Binding energy sketch the variation of binding energy

Binding energy • sketch the variation of binding energy per nucleon with nucleon number. • explain what is meant by nuclear fusion and nuclear fission. • explain the relevance of binding energy per nucleon to nuclear fusion and to nuclear fission. • Solve problems involving mass defect and binding energy.

Summary • Discussion: Introducing mass defect and atomic mass units. (10 minutes) • Discussion: Mass defect and binding energy (10 minutes) • Worked example: Calculating binding energy. (10 minutes) • Student questions: Calculations. (20 minutes) • Discussion: Fission and fusion linked to binding energy graph. (10 minutes) • Reading and further questions

Gneed two nough • 1 Students must be familiar with the units Me. V c − 2 and Ge. V c − 2 for mass. • 2 Students should be familiar with binding energies plotted as positive quantities.

Notice anything? proton mass, mp = 1. 673 10 -27 kg neutron mass, mn = 1. 675 10 -27 kg mass of a helium nucleus = 6. 643 10 -27 kg The mass of a nucleus is less than the sum of the masses of its parts; this is true for all nuclides. • So much for conservation of mass. • •

Atomic mass units • the atomic mass unit (amu, or u) is a convenient unit of nuclear mass. • 1 amu or 1 u = 1/12 the mass of a neutral 12 C atom (i. e. including its six electrons) = 1. 66056 10 -27 kg. Thus: • mp = 1. 0073 u • mn = 1. 0087 u • me = 0. 00055 u • mass of a neutral helium atom = 4. 0026 u

Mass defect and binding energy • What has happened to the missing mass – or mass defect – between the whole and the sum of the parts? • To separate the particles, they must be pulled apart against the attractive strong force. They thus have potential energy when they are separated. • When the particles come together to form a nucleus, their potential energy decreases. • So energy must be put in to separate the nucleons of a nucleus. • This energy is known as the binding energy, • This does not mean that energy is required to bind nucleons together. • As with chemical bonds, this is the opposite of the truth. Energy is needed to break bonds.

That equation • Einstein’s Special Theory of Relativity (1905) relates mass and energy via the equation E = mc 2 (where c is the speed of light in a vacuum). In this case, we have: • binding energy = mass defect c 2 or ΔE = Δ m c 2

Talking about mass and energy • It is not advisable to talk about mass being ‘converted to energy’. • It is better to say that, in measuring an object’s mass, we are determining its energy. • A helium nucleus has less mass than its constituent nucleons; in pulling them apart, we do work and so give them energy; hence their mass is greater.

Calculating binding energy • • mp = 1. 0073 u mn = 1. 0087 u me = 0. 00055 u mass of a neutral helium atom = 4. 0026 u • Calculate the mass defect and binding energy for A neutral helium atom. • Mass defect = (2 mp+2 mn+2 me) – m. He • Mass defect = (2. 0146+2. 0174+. 00110) – 4. 0026 • Mass defect = 0. 0305 • binding energy = 0. 0305 x 1. 661 x 10 -27 • 4. 56 10 -12 J

Another way • • • mp = 1. 0073 u mn = 1. 0087 u me = 0. 00055 u • • mass of a neutral helium atom = 4. 0026 u Mass defect = (2 mp+2 mn+2 me) – m. He Mass defect = (2. 0146+2. 0174+. 00110) – 4. 0026 Mass defect = 0. 0305 From data booklet 1 u = 931. 5 Me. Vc-2 E = mc 2 E = (931. 5 x 0. 0305) x c 2 E = 28. 411 Me. V ……. Check 4. 546 x 10 -12

Tu du • Complete changing energy changing mass worksheet. • Use the spreadsheets to investigate binding energies and binding energies per nucleon

Using a spreadsheet • On the website you can find a spreadsheet to do these calculations for you. • Student activity: Spreadsheet calculations. (20 minutes) • Student activity: Spreadsheet calculations of binding energy per nucleon. (20 minutes)
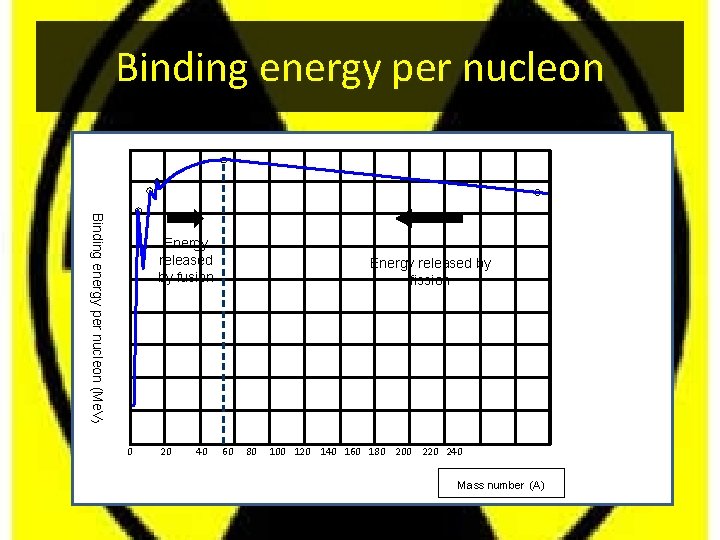
Binding energy per nucleon (Me. V) Energy released by fusion 0 20 40 Energy released by fission 60 80 100 120 140 160 180 200 220 240 Mass number (A)

Meaning of the graph • The part of the curve to the left shows that two light elements can produce energy by joining together - fusion • The part of the curve to the right shows that a heavy element can produce energy by breaking in to smaller pieces - fission. • If a reaction takes place where the products are closer to the peak than the original nucleus (nuclei) then energy is given out. • For helium the binding energy per nucleon is 28. 3/4 = 7. 1 Me. V. • The helium nucleus has a high binding energy per nucleon and is more stable than some of the other nuclei close to it in the periodic table. • A very useful web site containing a huge nuclear database is to be found at: • http: //nucleardata. nuclear. lu. se

Reading and Further questions • Adams and Allday P 374 +5 q 1 -5 • For fun http: //www. invisiblemoose. org/ald/site_mate rial/WALTA/Cosmic_Rays_CD/support_materi al/detectors/bubble_chamber/www. lalanet. gr. jp/nsm/E-radiation. html
- Slides: 15