Bioc 301 Inorganic biochemistry Course Outline and Objectives

Bioc 301 Inorganic biochemistry Course Outline and Objectives q Bioinorganic chemistry is concerned with the roles of inorganic elements in biological processes. q In bioc 301, we will apply fundamental principles of inorganic chemistry, particularly transition metal coordination chemistry and ligand field theory, to understand the structure and function of metal ion sites in biomolecules. q We will also consider bioinorganic topics including metal toxicity, the use of metal complexes as drugs and the bioregulatory functions of nitric oxide. 1

Biologically Important Elements Which “inorganic” elements are important biologically? 99% of human body is comprised of 11 elements Bulk biological elements: H, C, N, O, P, S, Cl (as PO 43 -, SO 42 -, Cl-) Bulk metal ion nutrients: Na, Mg, K, Ca Essential elements for a wide range of bacteria/plants/animals Transition metals: V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Mo Non-Metals: (B), F, (Si), Se I, F. 2

Periodic table Russian chemist Dmitri Mendeleev in 1869 • Standard table contains 117 elements (while element 118 has been synthesized, • element 117 has not). • Ninety-two elements are found naturally on Earth, and • the rest are synthetic elements that have been produced artificially in particle accelerators • A particle accelerator is a device that uses electromagnetic fields to propel charged particles to high speeds and to contain them in well-defined beams 3
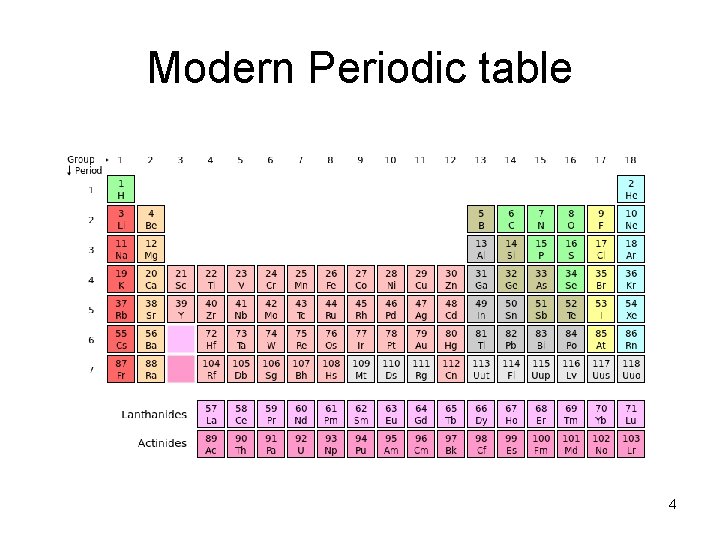
Modern Periodic table 4
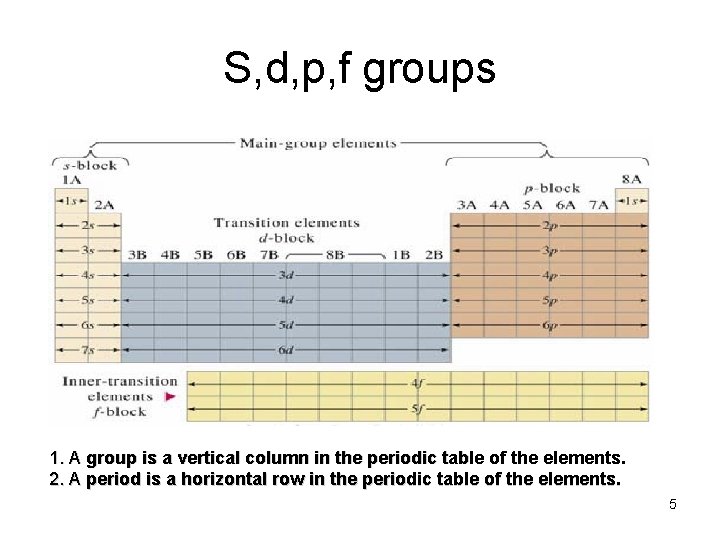
S, d, p, f groups 1. A group is a vertical column in the periodic table of the elements. 2. A period is a horizontal row in the periodic table of the elements. 5
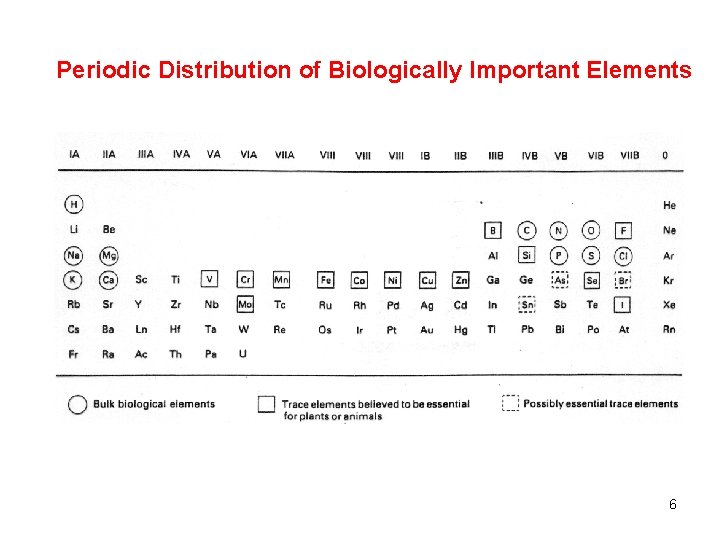
Periodic Distribution of Biologically Important Elements 6
Metals • Metals fall into groups in the periodic table determined by similar arrangements of their orbital electrons and a consequent similarity in chemical properties. • Groups of similar metals include the • • • alkali metals and the alkaline-earth metals (s-block), transition metals (d-block) and the rare-earth metals (the lanthanide and actinide series of Group 3 see f-block). • Most metals other than the alkali metals and the alkaline earth metals are called transition metals or transition elements. • 7

Non -metals and metalloids • Non-metals are found in the upper right hand side of the periodic table in the p-block region. • While metalloids are in the intersection between the metals and the non-metals and they roughly form a right hand leaning diagonal from top to bottom across the periodic table • A metalloid is a chemical element with properties that are in-between or a mixture of those of metals and nonmetals. • The six metalloids are boron(B), silicon(Si), germanium(Ge) , Arsenic (As), Antimony(Sb), and Tellurium(Te). The properties of the metalloids have characteristics in between that of the metals and the nonmetals. They are good conductors of heat and electricity, but they are not perfect good conductors or insulators. • • 8

Evolution of biological roles for essential metals Why have certain elements been "selected" for use in biological systems? a. their abundance (availability in the earth’s crust or oceans) b. their basic fitness (intrinsic chemical suitability) c. evolutionary adaption to realize critically required specificity. 9

• Lighter elements are more abundant in general and therefore utilized more. 3 d metals, rather than 4 d, are used as catalytic centers in metalloenzymes. • Why has Mo (4 d) rather than Cr (3 d) been utilized more biologically? Although Mo is rare in the earth’s crust, Mo is the most abundant transition metal in sea water as Mo. O 4 has fairly high solubility in water. Better correlation exists between the abundance of elements in in human body and in sea water than between the human body and the earth's crust. Taken as evidence for the oceans as the site of evolution of life. • Despite the high abundance of Si, Al and Ti (the 2 nd, 3 rd and 10 th most abundant elements on earth). Why are they are not utilized biologically? • Because of the insolubility of their naturally occurring oxides (Si. O 2, Al 2 O 3, Ti. O 2) under physiological conditions. A lower oxidation state is unavailable for Si and Al and unstable for Ti in an aerobic environment and is readily oxidized to Ti(IV) at p. H 7. 10 •

• Why has iron been used so widely in biology although Fe 3+, its most stable oxidation state, is highly insoluble at p. H 7 Complex biological mechanisms have been developed to accommodate the low solubility of Fe(OH)3 (Ksp = 1 x 10 38) ~ p. H 7, and take advantage of its high "availability". • Co 2+ and Zn 2+ have very similar coordination chemistry and ionic size and can be interchanged in many Zn enzymes without loss of activity. Why is Co not utilized more biologically? Zn is much more abundant and therefore has been utilized more. • Why has cobalt been given an essential role in cobalamins despite its very low availability? • The unique properties of cobalt (e. g. its oxidation states, redox potentials and coordination chemistry) is needed to achieve essential functions of B 12 coenzymes. 11

Indicators of Biologically Important Elements • Relative abundance of inorganic elements in earth's crust and in seawater. • Availability of elements from earth’s crust and sea water • Elemental composition of human body • Essential inorganic elements in food • Inorganic elements linked to deficiency symptoms 12
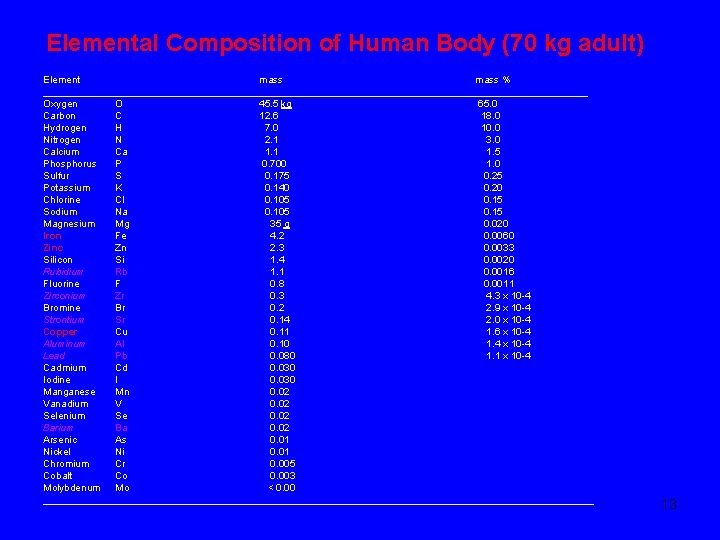
Elemental Composition of Human Body (70 kg adult) Element mass % _________________________________________________ Oxygen O 45. 5 kg 65. 0 Carbon C 12. 6 18. 0 Hydrogen H 7. 0 10. 0 Nitrogen N 2. 1 3. 0 Calcium Ca 1. 1 1. 5 Phosphorus P 0. 700 1. 0 Sulfur S 0. 175 0. 25 Potassium K 0. 140 0. 20 Chlorine Cl 0. 105 0. 15 Sodium Na 0. 105 0. 15 Magnesium Mg 35 g 0. 020 Iron Fe 4. 2 0. 0060 Zinc Zn 2. 3 0. 0033 Silicon Si 1. 4 0. 0020 Rubidium Rb 1. 1 0. 0016 Fluorine F 0. 8 0. 0011 Zirconium Zr 0. 3 4. 3 x 10 -4 Bromine Br 0. 2 2. 9 x 10 -4 Strontium Sr 0. 14 2. 0 x 10 -4 Copper Cu 0. 11 1. 6 x 10 -4 Aluminum Al 0. 10 1. 4 x 10 -4 Lead Pb 0. 080 1. 1 x 10 -4 Cadmium Cd 0. 030 Iodine I 0. 030 Manganese Mn 0. 02 Vanadium V 0. 02 Selenium Se 0. 02 Barium Ba 0. 02 Arsenic As 0. 01 Nickel Ni 0. 01 Chromium Cr 0. 005 Cobalt Co 0. 003 Molybdenum Mo < 0. 00 __________________________________________________ 13

q Mammals are believed to use only 25 of the known elements. q Eleven non-transition elements that make up 99. 9% of the human body (O, C, H, N, Ca, P, S, K, Cl, Na, Mg), q Three transition metals, Fe, Zn and Cu are needed in significant amounts. q “Trace quantities” of many other transition elements are required to maintain proper physical functioning. q Other elements in the human body (e. g. Rb, Zr, Sr, Al, Pb, Ba) are not essential but incorporated inadvertently because they share chemical and physical properties with essential elements. q Other elements are added to the list of elements thought to be essential as our knowledge of the chemistry of living systems increases. 14

Essential Inorganic Elements in Adult Diet ______________________________ Recommended Daily Allowance (mg) ______________________________ K Na Ca Mg Zn Fe Mn Cu Mo Cr Co Cl PO 43 SO 42 I Se F 2000 - 5500 1100 - 3300 800 - 1200 300 - 400 15 10 - 20 2. 0 - 5. 0 1. 5 - 3. 0 0. 075 - 0. 25 0. 05 - 0. 2 ~ 0. 2 3200 800 - 1200 10 0. 15 0. 05 - 0. 07 1. 5 - 4. 0 _______________________________ 15

Symptoms of Elemental Deficiency in Humans _____________________________ Ca Retarded skeletal growth Mg Muscle cramps Fe Anemia, immune disorders Zn Stunted growth, skin damage, retarded maturation Cu Liver disorders, secondary anemia Mn Infertility, impaired skeletal growth Mo Retarded cellular growth Co Pernicious anemia Ni Depressed growth, dermatitis Cr Diabetes symptoms Si Skeletal growth disorders F Dental disorders I Thyroid disorders Se Cardiac muscular weakness As Impaired growth (in animals) ____________________________ 16
17

Biological Roles of Metallic Elements. Structural Skeletal roles via biomineralization Ca 2+, Mg 2+, P, O, C, Si, S, F as anions, e. g. PO 43 , CO 32. Charge neutralization. Mg 2+, Ca 2+ to offset charge on DNA - phosphate anions Charge carriers: Na+, K+, Ca 2+ Transmembrane concentration gradients ("ion-pumps and channels") Trigger mechanisms in muscle contraction (Ca). Electrical impulses in nerves (Na, K) Heart rhythm (K). Hydrolytic Catalysts: Zn 2+ , Mg 2+ Lewis acid/Lewis base Catalytic roles. Small labile metals. Redox Catalysts: Fe(II)/Fe(IV), Cu(I)/Cu(II), Mn(II)/Mn(III)/(Mn(IV), Mo(IV)/Mo(VI), Co(I)/Co(III) Transition metals with multiple oxidation states facilitate electron transfer - energy transfer. Biological ligands can stabilize metals in unusual oxidation states and fine tune redox potentials. Activators of small molecules. Transport and storage of O 2 (Fe, Cu) Fixation of nitrogen (Mo, Fe, V) Reduction of CO 2 (Ni, Fe) Organometallic Transformations. Cobalamins, B 12 coenzymes (Co), Aconitase (Fe-S) 18

Transition Metals in Biomolecules Iron. Most abundant metal in biology, used by all plants and animals including bacteria. Some roles duplicated by other metals, while others are unique to Fe. Iron use has survived the evolution of the O 2 atmosphere on earth and the instability of Fe(II) with respect to oxidation to Fe(III). Zinc. Relatively abundant metal. Major concentration in metallothionein (which also serves as a reservoir for other metals, e. g. Cd, Cu, Hg). Many well characterized Zn proteins, including redox proteins, hydrolases and nucleic acid binding proteins. Copper Often participatse together with Fe in proteins or has equivalent redox roles in same biological reactions. Reversible O 2 binding, O 2 activation, electron transfer, O 2 - dismutation (SOD). Cobalt. Unique biological role in cobalamin (B 12 -coenzymes) isomerization reactions. Manganese Critical role in photosynthetic reaction centers, and SOD enzymes. Molybdenum Central role in nitrogenase enzymes catalyzing N 2 NH 3, NO 3 NH 3 Chromium, Vanadium and Nickel Small quantities, uncertain biological roles. Sugar metabolism (Cr); Ni only in plants and bacteria (role in CH 4 production) and SOD enzymes. 19

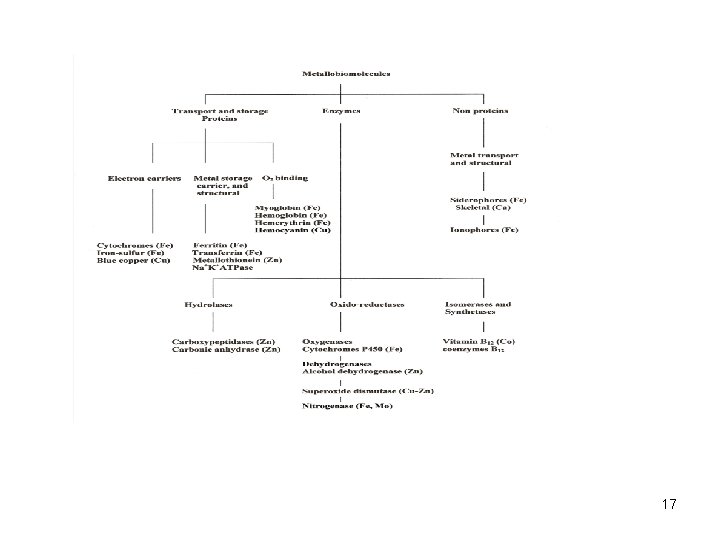
Biochemical Classification of Metallobiomolecules Transport and storage proteins : O 2 binding/transport: Transferrin (Fe) Ferritin (Fe) Metallothionein (Zn) Myoglobin (Fe) Hemerythrin (Fe) Hemocyanin (Cu) Enzymes (catalysts) Hydrolases: Oxido-Reductases: Isomerases: Carbonic anhydrase (Zn) Carboxypeptidase (Zn) Alcohol dehydrogenase (Zn) Superoxide dismutase (Cu, Zn, Mn, Ni) Catalase, Peroxidase (Fe) Nitrogenase (Fe, Mo) Cytochrome oxidase (Fe, Cu) Hydrogenase (Fe, Ni) B 12 coenzymes (Co) Aconitase (Fe-S) Oxygenases: Cytochrome P 450 (Fe) Nitric Oxide Synthases (Fe) Electron carriers: Electron transferases Cytochromes (Fe) Iron-sulfur (Fe) Blue copper proteins (Cu) Non Proteins Transport Agents: Siderophores (Fe) 20

21
- Slides: 21