Bio Boot Camp Biochemistry TOPIC 1 Bio A

Bio Boot Camp Biochemistry: TOPIC 1

Bio A. 2 The Chemical Basis for Life • Describe how the unique properties of water support life on Earth (freezing point, high specific heat, cohesion) • Describe and interpret relationships between structure and function at various levels of biochemical organization (atoms, molecules, macromolecules) – Explain how C is uniquely suited to form biological macromolecules – Describe how macromolecules form from monomers – Compare the structure and function of carbs, lipids, proteins and nucleic acids

Bio A. 2 The Chemical Basis for Life • Explain how enzymes regulate biochemical reactions within a cell – Describe the role of an enzyme as a catalyst in regulating a specific biochemical reaction – Explain how factors such as p. H, temp, and concentration can affect enzyme function
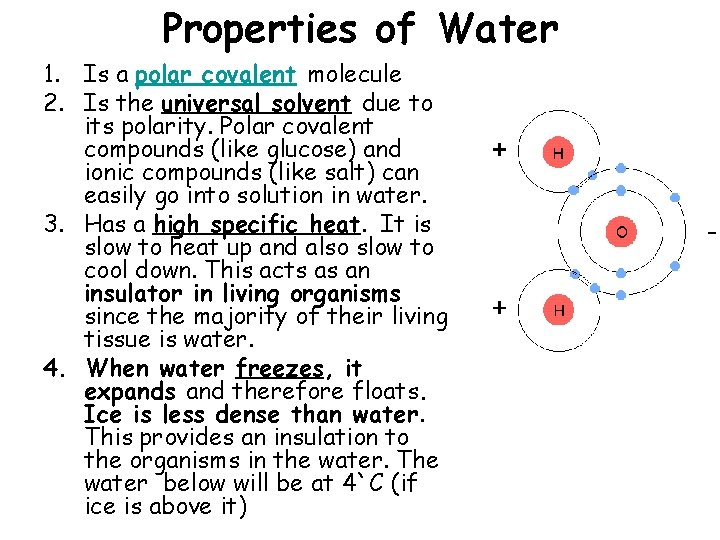
Properties of Water 1. Is a polar covalent molecule 2. Is the universal solvent due to its polarity. Polar covalent compounds (like glucose) and ionic compounds (like salt) can easily go into solution in water. 3. Has a high specific heat. It is slow to heat up and also slow to cool down. This acts as an insulator in living organisms since the majority of their living tissue is water. 4. When water freezes, it expands and therefore floats. Ice is less dense than water. This provides an insulation to the organisms in the water. The water below will be at 4`C (if ice is above it)

Water – Unique Properties • Adhesion: Water sticks to other substances • Cohesion: water sticks to water • Capillary actions: Water climbs up. – Ex. Plant roots to 400 feet up. • Surface Tension: Water spiders can walk on water. Water sticks together so tightly, causes a tension so certain things can “float”

Water • The cohesive forces between liquid molecules are responsible for the phenomenon known as surface tension. The molecules at the surface do not have other like molecules on all sides of them and consequently they cohere more strongly to those directly associated with them on the surface. This forms a surface "film" which makes it more difficult to move an object through the surface than to move it when it is completely submersed.

Facts about water • Water resists changes to temperature. • Water expands when it freezes • Water is less dense as a solid oppose to a liquid. Ice floats • High Specific Heat: able to absorb a lot of heat before it gets hot • Universal Solvent

Inorganic • Water is an inorganic molecule: chemical compounds that do not contain carbon as the principle element (exception CO 2)
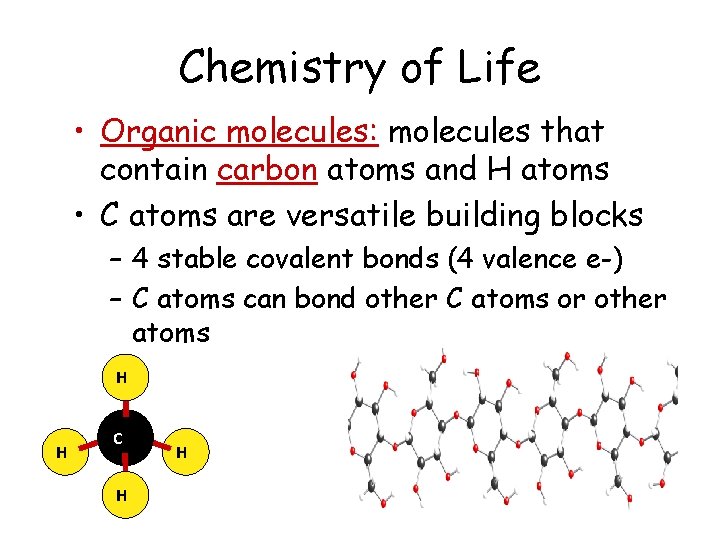
Chemistry of Life • Organic molecules: molecules that contain carbon atoms and H atoms • C atoms are versatile building blocks – 4 stable covalent bonds (4 valence e-) – C atoms can bond other C atoms or other atoms H H C H H
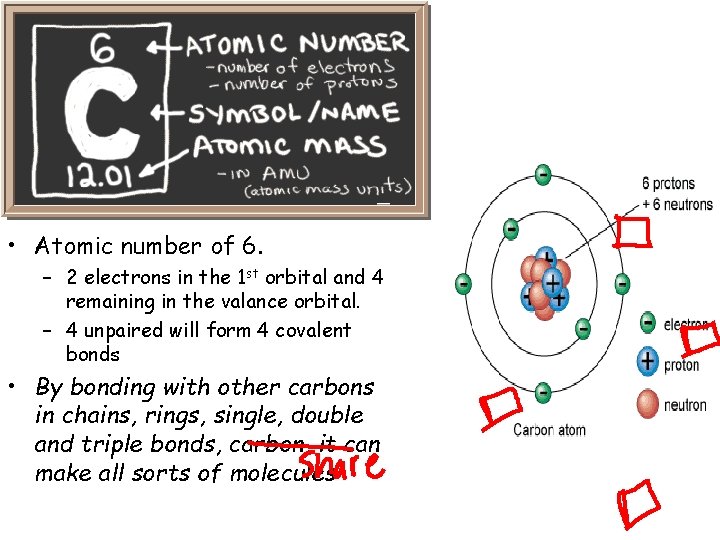
Carbon • Atomic number of 6. – 2 electrons in the 1 st orbital and 4 remaining in the valance orbital. – 4 unpaired will form 4 covalent bonds • By bonding with other carbons in chains, rings, single, double and triple bonds, carbon, it can make all sorts of molecules
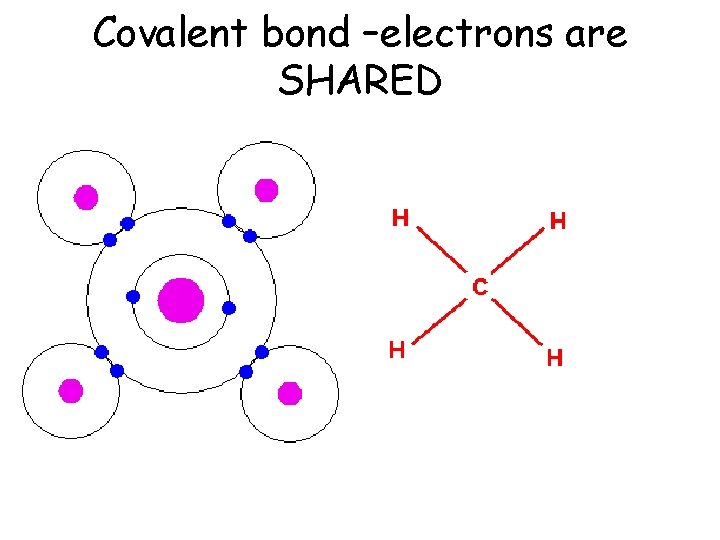
Covalent bond –electrons are SHARED
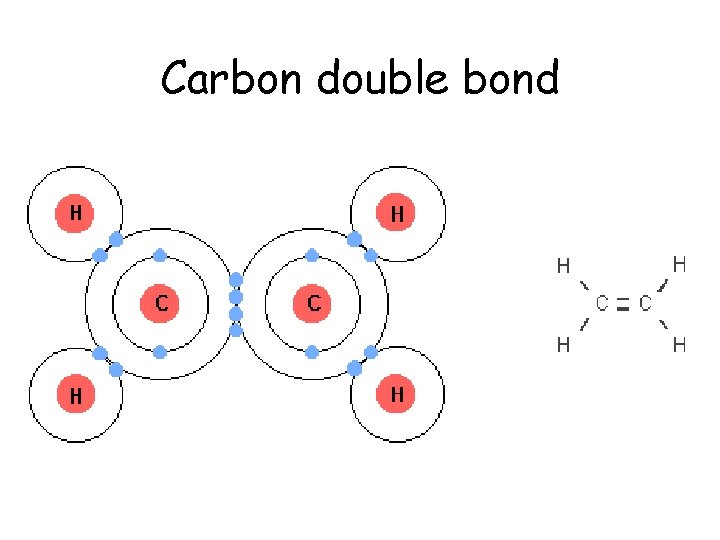
Carbon double bond
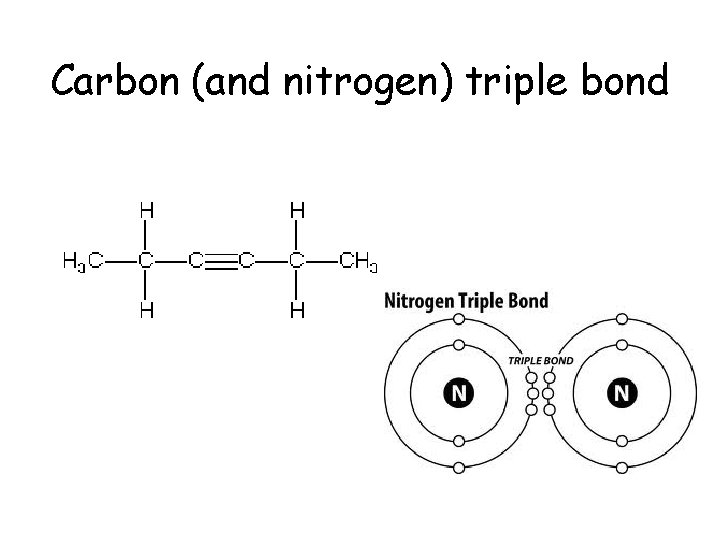
Carbon (and nitrogen) triple bond
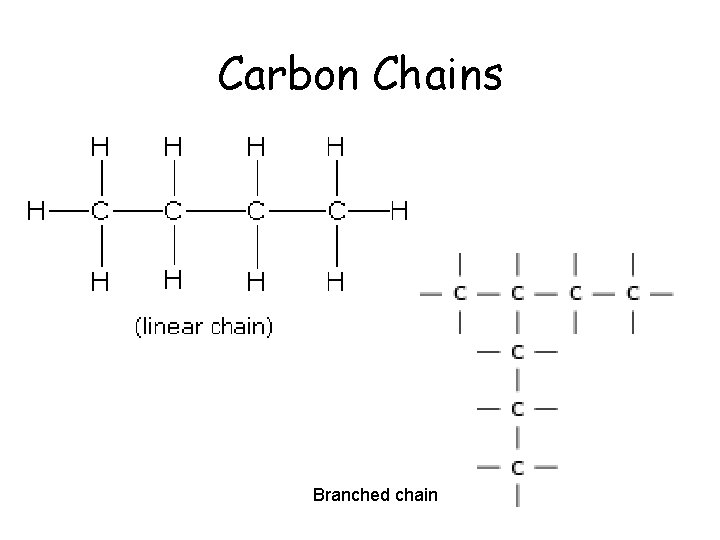
Carbon Chains Branched chain
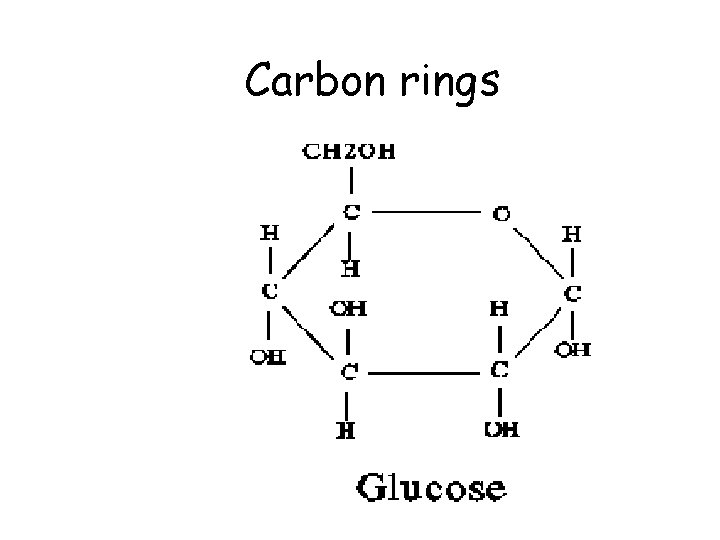
Carbon rings
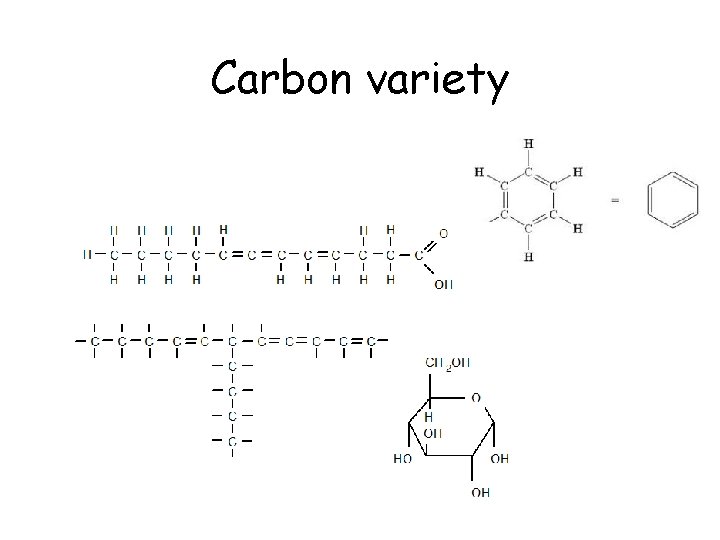
Carbon variety
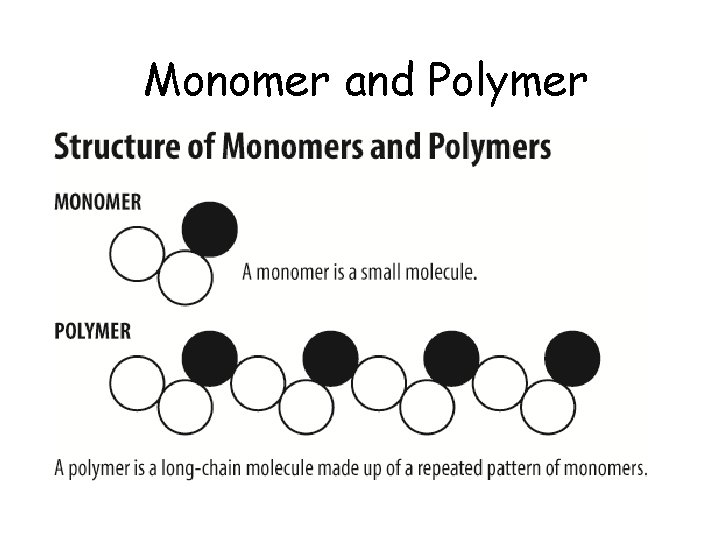
Monomer and Polymer

Monomers are like legos… • You can put them together in whatever way you dream up!

Macromolecules • Smaller organic molecules join together by covalent bonds to form larger molecules – built by linking repeating building blocks in a chain • 4 major classes of macromolecules: – – carbohydrates lipids proteins nucleic acids
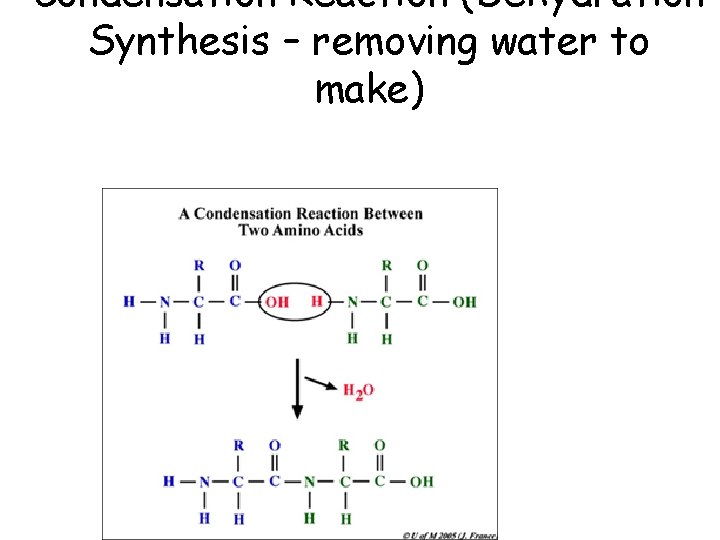
Condensation Reaction (Dehydration Synthesis – removing water to make)
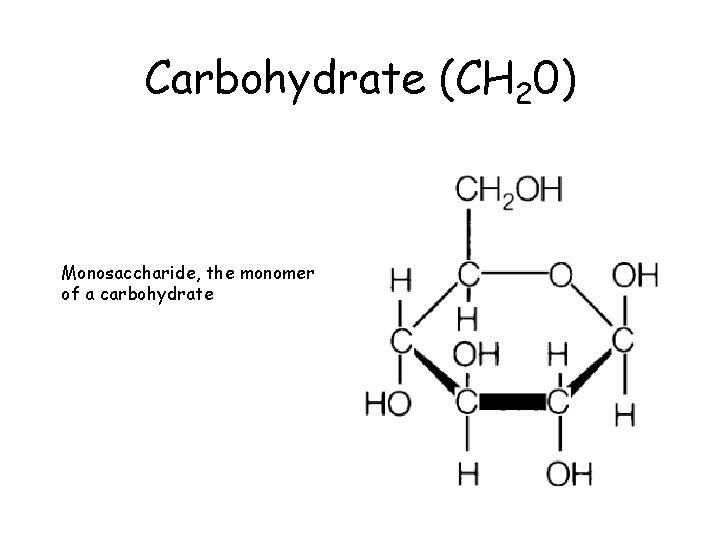
Carbohydrate (CH 20) Monosaccharide, the monomer of a carbohydrate
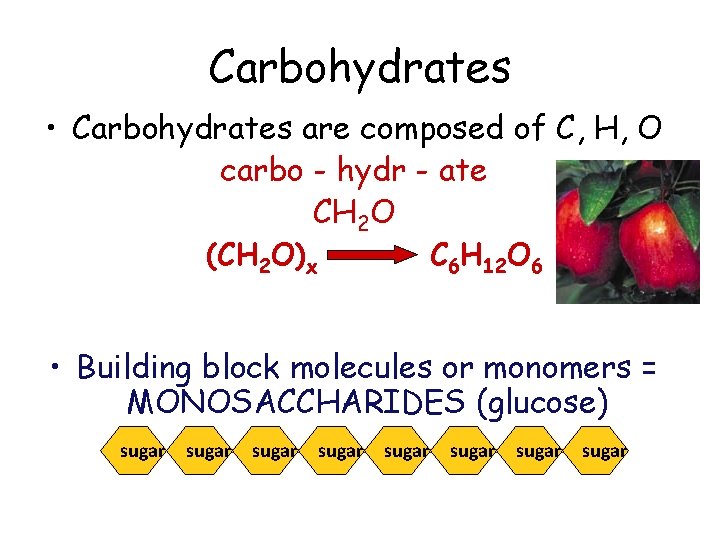
Carbohydrates • Carbohydrates are composed of C, H, O carbo - hydr - ate CH 2 O (CH H 12 O 66 (CH CC 66 H 2 O) 12 O 2 O) x x • Building block molecules or monomers = MONOSACCHARIDES (glucose) sugar sugar
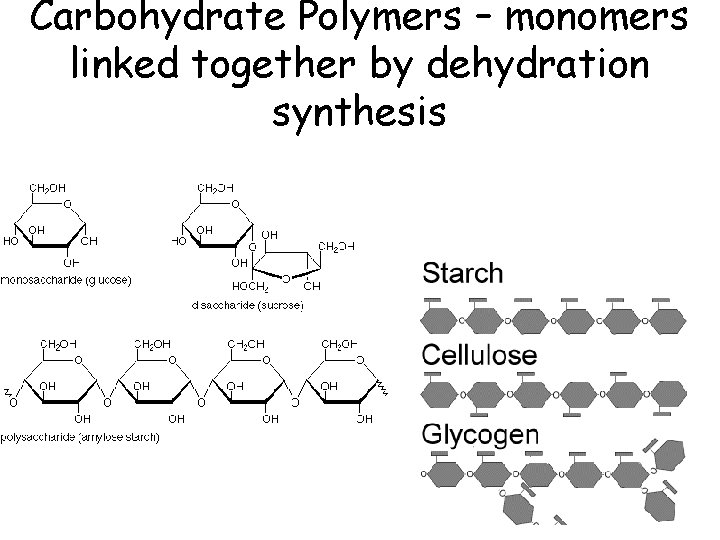
Carbohydrate Polymers – monomers linked together by dehydration synthesis

Functions of Carbohydrates • Monosaccharides – Glucose: quick energy • Polysaccharides – starch • energy storage in plants – glycogen • energy storage in animals – in liver & muscles – cellulose • structure in plants – cell walls
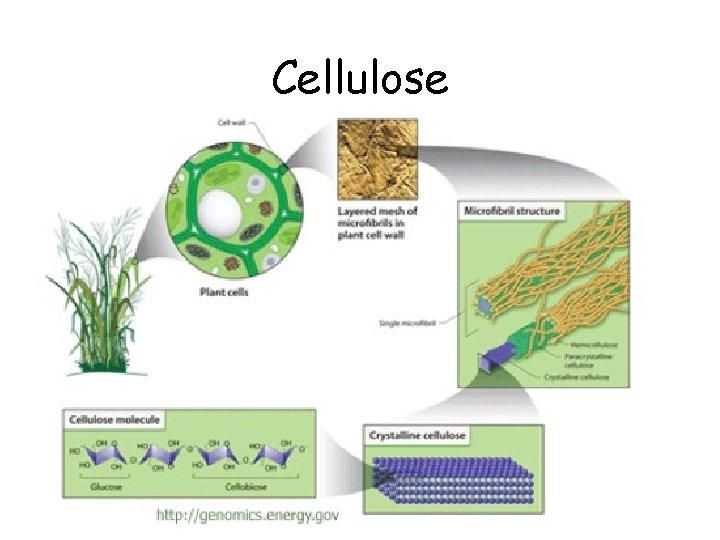
Cellulose
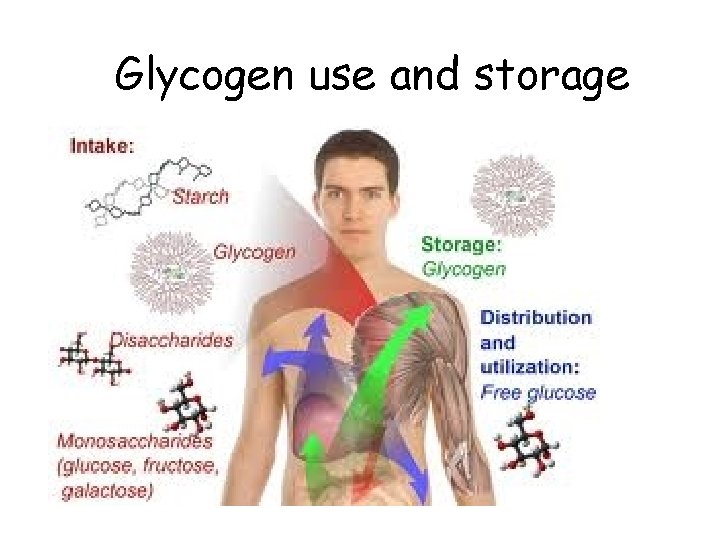
Glycogen use and storage
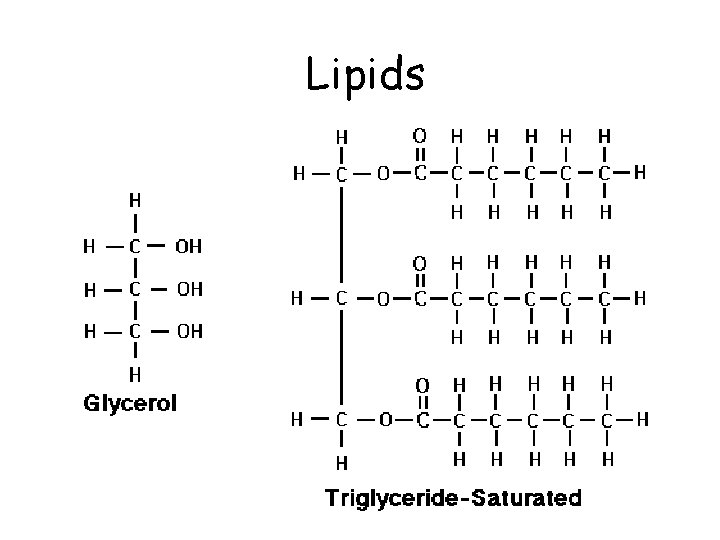
Lipids
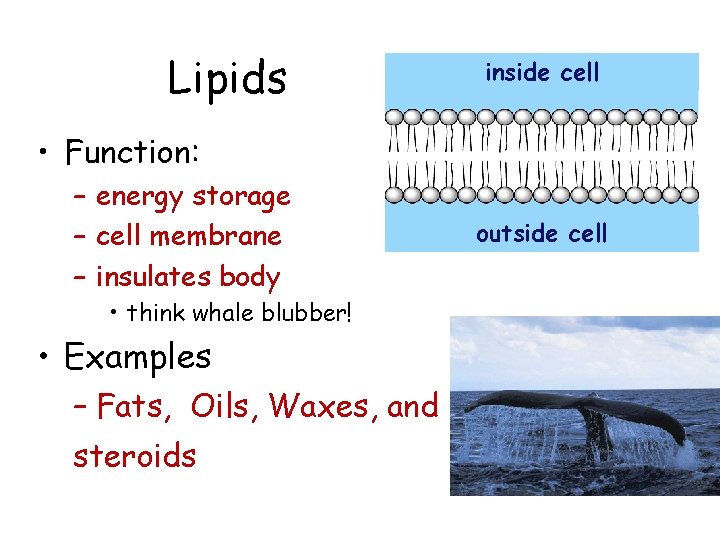
Lipids inside cell • Function: – energy storage – cell membrane – insulates body • think whale blubber! • Examples – Fats, Oils, Waxes, and steroids outside cell
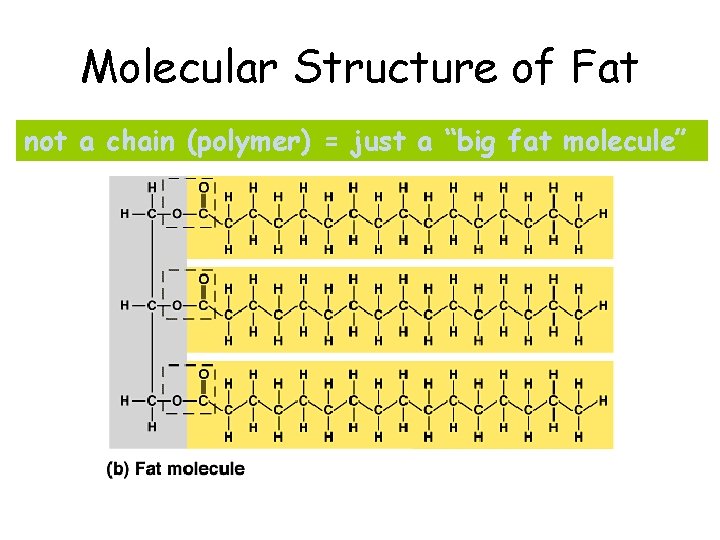
Molecular Structure of Fat not a chain (polymer) = just a “big fat molecule”
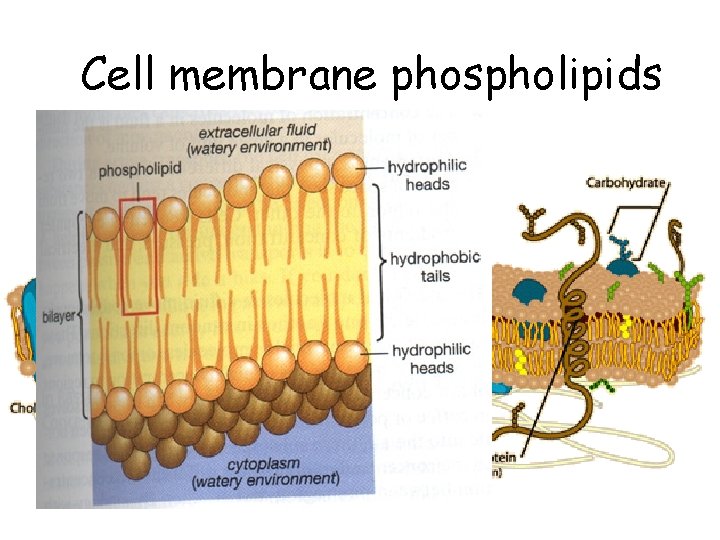
Cell membrane phospholipids
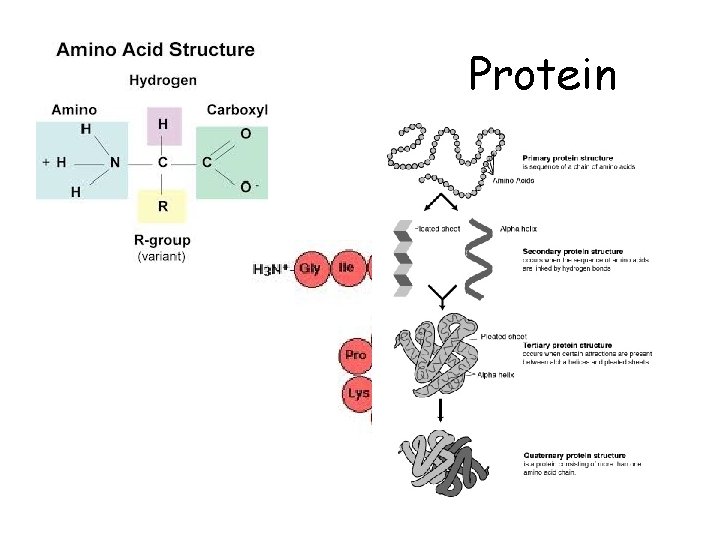
Protein
Proteins • Function: worker molecules – many, many functions • hormones – signals from one body system to another – insulin • movement – muscle • enzymes – help chemical reactions
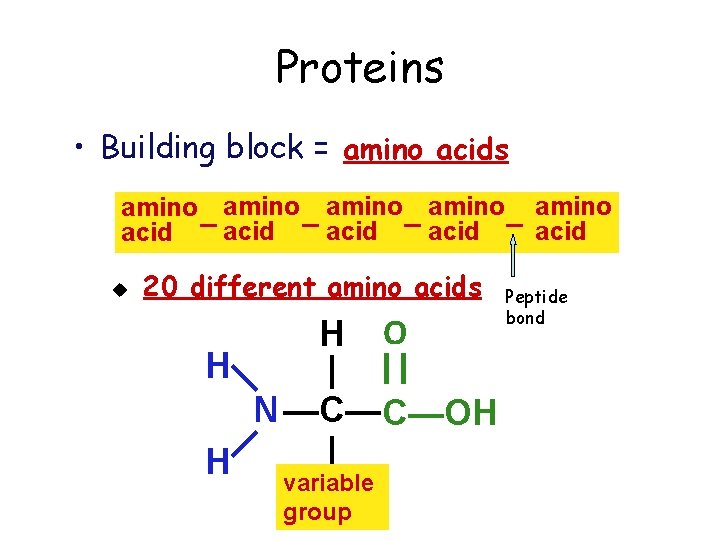
Proteins • Building block = amino acids amino amino acid – acid u 20 different amino acids H O H | || —N— —C—C—OH | H variable group Peptide bond
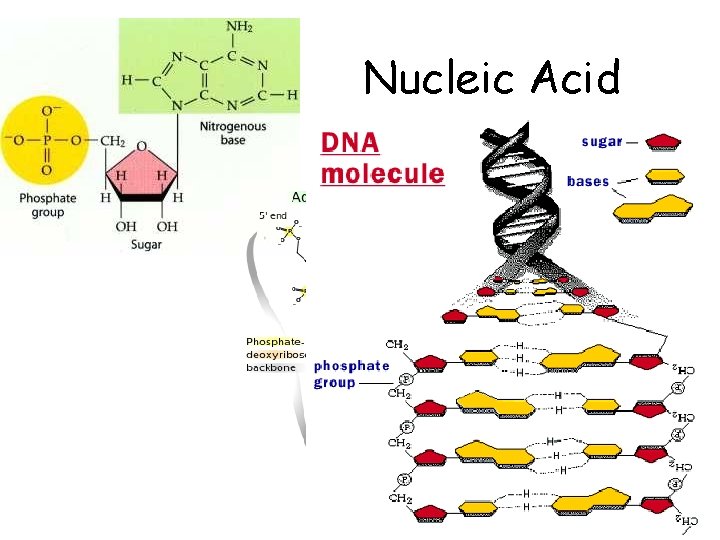
Nucleic Acid
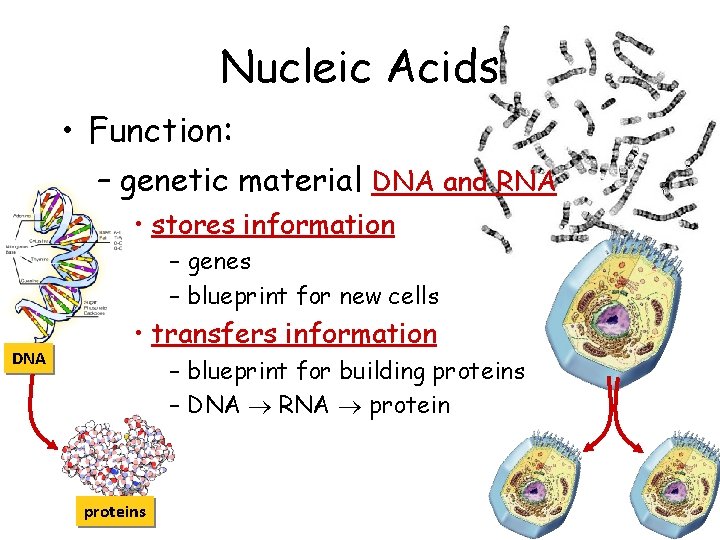
Nucleic Acids • Function: – genetic material DNA and RNA • stores information – genes – blueprint for new cells DNA • transfers information – blueprint for building proteins – DNA RNA proteins
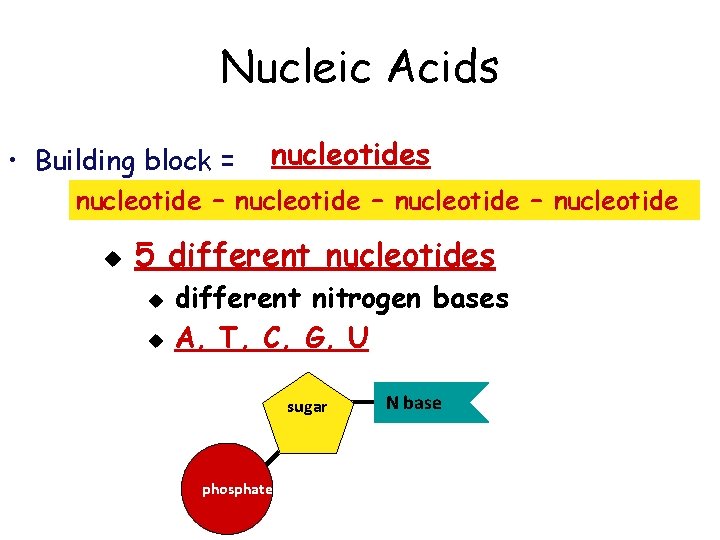
Nucleic Acids • Building block = nucleotides nucleotide – nucleotide u 5 different nucleotides u u different nitrogen bases A, T, C, G, U sugar phosphate N base
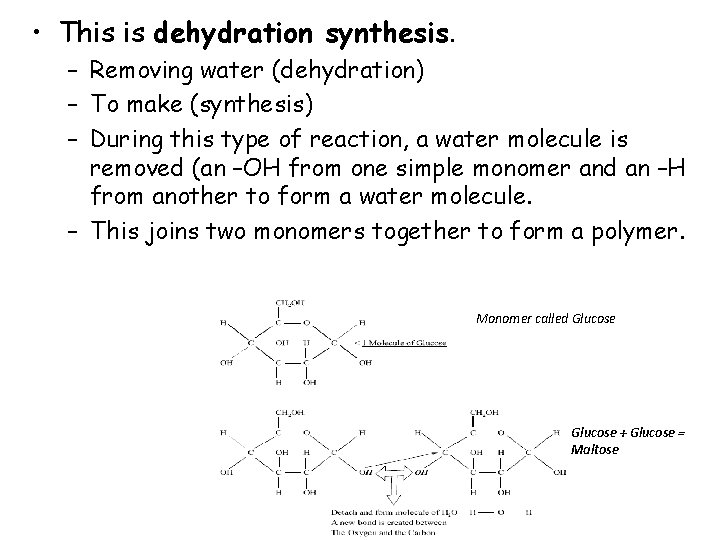
• This is dehydration synthesis. – Removing water (dehydration) – To make (synthesis) – During this type of reaction, a water molecule is removed (an –OH from one simple monomer and an –H from another to form a water molecule. – This joins two monomers together to form a polymer. Monomer called Glucose + Glucose = Maltose

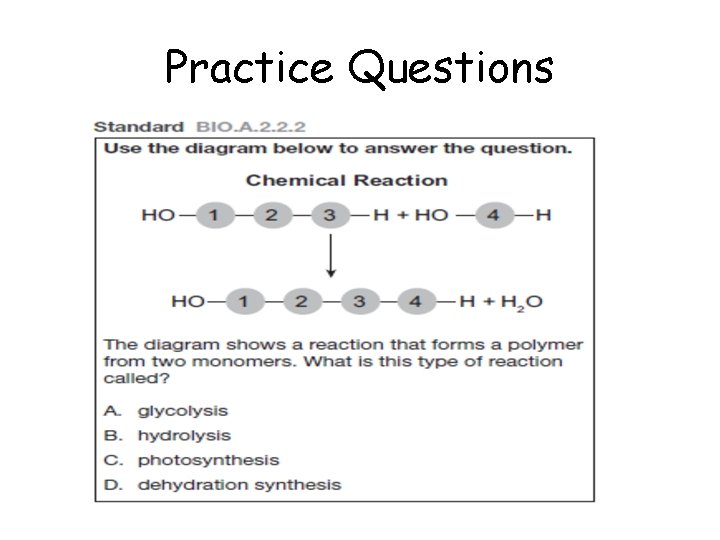
Use the diagram below to answer the question. Chemical Reaction HO – 1 – 2 – 3 – H + HO – 4 - H HO – 1 – 2 – 3 – 4 – H + H 2 O The diagram shows a reaction that forms a polymer from two monomers. What is this type of reaction called? A. B. C. D. Glycolysis Hydrolysis Photosynthesis Dehydration synthesis

Enzymes • Enzymes are not changed by the reaction – Enzymes are specific, they fit to their substrates like a lock and key – used only temporarily then reused – Provide a site for reaction (active site) substrate active site product enzyme
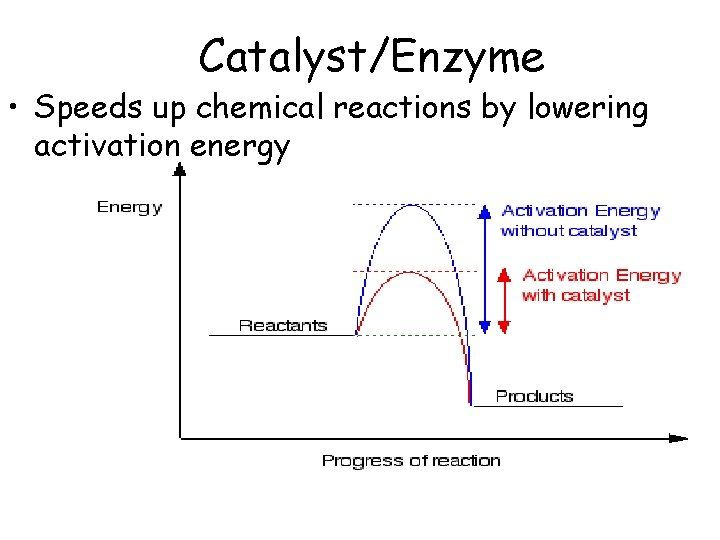
Catalyst/Enzyme • Speeds up chemical reactions by lowering activation energy
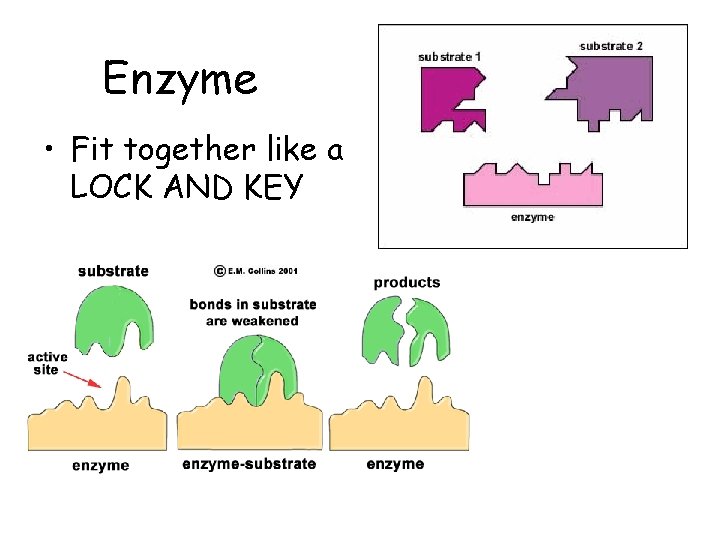
Enzyme • Fit together like a LOCK AND KEY
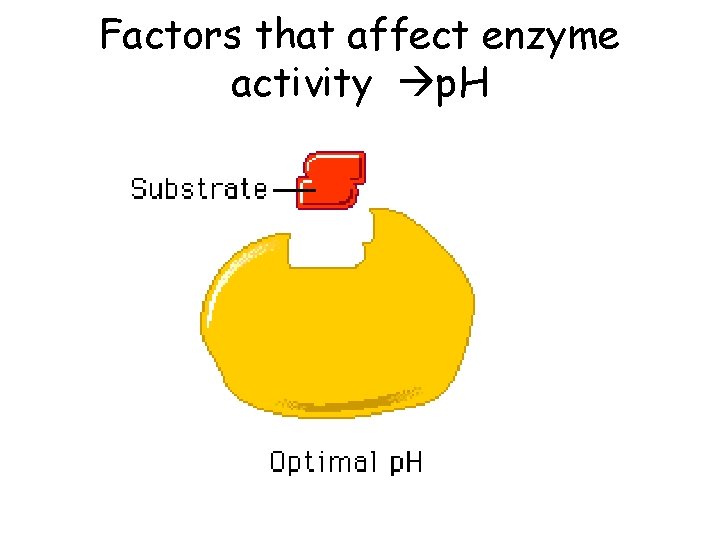
Factors that affect enzyme activity p. H
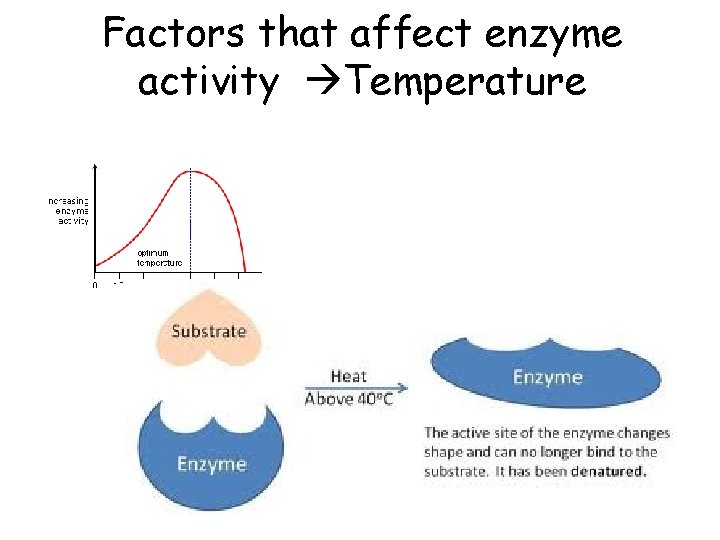
Factors that affect enzyme activity Temperature
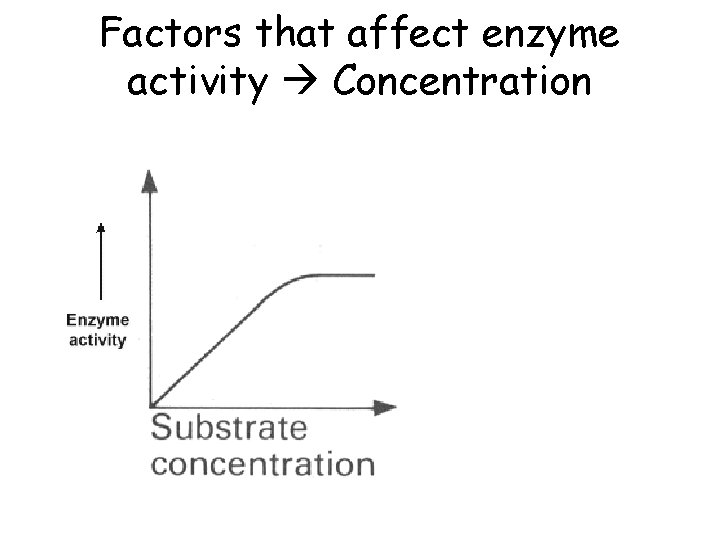
Factors that affect enzyme activity Concentration

Two molecules join together to make a macromolecule with the removal of water. This type of reaction is called? A. Glycolysis B. Hydrolysis C. Photosynthesis D. Dehydration synthesis.

A scientist observed that, when the p. H of the environment surrounding an enzyme is changed the rate the enzyme catalyzes a reaction greatly decreases. Which statement best describes how a change in p. H can affect an enzyme? A. A p. H change can cause the enzyme to change its shape B. A p. H change can remove energy necessary to activate an enzyme C. A p. H change can add new molecules to the structures of the enzyme D. A p. H change can cause an enzyme to react with a different substrate.

Substance A is converted to substance B in a metabolic reaction. Which statement best describes the role of an enzyme during this reaction? A. It adjusts the p. H of the reaction medium B. It provides energy to carry out the reaction C. It dissolves substance A in the reaction medium D. It speeds up the reaction without being consumed.

Carbohydrates and proteins are two types of macromolecules. Which functional characteristic of proteins distinguished them from carbohydrates? A. Large amounts of stored information B. Ability to catalyze biochemical reactions C. Efficient storage of usable chemical energy D. Tendency to make cell membranes hydrophobic

Practice Questions

Practice Questions
Practice Questions

Practice Questions

Practice Questions
Practice Questions

Practice Questions

Practice Questions
- Slides: 56