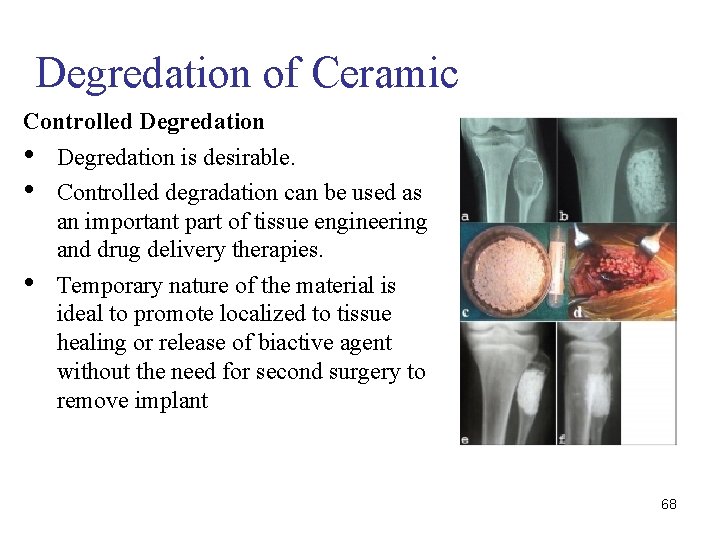
Degredation of Ceramic Controlled Degredation • Degredation is desirable. • Controlled degradation can be used as an important part of tissue engineering and drug delivery therapies. • Temporary nature of the material is ideal to promote localized to tissue healing or release of biactive agent without the need for second surgery to remove implant 68
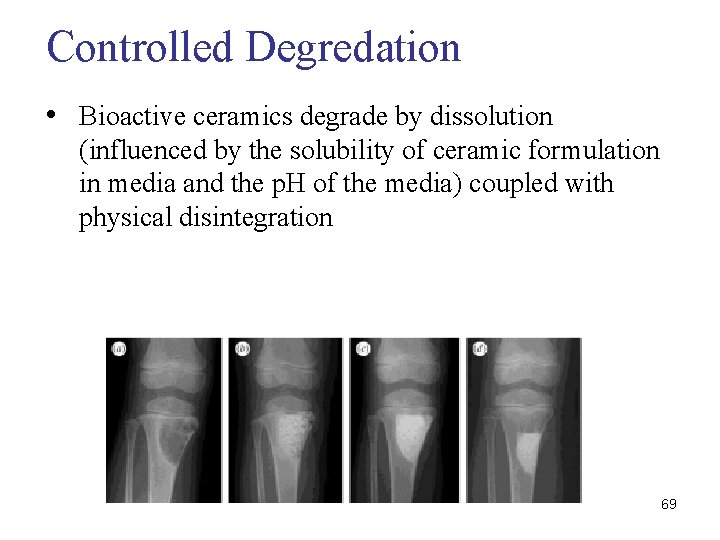
Controlled Degredation • Bioactive ceramics degrade by dissolution (influenced by the solubility of ceramic formulation in media and the p. H of the media) coupled with physical disintegration 69
- Slides: 69