Bio Sci D 145 Lecture 7 Bio Sci

Bio. Sci D 145 Lecture #7 • Bio. Sci D 145 lecture 1 page 1 ©copyright Bruce Blumberg 2020. All rights reserved
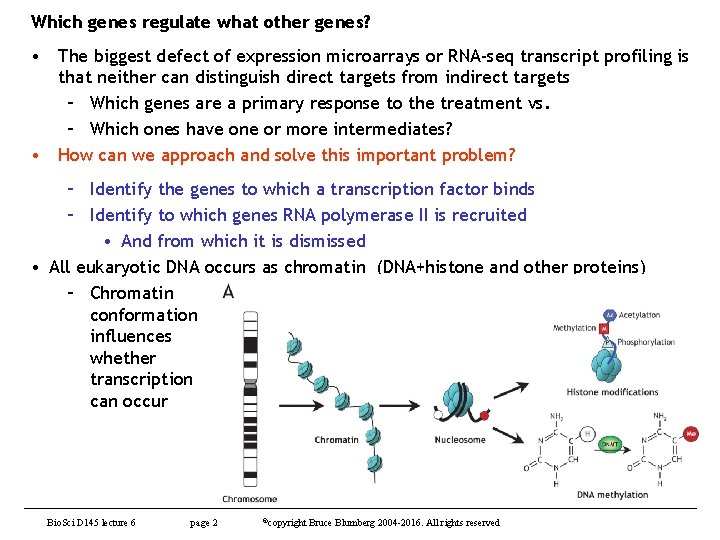
Which genes regulate what other genes? • The biggest defect of expression microarrays or RNA-seq transcript profiling is that neither can distinguish direct targets from indirect targets – Which genes are a primary response to the treatment vs. – Which ones have one or more intermediates? • How can we approach and solve this important problem? – Identify the genes to which a transcription factor binds – Identify to which genes RNA polymerase II is recruited • And from which it is dismissed • All eukaryotic DNA occurs as chromatin (DNA+histone and other proteins) – Chromatin conformation influences whether transcription can occur Bio. Sci D 145 lecture 6 page 2 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
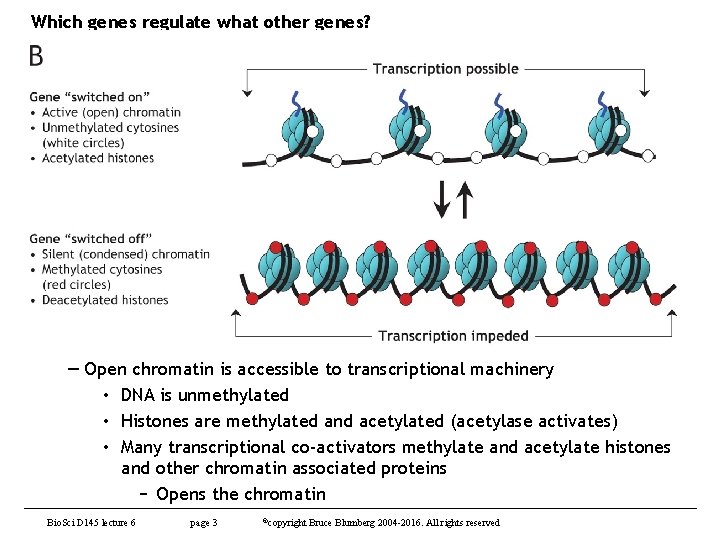
Which genes regulate what other genes? — Open chromatin is accessible to transcriptional machinery • DNA is unmethylated • Histones are methylated and acetylated (acetylase activates) • Many transcriptional co-activators methylate and acetylate histones and other chromatin associated proteins − Opens the chromatin Bio. Sci D 145 lecture 6 page 3 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

Which genes regulate what other genes? — Closed chromatin is inaccessible to transcriptional machinery • DNA is methylated • Some histone tails are methylated, others not • Transcriptional co-repressors recruit histone deacetylases (HDACs) which lead to chromatin condenstation • Chromatin condensation leads to gene silencing • Identification of chromatin-localized proteins is diagnostic for direct target genes of transcription factors − Most common application • Identification of promoters to which RNA Pol II is recruited upon some treatment is diagnostic for genes directly upregulated by the treatment − Trickier but very useful • Identification of promoters from which RNA Pol II is dismissed upon some treatment is diagnostic for genes downregulated by the treatment Bio. Sci D 145 lecture 6 page 4 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
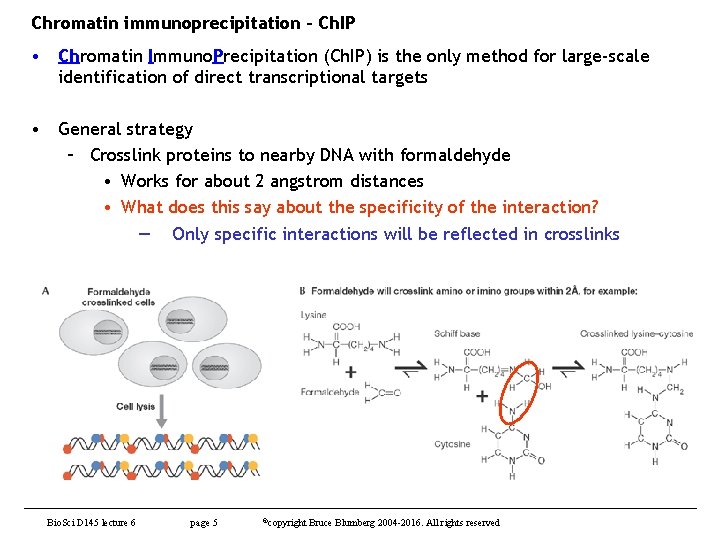
Chromatin immunoprecipitation - Ch. IP • Chromatin Immuno. Precipitation (Ch. IP) is the only method for large-scale identification of direct transcriptional targets • General strategy – Crosslink proteins to nearby DNA with formaldehyde • Works for about 2 angstrom distances • What does this say about the specificity of the interaction? — Only specific interactions will be reflected in crosslinks Bio. Sci D 145 lecture 6 page 5 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
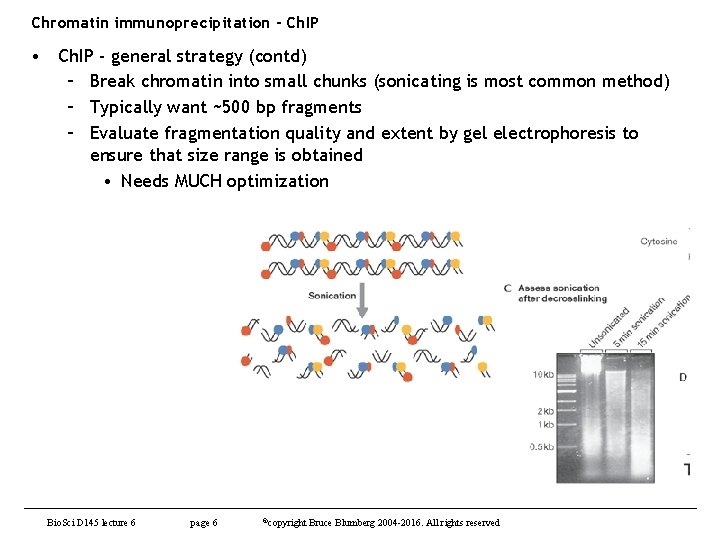
Chromatin immunoprecipitation - Ch. IP • Ch. IP - general strategy (contd) – Break chromatin into small chunks (sonicating is most common method) – Typically want ~500 bp fragments – Evaluate fragmentation quality and extent by gel electrophoresis to ensure that size range is obtained • Needs MUCH optimization Bio. Sci D 145 lecture 6 page 6 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
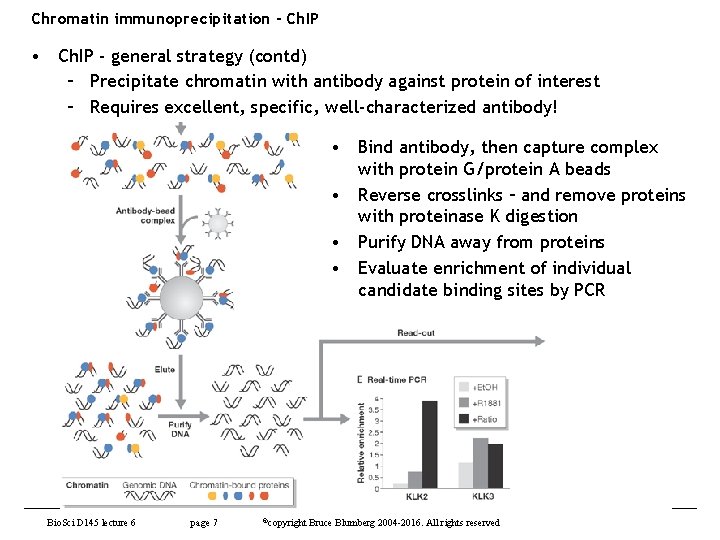
Chromatin immunoprecipitation - Ch. IP • Ch. IP - general strategy (contd) – Precipitate chromatin with antibody against protein of interest – Requires excellent, specific, well-characterized antibody! • Bind antibody, then capture complex with protein G/protein A beads • Reverse crosslinks – and remove proteins with proteinase K digestion • Purify DNA away from proteins • Evaluate enrichment of individual candidate binding sites by PCR Bio. Sci D 145 lecture 6 page 7 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
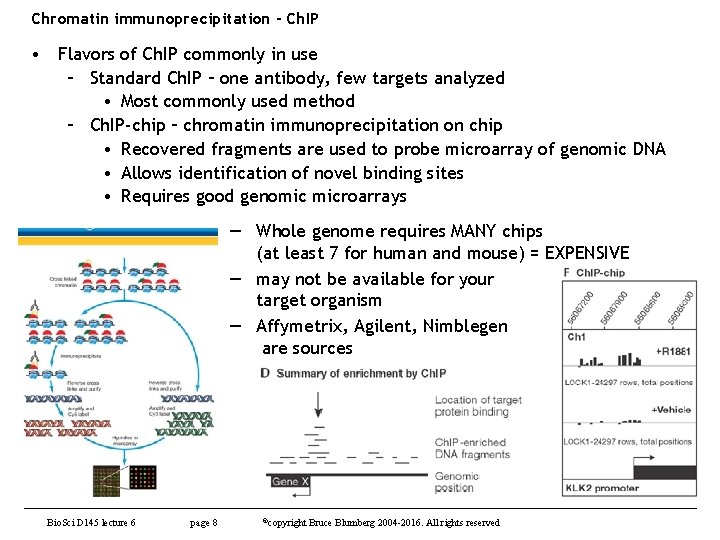
Chromatin immunoprecipitation - Ch. IP • Flavors of Ch. IP commonly in use – Standard Ch. IP – one antibody, few targets analyzed • Most commonly used method – Ch. IP-chip – chromatin immunoprecipitation on chip • Recovered fragments are used to probe microarray of genomic DNA • Allows identification of novel binding sites • Requires good genomic microarrays — Whole genome requires MANY chips (at least 7 for human and mouse) = EXPENSIVE — may not be available for your target organism — Affymetrix, Agilent, Nimblegen are sources Bio. Sci D 145 lecture 6 page 8 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
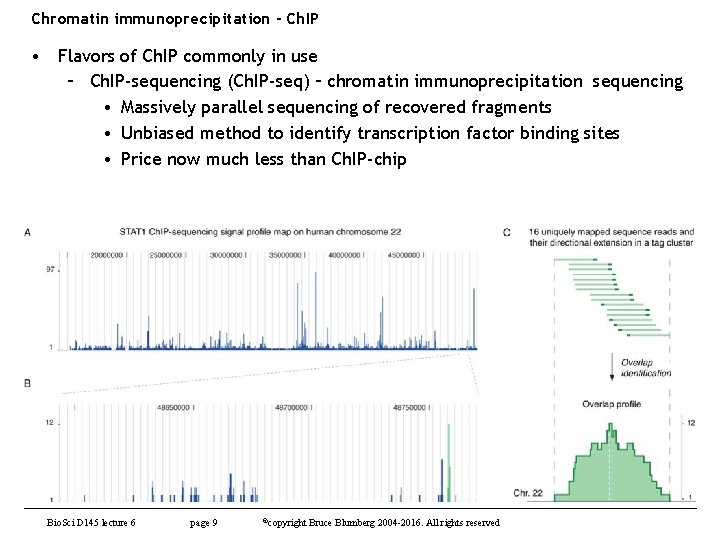
Chromatin immunoprecipitation - Ch. IP • Flavors of Ch. IP commonly in use – Ch. IP-sequencing (Ch. IP-seq) – chromatin immunoprecipitation sequencing • Massively parallel sequencing of recovered fragments • Unbiased method to identify transcription factor binding sites • Price now much less than Ch. IP-chip Bio. Sci D 145 lecture 6 page 9 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

Chromatin immunoprecipitation – other applications • Various methods of probing chromatin structure with Ch. IP, etc – Chromatin conformation capture and related methods – useful to study spatial organization of chromosomes (mostly looping) and how this influences gene expression • 3 C – chromatin conformation capture – Capture-C (most modern version) • 4 C – circularized chromosome conformation capture • 5 C – carbon-copy chromosome conformation capture • Ch. IP-loop • Hi-C (first truly genome wide 3 C method) – Ch. IA-PET – chromatin interaction analysis by paired-end tag sequencing • Useful to detect de novo long-range chromatin interactions – Methods differ in how much you must sequence to get the information you need to evaluate structure. Bio. Sci D 145 lecture 7 page 10 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
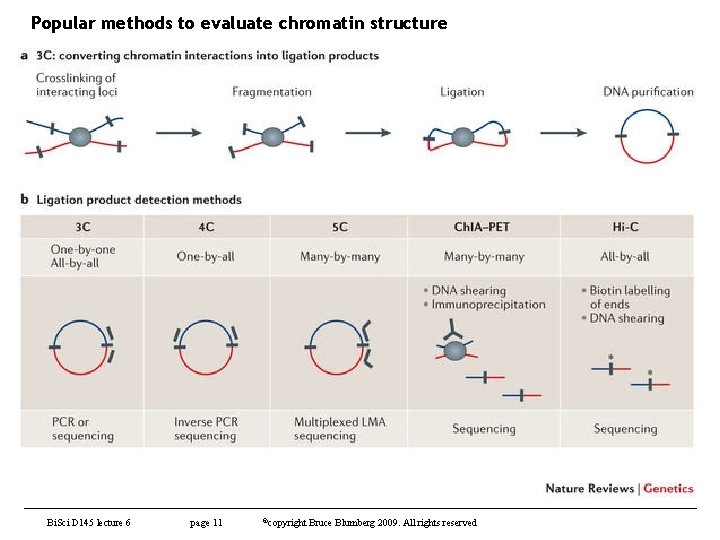
Popular methods to evaluate chromatin structure Bi. Sci D 145 lecture 6 page 11 ©copyright Bruce Blumberg 2009. All rights reserved
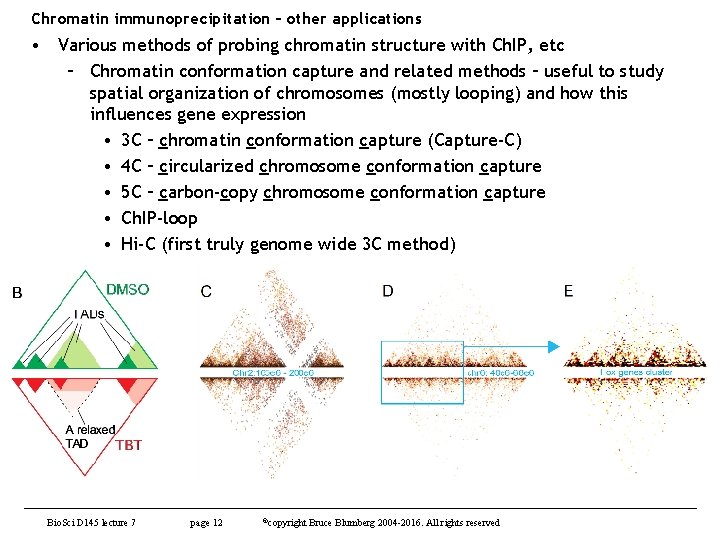
Chromatin immunoprecipitation – other applications • Various methods of probing chromatin structure with Ch. IP, etc – Chromatin conformation capture and related methods – useful to study spatial organization of chromosomes (mostly looping) and how this influences gene expression • 3 C – chromatin conformation capture (Capture-C) • 4 C – circularized chromosome conformation capture • 5 C – carbon-copy chromosome conformation capture • Ch. IP-loop • Hi-C (first truly genome wide 3 C method) Bio. Sci D 145 lecture 7 page 12 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
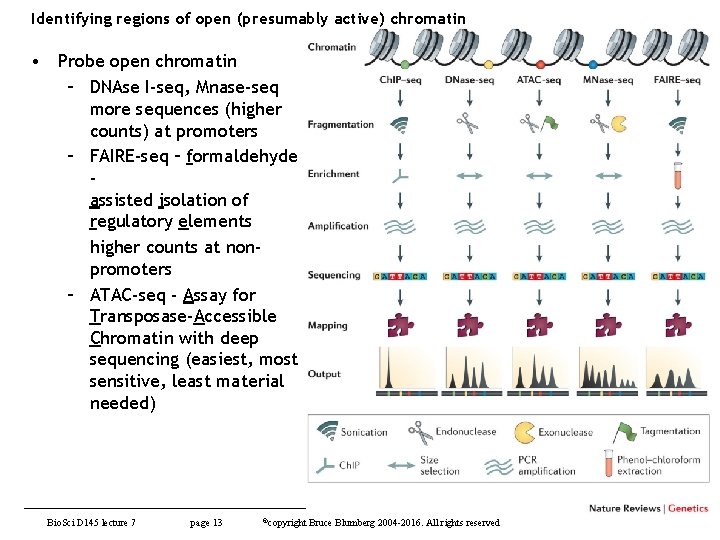
Identifying regions of open (presumably active) chromatin • Probe open chromatin – DNAse I-seq, Mnase-seq more sequences (higher counts) at promoters – FAIRE-seq – formaldehyde assisted isolation of regulatory elements higher counts at nonpromoters – ATAC-seq - Assay for Transposase-Accessible Chromatin with deep sequencing (easiest, most sensitive, least material needed) Bio. Sci D 145 lecture 7 page 13 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
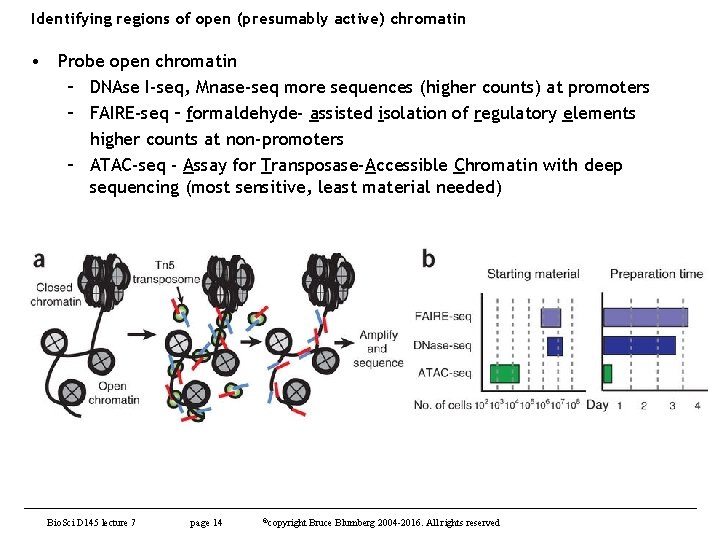
Identifying regions of open (presumably active) chromatin • Probe open chromatin – DNAse I-seq, Mnase-seq more sequences (higher counts) at promoters – FAIRE-seq – formaldehyde- assisted isolation of regulatory elements higher counts at non-promoters – ATAC-seq - Assay for Transposase-Accessible Chromatin with deep sequencing (most sensitive, least material needed) Bio. Sci D 145 lecture 7 page 14 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

What is Epigenetics ? • Literally “on top of genetics” – coined by C. H. Waddington in 1957 • Epigenetics involves changes in gene expression without changes in the DNA sequence – Heritable, maintained – Reversible – Encoded in chromatin – Cellular memory • What are some examples of epigenetic phenomena ? – X inactivation – Genomic imprinting – Cancer – widespread silencing or overexpression of genes Bio. Sci D 145 lecture 7 page 16 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

What is Epigenetics ? Bio. Sci D 145 lecture 7 page 17 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
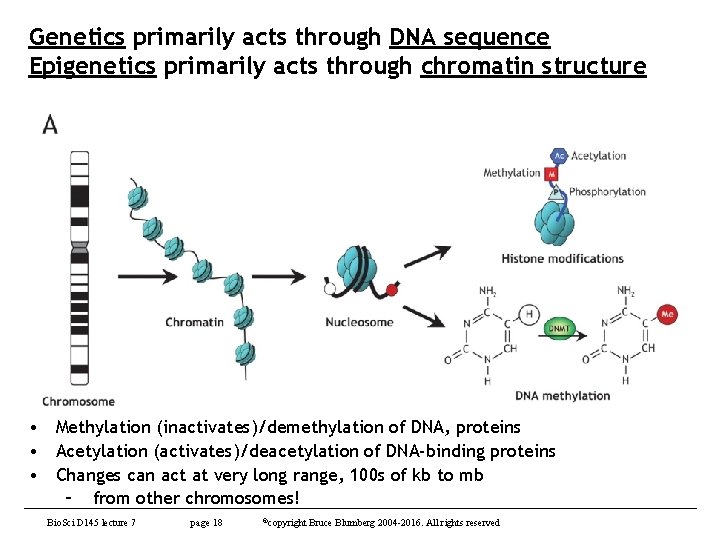
Genetics primarily acts through DNA sequence Epigenetics primarily acts through chromatin structure • Methylation (inactivates)/demethylation of DNA, proteins • Acetylation (activates)/deacetylation of DNA-binding proteins • Changes can act at very long range, 100 s of kb to mb – from other chromosomes! Bio. Sci D 145 lecture 7 page 18 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
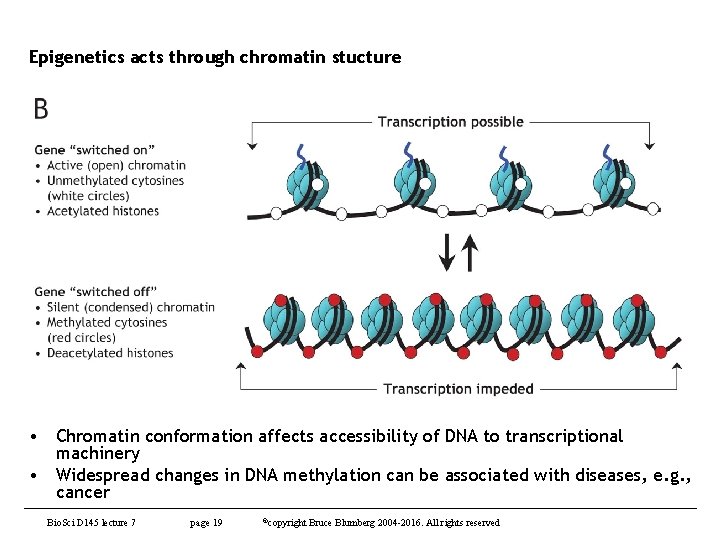
Epigenetics acts through chromatin stucture • Chromatin conformation affects accessibility of DNA to transcriptional machinery • Widespread changes in DNA methylation can be associated with diseases, e. g. , cancer Bio. Sci D 145 lecture 7 page 19 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
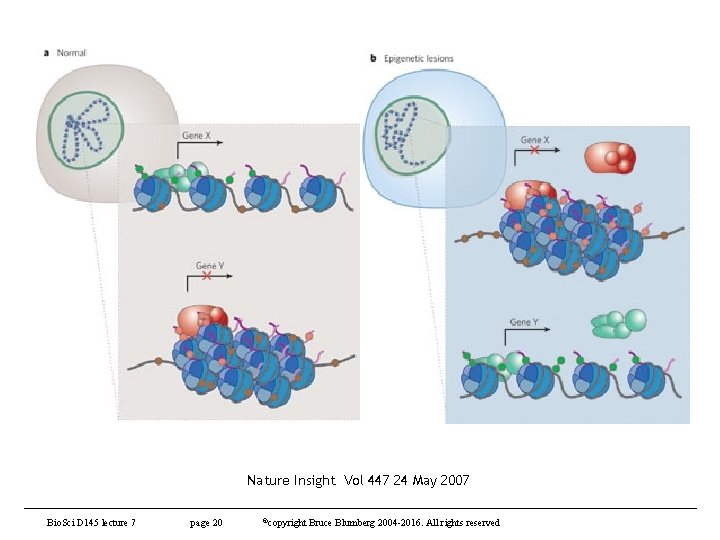
Nature Insight Vol 447 24 May 2007 Bio. Sci D 145 lecture 7 page 20 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
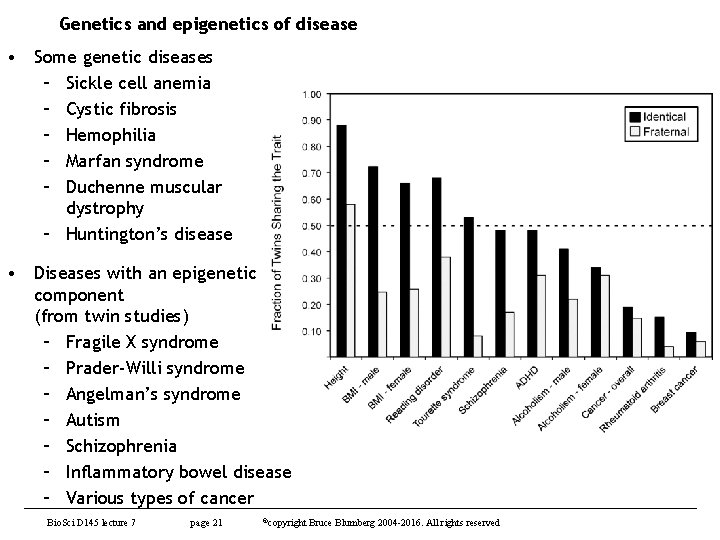
Genetics and epigenetics of disease • Some genetic diseases – Sickle cell anemia – Cystic fibrosis – Hemophilia – Marfan syndrome – Duchenne muscular dystrophy – Huntington’s disease • Diseases with an epigenetic component (from twin studies) – Fragile X syndrome – Prader-Willi syndrome – Angelman’s syndrome – Autism – Schizophrenia – Inflammatory bowel disease – Various types of cancer Bio. Sci D 145 lecture 7 page 21 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

The DOHa. D Hypothesis • Barker Hypothesis - gestational under-nutrition leads to a thrifty phenotype – reduced fetal growth is strongly associated with many cardiovascular disease and other related syndromes. – Increased susceptibility results from adaptations made by the fetus in an environment limited in its supply of nutrients • Developmental Origins of Health and Disease (Mark Hanson, Peter Gluckman) more generally proposes that development is very sensitive to perturbations that lead to permanent changes in susceptibility to disease – Birth defects, low birth weight, premature birth – Functional changes – individual appears normal but has molecular abnormalities that persist and lead to increased disease sensitivity later in life • Diseases with Developmental Origins (from animal models) – Cardiovascular, Pulmonary (asthma) – Neurological (ADHD, Neurodegenerative diseases), – Immune/autoimmune – Endocrine, reproductive/fertility, cancer – Obesity/diabetes Bio. Sci D 145 lecture 7 page 22 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

https: //ihavenotv. com/the-ghost-in-your-genes “How We're Already Killing Our Grandkids” “Sins of the fathers, and their fathers” “Men inherit hidden cost of Three generations from the dad's vices” Överkalix parish of Norrbotten Bio. Sci D 145 lecture 7 page 23 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
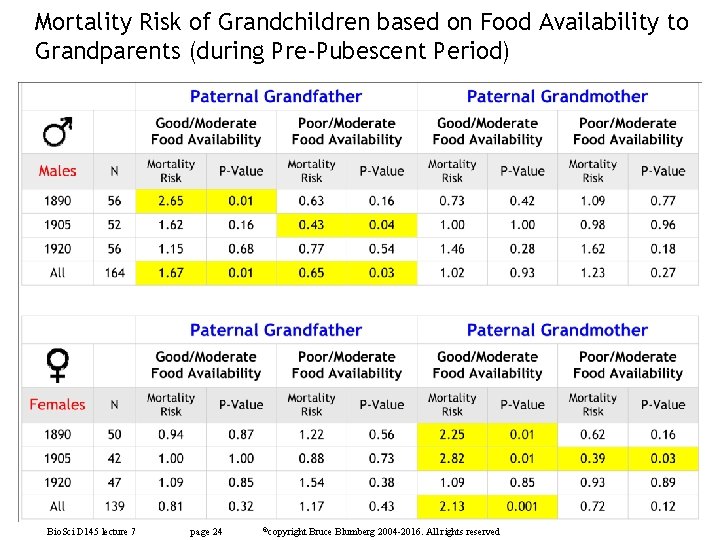
Mortality Risk of Grandchildren based on Food Availability to Grandparents (during Pre-Pubescent Period) Bio. Sci D 145 lecture 7 page 24 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
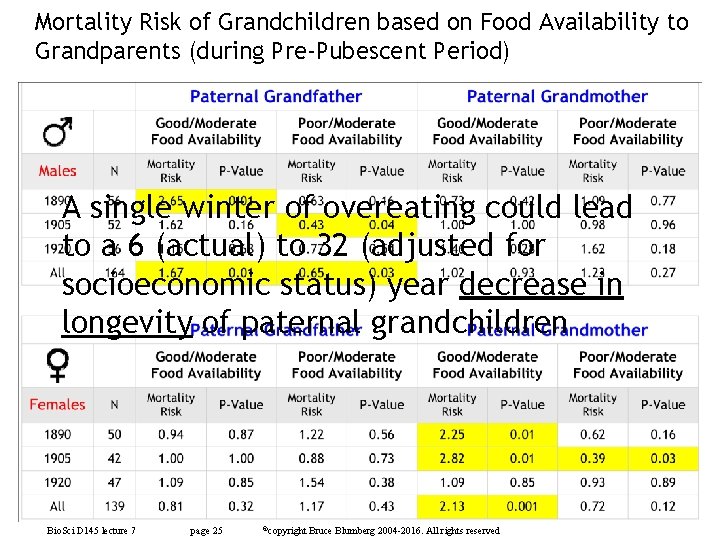
Mortality Risk of Grandchildren based on Food Availability to Grandparents (during Pre-Pubescent Period) A single winter of overeating could lead to a 6 (actual) to 32 (adjusted for socioeconomic status) year decrease in longevity of paternal grandchildren Bio. Sci D 145 lecture 7 page 25 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
Epigenetics can be changed by environment • Maternal care – licking studies in rats – (maternal) stress • Diet - folic acid, vitamins B 2, B 6, B 12 (influence DNA methylation) • Other dietary factors – e. g. , phytoestrogens • Toxins - heavy metals, arsenic, tobacco • Maternal caloric intake (paternal, too) • Changes in DNA methylation responsible Bio. Sci D 145 lecture 7 page 26 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

Types of epigenetic (epigenomic) phenomena • Epigenome – changes in chromatin states that may vary from cell to cell • Epigenetics – all processes that lead to heritable changes in gene expression (during development or across generations) – Without changes in DNA sequence – Usually involves changes in chromatin therefore epigenetic inheritance often involves epigenomics. • What are some ways that epigenetic inheritance can be transmitted? – Changes in DNA methylation – Changes in histone methylation/retention – Changes in nc. RNA partitioning – lnc. RNAs – Small RNAs – Modified t. RNAs – Changes in chromatin accessibility – Changes in 3 D-chromatin structure Bio. Sci D 145 lecture 7 page 27 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
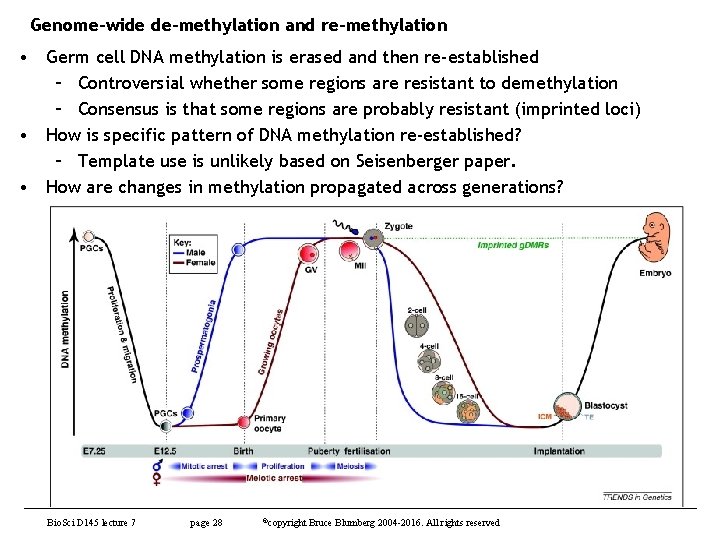
Genome-wide de-methylation and re-methylation • Germ cell DNA methylation is erased and then re-established – Controversial whether some regions are resistant to demethylation – Consensus is that some regions are probably resistant (imprinted loci) • How is specific pattern of DNA methylation re-established? – Template use is unlikely based on Seisenberger paper. • How are changes in methylation propagated across generations? Bio. Sci D 145 lecture 7 page 28 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
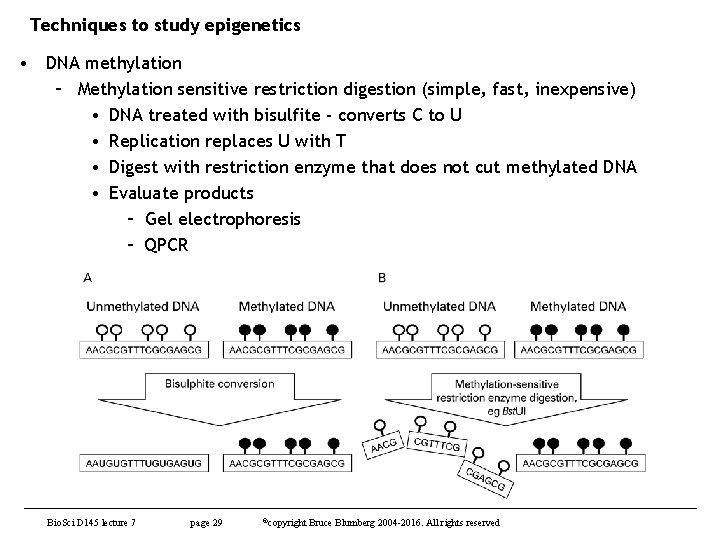
Techniques to study epigenetics • DNA methylation – Methylation sensitive restriction digestion (simple, fast, inexpensive) • DNA treated with bisulfite - converts C to U • Replication replaces U with T • Digest with restriction enzyme that does not cut methylated DNA • Evaluate products – Gel electrophoresis – QPCR Bio. Sci D 145 lecture 7 page 29 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

Techniques to study epigenetics (papers this week and next) • DNA methylation – Methylation sensitive restriction digestion (simple, fast, inexpensive) • Genome wide variation – cut with frequent digesting enzyme e. g, Mse. I – Add linker sequences with PCR primer – Cut with methylation sensitive restriction enzyme – PCR with primers – only detects sequences with methylated DNA between primers – Analyze by microarray or nextgen sequencing Bio. Sci D 145 lecture 7 page 30 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
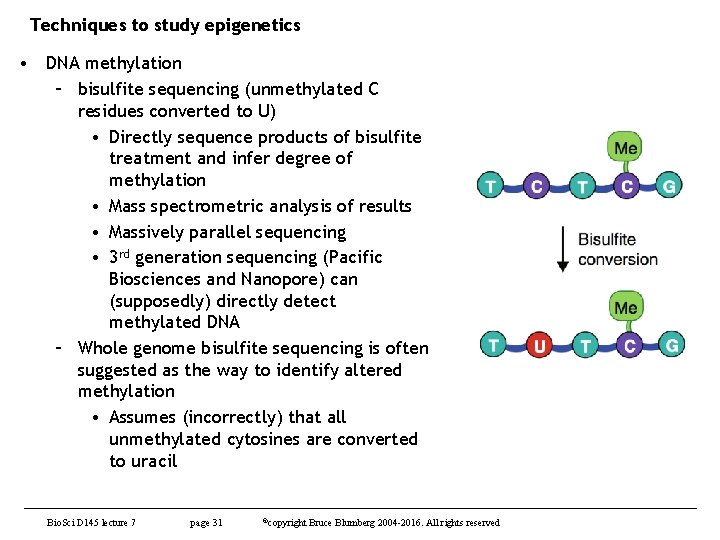
Techniques to study epigenetics • DNA methylation – bisulfite sequencing (unmethylated C residues converted to U) • Directly sequence products of bisulfite treatment and infer degree of methylation • Mass spectrometric analysis of results • Massively parallel sequencing • 3 rd generation sequencing (Pacific Biosciences and Nanopore) can (supposedly) directly detect methylated DNA – Whole genome bisulfite sequencing is often suggested as the way to identify altered methylation • Assumes (incorrectly) that all unmethylated cytosines are converted to uracil Bio. Sci D 145 lecture 7 page 31 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
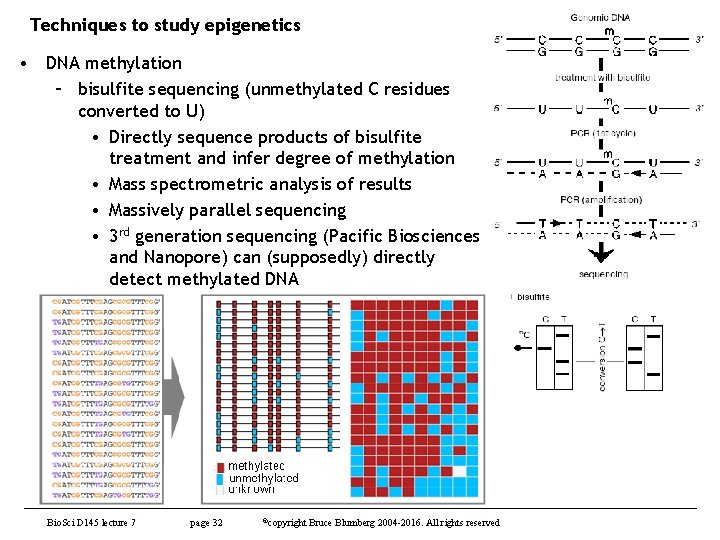
Techniques to study epigenetics • DNA methylation – bisulfite sequencing (unmethylated C residues converted to U) • Directly sequence products of bisulfite treatment and infer degree of methylation • Mass spectrometric analysis of results • Massively parallel sequencing • 3 rd generation sequencing (Pacific Biosciences and Nanopore) can (supposedly) directly detect methylated DNA Bio. Sci D 145 lecture 7 page 32 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
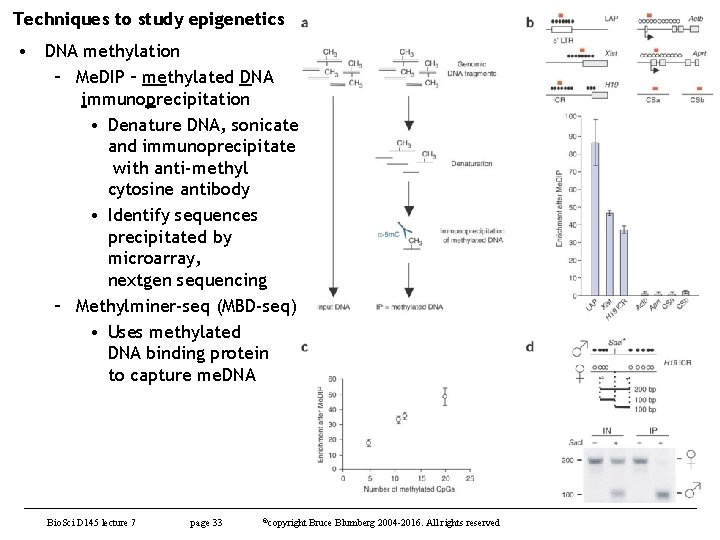
Techniques to study epigenetics • DNA methylation – Me. DIP – methylated DNA immunoprecipitation • Denature DNA, sonicate and immunoprecipitate with anti-methyl cytosine antibody • Identify sequences precipitated by microarray, nextgen sequencing – Methylminer-seq (MBD-seq) • Uses methylated DNA binding protein to capture me. DNA Bio. Sci D 145 lecture 7 page 33 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
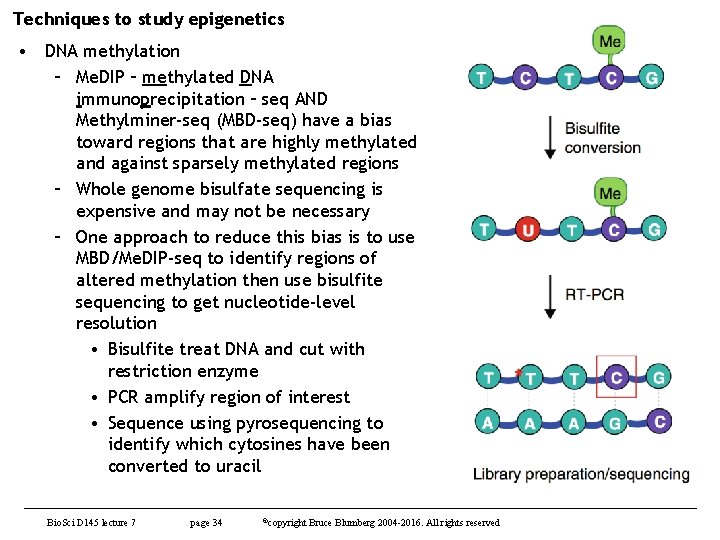
Techniques to study epigenetics • DNA methylation – Me. DIP – methylated DNA immunoprecipitation – seq AND Methylminer-seq (MBD-seq) have a bias toward regions that are highly methylated and against sparsely methylated regions – Whole genome bisulfate sequencing is expensive and may not be necessary – One approach to reduce this bias is to use MBD/Me. DIP-seq to identify regions of altered methylation then use bisulfite sequencing to get nucleotide-level resolution • Bisulfite treat DNA and cut with restriction enzyme • PCR amplify region of interest • Sequence using pyrosequencing to identify which cytosines have been converted to uracil Bio. Sci D 145 lecture 7 page 34 ©copyright Bruce Blumberg 2004 -2016. All rights reserved

Techniques to study epigenetics • Histone modification – Histones are considerably modified at many places and in a combinatorial fashion (many permutations) • Methylation • Phosphorylation • acetylation – Only methylation is heritable! – Methods of analysis • Ch. IP – precipitate chromatin with antibodies specific to particular methylation – Analyze by microarray – Ch. IP-CHIP – Or by high throughput sequencing – Ch. IP-seq Bio. Sci D 145 lecture 7 page 35 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
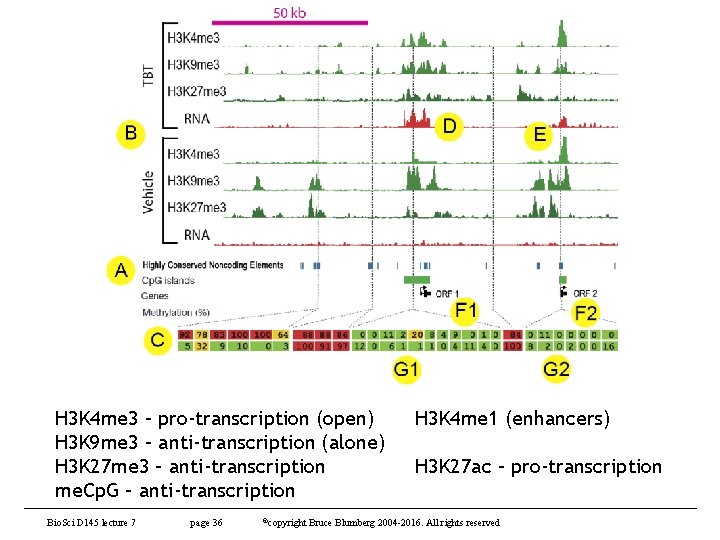
H 3 K 4 me 3 – pro-transcription (open) H 3 K 9 me 3 – anti-transcription (alone) H 3 K 27 me 3 – anti-transcription me. Cp. G – anti-transcription Bio. Sci D 145 lecture 7 page 36 ©copyright H 3 K 4 me 1 (enhancers) H 3 K 27 ac – pro-transcription Bruce Blumberg 2004 -2016. All rights reserved
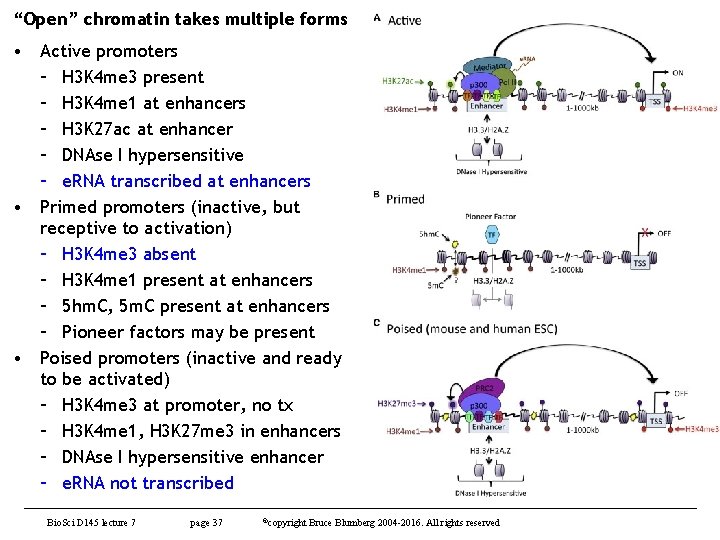
“Open” chromatin takes multiple forms • Active promoters – H 3 K 4 me 3 present – H 3 K 4 me 1 at enhancers – H 3 K 27 ac at enhancer – DNAse I hypersensitive – e. RNA transcribed at enhancers • Primed promoters (inactive, but receptive to activation) – H 3 K 4 me 3 absent – H 3 K 4 me 1 present at enhancers – 5 hm. C, 5 m. C present at enhancers – Pioneer factors may be present • Poised promoters (inactive and ready to be activated) – H 3 K 4 me 3 at promoter, no tx – H 3 K 4 me 1, H 3 K 27 me 3 in enhancers – DNAse I hypersensitive enhancer – e. RNA not transcribed Bio. Sci D 145 lecture 7 page 37 ©copyright Bruce Blumberg 2004 -2016. All rights reserved
- Slides: 36