Bio Sci 145 B Lecture 8 5252004 Bruce

Bio. Sci 145 B Lecture #8 5/25/2004 • Bruce Blumberg – 2113 E Mc. Gaugh Hall - office hours Wed 12 -1 PM (or by appointment) – phone 824 -8573 – blumberg@uci. edu • TA – Curtis Daly cdaly@uci. edu – 2113 Mc. Gaugh Hall, 924 -6873, 3116 – Office hours Tuesday 11 -12 • lectures will be posted on web pages after lecture – http: //eee. uci. edu/04 s/05705/ - link only here – http: //blumberg-serv. bio. uci. edu/bio 145 b-sp 2004 – http: //blumberg. bio. uci. edu/bio 145 b-sp 2004 • DON’T FORGET - TERM PAPERS ARE DUE BY 5 PM on FRIDAY JUNE 4 copyright 2000 Bruce Blumberg, all rights reserved

Methods of profiling gene expression – large scale • Global analysis of RNA expression – identifying all expressed sequences, a. k. a. the transcriptome – Array – micro or macro – Sequence sampling – SAGE – serial analysis of gene expression – Massively parallel signature sequencing • DNA microarray analysis is now totally dominant method – Two basic flavors • Spotted (spot DNA onto support) – c. DNA microarrays – Oligonucleotide arrays – Moderately expensive • Synthesized (use photolithography to synthesize oligos onto silicon or other suitable support – Affymetrix Gene Chips dominate – VERY expensive – Both are in wide use and suitable for whole genome analysis Bio. Sci 145 B lecture 8 page 2 ©copyright Bruce Blumberg 2004. All rights reserved
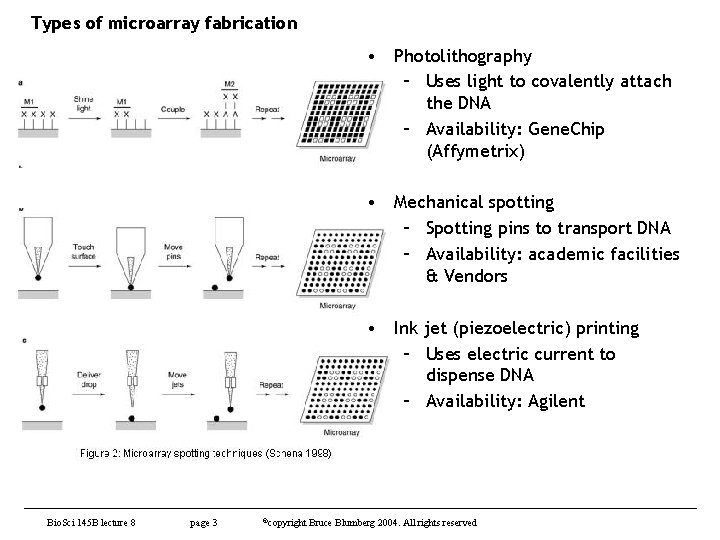
Types of microarray fabrication • Photolithography – Uses light to covalently attach the DNA – Availability: Gene. Chip (Affymetrix) • Mechanical spotting – Spotting pins to transport DNA – Availability: academic facilities & Vendors • Ink jet (piezoelectric) printing – Uses electric current to dispense DNA – Availability: Agilent Bio. Sci 145 B lecture 8 page 3 ©copyright Bruce Blumberg 2004. All rights reserved
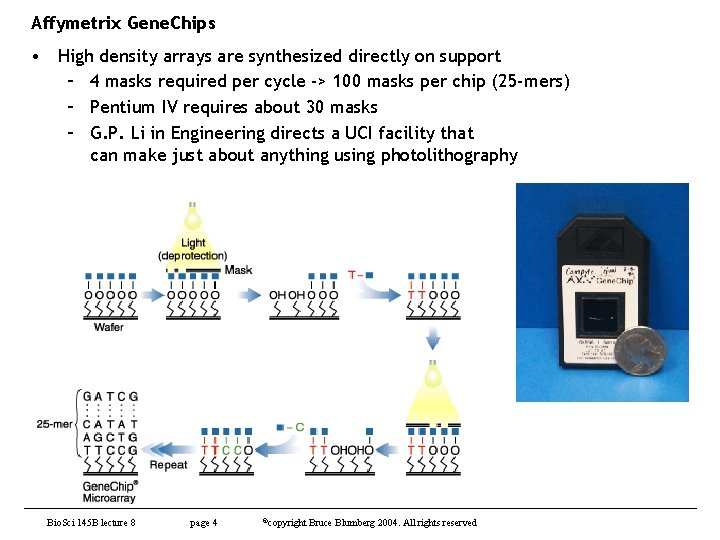
Affymetrix Gene. Chips • High density arrays are synthesized directly on support – 4 masks required per cycle -> 100 masks per chip (25 -mers) – Pentium IV requires about 30 masks – G. P. Li in Engineering directs a UCI facility that can make just about anything using photolithography Bio. Sci 145 B lecture 8 page 4 ©copyright Bruce Blumberg 2004. All rights reserved
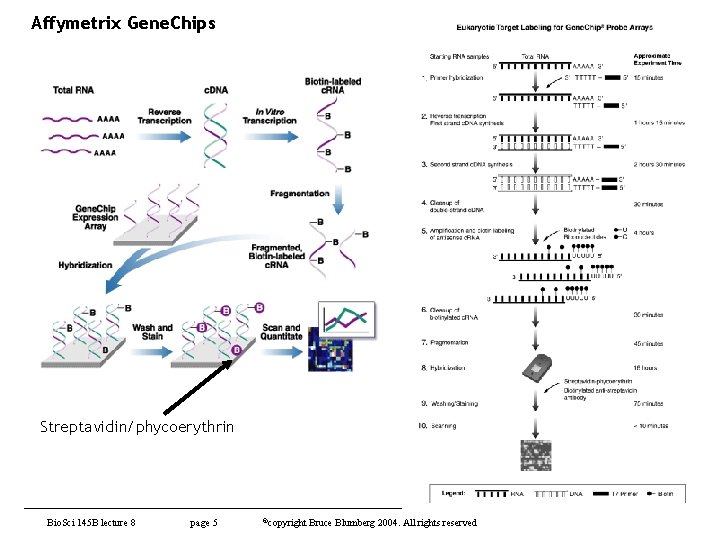
Affymetrix Gene. Chips Streptavidin/phycoerythrin Bio. Sci 145 B lecture 8 page 5 ©copyright Bruce Blumberg 2004. All rights reserved
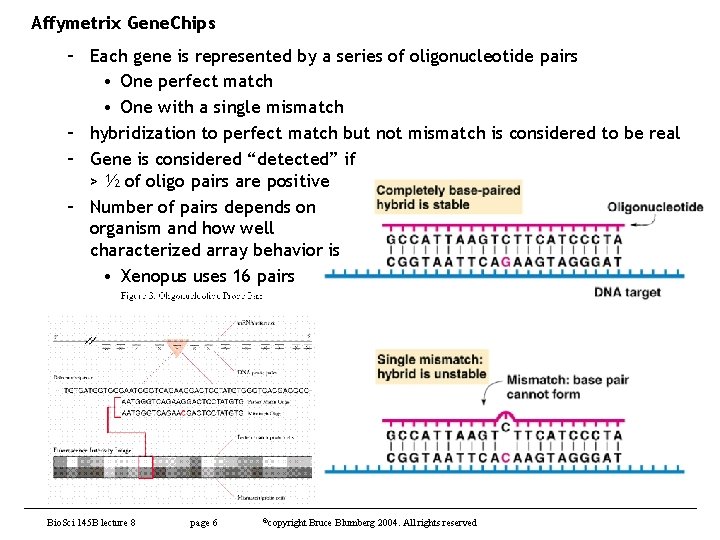
Affymetrix Gene. Chips – Each gene is represented by a series of oligonucleotide pairs • One perfect match • One with a single mismatch – hybridization to perfect match but not mismatch is considered to be real – Gene is considered “detected” if > ½ of oligo pairs are positive – Number of pairs depends on organism and how well characterized array behavior is • Xenopus uses 16 pairs Bio. Sci 145 B lecture 8 page 6 ©copyright Bruce Blumberg 2004. All rights reserved

Affymetrix Gene. Chips • Result is in single color – Need two chips – control and experimental for each condition • Advantages – Commercially available – Standardized • Disadvantages – About $1000 to buy, probe and process each chip at UC prices. • My Japanese collaborators pay > $3, 000/chip – May not be available for your organism of interest – No ability to compare probes directly on the same chip • Must rely on technology Bio. Sci 145 B lecture 8 page 7 ©copyright Bruce Blumberg 2004. All rights reserved
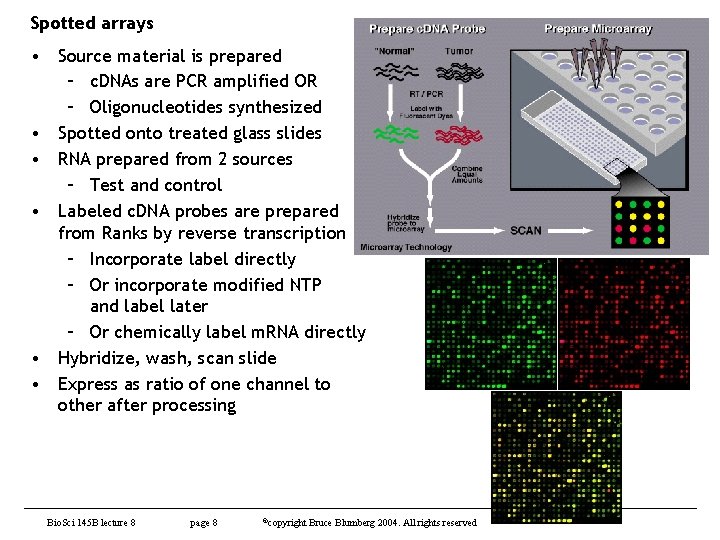
Spotted arrays • Source material is prepared – c. DNAs are PCR amplified OR – Oligonucleotides synthesized • Spotted onto treated glass slides • RNA prepared from 2 sources – Test and control • Labeled c. DNA probes are prepared from Ranks by reverse transcription – Incorporate label directly – Or incorporate modified NTP and label later – Or chemically label m. RNA directly • Hybridize, wash, scan slide • Express as ratio of one channel to other after processing Bio. Sci 145 B lecture 8 page 8 ©copyright Bruce Blumberg 2004. All rights reserved

DNA microarray types • Stanford type microarrayer – http: //cmgm. stanford. edu/pbrown/mguide/ index. html • Printing method – Reminiscent of fountain pen Bio. Sci 145 B lecture 8 page 9 ©copyright Bruce Blumberg 2004. All rights reserved
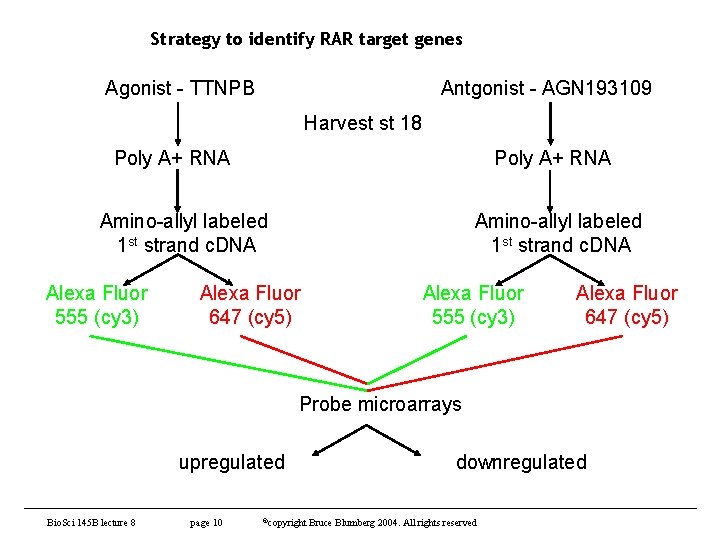
Strategy to identify RAR target genes Agonist - TTNPB Antgonist - AGN 193109 Harvest st 18 Poly A+ RNA Amino-allyl labeled 1 st strand c. DNA Alexa Fluor 555 (cy 3) Alexa Fluor 647 (cy 5) Probe microarrays upregulated Bio. Sci 145 B lecture 8 page 10 ©copyright downregulated Bruce Blumberg 2004. All rights reserved
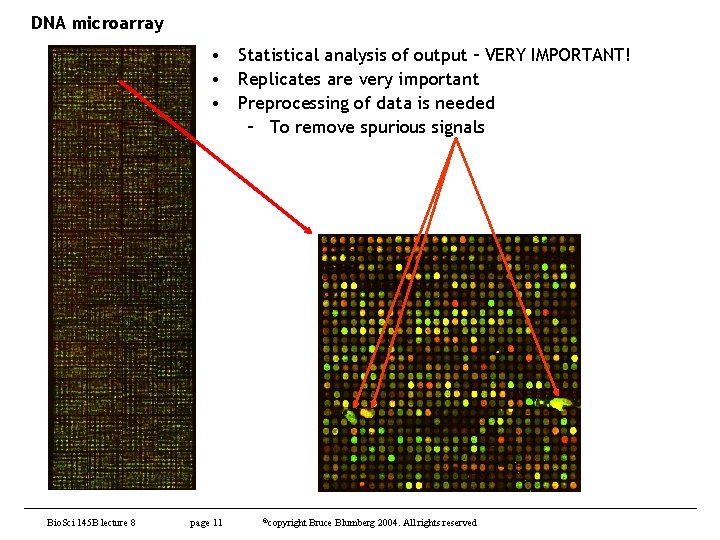
DNA microarray • Statistical analysis of output – VERY IMPORTANT! • Replicates are very important • Preprocessing of data is needed – To remove spurious signals Bio. Sci 145 B lecture 8 page 11 ©copyright Bruce Blumberg 2004. All rights reserved
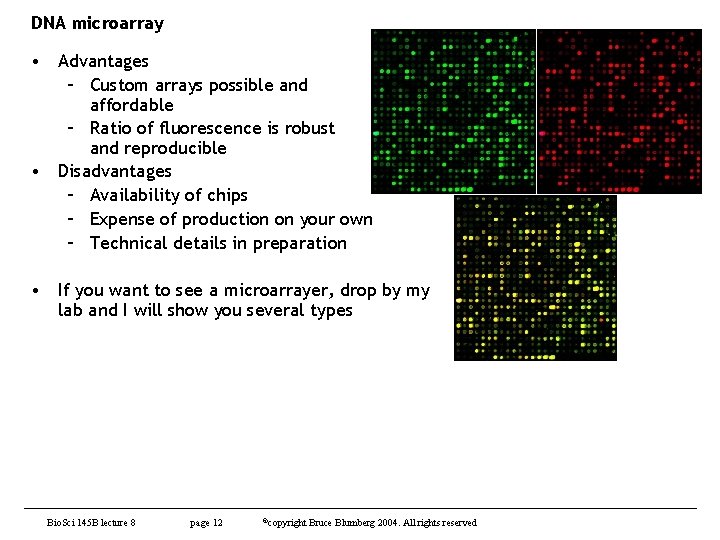
DNA microarray • Advantages – Custom arrays possible and affordable – Ratio of fluorescence is robust and reproducible • Disadvantages – Availability of chips – Expense of production on your own – Technical details in preparation • If you want to see a microarrayer, drop by my lab and I will show you several types Bio. Sci 145 B lecture 8 page 12 ©copyright Bruce Blumberg 2004. All rights reserved

DNA microarrays • What are they good for? – Identifying genes expressed in one condition vs another • One tissue vs another (heart vs liver) • Tissue vs tumor (liver vs hepatocarcinoma) • In response to a treatment (e. g. , RA) • In response to disease (after viral infection) – Building expression profiles • Tissues • Cancers • Developmental stages • Expressed genes on chromosomes – Identifying organisms • Array can identify which types of animals are present in a mix • http: //www. affymetrix. com/corporate/media/genechip_essentials/f oodexpert/Food. Expert_ID_Array. affx Bio. Sci 145 B lecture 8 page 13 ©copyright Bruce Blumberg 2004. All rights reserved

DNA microarrays • What are they good for? (contd) – Response of animal to drugs or chemicals • Toxicogenomics http: //www. niehs. nih. gov/nct/home. htm – Diagnostics • SNP analysis to identify disease loci • Specific testing for known diseases Bio. Sci 145 B lecture 8 page 14 ©copyright Bruce Blumberg 2004. All rights reserved

Global profiling of protein expression • Proteomics is broad name given to study of the proteome – Proteome -> a cell or organism’s complement of expressed proteins • Methods – 2 -D gel electrophoresis – Mass spectrometry of various sorts • All mass spec requires that molecules “fly” and measures mass/charge (m/z) ratio • MALDI-TOF – Matrix assisted laser desorption ionization – time of flight – Laser causes matrix to vaporize and molecules to fly, charge is applied and time molecule takes to fly to detector measured along with m/z • ESI – electrospray ionization – molecules are sprayed, ionized and detected • MS-MS – Tandem mass spec – has two mass analyzers - first detector shunts selected molecule to second – used for sequencing and structure analysis Bio. Sci 145 B lecture 8 page 15 ©copyright Bruce Blumberg 2004. All rights reserved
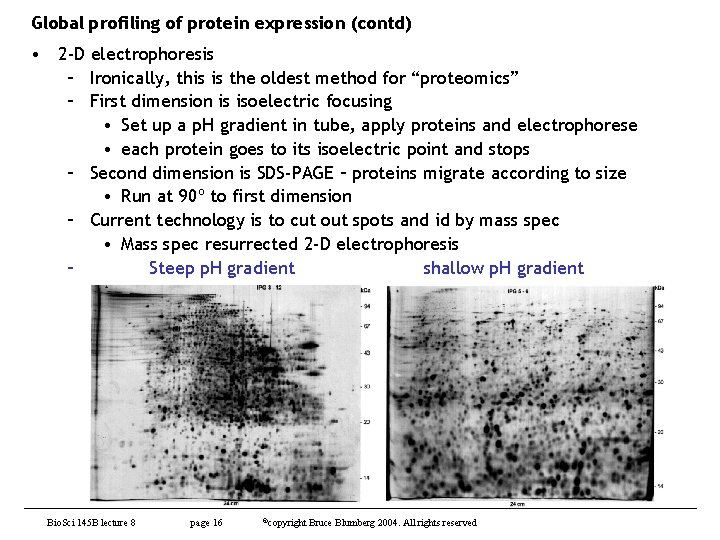
Global profiling of protein expression (contd) • 2 -D electrophoresis – Ironically, this is the oldest method for “proteomics” – First dimension is isoelectric focusing • Set up a p. H gradient in tube, apply proteins and electrophorese • each protein goes to its isoelectric point and stops – Second dimension is SDS-PAGE – proteins migrate according to size • Run at 90º to first dimension – Current technology is to cut out spots and id by mass spec • Mass spec resurrected 2 -D electrophoresis – Steep p. H gradient shallow p. H gradient Bio. Sci 145 B lecture 8 page 16 ©copyright Bruce Blumberg 2004. All rights reserved

Global profiling of protein expression (contd) • 2 -D electrophoresis (contd) – Good points • Straightforward separation • Can get good resolution with good isoelectric focusing gels – Downside • Protein may not be detectable as well-resolved spots that can be excised and characterized – Co-migrate – Abundance • Variation from experiment to experiment – Spot position on gel is very sensitive to small changes in p. H Bio. Sci 145 B lecture 8 page 17 ©copyright Bruce Blumberg 2004. All rights reserved
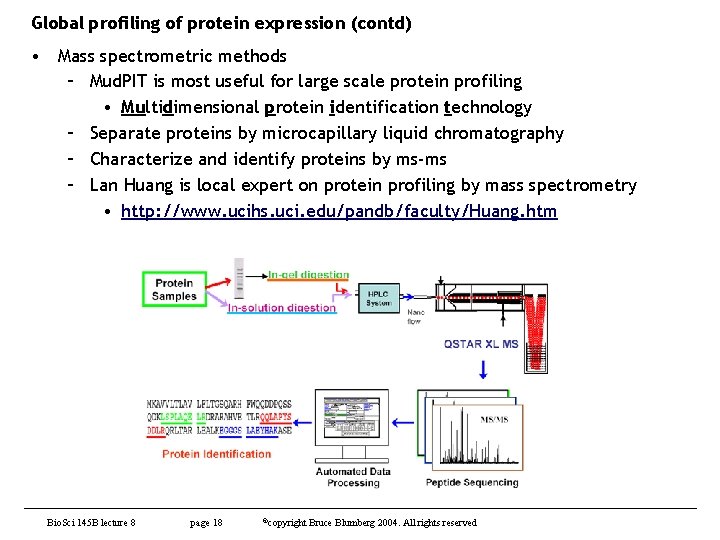
Global profiling of protein expression (contd) • Mass spectrometric methods – Mud. PIT is most useful for large scale protein profiling • Multidimensional protein identification technology – Separate proteins by microcapillary liquid chromatography – Characterize and identify proteins by ms-ms – Lan Huang is local expert on protein profiling by mass spectrometry • http: //www. ucihs. uci. edu/pandb/faculty/Huang. htm Bio. Sci 145 B lecture 8 page 18 ©copyright Bruce Blumberg 2004. All rights reserved
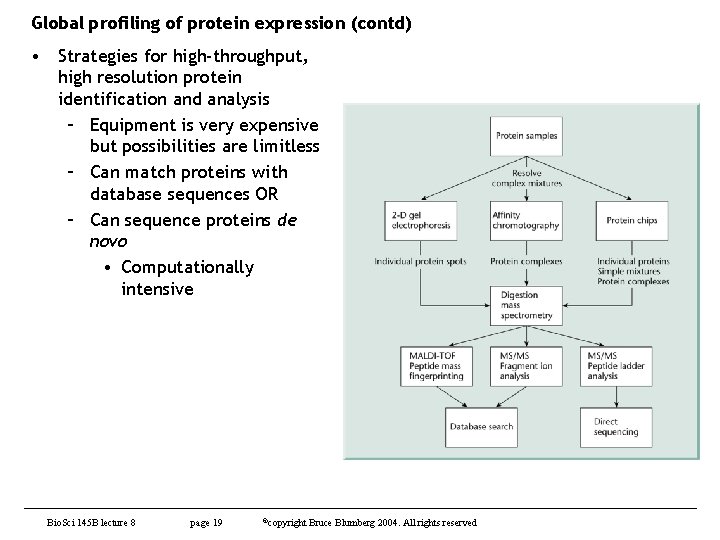
Global profiling of protein expression (contd) • Strategies for high-throughput, high resolution protein identification and analysis – Equipment is very expensive but possibilities are limitless – Can match proteins with database sequences OR – Can sequence proteins de novo • Computationally intensive Bio. Sci 145 B lecture 8 page 19 ©copyright Bruce Blumberg 2004. All rights reserved

Global profiling of protein expression (contd) • Protein arrays now available – Immobilized proteins • Spot proteins on slides and ask what interacts with particular ones • Luis Villareal runs a facility here that intends to produce all possible proteins for array generation – Antibody arrays • Antibodies spotted on arrays – test for presence of particular proteins in probe • Micro-ELISA or RIA – Antigen arrays • Known antigens spotted – tests for presence of antibodies in sample Bio. Sci 145 B lecture 8 page 20 ©copyright Bruce Blumberg 2004. All rights reserved

Genome wide analysis of gene function • Loss-of-function analysis is the most powerful way to identify gene function – Direct link between genotype and phenotype – Forward vs reverse genetics • Forward genetics-> random mutagenesis followed by phenotypic analysis – Identity of gene involved not known at the start • Reverse genetics -> associating functions with known genes – Directed mutagenesis of individual genes, phenotypic analysis – Reverse genetics is much more important than forward genetics in post genomic era • Because we have identified many genes from sequencing with no known functions, or even hints about function – Approaches • Mutagenesis (forward genetics) • Systematically mutating each gene (required genome sequence) • Random targeting with viruses or transposons, match genes later – Can id new genes as well as known genes • Generate phenocopies of mutant alleles – RNAi (si. RNA), morpholinos, virus induced gene silencing Bio. Sci 145 B lecture 8 page 21 ©copyright Bruce Blumberg 2004. All rights reserved

Construction of transgenic animals • standard transgenesis – Microinject DNA into a fertilized egg (mouse) or embryo (Drosophila) • some embryos undergo integration of DNA into genome • transgene goes to offspring in a few – Why do these mice have stubby tails? • Applications – rescue of a mutation – promoter analysis • ID elements required for expression • verify function of putative elements – model for dominant forms of human diseases – identify effects of misexpression – Large scale mutagenesis • Gene trap • Enhancer trap • RNAi Bio. Sci 145 B lecture 8 page 22 ©copyright Bruce Blumberg 2004. All rights reserved

Gene targeting • Transgenesis is mostly a gain of function technique – Loss-of-function preferred for identifying gene function • Targeted gene disruption is very desirable – to understand function of newly identified genes • e. g. , from genome projects • Or gene by gene – produce a mutation and evaluate the requirements for your gene of interest – good to create mouse models for human diseases • knockout the same gene disrupted in a human and may be able to understand disease better and develop efficacious treatments • excellent recent review is Müller (1999) Mechanisms of Development 82, 3 -21. Bio. Sci 145 B lecture 8 page 23 ©copyright Bruce Blumberg 2004. All rights reserved
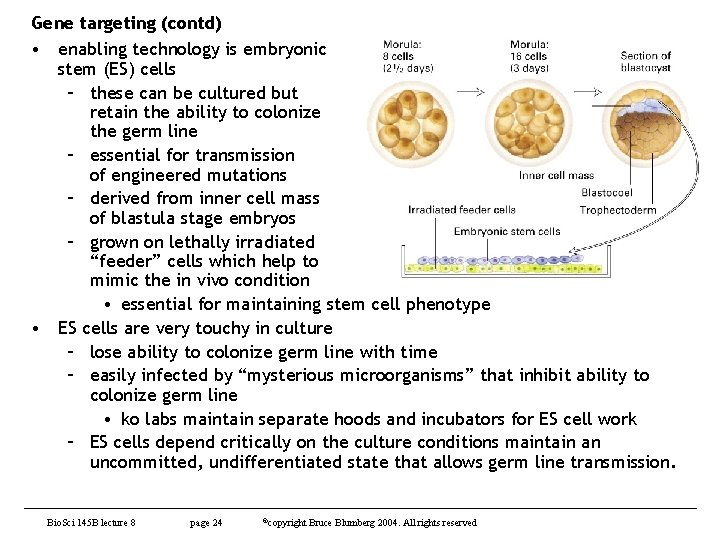
Gene targeting (contd) • enabling technology is embryonic stem (ES) cells – these can be cultured but retain the ability to colonize the germ line – essential for transmission of engineered mutations – derived from inner cell mass of blastula stage embryos – grown on lethally irradiated “feeder” cells which help to mimic the in vivo condition • essential for maintaining stem cell phenotype • ES cells are very touchy in culture – lose ability to colonize germ line with time – easily infected by “mysterious microorganisms” that inhibit ability to colonize germ line • ko labs maintain separate hoods and incubators for ES cell work – ES cells depend critically on the culture conditions maintain an uncommitted, undifferentiated state that allows germ line transmission. Bio. Sci 145 B lecture 8 page 24 ©copyright Bruce Blumberg 2004. All rights reserved
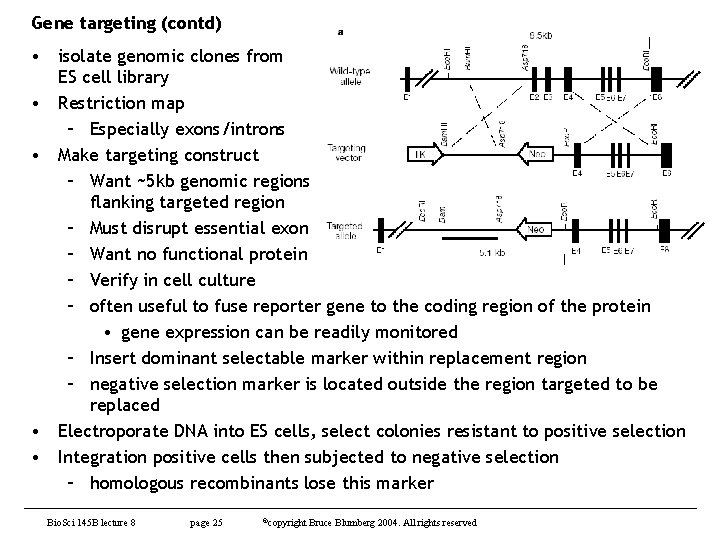
Gene targeting (contd) • isolate genomic clones from ES cell library • Restriction map – Especially exons/introns • Make targeting construct – Want ~5 kb genomic regions flanking targeted region – Must disrupt essential exon – Want no functional protein – Verify in cell culture – often useful to fuse reporter gene to the coding region of the protein • gene expression can be readily monitored – Insert dominant selectable marker within replacement region – negative selection marker is located outside the region targeted to be replaced • Electroporate DNA into ES cells, select colonies resistant to positive selection • Integration positive cells then subjected to negative selection – homologous recombinants lose this marker Bio. Sci 145 B lecture 8 page 25 ©copyright Bruce Blumberg 2004. All rights reserved
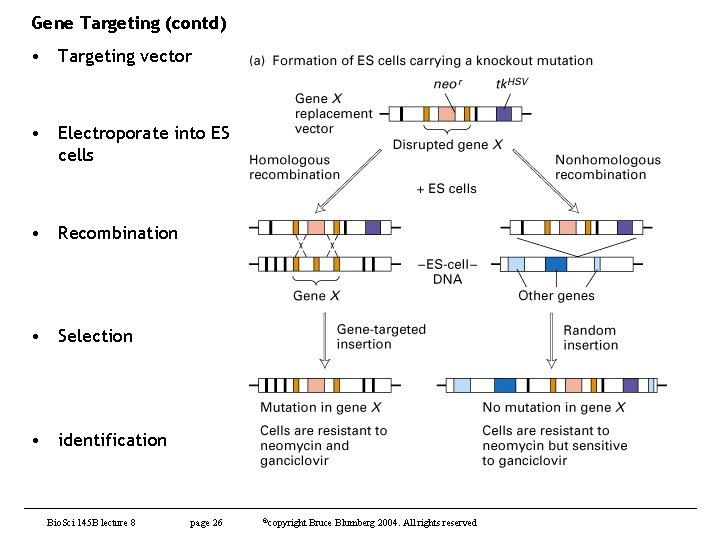
Gene Targeting (contd) • Targeting vector • Electroporate into ES cells • Recombination • Selection • identification Bio. Sci 145 B lecture 8 page 26 ©copyright Bruce Blumberg 2004. All rights reserved
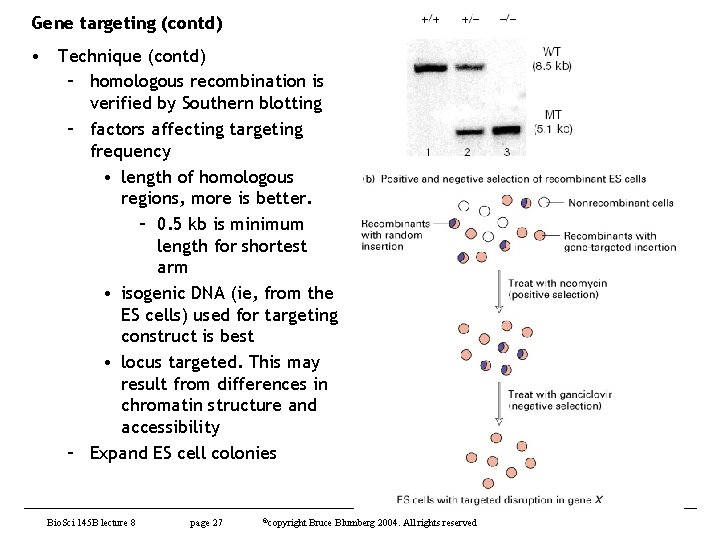
Gene targeting (contd) • Technique (contd) – homologous recombination is verified by Southern blotting – factors affecting targeting frequency • length of homologous regions, more is better. – 0. 5 kb is minimum length for shortest arm • isogenic DNA (ie, from the ES cells) used for targeting construct is best • locus targeted. This may result from differences in chromatin structure and accessibility – Expand ES cell colonies Bio. Sci 145 B lecture 8 page 27 ©copyright Bruce Blumberg 2004. All rights reserved
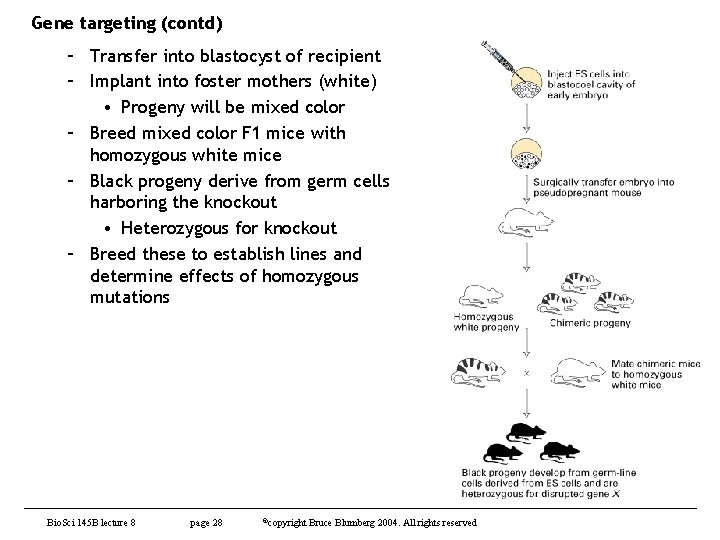
Gene targeting (contd) – Transfer into blastocyst of recipient – Implant into foster mothers (white) • Progeny will be mixed color – Breed mixed color F 1 mice with homozygous white mice – Black progeny derive from germ cells harboring the knockout • Heterozygous for knockout – Breed these to establish lines and determine effects of homozygous mutations Bio. Sci 145 B lecture 8 page 28 ©copyright Bruce Blumberg 2004. All rights reserved

Gene targeting (contd) • problems and pitfalls – incomplete knockouts, ie, protein function is not lost • but such weak alleles may be informative – alteration of expression of adjacent genes • region removed may contain regulatory elements • may remove unintended genes (e. g. on opposite strand) – interference from selection cassette • strong promoters driving these may cause phenotypes Bio. Sci 145 B lecture 8 page 29 ©copyright Bruce Blumberg 2004. All rights reserved

Gene targeting (contd) • Applications – creating loss-of-function alleles – introducing subtle mutations – chromosome engineering – marking gene with reporter, enabling whole mount detection of expression pattern (knock-in) • advantages – can generate a true loss-of-function alleles – precise control over integration sites – prescreening of ES cells for phenotypes possible – can also “knock in” genes • disadvantages – not trivial to set up – may not be possible to study dominant lethal phenotypes – non-specific embryonic lethality is common (~30%) – difficulties related to selection cassette Bio. Sci 145 B lecture 8 page 30 ©copyright Bruce Blumberg 2004. All rights reserved

Conditional gene targeting • Many gene knockouts are embryonic lethal – some of these are appropriate and expected • gene activity is required early – others result from failure to form and/or maintain the placenta • ~30% of all knockouts • Clearly a big obstacle for gene analysis • How can this be overcome? – Generate conditional knockouts either in particular tissues or after critical developmental windows pass – Sauer (1998) Methods 14, 381 -392. Bio. Sci 145 B lecture 8 page 31 ©copyright Bruce Blumberg 2004. All rights reserved
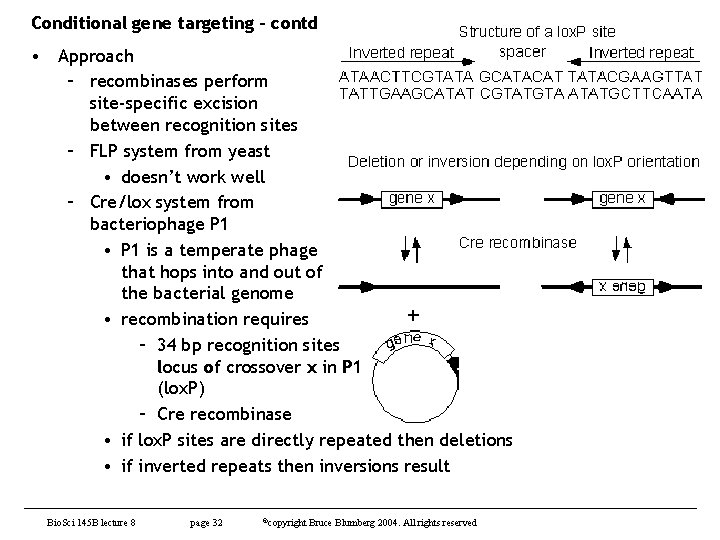
Conditional gene targeting - contd • Approach – recombinases perform site-specific excision between recognition sites – FLP system from yeast • doesn’t work well – Cre/lox system from bacteriophage P 1 • P 1 is a temperate phage that hops into and out of the bacterial genome • recombination requires – 34 bp recognition sites locus of crossover x in P 1 (lox. P) – Cre recombinase • if lox. P sites are directly repeated then deletions • if inverted repeats then inversions result Bio. Sci 145 B lecture 8 page 32 ©copyright Bruce Blumberg 2004. All rights reserved
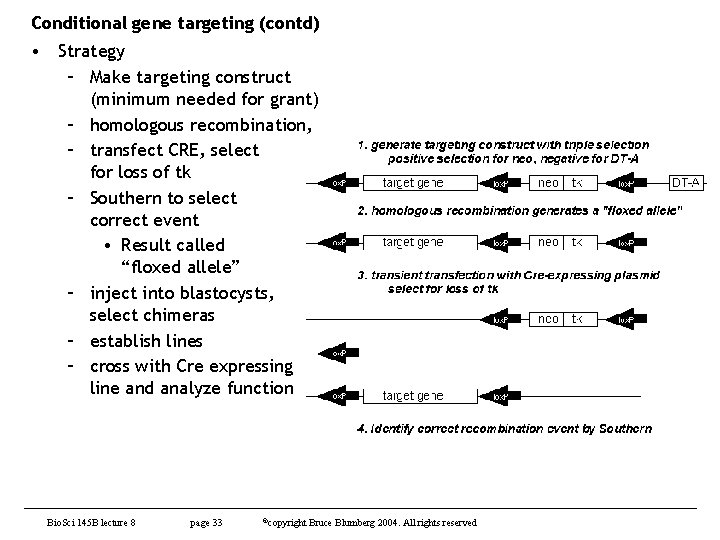
Conditional gene targeting (contd) • Strategy – Make targeting construct (minimum needed for grant) – homologous recombination, – transfect CRE, select for loss of tk – Southern to select correct event • Result called “floxed allele” – inject into blastocysts, select chimeras – establish lines – cross with Cre expressing line and analyze function Bio. Sci 145 B lecture 8 page 33 ©copyright Bruce Blumberg 2004. All rights reserved
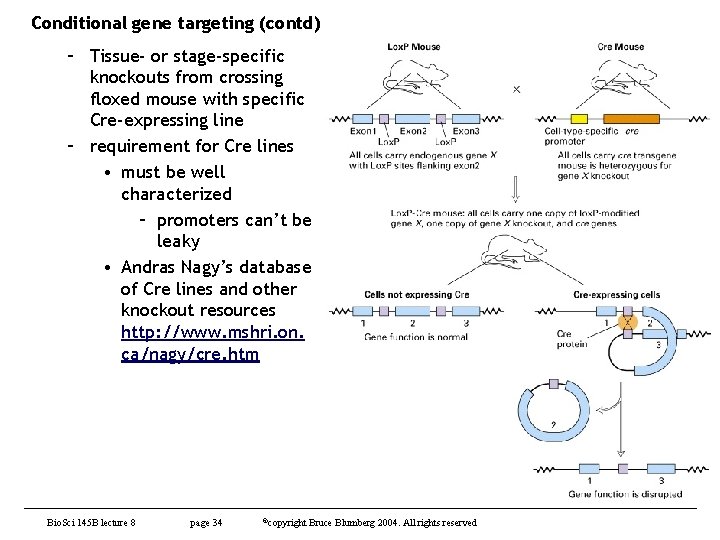
Conditional gene targeting (contd) – Tissue- or stage-specific knockouts from crossing floxed mouse with specific Cre-expressing line – requirement for Cre lines • must be well characterized – promoters can’t be leaky • Andras Nagy’s database of Cre lines and other knockout resources http: //www. mshri. on. ca/nagy/cre. htm Bio. Sci 145 B lecture 8 page 34 ©copyright Bruce Blumberg 2004. All rights reserved

Conditional gene targeting (contd) • advantages – can target recombination to specific tissues and times – can study genes that are embryonic lethal when disrupted – can use for marker eviction – can study the role of a single gene in many different tissues with a single mouse line – can use for engineering translocations and inversions on chromosomes • disadvantages – not trivial to set up, more difficult than std ko but more information possible – requirement for Cre lines • must be well characterized regarding site and time of expression • promoters can’t be leaky (expressed when not intended) Bio. Sci 145 B lecture 8 page 35 ©copyright Bruce Blumberg 2004. All rights reserved
- Slides: 35