Biobased Materials for Li ion Batteries Duncan J

Biobased Materials for Li ion Batteries Duncan J Macquarrie Xiao Wu, Mario De bruyn, Green Chemistry Centre of Excellence, Department of Chemistry, University of York P Hubert Mutin, Sanghoon Kim, Nicholas Brun, Nicholas Louvain, Johan Alauzin, Bruno Boury, Laure Monconduit, L Stievano, Inst Charles Gerhardt Montpellier, CNRS UMR 5253, CMOS TEAM, Montpellier, Peter Hurst, Mark Gronnow, Philip Lawless Biorenewables Development Centre, , York UK This project has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No 686163 www. greenchemistry. net

Introduction • Introduction to the project • Biobased mesoporous materials • Application range • Uses in Li ion batteries www. greenchemistry. net

Green Chemistry Centre of Excellence £ 5 M purpose-built Centre with Industrial Engagement Facility Currently at: ~100 Staff and Students www. greenchemistry. net

Multi partner EU project • Scale up to 20 kg / day (York, BDC, Leitat) • Catalysis applications (Johnson-Matthey, IBERcat) • Battery / energy storage applications ( Inst Charles Gerhard, Johnson Matthey, Varta Micro. Innovation) Current status: 4. 5 kg/hour expansion, ca. 1. 5 kg/day drying and 300 g/day pyrolysis This project has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No 686163 4 www. greenchemistry. net
Starbons – tunable mesoporous materials • Now being produced in multi-kg/day scale • Registered company successfully spun out from the University • Current customers Merck and Trio Healthca • 3 Patents and over 20 research articles • Scale-up development through Porous 4 Ap H 2020 commercialisation grant Enhanced CO 2 capture by waste-derived mesoporous carbons. Angewandte Chemie, 55, 9173 Treatment of laundrette wastewater using Starbon and Fenton’s reagent. J Env Sci and Health Revisiting the structure of mesoporous α-D-polysaccharide gels. Chem. Sus. Chem. 9(3), 280 -288. www. greenchemistry. net
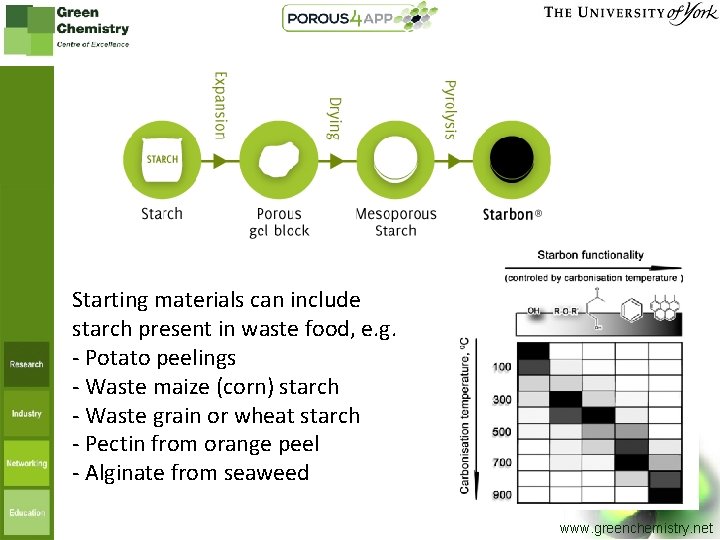
Starting materials can include starch present in waste food, e. g. - Potato peelings - Waste maize (corn) starch - Waste grain or wheat starch - Pectin from orange peel - Alginate from seaweed www. greenchemistry. net
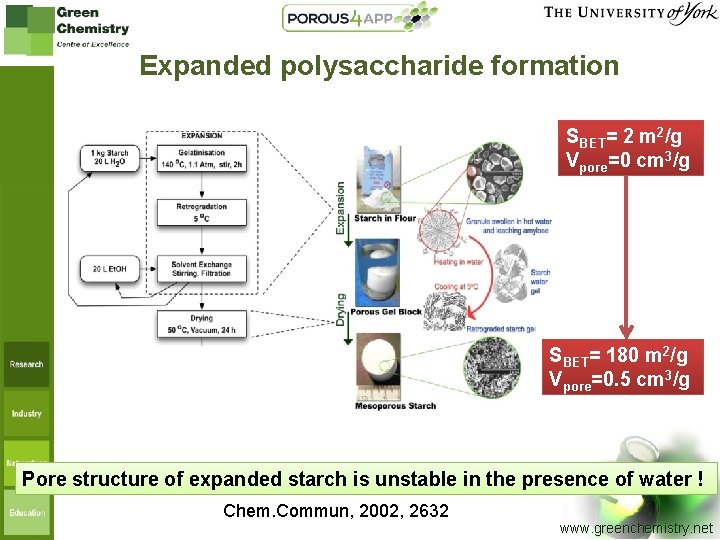
Expanded polysaccharide formation SBET= 2 m 2/g Vpore=0 cm 3/g SBET= 180 m 2/g Vpore=0. 5 cm 3/g Pore structure of expanded starch is unstable in the presence of water ! Chem. Commun, 2002, 2632 7 www. greenchemistry. net
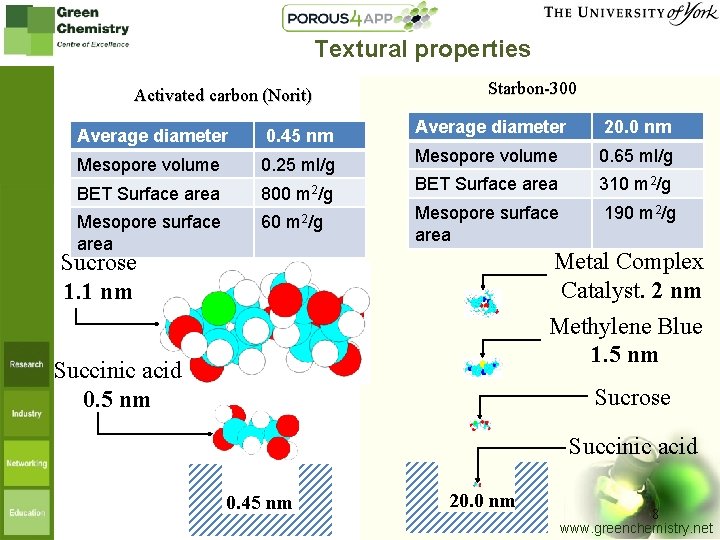
Textural properties Activated carbon (Norit) Average diameter 0. 45 nm Mesopore volume 0. 25 ml/g BET Surface area 800 m 2/g Mesopore surface area 60 m 2/g Starbon-300 Average diameter 20. 0 nm Mesopore volume 0. 65 ml/g BET Surface area 310 m 2/g Mesopore surface area 190 m 2/g Metal Complex Catalyst. 2 nm Methylene Blue 1. 5 nm Sucrose 1. 1 nm Succinic acid 0. 5 nm Sucrose Succinic acid 0. 45 nm 20. 0 nm 8 www. greenchemistry. net

Kilo scale production Robo. Qbo processor Ca. 15 kg / hour of polysaccharide This project has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No 686163 www. greenchemistry. net

Freeze dryer – ca. 1. 5 kg/day www. greenchemistry. net
3 stage continuous rotary furnace Capacity 30 kg/day Will arrive this week www. greenchemistry. net

Applications • Recovery of precious metals from waste streams through reductive adsorption (Starbon® R Series) • Purification, in particular removal of harmful organics and heavy metals to purify water and clean up waste streams (Starbon® P Series) • Separation of complex mixtures for production and analysis with Starbon® as the stationary phase in chromatographic systems (Starbon® S Series) • Catalysis of bio-refinery downstream processes including esterification reactions in aqueous systems (Starbon® C Series) • Catalysis using adsorbed metal complexes and nanoparticles • As a hybrid with mixed metal oxides for energy storage 12 www. greenchemistry. net
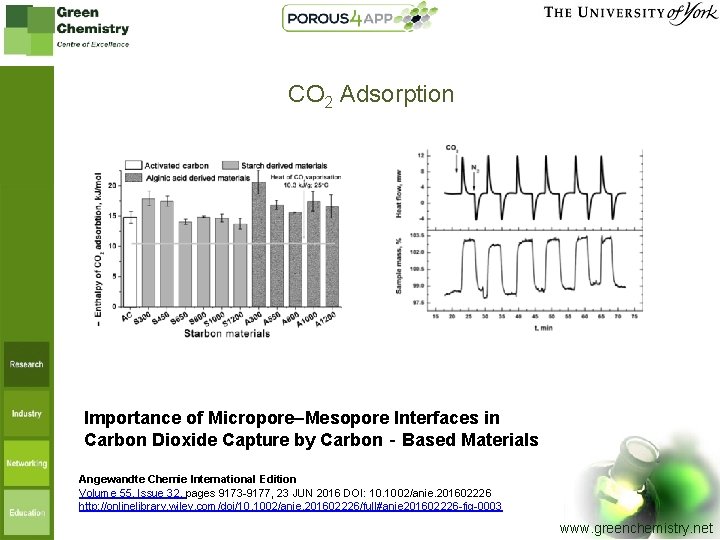
CO 2 Adsorption Importance of Micropore–Mesopore Interfaces in Carbon Dioxide Capture by Carbon‐Based Materials Angewandte Chemie International Edition Volume 55, Issue 32, pages 9173 -9177, 23 JUN 2016 DOI: 10. 1002/anie. 201602226 http: //onlinelibrary. wiley. com/doi/10. 1002/anie. 201602226/full#anie 201602226 -fig-0003 www. greenchemistry. net

Applications in Li ion batteries 1. Preparation of nanostructured Li. Mn 2 O 4 2. Use as a negative electrode material Focussed on Alginate based materials www. greenchemistry. net

Textural features of A 800 www. greenchemistry. net

Nano LMO synthesis Algibon as template and sacrificial reductant for nano LMO synthesis 4 Mn. O 4 - + 3 C 4 Mn. O 2 + CO 32 - + 2 HCO 3 – Carried out within the pore structure of the Algibon Reduction of KMn. O 4 known using organic reductants Little structural control Can we use mesoporous polysaccharides as structurants AND reductants? www. greenchemistry. net
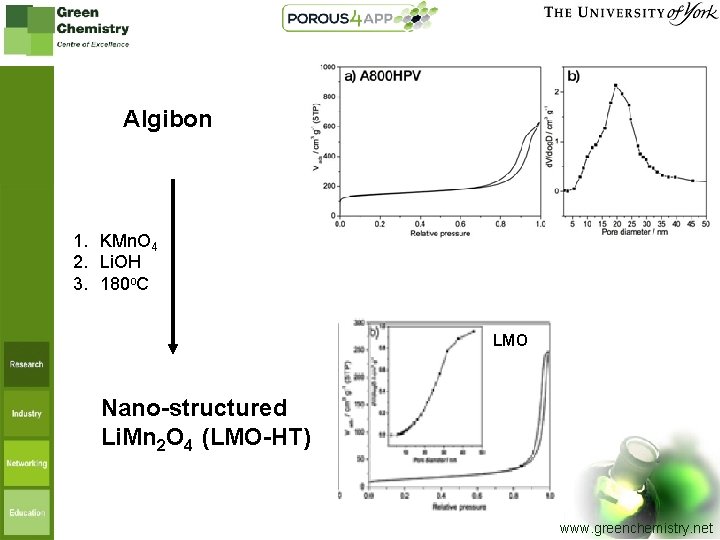
Algibon 1. KMn. O 4 2. Li. OH 3. 180 o. C LMO Nano-structured Li. Mn 2 O 4 (LMO-HT) www. greenchemistry. net
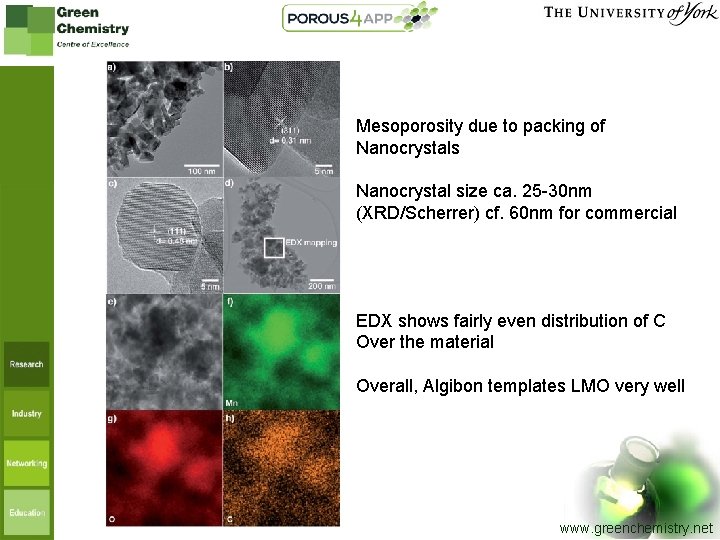
Mesoporosity due to packing of Nanocrystals Nanocrystal size ca. 25 -30 nm (XRD/Scherrer) cf. 60 nm for commercial EDX shows fairly even distribution of C Over the material Overall, Algibon templates LMO very well www. greenchemistry. net
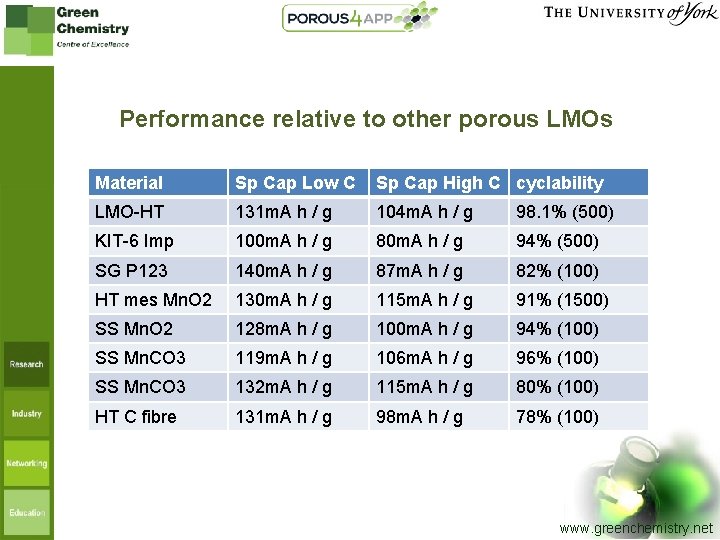
Performance relative to other porous LMOs Material Sp Cap Low C Sp Cap High C cyclability LMO-HT 131 m. A h / g 104 m. A h / g 98. 1% (500) KIT-6 Imp 100 m. A h / g 80 m. A h / g 94% (500) SG P 123 140 m. A h / g 87 m. A h / g 82% (100) HT mes Mn. O 2 130 m. A h / g 115 m. A h / g 91% (1500) SS Mn. O 2 128 m. A h / g 100 m. A h / g 94% (100) SS Mn. CO 3 119 m. A h / g 106 m. A h / g 96% (100) SS Mn. CO 3 132 m. A h / g 115 m. A h / g 80% (100) HT C fibre 131 m. A h / g 98 m. A h / g 78% (100) www. greenchemistry. net
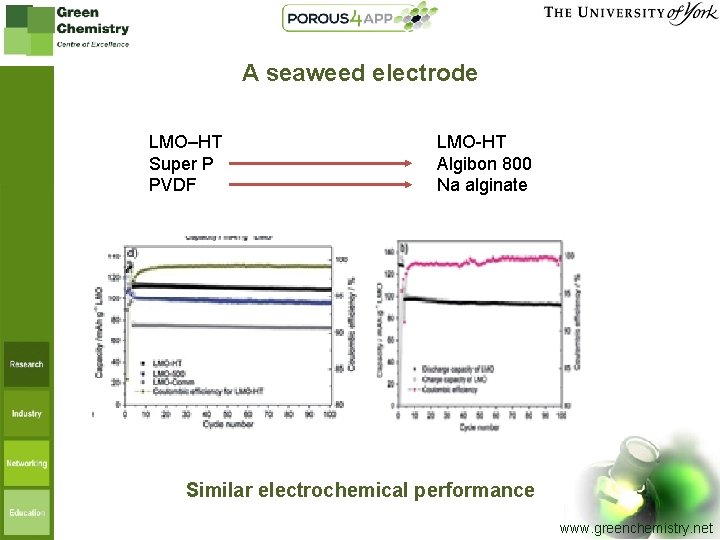
A seaweed electrode LMO–HT Super P PVDF LMO-HT Algibon 800 Na alginate Similar electrochemical performance www. greenchemistry. net
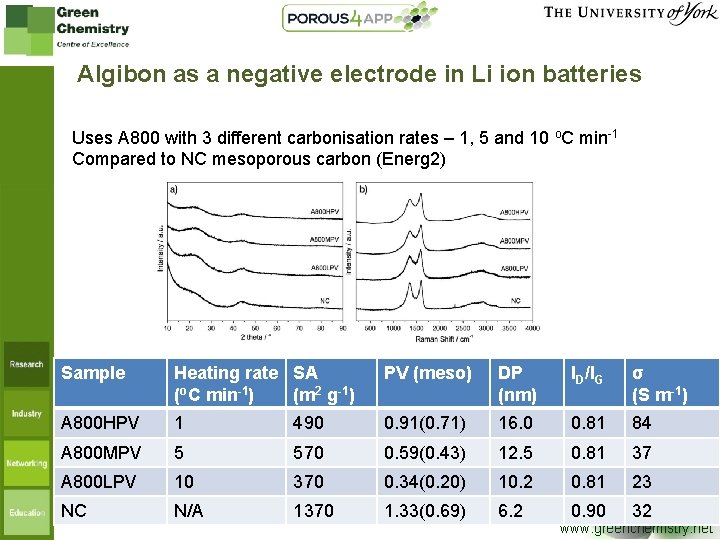
Algibon as a negative electrode in Li ion batteries Uses A 800 with 3 different carbonisation rates – 1, 5 and 10 o. C min-1 Compared to NC mesoporous carbon (Energ 2) Sample Heating rate SA (o. C min-1) (m 2 g-1) PV (meso) DP (nm) ID/IG σ (S m-1) A 800 HPV 1 490 0. 91(0. 71) 16. 0 0. 81 84 A 800 MPV 5 570 0. 59(0. 43) 12. 5 0. 81 37 A 800 LPV 10 370 0. 34(0. 20) 10. 2 0. 81 23 NC N/A 1370 1. 33(0. 69) 6. 2 0. 90 32 www. greenchemistry. net
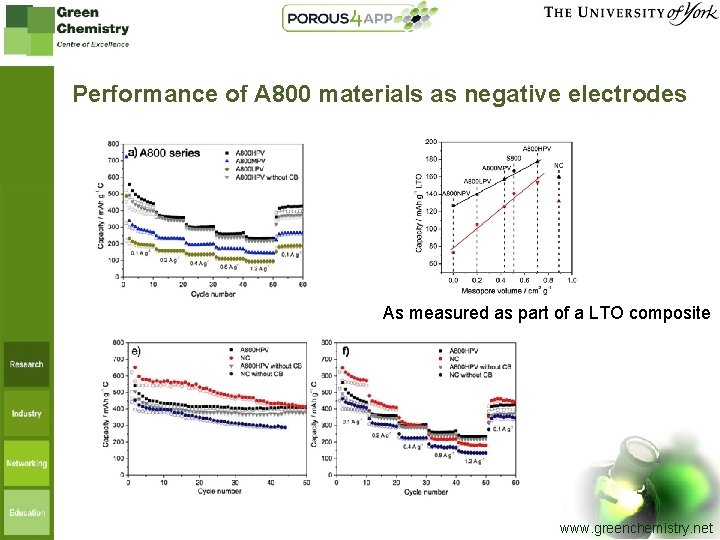
Performance of A 800 materials as negative electrodes As measured as part of a LTO composite www. greenchemistry. net

Conclusions • Porous polysaccharides can be scaled up to multi-kg scales • Initial work on using them as components in Li ion batteries promising • Pore volume is important • Currently initiating larger scale testing www. greenchemistry. net
- Slides: 23