Bio Pharma Industry Overview Myron Pyzyk BSc MS

Bio. Pharma Industry Overview Myron Pyzyk, BSc, MS Principal & CEO Marenon Consulting June 20, 2011 7 MARENON HEALTH GROUP 3/3/2021

Bio. Pharma Industry Overview • Current dynamics of the Bio. Pharma market: implications for processes and organizational structures • Trends in the industry: impact on existing business models • Current dynamics of the global Bio. Pharma market competencies • Economic, regulatory, political and technological forces shaping Bio. Pharma industry • Evolving business competencies required of Bio. Pharma Executives

Bioscience Sectors Bio. Science & Health. Care Private Sector Non-Gov’t Organisations (NGOs) Presentation to Investment Conference (UKraine), Nov. 1/2, 2007 Government MARENON HEALTH GROUP 3/3/2021

Bioscience Sectors Bio. Science Industry • Biologics and pharmaceuticals • Medical devices and equipment • Research, testing and medical laboratories • Agricultural feedstocks and chemicals MARENON HEALTH GROUP 3/3/2021

History of Healthcare • Early Medicine - Early `Bio. Pharma`: • Mesopotamia (Hammurabi; 3500 BCE) • Egyptians (1500 BCE) • Greeks (450 BCE) • Hippocrates: established medicine as a separate profession • Romans (140 BCE) • • Philosophers - Physicians Supernatural – Disease – Health Placebo effect Ceremonies, initiations, customs and rites
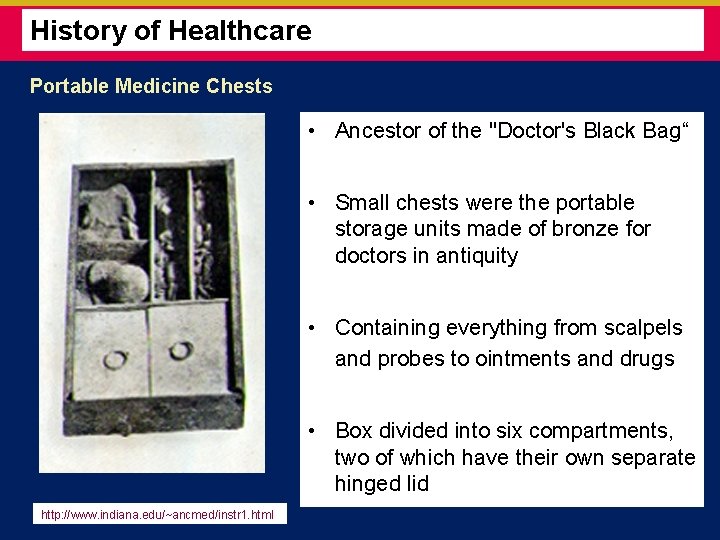
History of Healthcare Portable Medicine Chests • Ancestor of the "Doctor's Black Bag“ • Small chests were the portable storage units made of bronze for doctors in antiquity • Containing everything from scalpels and probes to ointments and drugs • Box divided into six compartments, two of which have their own separate hinged lid http: //www. indiana. edu/~ancmed/instr 1. html

History of Healthcare Scientific Medicine • • • Physician – Scientists Evidence-based Health – Disease – Health; Health promotion Placebo effect Re-emphasis of ‘natural` health maintenance/healing Social Development • Complex social systems • Complex organizations • Complex roles • Complex technology
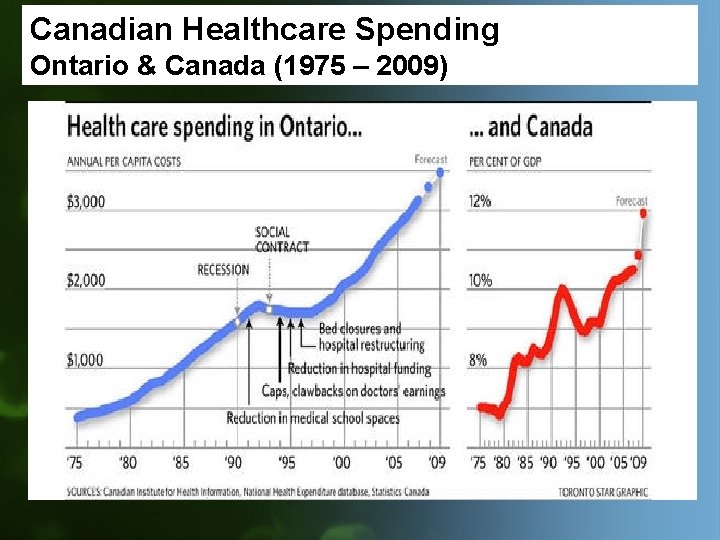
Canadian Healthcare Spending Ontario & Canada (1975 – 2009)
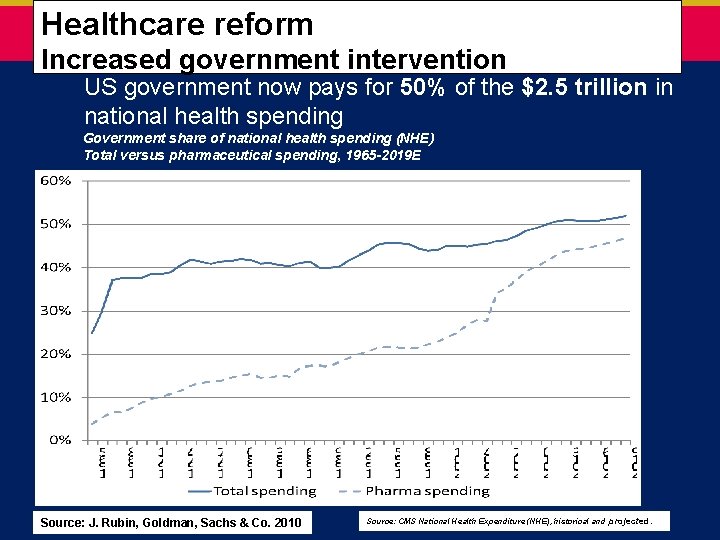
Healthcare reform – increased government intervention Healthcare reform Increased government intervention US government now pays for 50% of the $2. 5 trillion in national health spending Government share of national health spending (NHE) Total versus pharmaceutical spending, 1965 -2019 E Source: J. Rubin, Goldman, Sachs & Co. 2010 Source: CMS National Health Expenditure (NHE), historical and projected.
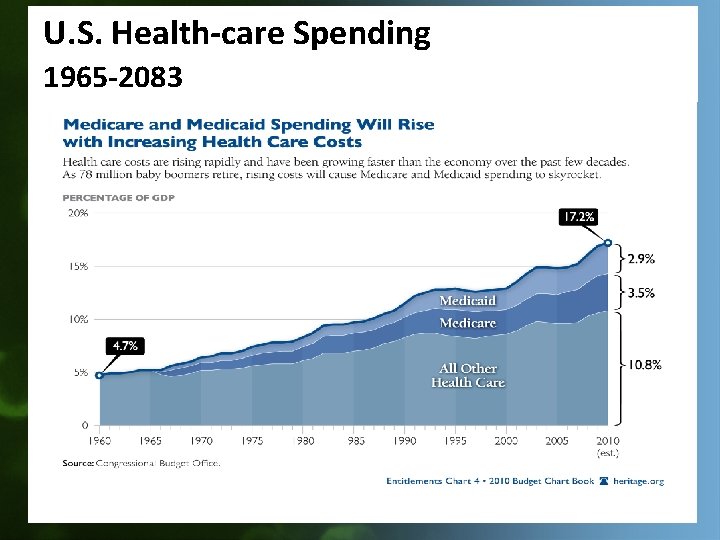
U. S. Health-care Spending 1965 -2083
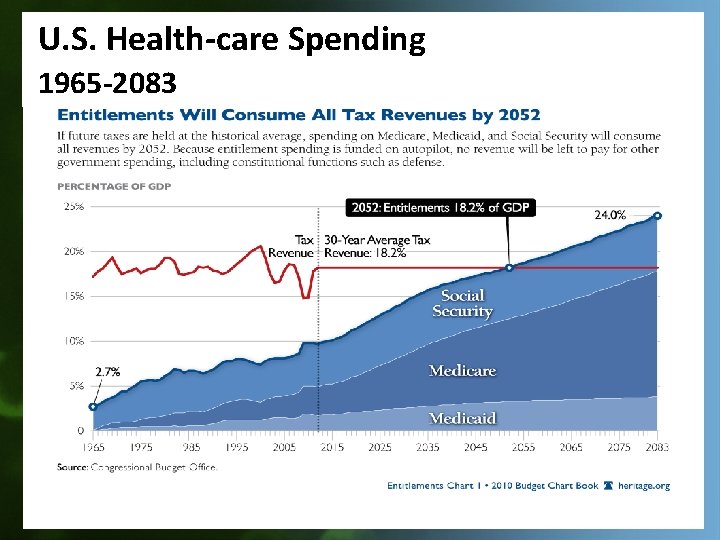
U. S. Health-care Spending 1965 -2083

Health Warning! MARENON HEALTH GROUP 3/3/2021
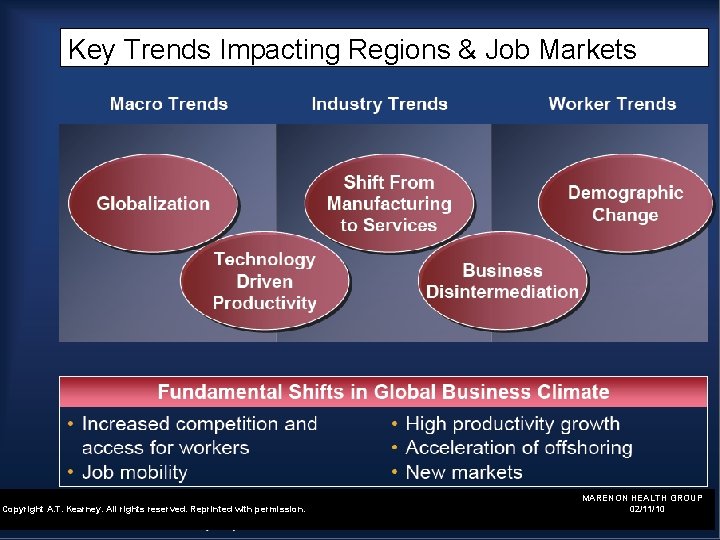
Key Trends Impacting Regions & Job Markets Copyright A. T. Kearney. All rights reserved. Reprinted with permission. MARENON HEALTH GROUP 02/11/10

Five “Generations” of Drug Development 1880 – Present • 1. Discovery of “active principles” in natural products, fermentations, and simple coal-tar derivatives: analgesics, antipyretics, anesthetics, hypnotics, sedatives (1820 - 1880) • 2. Experimental therapeutics and chemotherapy. Use of synthetic organic dyes to identify pathogenic microorganisms and to manufacture antiprotozoal medicines, serums, toxins, and vaccines (1880 - 1930) • 3. Introduction of sulfa drugs, antibiotics, antihistamines, vitamins, corticosteroids, and sex hormones (1930 - 1960) • 4. Drugs to treat hypertension and other cardiovascular diseases; antianxiety drugs, antidepressants, other CNS; oral contraceptives; semisynthetic penicillins, cephalosporins; and NSAIDS (1960 - 1980) • 5. Bio-engineered proteins, antineoplastics, antivirals; new drug delivery systems, and diagnostic tests based on recombinant DNA and monoclonal antibodies (1980 - ? ). 14
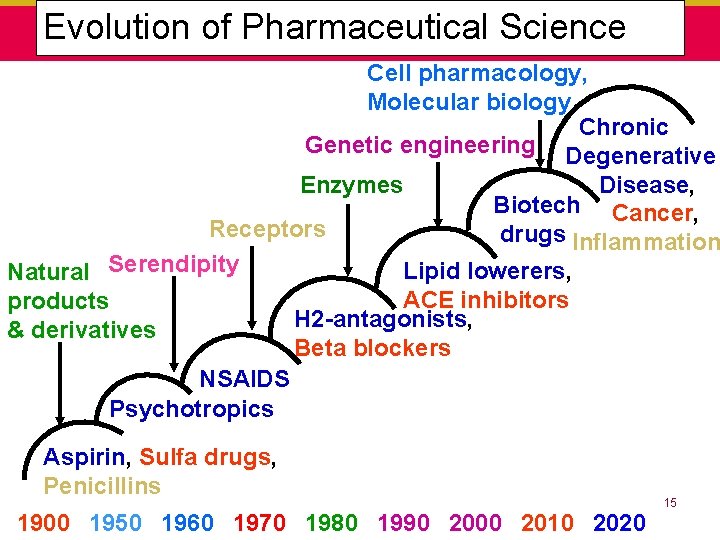
Evolution of Pharmaceutical Science Cell pharmacology, Molecular biology Chronic Genetic engineering Degenerative Disease, Enzymes Biotech Cancer, Receptors drugs Inflammation Lipid lowerers, Natural Serendipity ACE inhibitors products H 2 -antagonists, & derivatives Beta blockers NSAIDS Psychotropics Aspirin, Sulfa drugs, Penicillins 1900 1950 1960 1970 1980 1990 2000 2010 2020 15

Types of Radical Innovations • Result in new industries or subsectors of an existing industry: – – – Smallpox vaccine (1796); Morphine, 1 st alkaloid (1806); Carbolic acid (phenol), 1 st antiseptic (1860); Phenazone (Antipyrin), 1 st synthetic drug (1884); Arsphenamine (Salvarsan), 1 st chemotherapeutic agent (1911); – Sulfamidochrysoidine (Prontosil), 1 st antibacterial (1935); – Penicillin, 1 st antibiotic (1942); – Process for recombinant DNA, beginning of biotechnology (1975). 16

Types of Radical Innovations • Widen the scope and markets of existing sectors or subsectors • Apply new scientific principles, technology, or materials to displace existing products or processes • Serve as models for further innovation by imitation: – Barbital (Veronal), 1 st barbiturate hypnotic (1903) – 32 imitations; – Chlorothiazide (Diuril), 1 st antihypertensive diuretic (1958) – 15 i’s; – Chlordiazepoxide (Librium), 1 st benzodiazepine anxiolytic (1960) – 37 i’s; – Propranolol (Inderal), 1 st antihypertensive ß-blocker (1964) – 24 i’s; – Cimetidine (Tagamet), 1 st treatment for peptic ulcers (1977) – 7 i’s. 17

Incremental Innovation • Preeminent vehicle for diffusing innovation among competing companies. • Can be big money makers. • Designed on models of existing products or processes • modest differences in science, technology, materials, etc. ; • do not provide scope for further innovation by imitation. • • • Trifluoperazine (Stelazine), tranquilizer (1959); Cefaclor (Ceclor), antibacterial (1979); Enalapril (Vasotec), ACE inhibitor (1985); Ranitidine (Zantac), antiulcer (1982); Atorvastatin (Lipitor) cholesterol reducer (1997). 18

Time Horizons for Structural Change • Perceptions of the short- and medium-term outlook for an industry can change almost overnight • Structural change to diversify feedstocks and supply lines of intermediates can take years, if not decades to accomplish. 19

The Situation Economies 2008 - 2010: • The Market Presentation to University of Alberta March 18, 2009 MARENON HEALTH GROUP 3/3/2021

The Situation • Economies 2008 - 2011: • Short-term impact: • startup biotech companies face credit vacuum • Big Bio. Pharma reduces spend • Big Bio. Pharma restructures • Long-term impact: • Rethink how we approach innovation and risk • People drive innovation, innovation driven by shared commitment, not heavy system planning & regulation • New business model(s) offering new system incentives MARENON HEALTH GROUP 3/3/2021
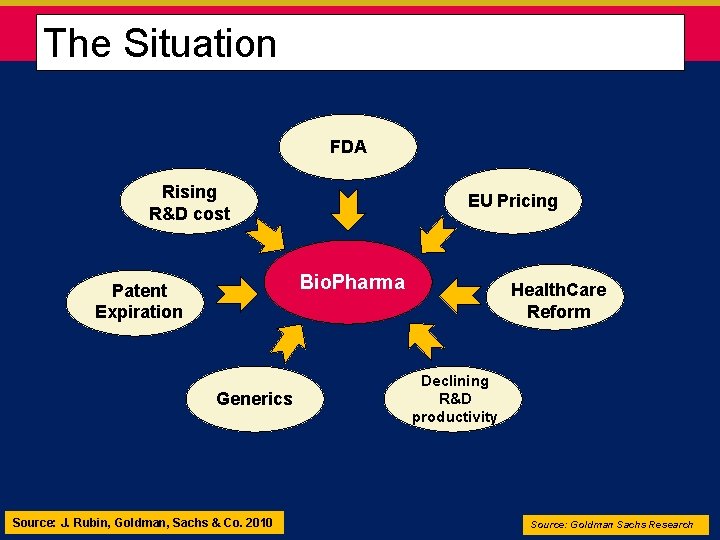
The Situation FDA Rising R&D cost EU Pricing Bio. Pharma Patent Expiration Generics Source: J. Rubin, Goldman, Sachs & Co. 2010 Health. Care Reform Declining R&D productivity Source: Goldman Sachs Research
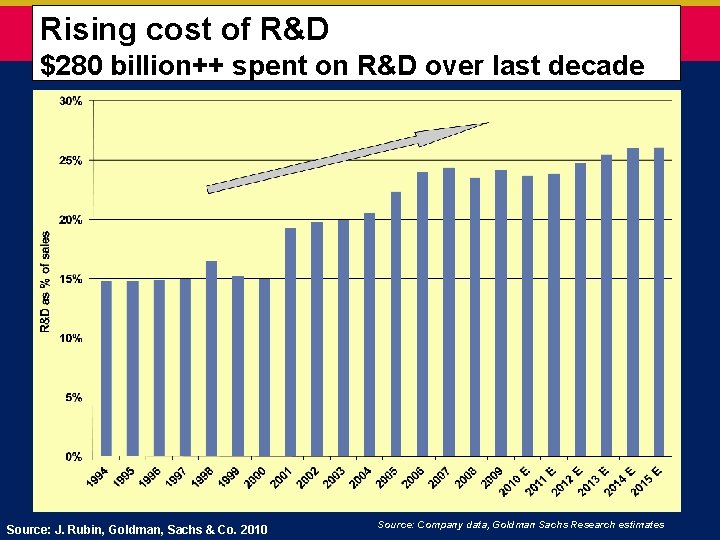
Rising cost of R&D $280 billion++ spent on R&D over last decade Source: J. Rubin, Goldman, Sachs & Co. 2010 Source: Company data, Goldman Sachs Research estimates
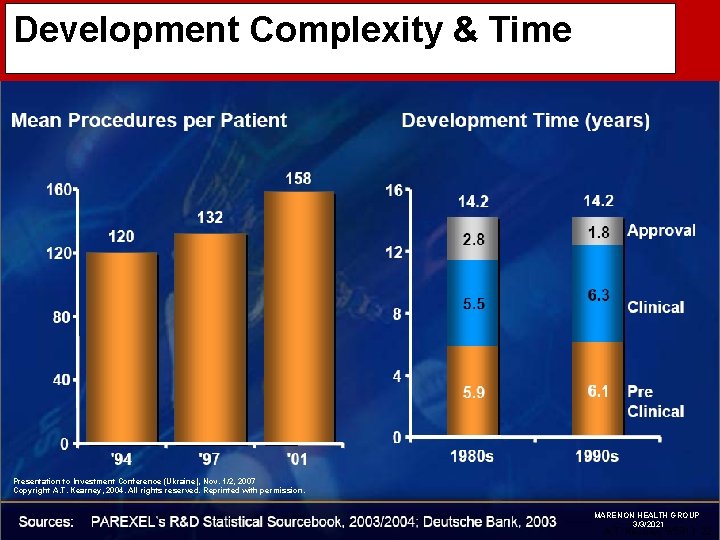
Development Complexity & Time Presentation to Investment Conference (Ukraine), Nov. 1/2, 2007 Copyright A. T. Kearney, 2004. All rights reserved. Reprinted with permission. MARENON HEALTH GROUP 3/3/2021
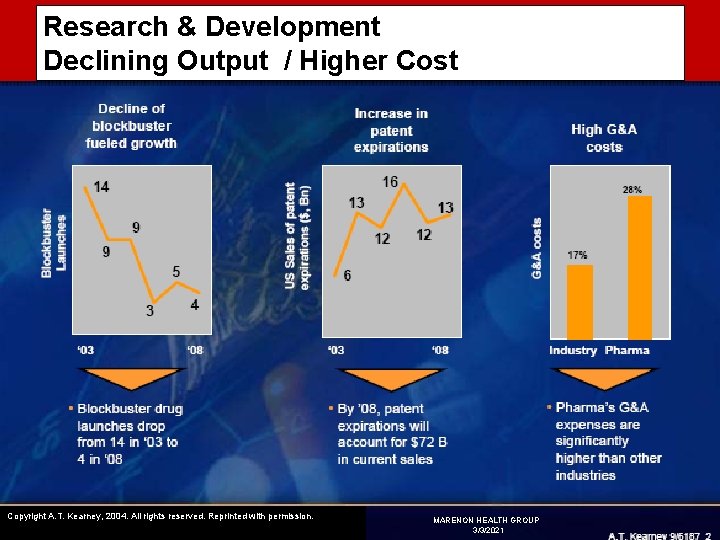
Research & Development Declining Output / Higher Cost Copyright A. T. Kearney, 2004. All rights reserved. Reprinted with permission. MARENON HEALTH GROUP 3/3/2021
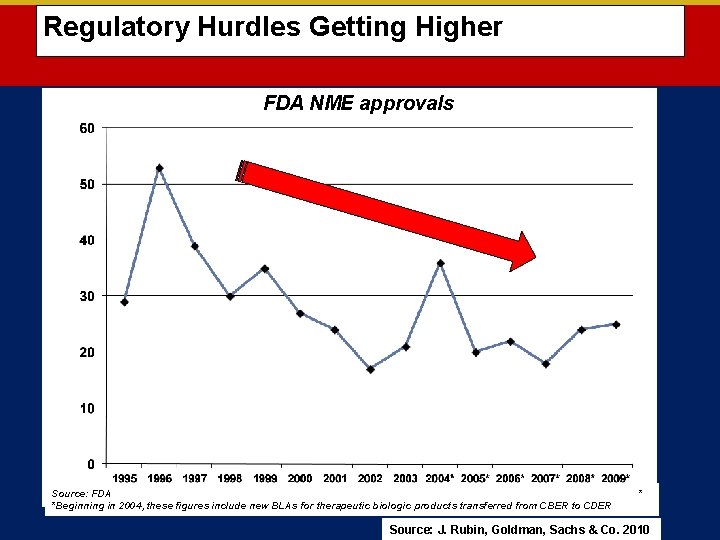
Regulatory Hurdles Getting Higher FDA NME approvals Source: FDA *Beginning in 2004, these figures include new BLAs for therapeutic biologic products transferred from CBER to CDER * Source: J. Rubin, Goldman, Sachs & Co. 2010
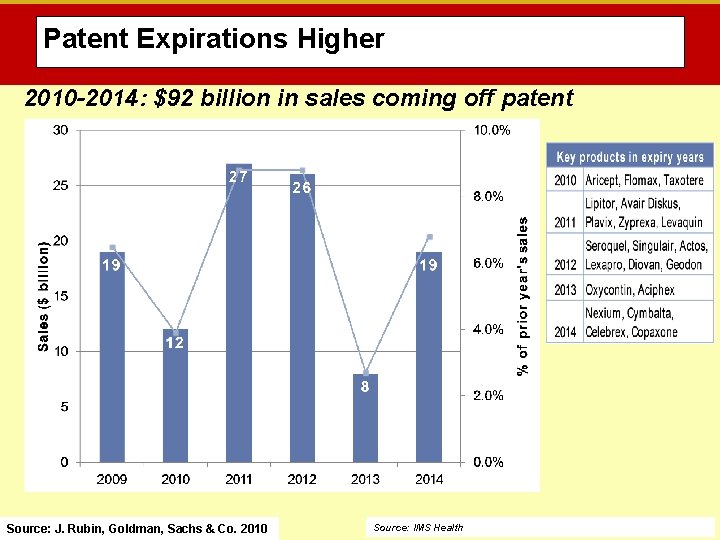
Patent Expirations Higher 2010 -2014: $92 billion in sales coming off patent Source: J. Rubin, Goldman, Sachs & Co. 2010 Source: IMS Health
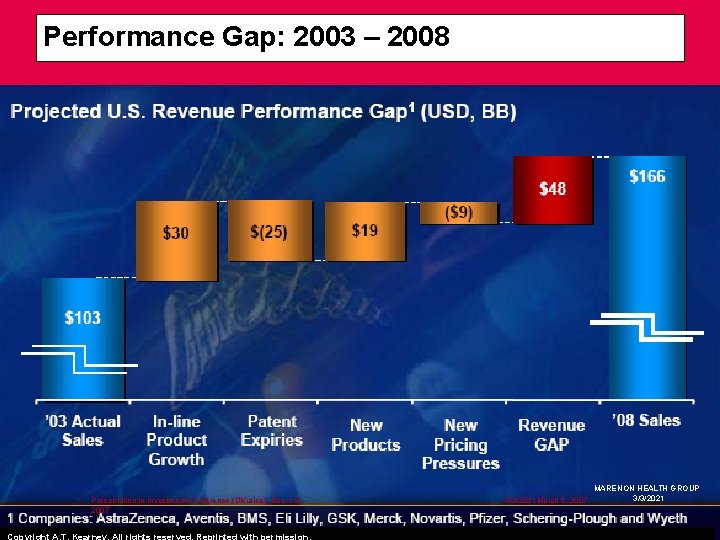
Performance Gap: 2003 – 2008 Presentation to Investment Conference (UKraine), Nov. 1/2, 2007 Copyright A. T. Kearney. All rights reserved. Reprinted with permission. 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021

The Situation • Easy targets are gone (eg. Losec, Prozac) • Chronic disease harder to study (eg. Coronary Artery Disease) • Slow integration of newer predictive and evaluative tools MARENON HEALTH GROUP 3/3/2021

Evolution of Best-Practices in Medicine Impact on Drug Development Copyright A. T. Kearney. All rights reserved. Reprinted with permission. MARENON HEALTH GROUP Source: AT Kearney 2/11/10

The Situation Drug Developer Uncertainty • Flux in endpoints/trial designs – What should I measure to support my label claim? – What type of trial design would best support my label claims? • Mandated trials too large/complex * – Imposition of multiple endpoints * Can also be an excellent ‘competitive block’ as part of an R&D strategy by raising the bar for entry’ for any new competitor into a drug class or indication i. e. more complex data, more expensive R&D MARENON HEALTH GROUP 3/3/2021

Similar Comparative Effectiveness Drugs based on performance and outcome • Shift from surrogate endpoints (i. e, LDL, Hba 1 C) to clinical outcomes (i. e, heart attacks, mortality) • Cholesterol drugs (Vytorin has been significantly hurt by lack of outcomes) • Diabetes drugs (need cardiovascular outcomes trials; has slowed the development of PPAR gammas, etc. ) • Oncology drugs must show overall survival benefits Source: J. Rubin, Goldman, Sachs & Co. 2010

The Situation • Basic Science ≠ New Meds / Therapies • Bench to Bedside – Expen$$$ive development – Increasingly longer timeframes – ↑ investment : : ↓ Po. S • Regulatory system slow(er) to evolve and respond re: standards and models MARENON HEALTH GROUP 3/3/2021

Bio. Pharma Innovation Issues Slowing Innovation Legal Issues • IP = basic discoveries e. g. biosimilars Biomedical Research `Un-Collaboration` • Industry and academia: • Reluctant / infrequent partners • Don't share data MARENON HEALTH GROUP 3/3/2021

Entrepreneurship and Business, Emerging Science and Technology by webgoddesscathy@Ma. RS February 26 th, 2007 Biotechnology has been “one of the biggest money-losing industries in the history of mankind” Genentech CEO Arthur D. Levinson • Estimates biotech industry lost nearly $100 billion in the past three decades • Only 54 of 342 publicly traded American biotech companies were profitable in 2006 MARENON HEALTH GROUP 3/3/2021
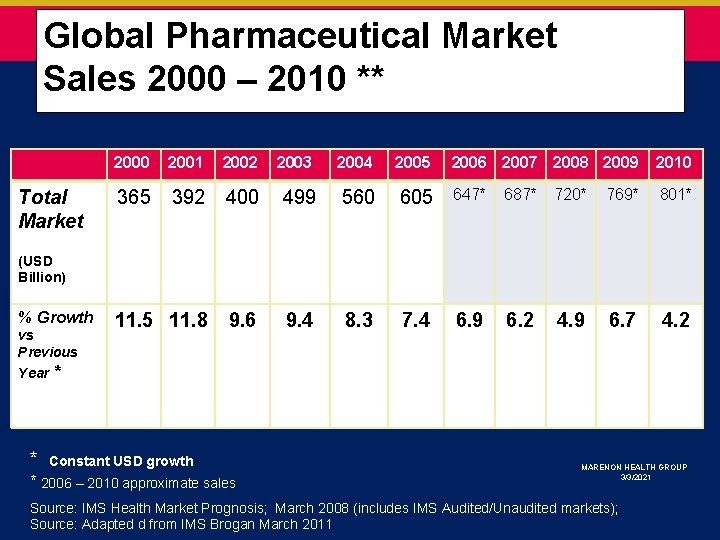
Global Pharmaceutical Market * Sales 2000 – 2010 ** Total Market 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 365 392 400 499 560 605 647* 687* 720* 769* 801* 11. 5 11. 8 9. 6 9. 4 8. 3 7. 4 6. 9 6. 2 4. 9 6. 7 4. 2 (USD Billion) % Growth vs Previous Year * * Constant USD growth * 2006 – 2010 approximate sales MARENON HEALTH GROUP 3/3/2021 Source: IMS Health Market Prognosis; March 2008 (includes IMS Audited/Unaudited markets); Source: Adapted d from IMS Brogan March 2011
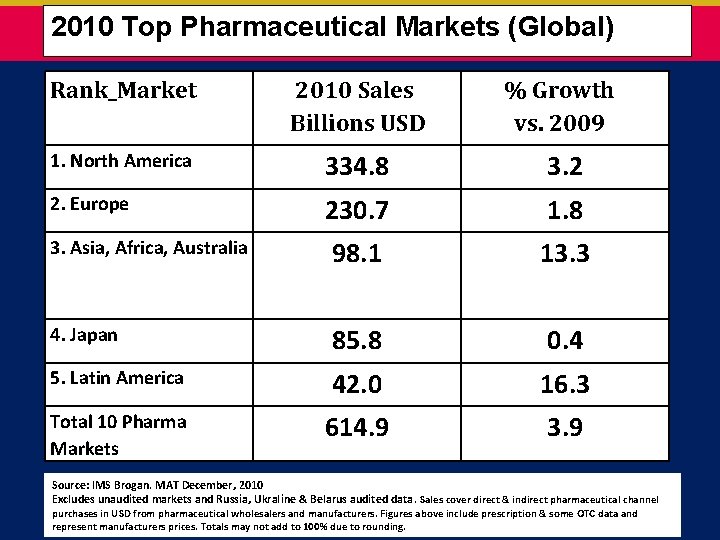
2010 Top Pharmaceutical Markets (Global) Rank_Market 2010 Sales Billions USD % Growth vs. 2009 1. North America 334. 8 3. 2 2. Europe 230. 7 1. 8 3. Asia, Africa, Australia 98. 1 13. 3 4. Japan 85. 8 0. 4 5. Latin America 42. 0 16. 3 Total 10 Pharma Markets 614. 9 3. 9 Source: IMS Brogan. MAT December, 2010 Excludes unaudited markets and Russia, Ukra. Iine & Belarus audited data. Sales cover direct & indirect pharmaceutical channel purchases in USD from pharmaceutical wholesalers and manufacturers. Figures above include prescription & some OTC data and represent manufacturers prices. Totals may not add to 100% due to rounding.
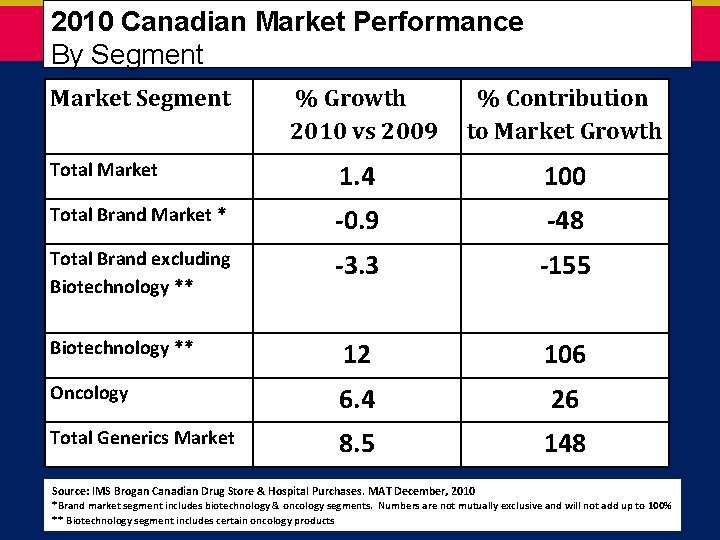
2010 Canadian Market Performance By Segment Market Segment % Growth 2010 vs 2009 % Contribution to Market Growth Total Market 1. 4 100 Total Brand Market * -0. 9 -48 Total Brand excluding Biotechnology ** -3. 3 -155 Biotechnology ** 12 106 Oncology 6. 4 26 Total Generics Market 8. 5 148 Source: IMS Brogan Canadian Drug Store & Hospital Purchases. MAT December, 2010 *Brand market segment includes biotechnology & oncology segments. Numbers are not mutually exclusive and will not add up to 100% ** Biotechnology segment includes certain oncology products
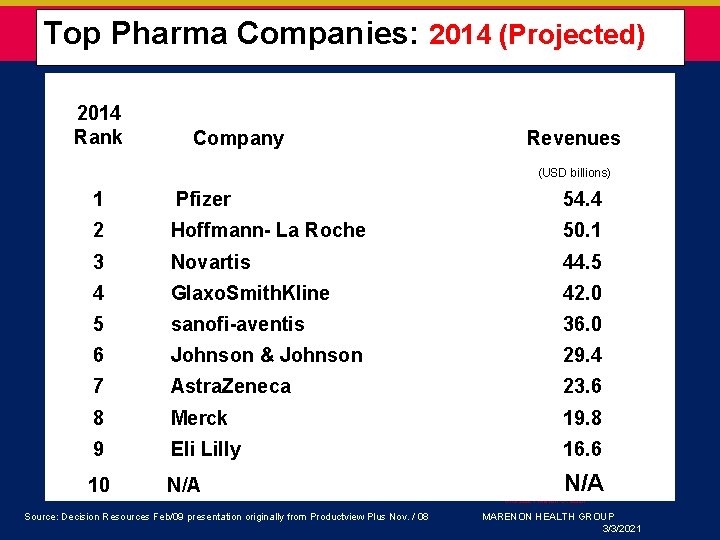
Top Pharma Companies: 2014 (Projected) 2014 Rank Company Revenues (USD billions) 1 Pfizer 54. 4 2 Hoffmann- La Roche 50. 1 3 Novartis 44. 5 4 Glaxo. Smith. Kline 42. 0 5 sanofi-aventis 36. 0 6 Johnson & Johnson 29. 4 7 Astra. Zeneca 23. 6 8 Merck 19. 8 9 Eli Lilly 16. 6 10 N/A 3/3/2021 March 6, 2007 Source: Decision Resources Feb/09 presentation originally from Productview Plus Nov. / 08 MARENON HEALTH GROUP 3/3/2021
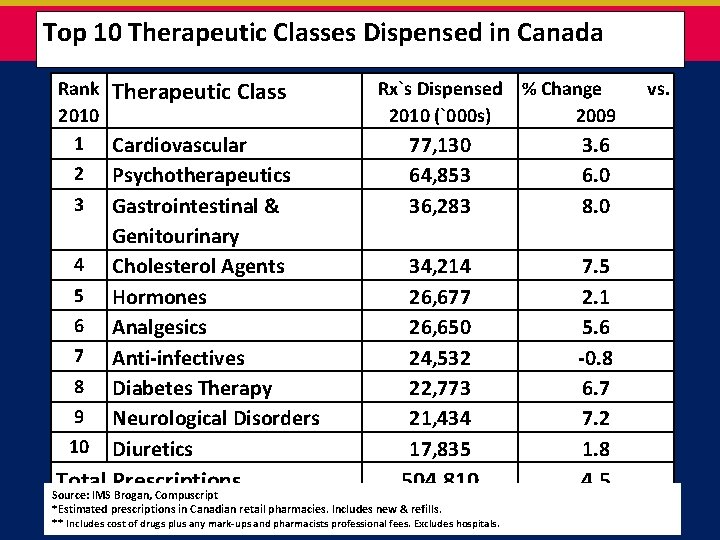
Top 10 Therapeutic Classes Dispensed in Canada Rank Therapeutic Class 2010 1 Cardiovascular 2 Psychotherapeutics 3 4 5 6 7 8 9 10 Gastrointestinal & Genitourinary Cholesterol Agents Hormones Analgesics Anti-infectives Diabetes Therapy Neurological Disorders Diuretics Total Prescriptions Rx`s Dispensed % Change 2010 (`000 s) 2009 77, 130 64, 853 36, 283 3. 6 6. 0 8. 0 34, 214 26, 677 26, 650 24, 532 22, 773 21, 434 17, 835 7. 5 2. 1 5. 6 -0. 8 6. 7 7. 2 1. 8 504, 810 4. 5 Source: IMS Brogan, Compuscript *Estimated prescriptions in Canadian retail pharmacies. Includes new & refills. ** Includes cost of drugs plus any mark-ups and pharmacists professional fees. Excludes hospitals. vs.
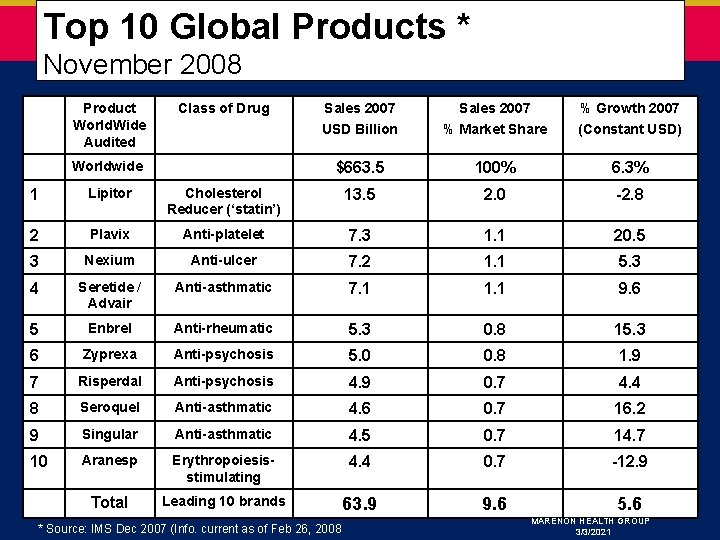
Top 10 Global Products * November 2008 Product World. Wide Audited Class of Drug Worldwide Sales 2007 USD Billion Sales 2007 % Market Share % Growth 2007 (Constant USD) $663. 5 100% 6. 3% 1 Lipitor Cholesterol Reducer (‘statin’) 13. 5 2. 0 -2. 8 2 Plavix Anti-platelet 7. 3 1. 1 20. 5 3 Nexium Anti-ulcer 7. 2 1. 1 5. 3 4 Seretide / Advair Anti-asthmatic 7. 1 1. 1 9. 6 5 Enbrel Anti-rheumatic 5. 3 0. 8 15. 3 6 Zyprexa Anti-psychosis 5. 0 0. 8 1. 9 7 Risperdal Anti-psychosis 4. 9 0. 7 4. 4 8 Seroquel Anti-asthmatic 4. 6 0. 7 16. 2 9 Singular Anti-asthmatic 4. 5 0. 7 14. 7 10 Aranesp Erythropoiesisstimulating 4. 4 0. 7 -12. 9 Total Leading 10 brands 63. 9 9. 6 5. 6 * Source: IMS Dec 2007 (Info. current as of Feb 26, 2008 MARENON HEALTH GROUP 3/3/2021
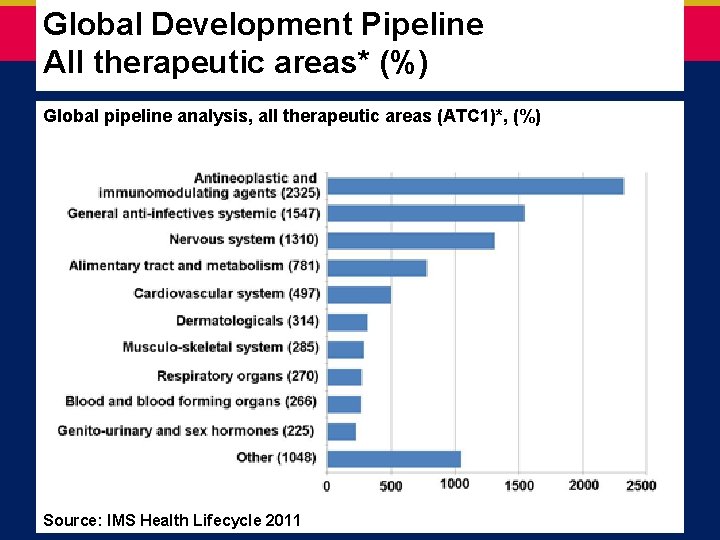
Global Development Pipeline All therapeutic areas* (%) Global pipeline analysis, all therapeutic areas (ATC 1)*, (%) Source: IMS Health Lifecycle 2011
Producing Movie! Many Concepts - Many Risks – Few Investors Directed by Tom Hooper Produced by Iain Canning, Emile Sherman, Gareth Unwin Screenplay by David Seidler Starring Colin Firth, Geoffrey Rush, Helena Bonham Carter Music by Alexandre Desplat Cinematography Danny Cohen Editing by Tariq Anwar Studio The Weinstein Company, UK Film Council, See-saw Films Bedlam Productions Distributed by The Weinstein Company Release date(s) 2010 -09 -06 (Telluride Film Festival), 2011 -01 -07 (UK) Budget £ 8 million] Gross. Presentation to University of Alberta revenue $401, 429, 179[ MARENON HEALTH GROUP 3/3/2021 07

Developing New Drugs Many Concepts – Many Risks – Few Investors Shared Risk or Risk Reduction • Small biotech companies – – Develop concept / molecule / IP Raise VC financing Develop drug to phase II or III Business Development: partnering opportunities • Big Pharma (eg. Pfizer) – Partnering or licensing – Royalties / Milestone payments – Marketing & distribution & sales muscle 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021
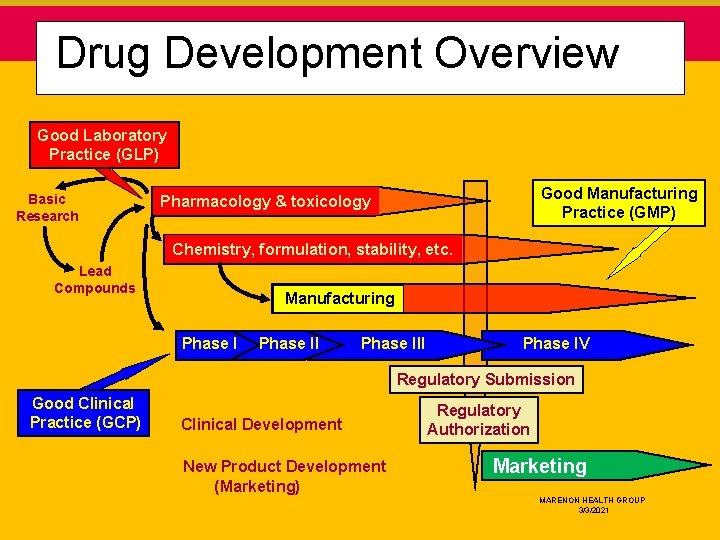
Drug Development Overview Good Laboratory Practice (GLP) Basic Research Good Manufacturing Practice (GMP) Pharmacology & toxicology Chemistry, formulation, stability, etc. Lead Compounds Manufacturing Phase III Phase IV Regulatory Submission Good Clinical Practice (GCP) Clinical Development New Product Development (Marketing) Regulatory Authorization Marketing MARENON HEALTH GROUP 3/3/2021
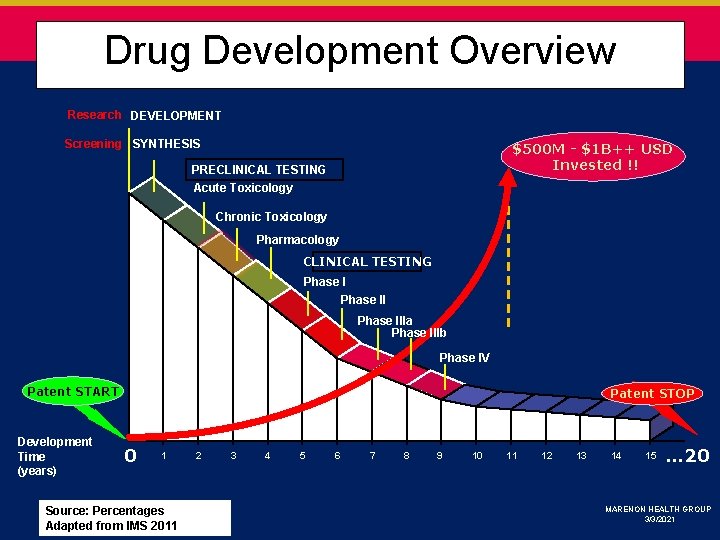
Drug Development Overview Research DEVELOPMENT Screening SYNTHESIS $500 M - $1 B++ USD Invested !! PRECLINICAL TESTING Acute Toxicology Chronic Toxicology Pharmacology CLINICAL TESTING Phase IIIa Phase IIIb Phase IV Patent START Development Time (years) Patent STOP 0 1 Source: Percentages Adapted from IMS 2011 2 3 4 5 6 7 8 9 10 11 12 13 14 15 … 20 MARENON HEALTH GROUP 3/3/2021
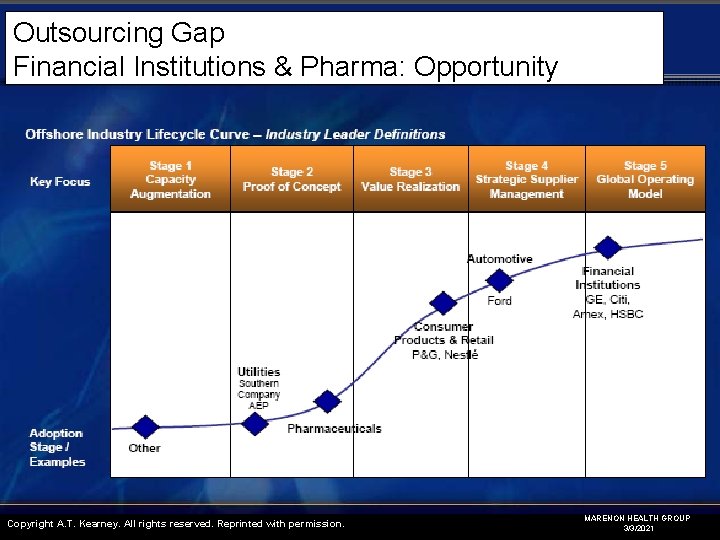
Outsourcing Gap Financial Institutions & Pharma: Opportunity Copyright A. T. Kearney. All rights reserved. Reprinted with permission. MARENON HEALTH GROUP 3/3/2021
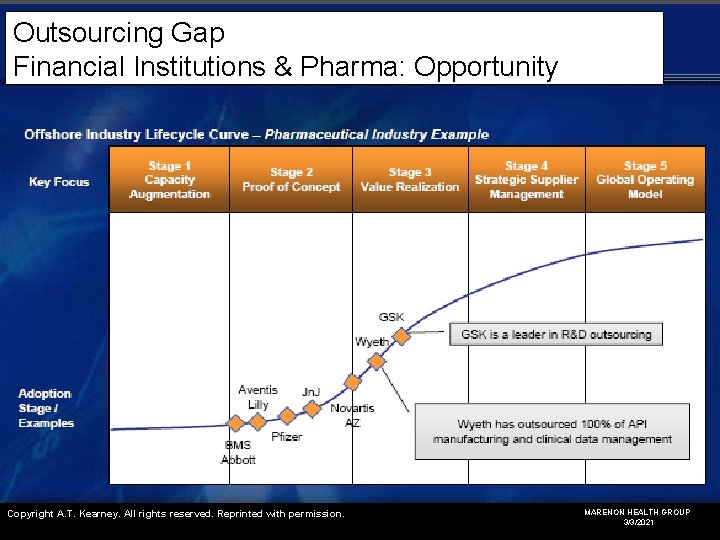
Outsourcing Gap Financial Institutions & Pharma: Opportunity Copyright A. T. Kearney. All rights reserved. Reprinted with permission. MARENON HEALTH GROUP 3/3/2021

Outsourcing Gap Financial Institutions & Pharma: Opportunity Advantages • Expertise • Speed / flexibility • Better process management • Better fixed/variable cost ratio • Better ROI Industry Concerns • Pharma is Conservative (late or even last adopters) • Business continuity (pipeline delays) • Regulatory demands • IP protection • Patient Confidentiality • Outsource reliability (quality, on-time, on-budget) MARENON HEALTH GROUP 3/3/2021

Conclusions and Recommendations MARENON HEALTH GROUP 3/3/2021

Global Pharma - Critical Steps • Consider New Ways of Doing Business • What Business do you want to be in • Are You Really In that business • Business model(s) • Get a handle on costs (financial controls) • Accelerate product development • Greater breadth / depth of skills • Greater flexibility to structure and processes eg. Outsourcing… MARENON HEALTH GROUP 3/3/2021

Strategic Questions to Ask • In what direction is the life sciences industry heading globally? • In what direction is the healthcare heading globally? • What are the key determining factors that will affect the future structure? • What impact will the future structure have on planning needs and functions? 52

In what direction is the pharmaceutical industry heading globally? Industry simultaneously pursuing three macro-objectives: • Increased specialization; – complex and highly technical nature of virtually all aspects of the discovery, development, manufacturing and marketing of pharmaceutical products • Global consolidation; – economies of scale, eliminating redundancies, reducing costs, streamlining operations, garnering larger shares of emerging markets, and monopolizing intellectual property. • Bio-integration; – growing potential for natural and/or engineered biological systems (e. g. , botanical, microbial, mammalian cell cultures, etc. ) to produce economic (large-scale, low-cost) quantities of active pharmaceutical ingredients or their intermediates – particularly (though not exclusively) novel targets and/or 53 peculiar disease states.

Key determining factors Impact on future structure • Managed care, formularies, and the worldwide trend toward socialized medicine • Growth of generics • Social media and better brand management • Availability of capital • Better drug delivery 54

Key determining factors Impact on future structure • biotechnology • economic geography • improved chemical engineering, industrial processes, and better yields • new forms of leadership, and superior managerial ability • patent and tax reform, other legal inducements or obstacles, and moral impediments 55

Role of the Bio. Pharma Executive Impact of structure on planning needs & functions • need for generalists; broad backgrounds and experiences to understand – Manage growing “cat-like” herds of specialization and entrepreneurship that will continue to characterize the industry • better methods of monitoring, analyzing, and interpreting emergent and potential new innovations • increased quality of communication and paradigm sharing among firms and operating units – represent various areas of specialization within the organization or channel of distribution • better methods of conceptualizing and operationalizing the consolidation and integration of discovery, development, manufacturing, and marketing processes in 56 order to minimize costs

Organizational Considerations • Con$olidation of the Pharma / Biotech industry • M & A • How big is too big? • Break up larger organizations? Eg. Pfizer CEO comment • To Outsource / Offshore or Not ? MARENON HEALTH GROUP 3/3/2021

Healthcare Trends • ‘Personalized medicine’ – Reflect clinical response to treatment e. g. Imaging, serum or genetic assays, or psychological tests – Surrogate endpoints – optimize use of biomarkers to predict clinical benefit • At-risk populations eg. Pediatrics MARENON HEALTH GROUP 3/3/2021

Public-Private Initiatives Innovators and Leaders • Develop novel tools • health-care delivery systems, regulatory support and private sector collaboration in the 21 st century • Automating and streamlining clinical trials • Standardizing terminology • Developing data standards • Technology (eg. tablets computers - patients) • EHR – EDC (a possible new paradigm) • Accelerate regulatory Approval eg. Canada Infoway MARENON HEALTH GROUP 3/3/2021

Drug Development Innovation • Improved animal models • New biomarkers • Model-based drug development • Opportunities for genomics, proteomics 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021

Drug Development Innovation • Data libraries • Harnessing bioinformatics / data mining • Better evaluation tools eg. EHR in Edmonton, Canada Infoway • Manufacturing processes (21 st century) 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021

Impact – Short-term • Post-approval studies will grow in importance • Conduct real-world studies to validate biomarkers / surrogate endpoints using existing drugs • Proliferation of patient registries and long- term outcomes studies 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021

Challenges to Initiative • Largely dependent on industry willingness to fund research, share data • Need greater collaboration among government, academia and industry, and between competitors • Greate clarity on questions of privacy and proprietary information 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021

Contract Research Organizations Need to be Innovators and Leaders • Embrace technology (e. g. EDC) • Cross-pollinate - bring range of experiences to the table • Identify efficiencies, reduce waste – Data management automation (market research, clinical trials, manufacturing, discovery, etc. ) • Develop novel tools – Patient registries, observational studies – Social media and clinical trials – *Insert Other Novel Tools Here* 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021

Regulatory Agency Perspective on Scientific & Clinical Considerations Successful Drug Development Use each new knowledge acquired as a building block for the next step Scientific • Is this product entirely novel? - Consider preclinical data and appropriateness of model to the human situation - Exercise maximal caution for "first in man": go slow, . . . • What if the product is not novel? - Apply class properties Clinical • How to apply lessons learned from animals and animal models (consider their limitations) 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021 Agnes V. Klein, Health Canada 2007

Regulatory Agency Perspective on Conclusion Successful Drug Development • Ongoing consultation with regulator needed • Consult as often as necessary and ask questions • Go slow to arrive fast!! 3/3/2021 March 6, 2007 MARENON HEALTH GROUP 3/3/2021 Agnes V. Klein, Health Canada 2007
• • • It's that time of year again; time to visit the biotech graveyard to reflect on the developers whose businesses have in one way or another failed. There's a common theme among many of this year's graveyard companies: most of their problems started in 2009, which was one of the toughest years on record for biotech. (It was also the first year of the biotech graveyard; we said farewell to a record 16 developers that year). Many of these companies are reminders of how difficult the biotech game is; one failure in the clinic nowadays can ruin a small drug developer, and limited supplies of capital have made some investors extremely conservative with their bets on teams that have faltered with previous R&D programs. Yet what makes this industry difficult also makes it great; success in biotech beats the odds and can improve the lives of patients. Advanced Life Sciences Altair Therapeutics Ambrilia Biopharma ARYx Therapeutics Peptimmune Phenomix Corporation Tolerx Transdel Read more: The 2011 Biotech Graveyard - Fierce. Biotech http: //www. fiercebiotech. com/special. Presentation to University of Alberta March 18, 2009 reports/2011 -biotech-graveyard#ixzz 1 c. Ok 9 Zb 7 m Subscribe: http: //www. fiercebiotech. com/signup? sourceform=Viral-Tynt-Fierce. Biotech-

Issues for Discussion • How to Drive Innovation when Safety Comes First? • How to Improve Competitiveness? – New business models & processes – Canadian Bio. Pharma Industry • Show me the money! – Where in the world is the investment capital? MARENON HEALTH GROUP 3/3/2021

Marenon Consulting Myron Pyzyk Principal Consultant & CEO 2098 Mississauga Rd Mississauga, ON Canada L 5 H 2 K 6 tel 416 -571 -8504 fax 905 -891 -8504 e-mail: "Myron Pyzyk" <myron@marenon. com> website: http: //marenon. com
- Slides: 69