Bio 260 summer 2016 Chapter 5 Metabolism Chapter

Bio 260 summer 2016 Chapter 5 Metabolism

Chapter 5 Metabolism: cellular life support

Human implications • Microbes in the lab – Our metabolism in an easier model • In the clinic – Differences exploited for drug design and treatment • In the wild – Making energy; recycling nutrients • Technology – Synthesis of useful products; degradation pollutants. • In the classroom – Ch 4: focused on structure that allows us to study, ID, kill – Now Ch 5: chemical reactions aka metabolism

Cell’s perspective • All cells need to accomplish two fundamental tasks • Synthesize new parts – Cell wall, membranes, ribosomes, proteins, nucleic acids • Harvest energy to power reactions – Make their own energy – Or use nutrients made by other organisms • Sum total of these is called metabolism – Catabolism – Anabolism

How do WE • Get Carbon? • Get energy? • Make stuff? • Do all organisms- eg. plants, fungi, other animals - do this the same way? • What about microbes? Why not?

Microbes – how they do it • Photoautotrophs (Photosynthetic – like plants…)) – Harvest energy in sunlight – Synthesize organic compounds from CO 2 – Convert kinetic energy of photons to potential energy of chemical bonds • Chemoorganoheterotrophs (like us) – Obtain energy by digesting organic compounds – sometimes to CO 2 and H 2 O – Dependent on autotrophic organisms • Lithoautotrophs (“rock eaters”) – Harvest energy in inorganic chemicals – Synthesize organic compounds from CO 2

Energy • What do you know? • What kinds of energy are there? • Kinetic (motion) – heat (a form of kinetic), • Potential (stored) – chemical ( a form of stored) • Can one turn into another? How?

Chemical energy • Free energy is energy available to do work – E. g. , energy released when chemical bond is broken – Compare free energy of reactants, products • Change in free energy is same regardless # of steps – (e. g. , converting glucose to CO 2 + H 2 O) – boulder analogy • Cells use multiple steps to degrade compounds – Energy released from degradation reactions powers cell

How do chemical reactions occur? • Collision theory – chemical reactions can occur when atoms, ions, and molecules collide • Activation energy – Energy needed to disrupt electronic configurations • Reaction rate – frequency of collisions with enough energy to bring about a reaction. – can be increased by enzymes or by increasing temperature or pressure • What happens in cells? – Normal rates are too slow to support life. – Increasing temperature ?
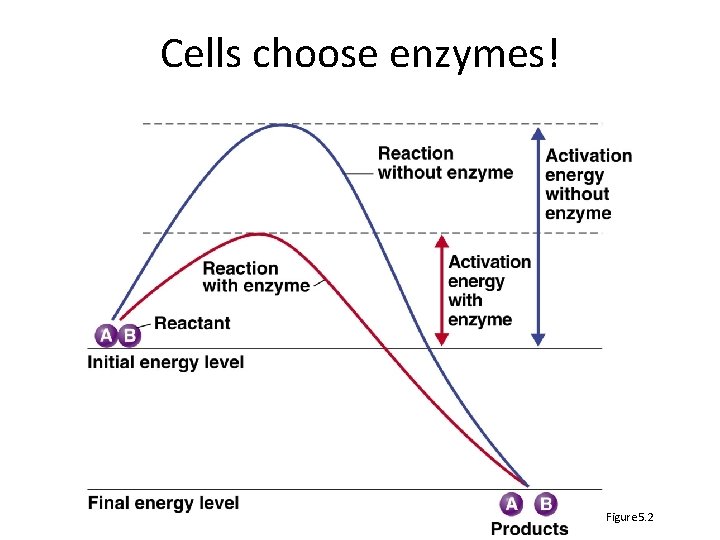
Cells choose enzymes! Figure 5. 2

One second on how enzymes work Bring things close together so they don’t need a lot of energy to collide - Whisper and Matchmaker analogies Figure 5. 2
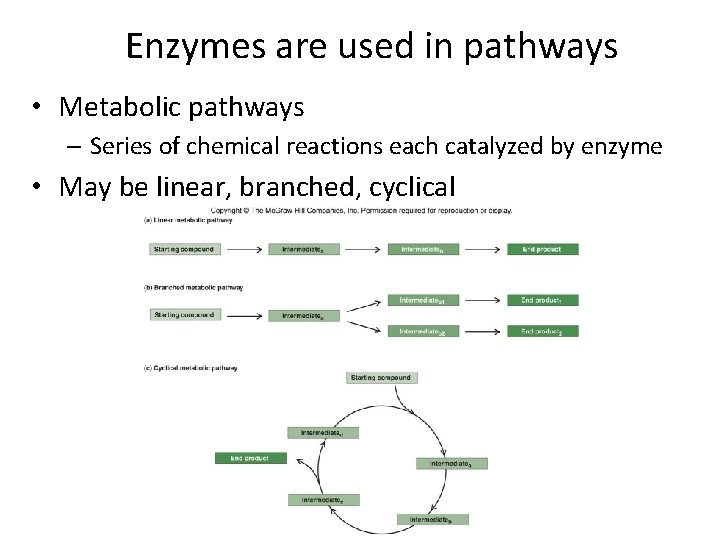
Enzymes are used in pathways • Metabolic pathways – Series of chemical reactions each catalyzed by enzyme • May be linear, branched, cyclical

Why use pathways? • Protection • Efficiency • Versatility

Enzyme features • Biological catalyst – Reduce energy of activation of reaction – Existing substrate bonds destabilized, new ones form – Not used up or altered in the reaction • Highly specific – Has active site to which substrate binds weakly – Causes enzyme shape to change slightly – Product no longer “fits” so is released
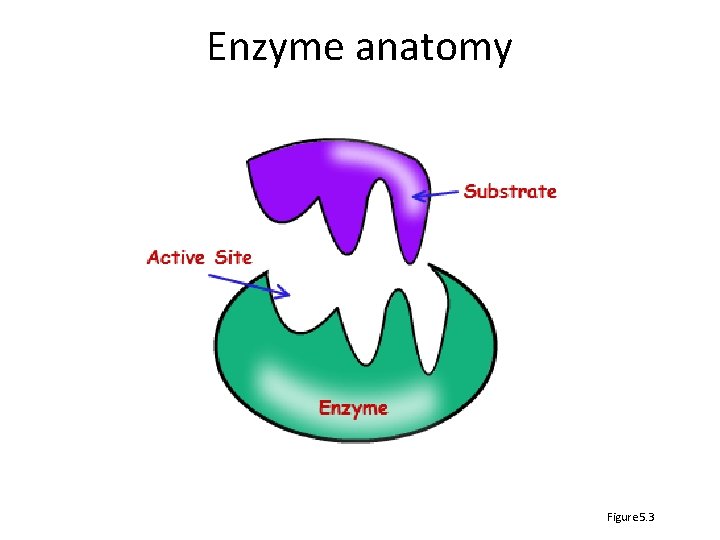
Enzyme anatomy Figure 5. 3
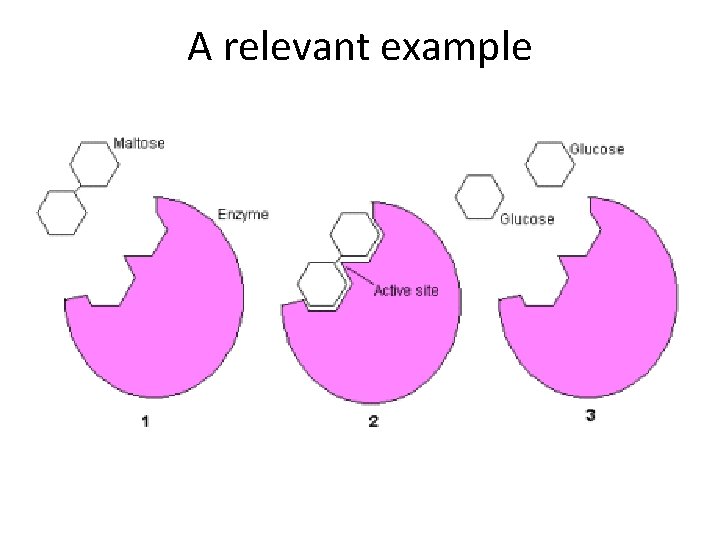
A relevant example
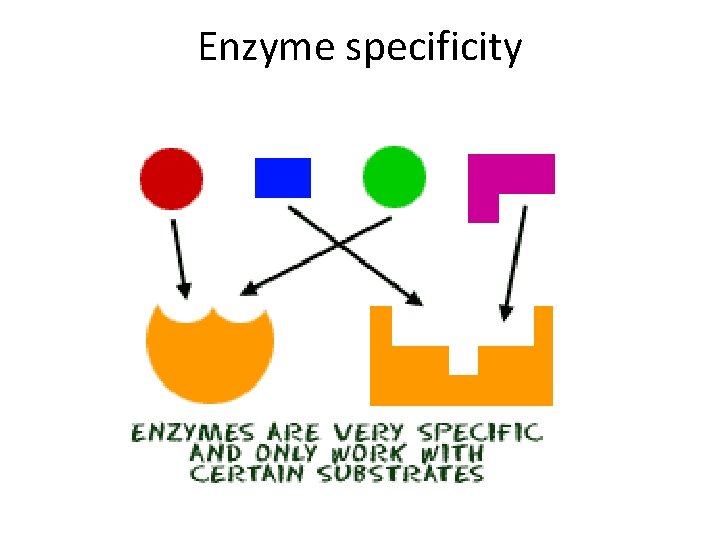
Enzyme specificity

Enzyme Types • • End in –ase: Oxidoreductase: oxidation-reduction reactions (redox) Transferase: Transfer functional groups Hydrolase: Hydrolysis Lyase: Removal of atoms without hydrolysis Isomerase: Rearrangement of atoms Ligase: Joining of molecules, uses ATP
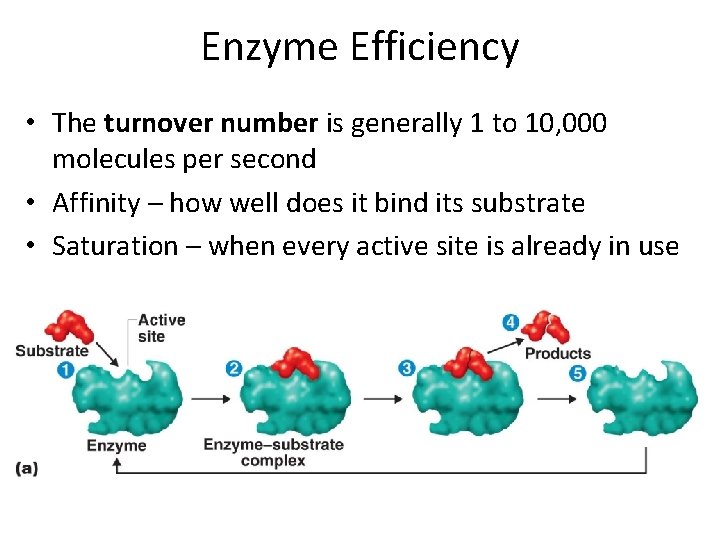
Enzyme Efficiency • The turnover number is generally 1 to 10, 000 molecules per second • Affinity – how well does it bind its substrate • Saturation – when every active site is already in use

Factors Influencing Enzyme Activity • What do you think (HINT: enzymes are proteins)
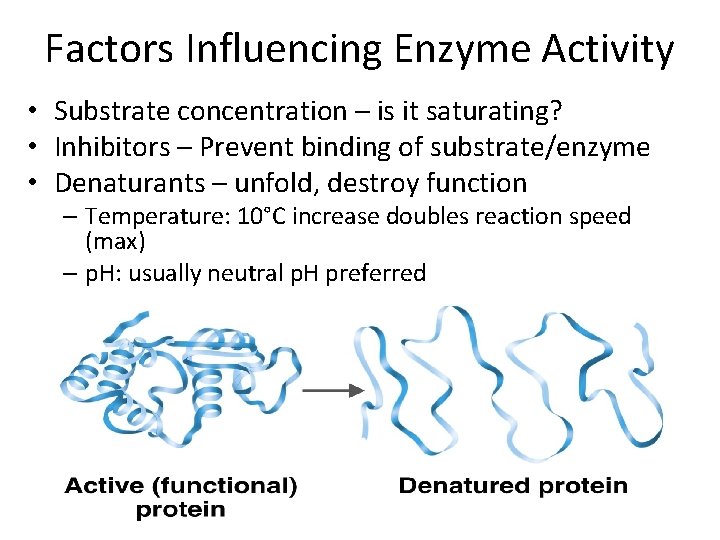
Factors Influencing Enzyme Activity • Substrate concentration – is it saturating? • Inhibitors – Prevent binding of substrate/enzyme • Denaturants – unfold, destroy function – Temperature: 10°C increase doubles reaction speed (max) – p. H: usually neutral p. H preferred
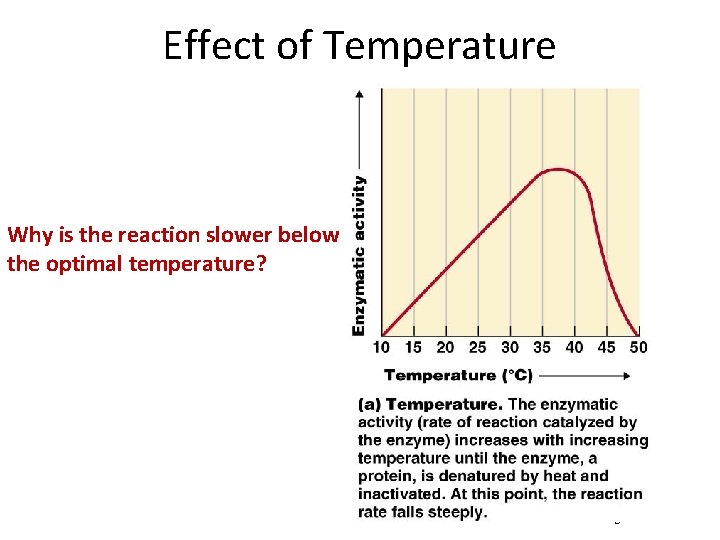
Effect of Temperature Why is the reaction slower below the optimal temperature? Figure 5. 5 a
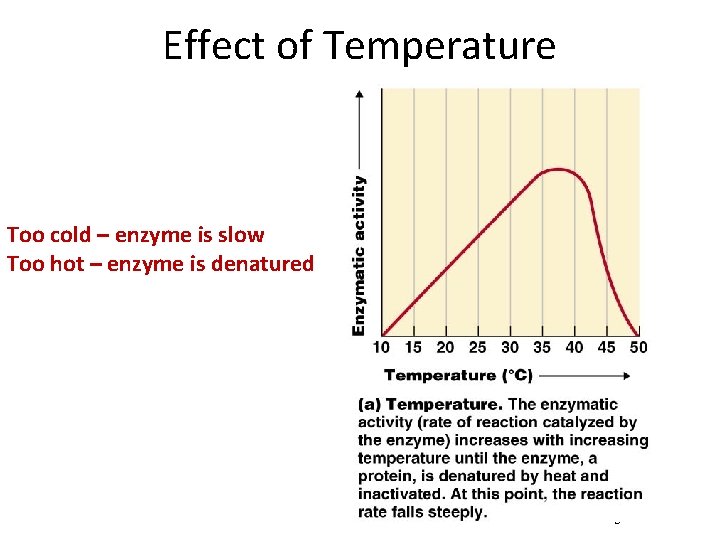
Effect of Temperature Too cold – enzyme is slow Too hot – enzyme is denatured Figure 5. 5 a
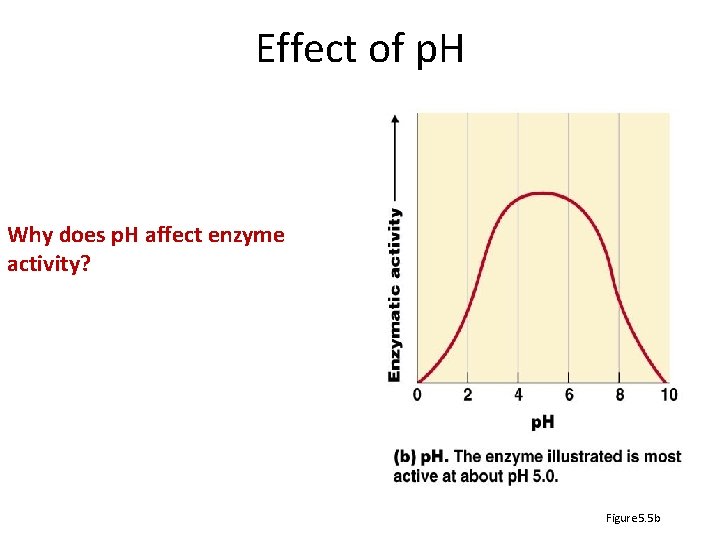
Effect of p. H Why does p. H affect enzyme activity? Figure 5. 5 b
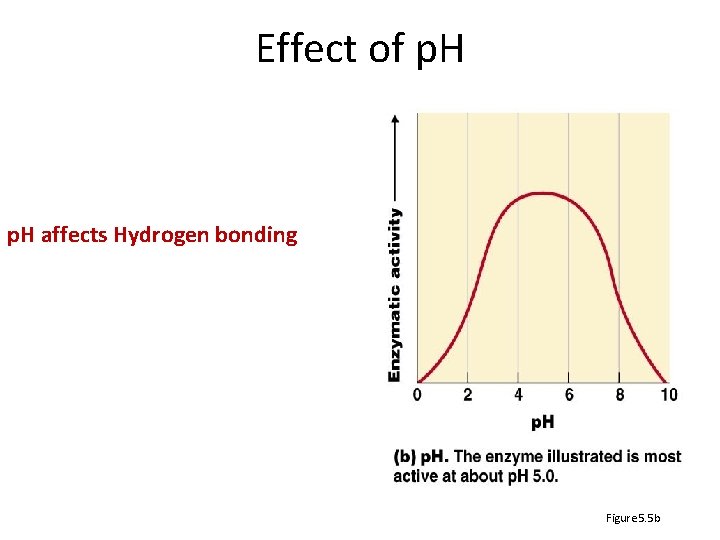
Effect of p. H affects Hydrogen bonding Figure 5. 5 b

Regulate enzymes
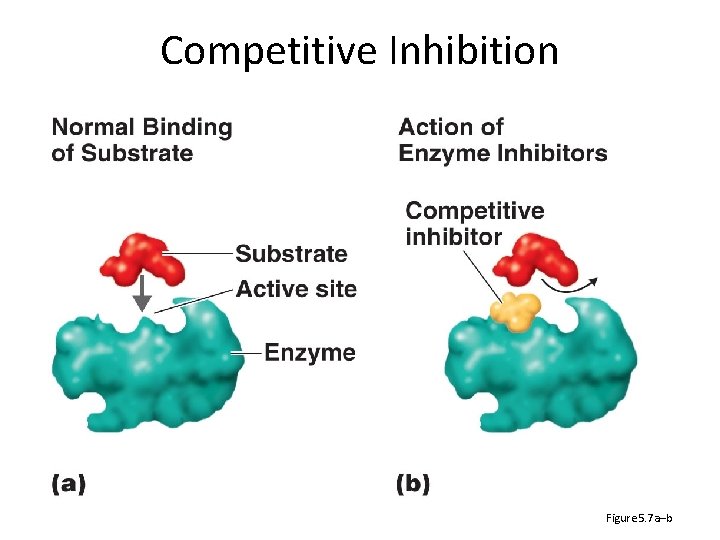
Competitive Inhibition Figure 5. 7 a–b
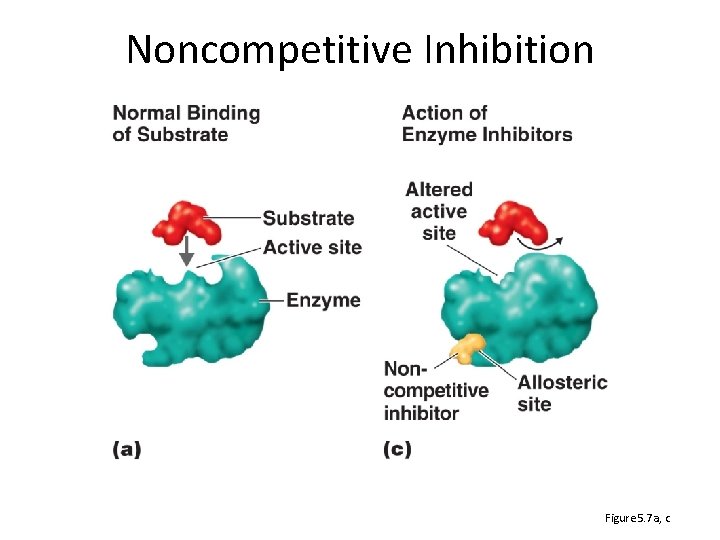
Noncompetitive Inhibition Figure 5. 7 a, c
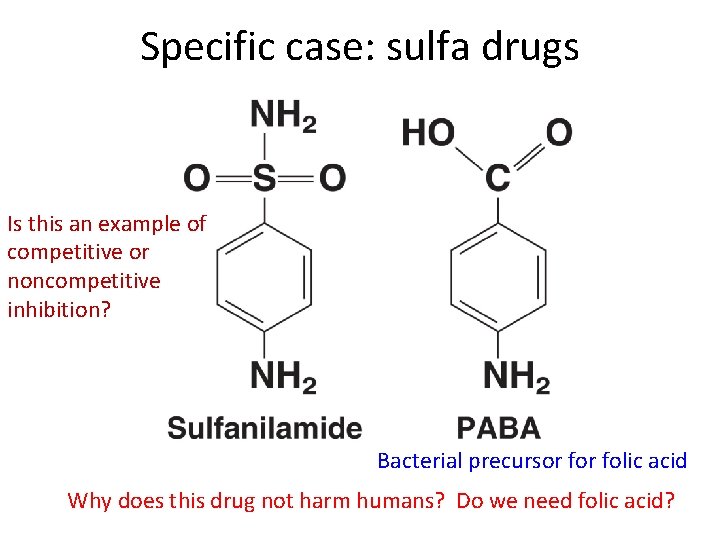
Specific case: sulfa drugs Is this an example of competitive or noncompetitive inhibition? Bacterial precursor folic acid Why does this drug not harm humans? Do we need folic acid?

Specific case: Feedback Inhibition QUESTION: Is feedback inhibition competitive or noncompetitive inhibition? Why is this useful/important? Figure 5. 8

Specific case: poisons • non-competitive inhibitors – may be reversible or irreversible • cellular inhibitors – generally reversible vs. • Examples of poisons often irreversible; may be used as antimicrobials – Arsenic • Disrupts ATP produciton – enzymes in TCA and oxphos; multiorgan failure – Cyanide • Inhibits cytochrome C oxidase in mitochondria (oxphos) – Mercury (methylmercury) • oxidizes the S-H groups of amino acid cysteine, cystine • No di-S bonds; enzyme shape change – Fluoride • Inhibits phosphatase (important for calcium metabolism) Figure 5. 8
Do enzymes require energy? • It depends on the reaction they are catalyzing

What energy form do they use? • Typically, they use ATP

Where do they get ATP? • Other reactions produce ATP….

Is this frustratingly circular? • Yes…
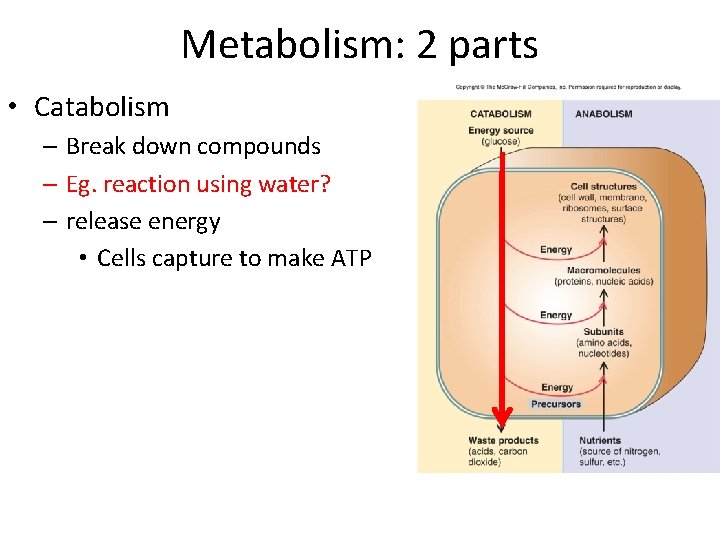
Metabolism: 2 parts • Catabolism – Break down compounds – Eg. reaction using water? – release energy • Cells capture to make ATP
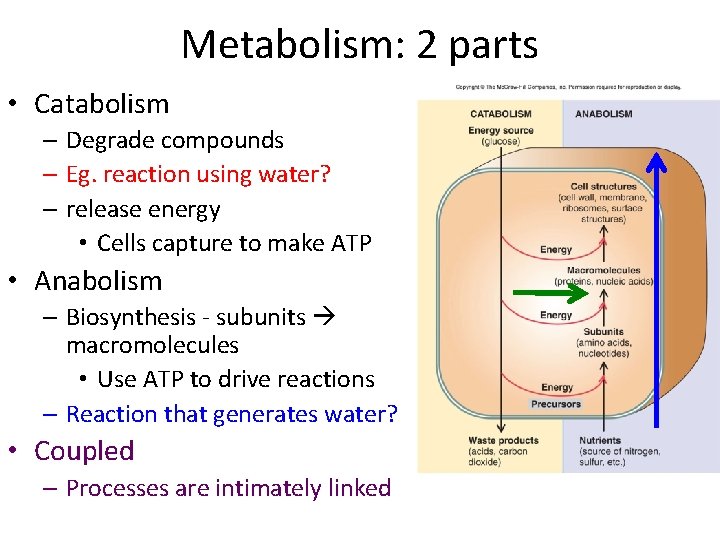
Metabolism: 2 parts • Catabolism – Degrade compounds – Eg. reaction using water? – release energy • Cells capture to make ATP • Anabolism – Biosynthesis - subunits macromolecules • Use ATP to drive reactions – Reaction that generates water? • Coupled – Processes are intimately linked
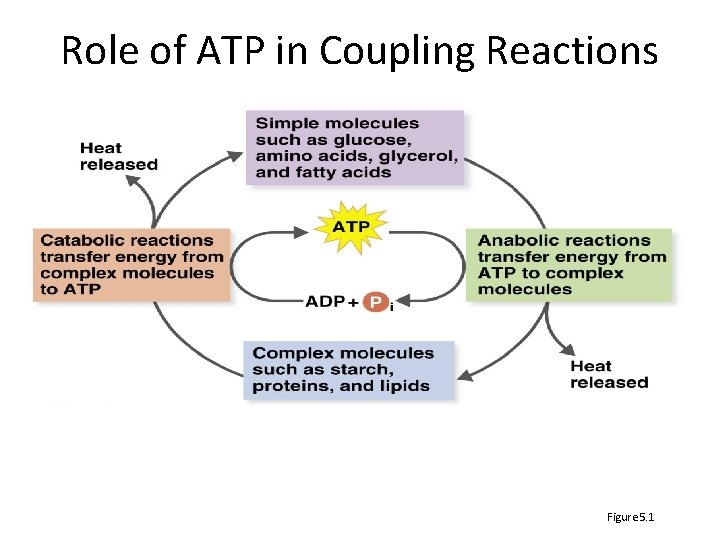
Role of ATP in Coupling Reactions Figure 5. 1

ATP drives cell metabolism • The Role of ATP – Adenosine triphospate (ATP) is energy currency • WHY IS ATP A GOOD ENERGY CURRENCY? • Three processes to generate ATP – Substrate-level phosphorylation – Oxidative phosphorylation – Photophosphorylation
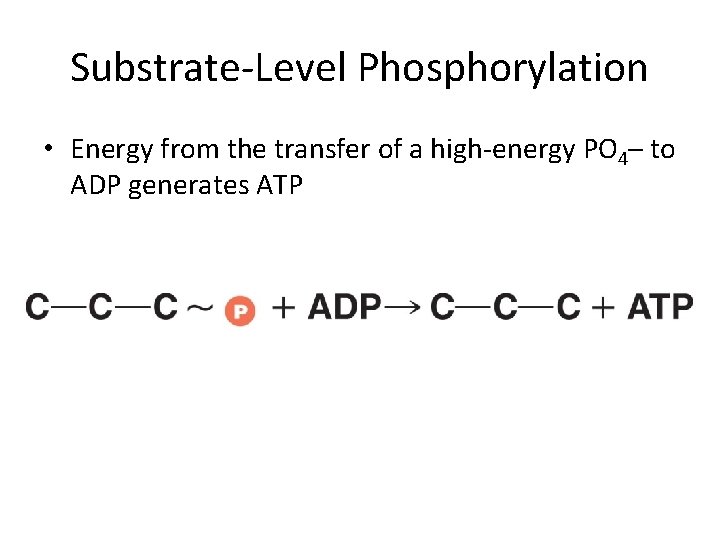
Substrate-Level Phosphorylation • Energy from the transfer of a high-energy PO 4– to ADP generates ATP

Oxidative Phosphorylation • Driven by “proton motive force” • Energy released from transfer of electrons (oxidation) of one compound to another (reduction) is used to move H+ ions via an “electron transport chain” • Energy released from allowing H+ ions to return is used to generate ATP in a protein turbine machine called ATP SYNTHAS

Photophosphorylation • Sunlight used to create “proton motive force” • Light energizes chlorophyll electrons. Transfer of high energy electrons (oxidation) via an “electron transport chain” drives H+ ion transport • Return of H+ ions releases energy ATP

Role of electrons in reaction • Electronegativity – Different atoms have different affinities for electrons – Which two atoms are highly e-negative (week 1)? • Energy released – When electrons move from low affinity molecule to high • (E. g. , glucose to O 2) – More energy released when difference in electronegativity is greater
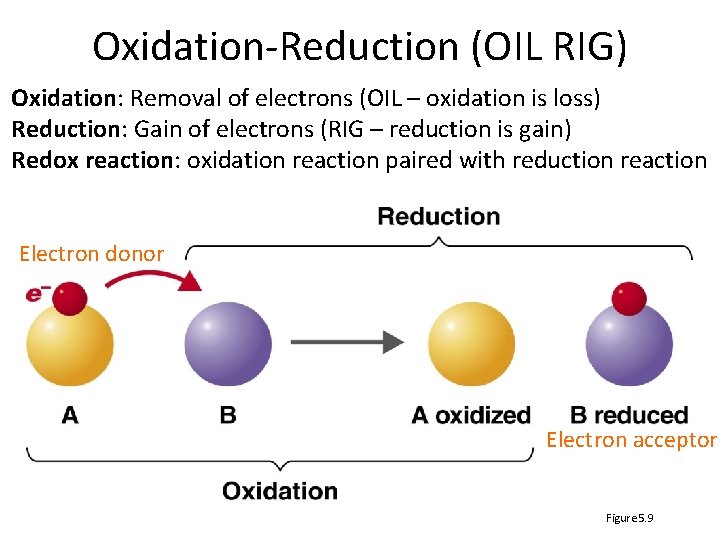
Oxidation-Reduction (OIL RIG) Oxidation: Removal of electrons (OIL – oxidation is loss) Reduction: Gain of electrons (RIG – reduction is gain) Redox reaction: oxidation reaction paired with reduction reaction Electron donor Electron acceptor Figure 5. 9

Terminal electron acceptor • Metabolic pathways – multi-step • Terminal electron acceptor – lowest energy • In eukaryotes and many organisms this is?

Prokaryotic diversity • Eukaryotic metabolism generally studied • Prokaryotes use diverse energy sources – Organic, inorganic compounds • And terminal electron acceptors – O 2, but also other molecules

Topics we’ll cover • • Major metabolic pathways of the cell Photosynthesis light and dark reactions Innovations and features in microbes - chemolithoautotrophy

Overview of Catabolism • Cellular respiration – Glycolysis – TCA (Krebs) cycle – Electron transport chain • Key outcomes – ATP – Precursor metabolites

Respiration glucose electron carriers electron transport chain aerobic respiration: O 2 is terminal electron acceptor anaerobic respiration molecule other than O 2 modified version of TCA/Krebs cycle

Fermentation • If cells cannot respire, will run out of electron carriers • Glycolysis will stop • Fermentation uses pyruvate or derivative as terminal electron acceptor to regenerate NAD+ • Glycolysis can continue

Carbohydrate Catabolism if you’ve got sugar, use it • The breakdown of carbohydrates to release energy – Glycolysis – Krebs cycle – Electron transport chain
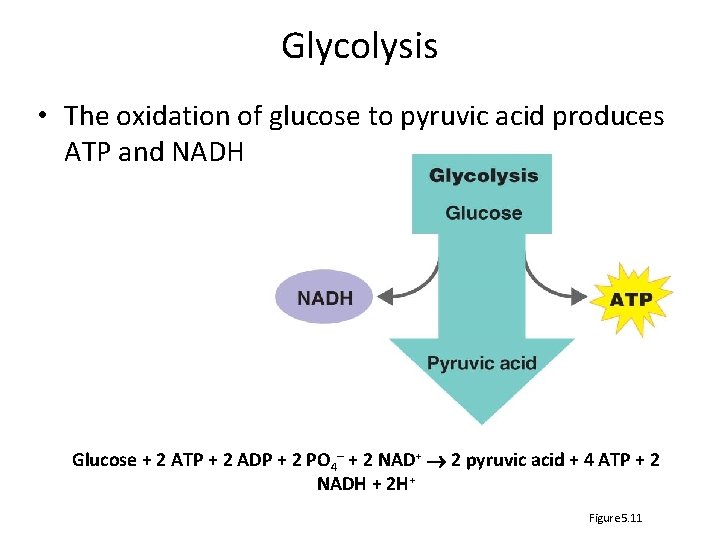
Glycolysis • The oxidation of glucose to pyruvic acid produces ATP and NADH Glucose + 2 ATP + 2 ADP + 2 PO 4– + 2 NAD+ 2 pyruvic acid + 4 ATP + 2 NADH + 2 H+ Figure 5. 11
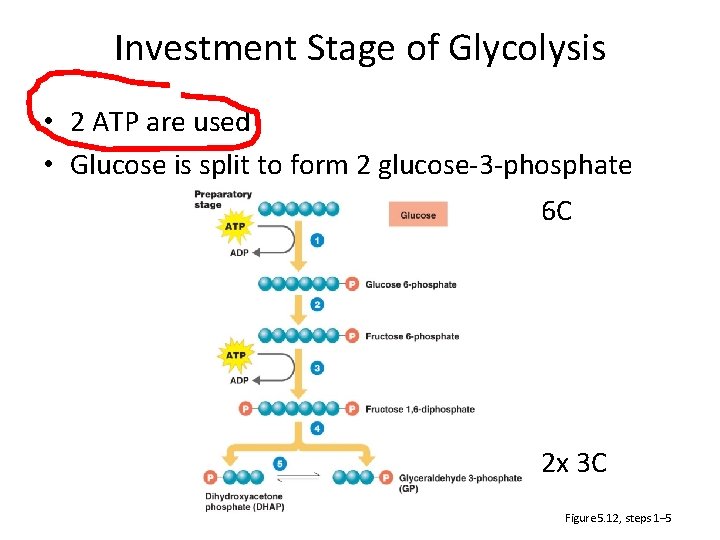
Investment Stage of Glycolysis • 2 ATP are used • Glucose is split to form 2 glucose-3 -phosphate 6 C 2 x 3 C Figure 5. 12, steps 1– 5
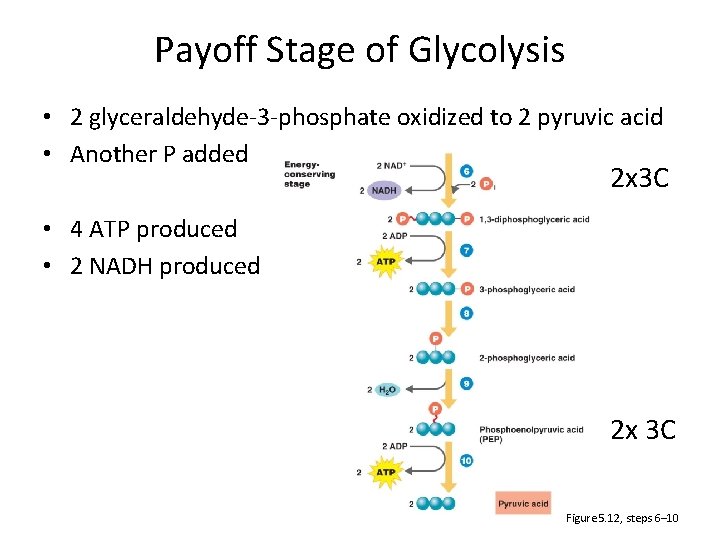
Payoff Stage of Glycolysis • 2 glyceraldehyde-3 -phosphate oxidized to 2 pyruvic acid • Another P added 2 x 3 C • 4 ATP produced • 2 NADH produced 2 x 3 C Figure 5. 12, steps 6– 10
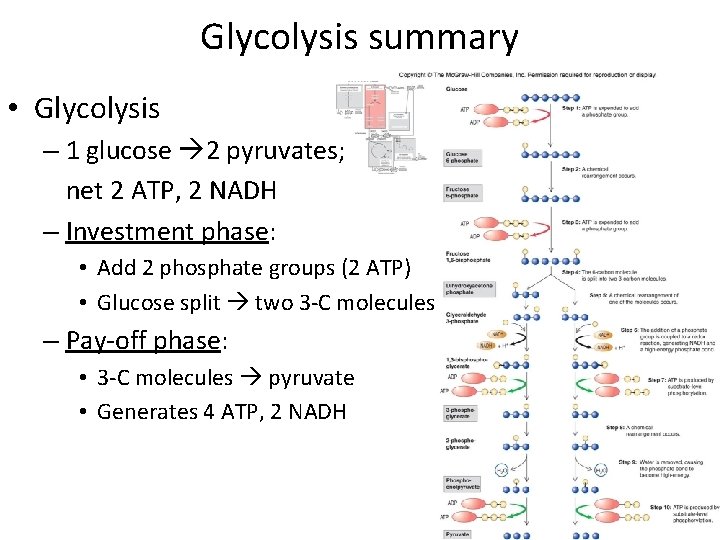
Glycolysis summary • Glycolysis – 1 glucose 2 pyruvates; net 2 ATP, 2 NADH – Investment phase: • Add 2 phosphate groups (2 ATP) • Glucose split two 3 -C molecules – Pay-off phase: • 3 -C molecules pyruvate • Generates 4 ATP, 2 NADH

Alternatives to Glycolysis • Pentose phosphate pathway – Uses pentoses, generates 1 ATP/glucose – Operates with glycolysis – Reversible pentoses eg. for RNA, DNA, some aas • Entner-Doudoroff pathway – Uses glucose, produces 1 ATP/glucose – Is not glycolysis – different enzymes – Lack glycolysis enzymes – Pseudomonas, Rhizobium, Agrobacterium What is the value of these pathways if they produce only one ATP molecule?

The destiny of glucose… • Oxidized – partially (fermentation) – fully to CO 2 (respiration) • Generate NADH, FADH 2, NADPH and ATP • Precursor metabolites – Can be siphoned off for use in biosynthesis • Let’s continue respiring
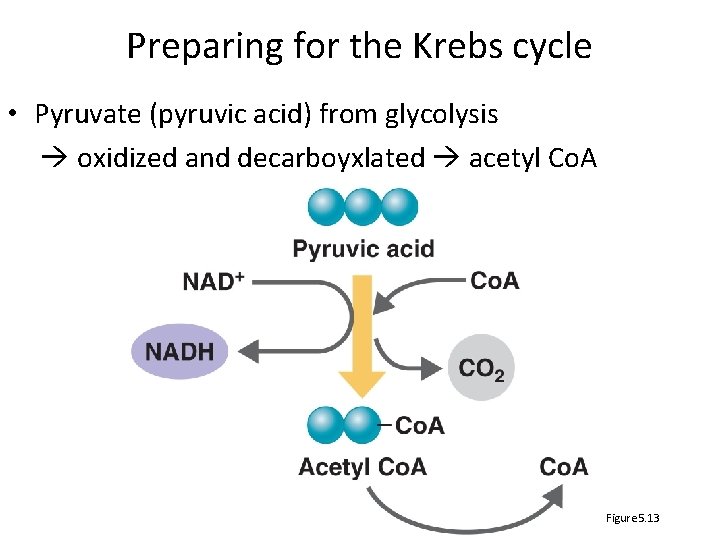
Preparing for the Krebs cycle • Pyruvate (pyruvic acid) from glycolysis oxidized and decarboyxlated acetyl Co. A Figure 5. 13
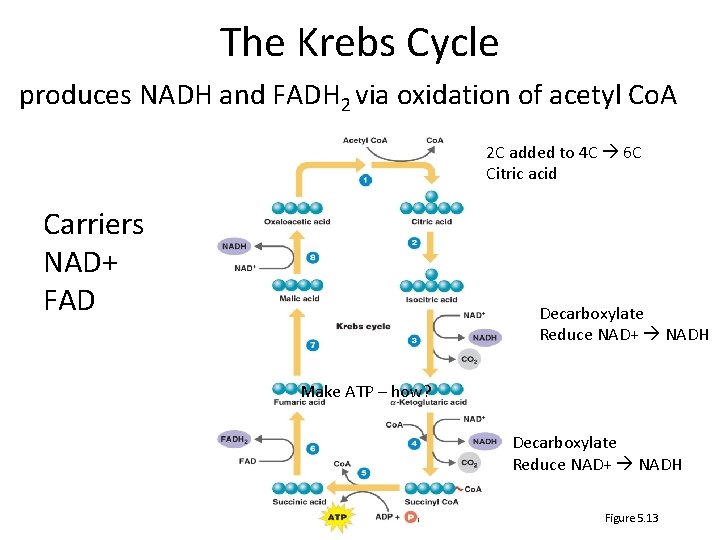
The Krebs Cycle produces NADH and FADH 2 via oxidation of acetyl Co. A 2 C added to 4 C 6 C Citric acid Carriers NAD+ FAD Decarboxylate Reduce NAD+ NADH Make ATP – how? Decarboxylate Reduce NAD+ NADH Figure 5. 13

Where do the electrons go next? Where do the carriers drop their cargo? • NADH • FADH 2
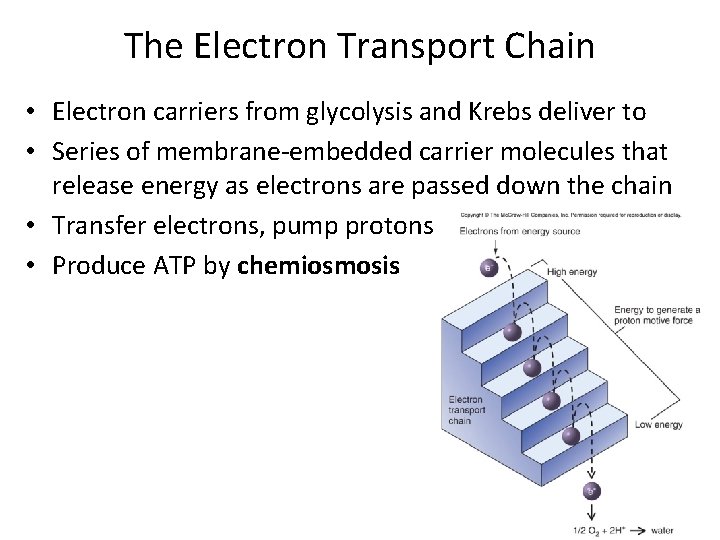
The Electron Transport Chain • Electron carriers from glycolysis and Krebs deliver to • Series of membrane-embedded carrier molecules that release energy as electrons are passed down the chain • Transfer electrons, pump protons • Produce ATP by chemiosmosis

ETC components • Most carriers grouped into large protein complexes • Serve as proton pumps • Three types of – Quinones • Lipid-soluble, move freely, transfer electrons between complexes – Cytochromes • Contain heme, molecule with iron atom at center – Flavoproteins • Proteins to which a flavin is attached • FAD, other flavins synthesized from riboflavin
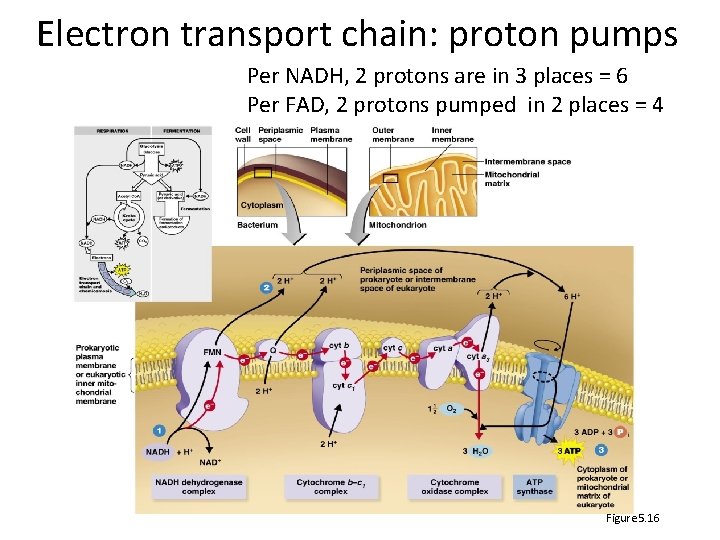
Electron transport chain: proton pumps Per NADH, 2 protons are in 3 places = 6 Per FAD, 2 protons pumped in 2 places = 4 Figure 5. 16
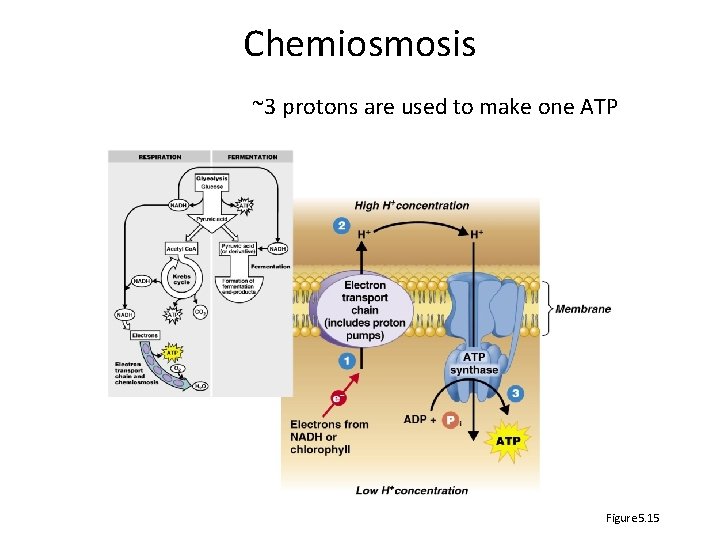
Chemiosmosis ~3 protons are used to make one ATP Figure 5. 15

Respiration review • Draw the three pathways summary • What is the terminal electron acceptor? • What is NAD+ and NADH doing?

Aerobic v. Anaerobic respiration -terminal electron acceptor • Aerobic respiration: Terminal electron acceptor in the electron transport chain is oxygen (O 2). • Anaerobic respiration: Terminal electron acceptor in the electron transport chain is not O 2. • Which is better? Anaerobic yields less energy than aerobic because only part of the Krebs cycle and only some ETC carriers operate under anaerobic conditions.

Anaerobic Respiration If O 2 is terminal electron acceptor, product is ? ? ? Electron Acceptor Products NO 3– NO 2–, N 2 + H 2 O SO 4– H 2 S + H 2 O CO 32 – CH 4 + H 2 O
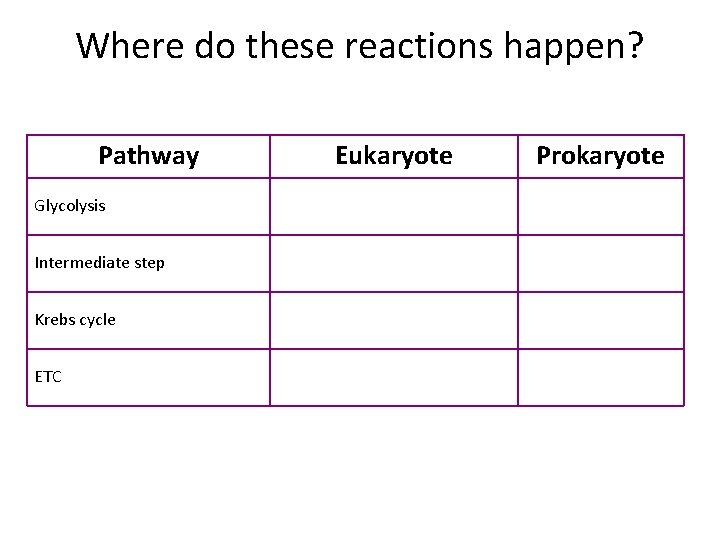
Where do these reactions happen? Pathway Glycolysis Intermediate step Krebs cycle ETC Eukaryote Prokaryote

Where do these reactions happen? Pathway Eukaryote Prokaryote Glycolysis Cytoplasm Intermediate step Mitochondrial matrix Cytoplasm Krebs cycle Mitochondrial matrix Cytoplasm ETC Mitochondrial inner membrane Plasma membrane

The cell • Eukaryotes – Which part of mitochondrion is acidic – How much ATP per glucose? • Prokaryotes – Where do the protons go? – How much ATP per glucose? – Why is it more?

Fermentation • Any spoilage of food by microorganisms (general use) • Any process that produces alcoholic beverages or acidic dairy products (general use) • Any large-scale microbial process occurring with or without air (common definition used in industry)

Fermentation • Scientific definition: – Releases energy from oxidation of organic molecules – Does not require oxygen – Does not use the Krebs cycle or ETC – Uses an organic molecule as the final electron acceptor
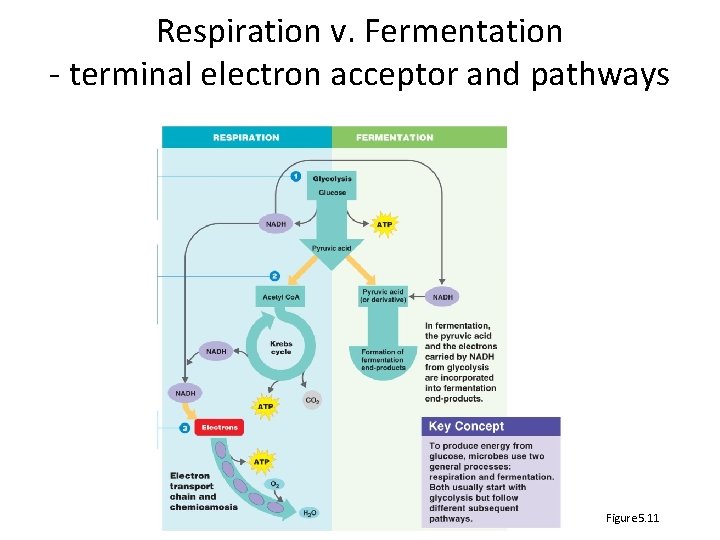
Respiration v. Fermentation - terminal electron acceptor and pathways Figure 5. 11

When would this happen? • Fermentation used when respiration not an option – E. coli is facultative anaerobe • Aerobic respiration, • Anaerobic respiration, • and fermentation – Streptococcus pneumoniae lacks electron transport chain • Lives on sugar; Fermentation only option – ATP is ONLY generated during glycolysis • Additional steps regenerate electron carrier ie. NAD+

Do we do fermentation? • Not USUALLY except… • muscle • cancer
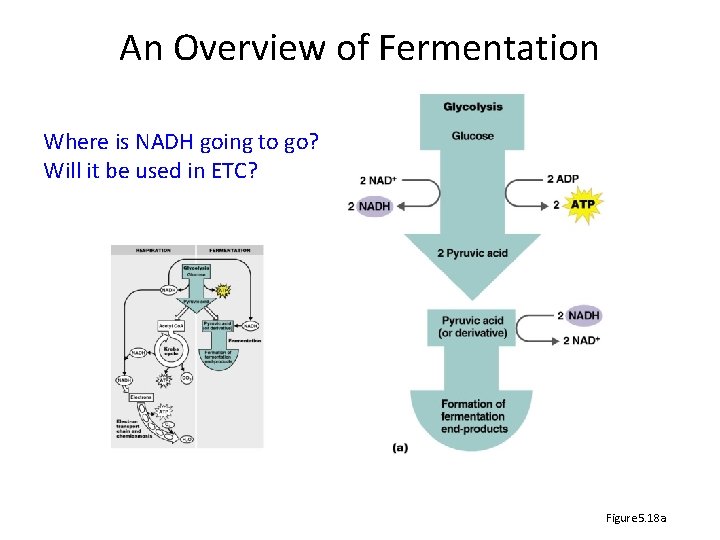
An Overview of Fermentation Where is NADH going to go? Will it be used in ETC? Figure 5. 18 a

Fermentation – what are the end products? • Alcohol fermentation: Produces ethanol + CO 2 • Lactic acid fermentation: Produces lactic acid – Homolactic fermentation: Produces lactic acid only – Heterolactic fermentation: Produces lactic acid and other compounds
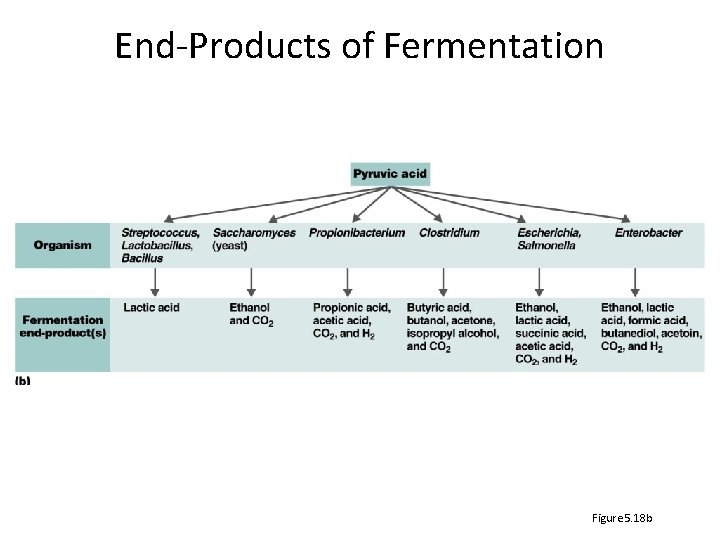
End-Products of Fermentation Figure 5. 18 b
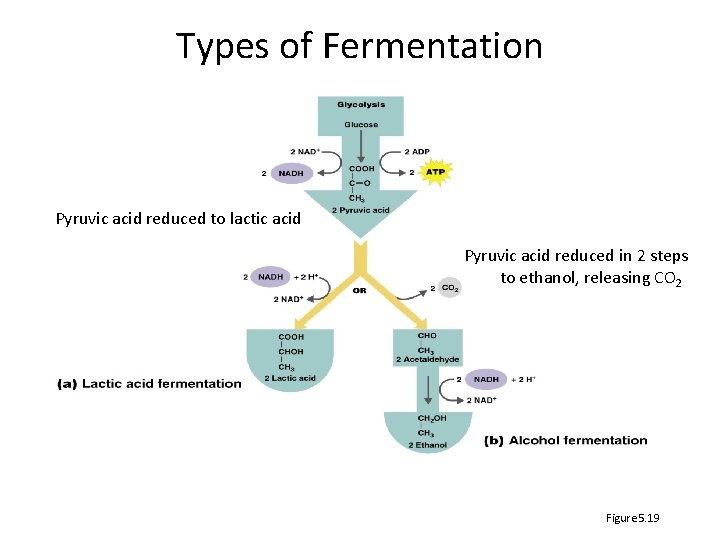
Types of Fermentation Pyruvic acid reduced to lactic acid Pyruvic acid reduced in 2 steps to ethanol, releasing CO 2 Figure 5. 19

Why should we care? • Identify groups, species, pathogens • Harness abilities for food

In the lab: Fermentation Test Control (mannitol) Staph epidermidis (negative) Acid formation – p. H indicator Gas formation – Durham tube Staph aureus (acid +) E coli (acid/gas) YELLOW is positive BUBBLE is positive Figure 5. 23

Speed quiz!!

Making ATP • List 3 types of phosphorylation that make ATP in cells? – Substrate-level, oxidative, photo-phosphorylation • List 3 metabolic strategies cells use to make ATP from oxidizing glucose? – Aerobic respiration, anaerobic respiration, fermentation • What three metabolic pathways are required to perform cellular respiration? – Glycolysis, Kreb’s cycle, electron transport chain
Focusing on respiration ETC, ATP synthase NADH, FADH 2

Respiration - glycolysis • How many Carbons are in the glucose molecule? – Six • How much ATP is produced during glycolysis per starting molecule of glucose? – 2 molecules (net) • ATP made by which type of phosphorylation? – Substrate level • What are the other products of glycolysis – 2 NADH and 2 pyruvate (3 carbon organic molecule)
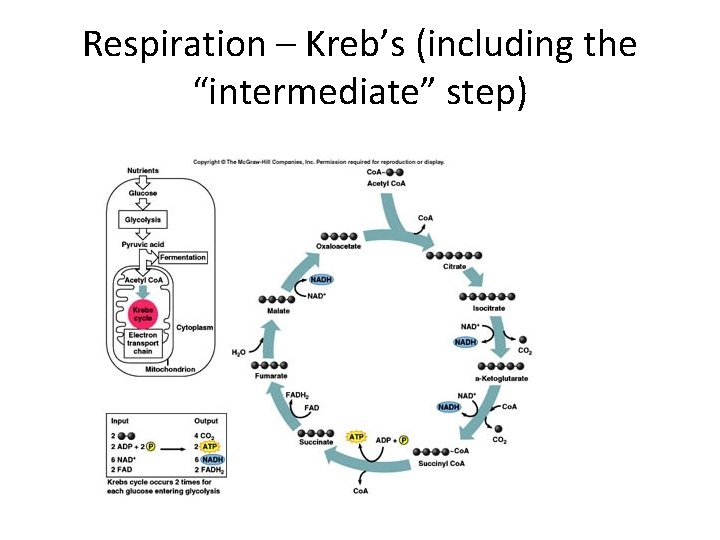
Respiration – Kreb’s (including the “intermediate” step)

Respiration – Kreb’s (including the “intermediate” step) • What is the starting molecule and how is it derived from pyruvate? – Acetyl which is added to Co. A; derived by decaroxylating (removing a C) from 3 C pyruvate 2 C acetyl • How much ATP is produced during the Kreb’s cycle for every pair of pyruvates? – 2 molecules (net) • ATP made by which type of phosphorylation? – Substrate level • What are the other products of glycolysis • 8 NADH, 2 FADH 2 and 6 CO 2

Respiration - ETC • When electrons are delivered, what happens to the electron carriers NADH or FADH 2? – They are oxidized back to NAD+ or FAD and return to glycolysis and/or Kreb’s cycle to pick up more electrons • Why does transferring electrons down the chain release energy? – Each carrier has a higher electronegativity than the last; transferring from a lower to a higher e-neg complex releases energy • What is that energy used for? • Several of the electron acceptor complexes are also proton pumps thatuse the released energy of electron transfer to drive H+ ions across the membrane • What is the “terminal electron acceptor” in aerobic respiration? Anaerobic? – In aerobic respiration it’s Oxygen gas; in anaerobic it’s some other inorganic molecule like nitrate, sulfate, carbonate etc • What is the “proton motive force” used for? – Protons are allowed back across the membrane through ATP synthase; energy released during this process drives ATP production • How much ATP is generated during the ETC? – Up to 32 ATP (3 per NADH and 2 per FADH 2 approximately)

Compare – in groups • Respiration in eukaryotes to prokaryotes • Are both cell types capable of respiration? • Where is glycolysis, Kreb’s cycle, ETC happening? • What region of the cell is becoming acidic due to the action of the ETC • What is the ATP yield (approx. ) after aerobic cellular respiration in each cell type

Compare – in groups • Aerobic to Anaerobic respiration to Fermentation • Do they all oxidize glucose? Y/N? • Which of the three require oxygen? • Which of the three metabolic pathways are used (glycolysis, Kreb’s, ETC) • What is the terminal electron acceptor in each? • What is the ATP yield (approx. ) for each • Can our cells do all three? • Can microbes do all three?

Fermentation • Where is all the energy created during fermentation? – glycolysis • ATP yield is? What else is made? – 2 ATP (net); 2 NADH and 2 pyruvate • What are the two most common next steps in fermentation? – Lactic acid fermentation - NADH dumps its electrons onto pyruvate creating lactic acid OR – Ethanol fermentation – pyruvate is decarboxylated to acetate, which then receives electrons from NADH creating ethanol • Why does the NADH need to be oxidized in some way? – Because we don’t have an ETC and we need NAD+ in glycolysis
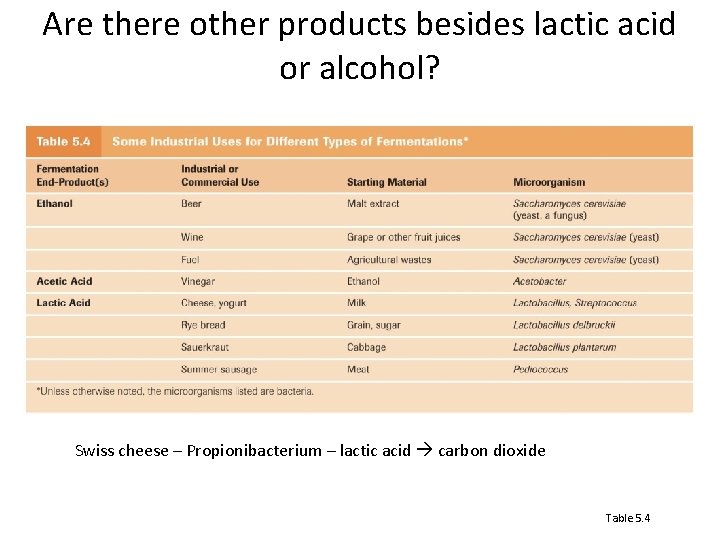
Are there other products besides lactic acid or alcohol? Swiss cheese – Propionibacterium – lactic acid carbon dioxide Table 5. 4

Are carbohydrates the only fuel? • If there’s no sugar in the house… • Use dietary fat or protein

Using fat • Triglyceride is made of? • Where can we use these pieces? • Glycerol can be fed into second half of glycolysis • Fatty acids can be broken into 2 C chunks by of acetyl-Co. A ?
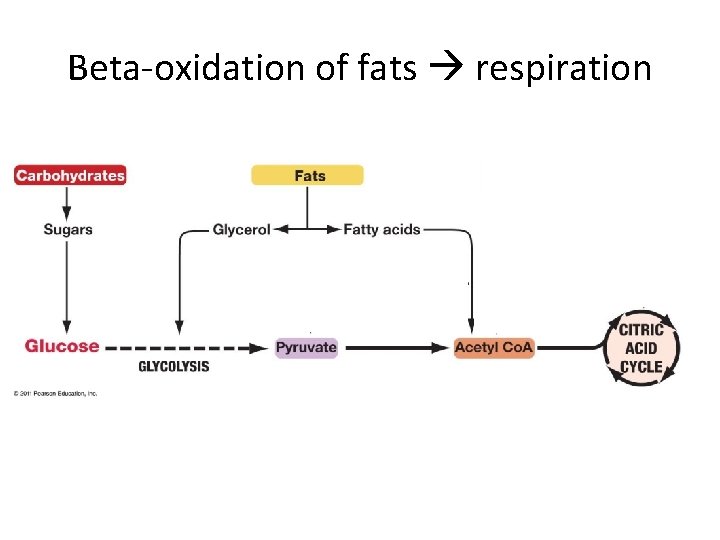
Beta-oxidation of fats respiration

Using protein Protein is made of? Are there 2 C units in these pieces? What do we need to do first? Proteins can be broken into amino acids Amino acids need to be DE-AMINATED (remove the amino group) • Depending on the R group, go into Kreb’s cycle at various places • • •
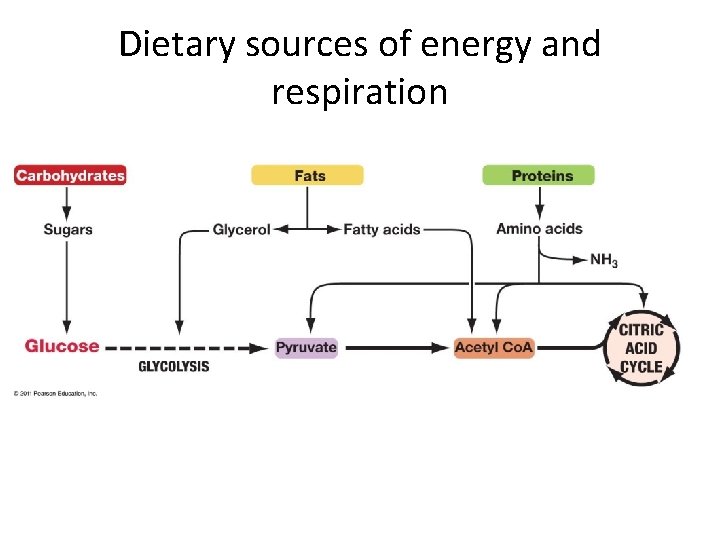
Dietary sources of energy and respiration

How do microbes do it? • Microbes can use variety of compounds – Excrete hydrolytic enzymes; transport subunits into cell • Degrade further to appropriate precursor metabolites – Polysaccharides and disaccharides • Amylases digest starch; cellulases digest cellulose • Disaccharides hydrolyzed by specific disaccharidases – Lipids • Fats hydrolyzed by lipases; glycerol converted to dihydroxyacetone phosphate, enters glycolysis • Fatty acids degraded by β-oxidation to enter TCA cycle – Proteins • Hydrolyzed by proteases; amino group deaminated • Carbon skeletons converted into precursor molecules

OK where we… • • Major metabolic pathways of the cell Photosynthesis light and dark reactions Innovations and features in microbes - chemolithoautotrophy

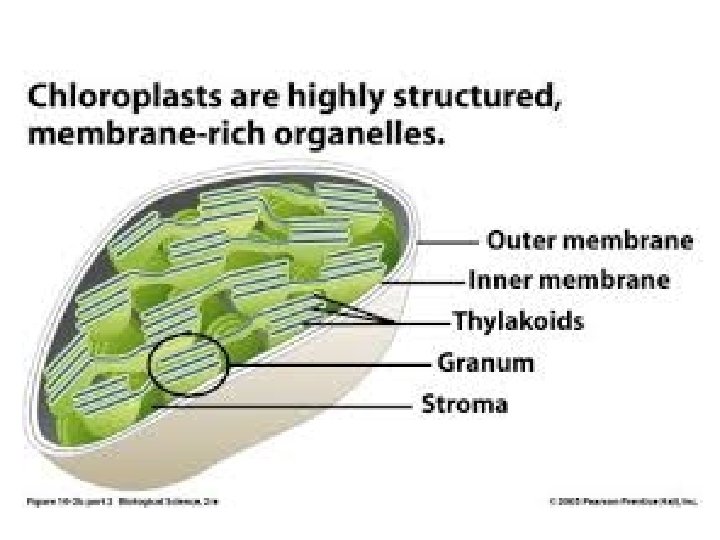
Photosynthesis • What organelle does this?

Photosynthesis in 2 EZ steps • Are plants the only ones who do this? (HINT: you are taking a microbiology class…) • NO! photosynthetic bacteria and algae too! • Step one: photophosphorylation • Step two: carbon fixation • What do you think this means? ?

Photophosphorylation – aka “light reactions” • Dependent on? – LIGHT! • Also you need: – Pigments • Plants, algae and blue-green bacteria: chlorphoyll) • In green and purple bacteria: bacteriochlorophyll – Proton pumps in an electron transport chain – ATP synthase – Hmmmm…. This sounds awfully familiar… • Where are the electrons coming from? – H 2 O (oxygenic photosynthesis) – Other… eg. H 2 S (anoxygenic photosynthesis) • What is produced? – ATP! And either Oxygen gas or something else like Sulfur waste
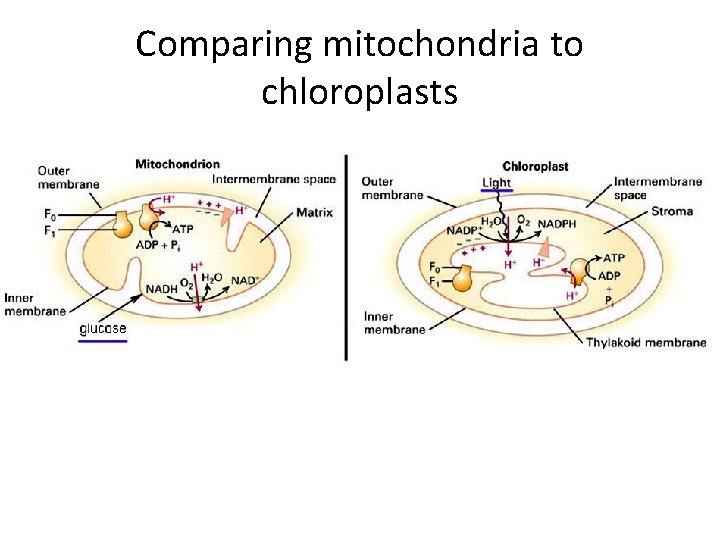
Comparing mitochondria to chloroplasts

Where are the light reactions happening? • Plants and algae: – Chloroplast thylakoid membranes • Photosynthetic bacteria: – Plasma membrane

Carbon fixation – aka “dark reactions” • Dependent on light? • Use? – 18 ATP, 6 CO 2, 6 H 2 O • Produce? – Glucose C 6 H 12 O 6

Carbon fixation – aka “dark reactions”
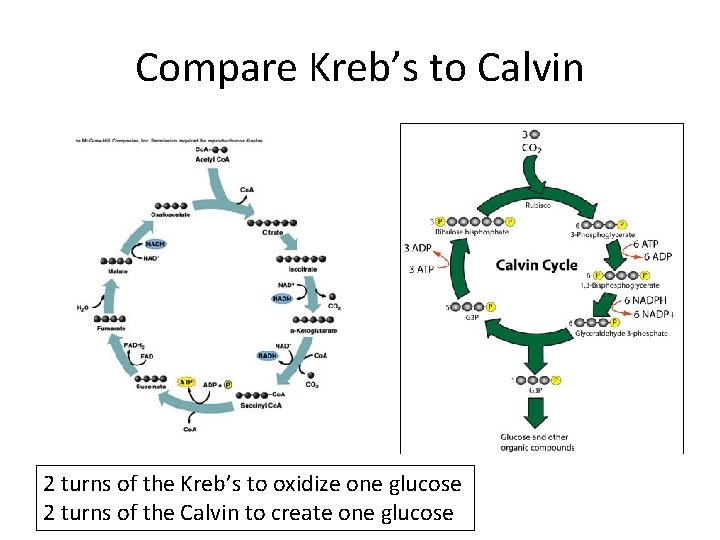
Compare Kreb’s to Calvin 2 turns of the Kreb’s to oxidize one glucose 2 turns of the Calvin to create one glucose

Where are the dark reactions happening? • Plants and algae: – Chloroplast “stroma” • Photosynthetic bacteria: – Cytoplasm!

Almost done! • • Finish metabolic pathways of the cell Photosynthesis light and dark reactions Innovations and features in microbes - chemolithoautotrophy

chemolithoautotrophy

chemolithoautotrophy • “rock eater” – in mine, soil, ocean floor near hydrothermal vents • Able to “fix carbon” - ie use CO 2 glucose • ? LIGHT? so where do they get energy? ? • Oxidize INORGANIC compounds – H 2 S and metal ions • High energy electrons -ETC ATP Calvin cycle glucose energy and growth • In mines and vents, organisms rely on them the way we rely on photosynthesizers • Variations on this theme….

THIS WEEK IN LAB • Metabolism I/II • Overview of metabolism • Pathogenesis – often related to metabolism

Lab procedures - carbohydrates • Sugar tubes (fermentation of mono/disacc) – Glucose, sucrose, lactose – why? What’s being tested? – What will a negative look like? A positive?

In the lab: Fermentation Test Control (mannitol) Staph epidermidis (negative) Acid formation – p. H indicator Gas formation – Durham tube Staph aureus (acid +) E coli (acid/gas) YELLOW is positive BUBBLE is positive Figure 5. 23

Lab procedures - protein • Litmus milk – Lactose fermentation? – Caseinase? (clearing) – Rennetase? (coagulation)
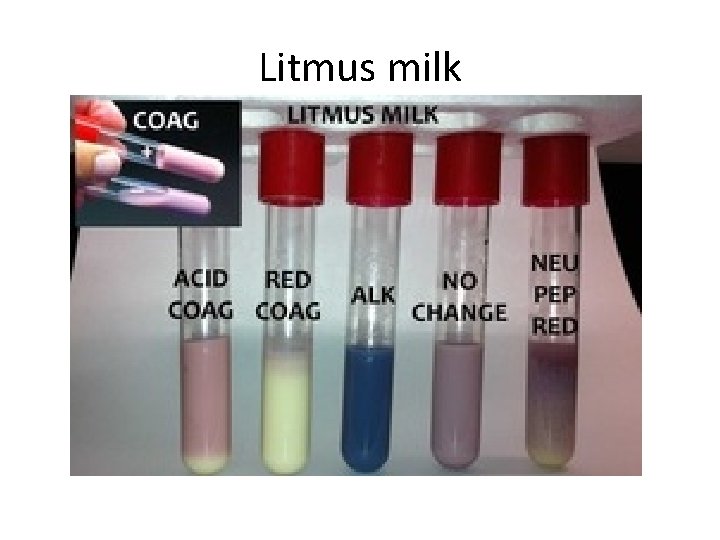
Litmus milk

Continue unknown ID • Hand in reserve • Grade T streaks and Gram stain if ready • Begin traditional ID method – dichotomous charts • Begin molecular ID method – inoculate TSB for DNA isolation
- Slides: 118