BINDING ENERGY Contents Binding Energy Mass Defect Packing

BINDING ENERGY
Contents Binding Energy Mass Defect Packing Fraction

Binding Energy Binding energy is the energy required to disassemble or break a whole system into separate parts. Nuclear binding energy is the energy that would be required to disassemble or break the nucleus of an atom into its component parts. These component parts are neutrons and protons, which are collectively called nucleons.

Mass defect The mass of an atomic nucleus is usually less than the sum of the individual masses of the constituent protons and neutrons (according to Einstein's equation E=mc 2) and this 'missing mass' is known as the mass defect, and represents the energy that was released when the nucleus was formed.

For the alpha particle Dm= 0. 0304 u which gives 28. 3 Me. V binding energy!

Binding fraction f. B • If the binding energy EB of a nucleus divided by the mass number A , we get the binding energy per nucleon in the nucleus which is known as binding fraction f. B • Here we assumed that the masses are expressed in energy unit so that c 2 has been omitted. Binding fraction f. B of different nuclei represent relative strength of their binding

Binding Energy � When the protons and neutrons combine to form a nucleus, the mass that disappears (mass defect, ∆m) is converted into an equivalent amount of energy (∆mc 2). This energy is called the binding energy of the nucleus. Binding energy = [Zm. P + Nmn - m] c 2 = ∆m c 2 It determines its stability against disintegration. � � If the binding energy is large, the nucleus is stable. The binding energy per nucleon varies from element to element.
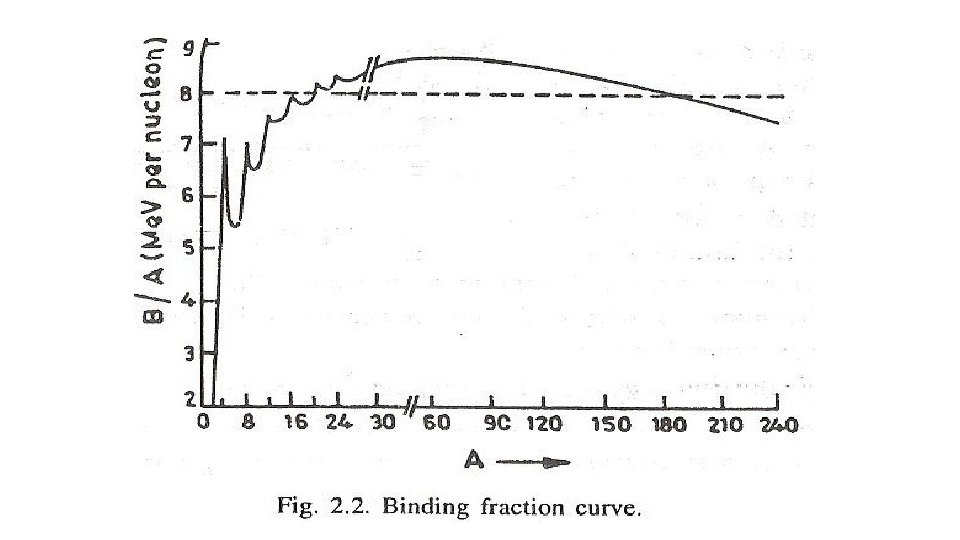
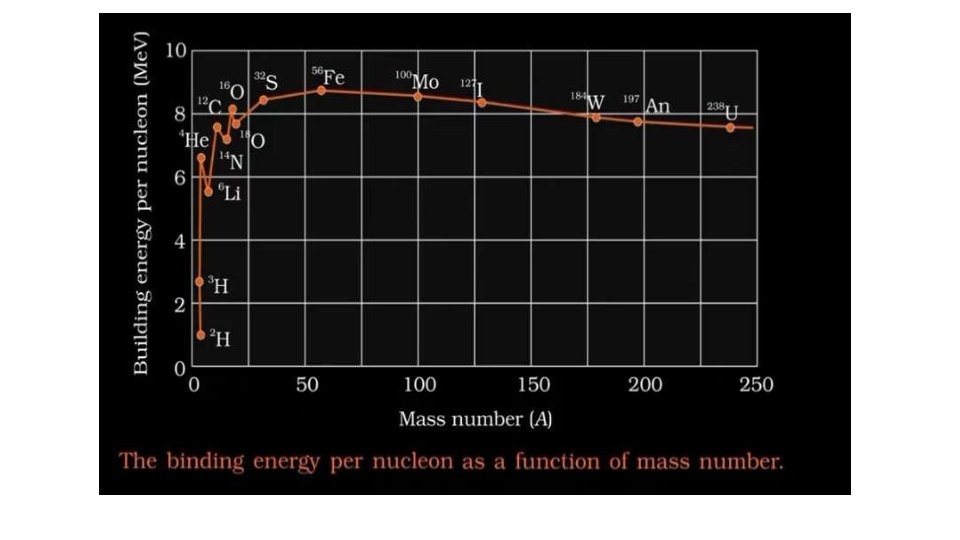

Important points about f. B 1. f. B For very light nuclei is very small and rises rapidly with A attaining the value of 8 Me. V/nucleon for A~20, then it rises slowly with attains 8. 7 Me. V/nucleon for at A~56 for higher it decreases slowly 2. For 20<A<180 the variation of f. B is very slight and it may be constant in this region having a mean value 8. 5 Me. V/nucleon 3. For very heavy nuclei A>180 f. B decreases with the increase of A , for heaviest nuclei f. B is about 7. 5 Me. V/nucleon 4. For very light nuclei there are rapid fluctuations in f. B , in particular for even-even nuclei for which A=4 n , n is an integer. similar but less prominent peaks are observed at values of Z or N=20, 28, 50, 82, 126 magic numbers, peaks means greater stability

The binding energy per nucleonincreases � i) sharply with mass number A upto 20. It increases slowly after A = 20. For A<20, there exists recurrence of peaks corresponding to those nuclei, whose mass numbers are multiples of four and they contain not only equal but also even number of protons and neutrons. Example: 2 He 4, 4 Be 8, 12 16 6 C , 8 O , and 10 Ne 20. The curve becomes almost flat for mass number between 40 and 120. Beyond 120, it decreases slowly as A increases.

�ii)The binding energy per nucleon reaches a maximum of Me. V at A=56, corresponding to the iron nucleus (26 Fe 56). Hence, iron nucleus is the most stable. � iii) The average binding energy per nucleon is about 8. 5 Me. V for nuclei having mass number ranging between 40 and 120. These elements are comparatively more stable and non radioactive.

Packing fraction f • The mass defect of an atom divided by its mass number • packing fraction has the same sign of mass defect



• From the figure it is seen that f varies in a systematic manner with the mass number A • For very light nuclei and very heavy nuclei f is positive (20>A>180) • For nuclei with mass numbers between 20 and 180 , f is negative

Thank. You! Any questions?
- Slides: 17