Binary Multistage Distillation Condenser At the top of
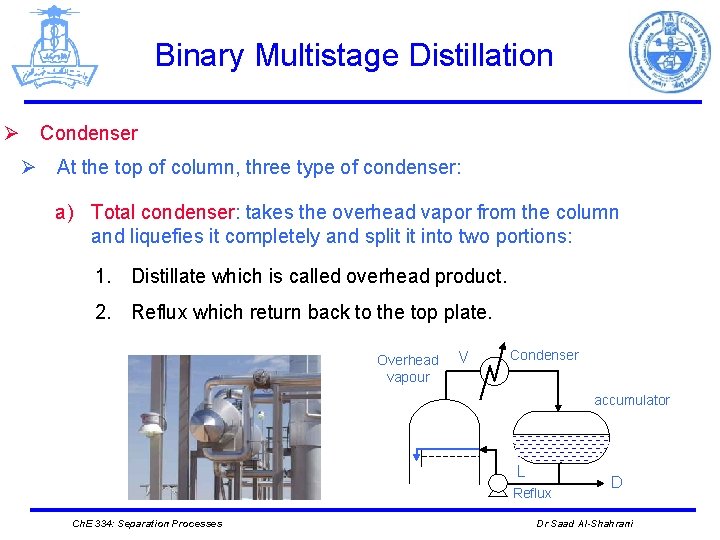
Binary Multistage Distillation Ø Condenser Ø At the top of column, three type of condenser: a) Total condenser: takes the overhead vapor from the column and liquefies it completely and split it into two portions: 1. Distillate which is called overhead product. 2. Reflux which return back to the top plate. Overhead vapour V Condenser accumulator L Reflux Ch. E 334: Separation Processes D Dr Saad Al-Shahrani
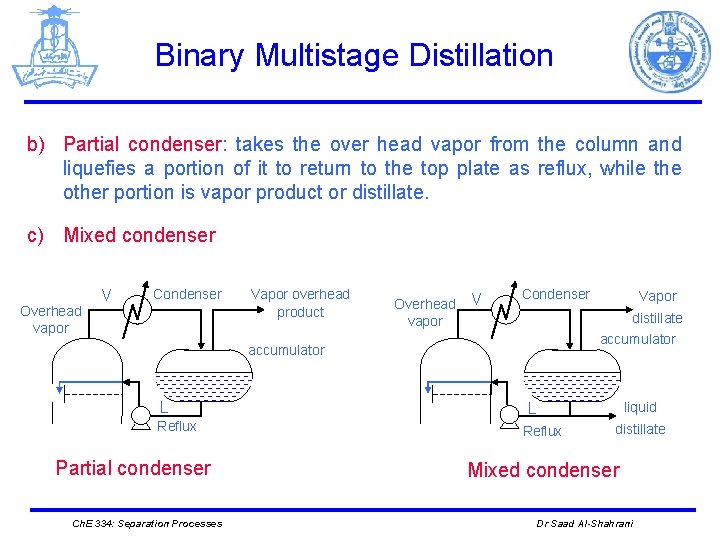
Binary Multistage Distillation b) Partial condenser: takes the over head vapor from the column and liquefies a portion of it to return to the top plate as reflux, while the other portion is vapor product or distillate. c) Mixed condenser Overhead vapor V Condenser Vapor overhead product Overhead vapor V Condenser distillate accumulator L Reflux Partial condenser Ch. E 334: Separation Processes Vapor liquid L Reflux distillate Mixed condenser Dr Saad Al-Shahrani
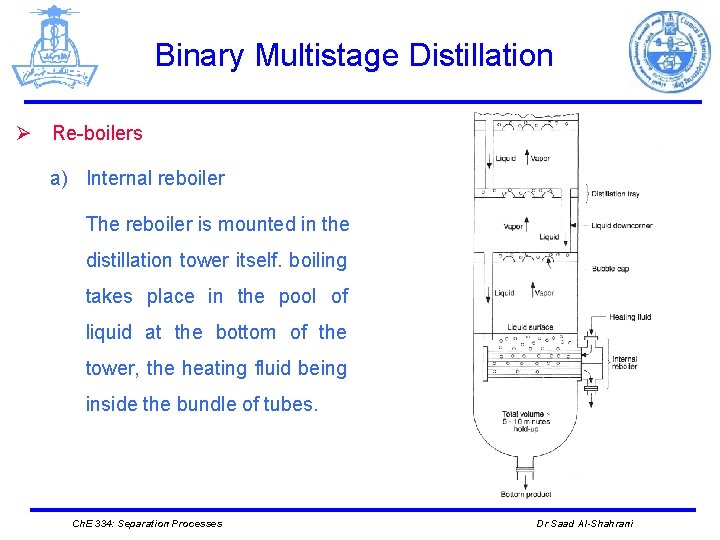
Binary Multistage Distillation Ø Re-boilers a) Internal reboiler The reboiler is mounted in the distillation tower itself. boiling takes place in the pool of liquid at the bottom of the tower, the heating fluid being inside the bundle of tubes. Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Binary Multistage Distillation Ø Re-boilers b) Kettle type re-boiler At the bottom of column which heat the bottom liquid to produce vapor partially and return it back to column, while the rest liquid is withdrawn as a bottom product. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
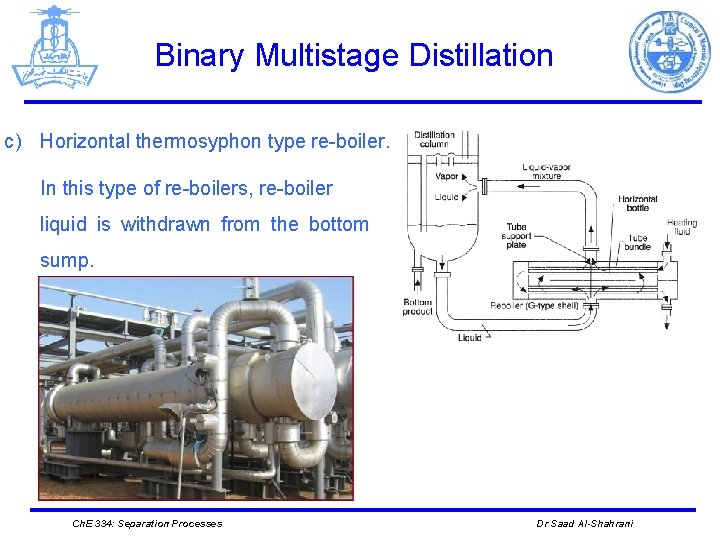
Binary Multistage Distillation c) Horizontal thermosyphon type re-boiler. In this type of re-boilers, re-boiler liquid is withdrawn from the bottom sump. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
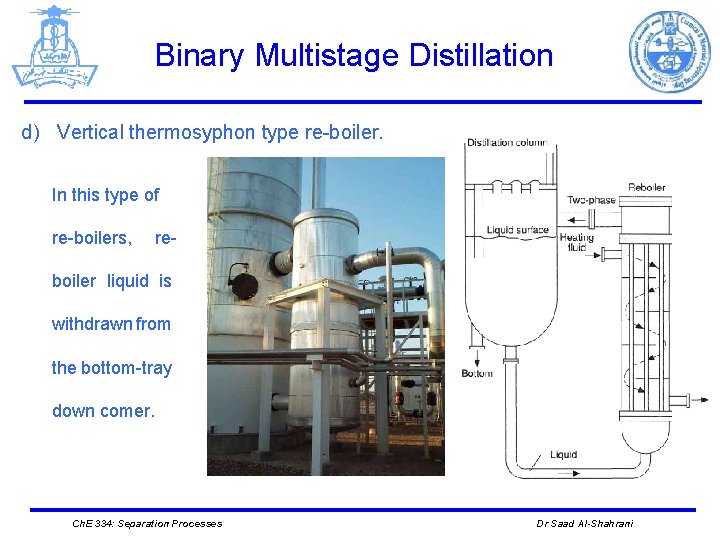
Binary Multistage Distillation d) Vertical thermosyphon type re-boiler. In this type of re-boilers, re- boiler liquid is withdrawn from the bottom-tray down comer. Ch. E 334: Separation Processes Dr Saad Al-Shahrani
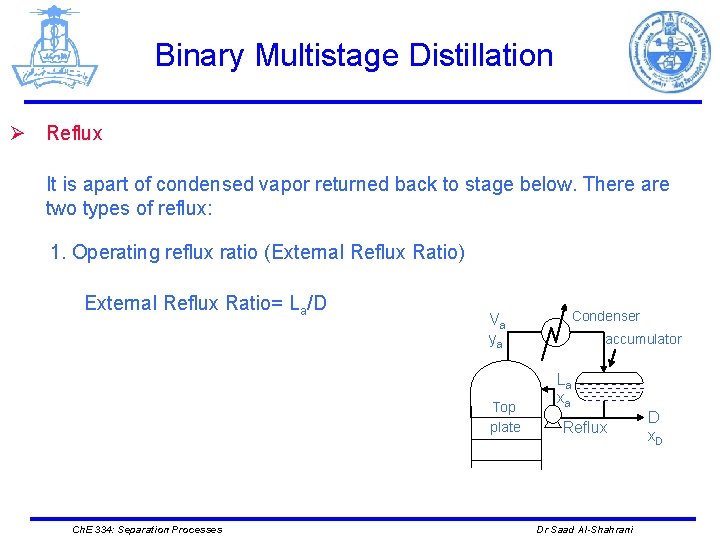
Binary Multistage Distillation Ø Reflux It is apart of condensed vapor returned back to stage below. There are two types of reflux: 1. Operating reflux ratio (External Reflux Ratio) External Reflux Ratio= La/D Va ya Top plate Ch. E 334: Separation Processes Condenser accumulator La xa Reflux Dr Saad Al-Shahrani D x. D
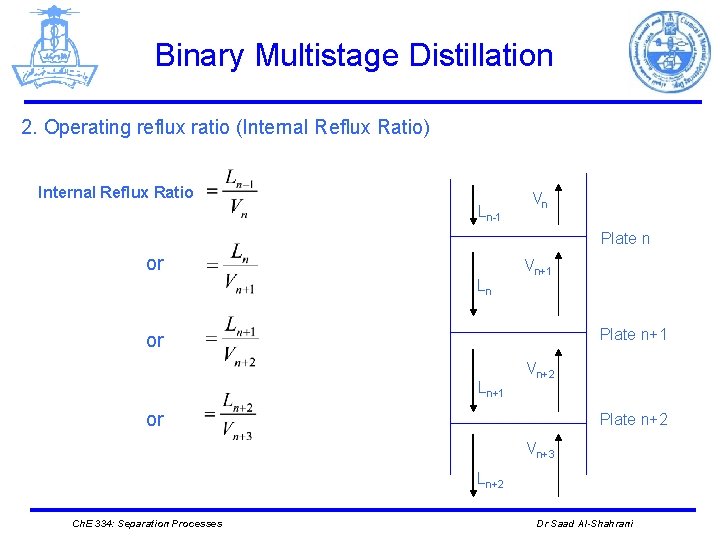
Binary Multistage Distillation 2. Operating reflux ratio (Internal Reflux Ratio) Internal Reflux Ratio Ln-1 Vn Plate n or Ln Vn+1 Plate n+1 or Ln+1 Vn+2 or Plate n+2 Vn+3 Ln+2 Ch. E 334: Separation Processes Dr Saad Al-Shahrani
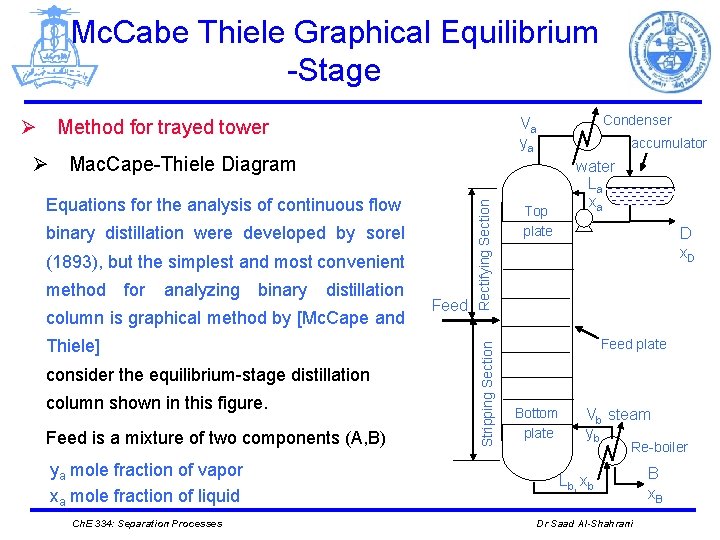
Mc. Cabe Thiele Graphical Equilibrium -Stage binary distillation were developed by sorel (1893), but the simplest and most convenient for analyzing binary distillation column is graphical method by [Mc. Cape and Thiele] consider the equilibrium-stage distillation column shown in this figure. Feed is a mixture of two components (A, B) ya mole fraction of vapor xa mole fraction of liquid Ch. E 334: Separation Processes Feed Stripping Section Equations for the analysis of continuous flow Rectifying Section Ø Mac. Cape-Thiele Diagram method Condenser Va ya Ø Method for trayed tower accumulator Top plate water La xa D x. D Feed plate Bottom plate Vb steam yb Re-boiler Lb, xb Dr Saad Al-Shahrani B x. B
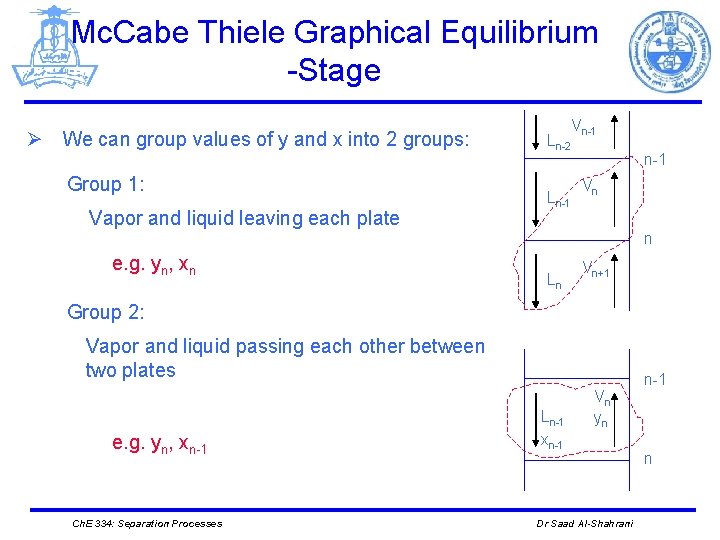
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø We can group values of y and x into 2 groups: Group 1: Vapor and liquid leaving each plate Ln-2 Ln-1 Vn-1 Vn n e. g. yn, xn Ln Vn+1 Group 2: Vapor and liquid passing each other between two plates e. g. yn, xn-1 Ch. E 334: Separation Processes Ln-1 xn-1 Vn yn Dr Saad Al-Shahrani n-1 n
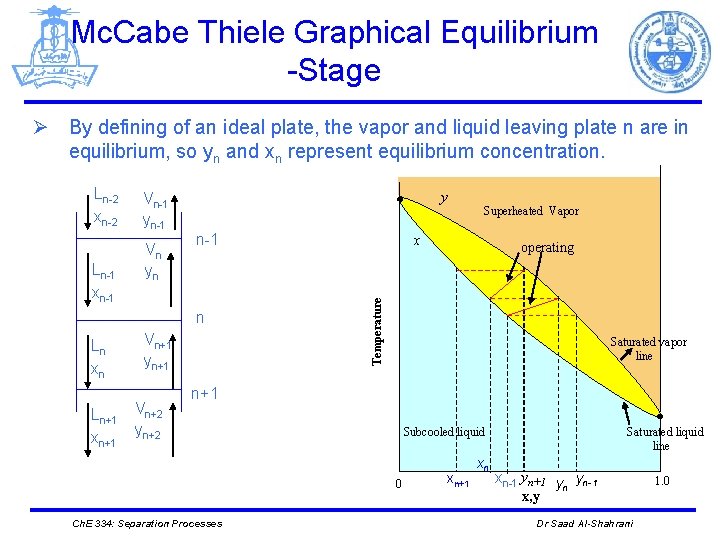
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø By defining of an ideal plate, the vapor and liquid leaving plate n are in equilibrium, so yn and xn represent equilibrium concentration. Ln-1 xn-1 Vn-1 yn-1 Vn yn y n-1 n Ln xn Ln+1 xn+1 Vn+1 yn+1 Vn+2 yn+2 Superheated Vapor x operating Temperature Ln-2 xn-2 Saturated vapor line n+1 Subcooled liquid 0 Ch. E 334: Separation Processes xn+1 xn Saturated liquid line xn-1 yn+1 y yn-1 n x, y Dr Saad Al-Shahrani 1. 0

Mc. Cabe Thiele Graphical Equilibrium -Stage Ø Since the concentration in both phases (vapor, liquid) increases with the hight of the column, xn-1 > xn+1 yn > yn+1 > yn+2 Ø In spite of xn, yn (streams leaving plate n) are in equilibrium, xn-1, yn+1( streams entering plate n) are not in equilibrium (see the last figure) Ch. E 334: Separation Processes Dr Saad Al-Shahrani
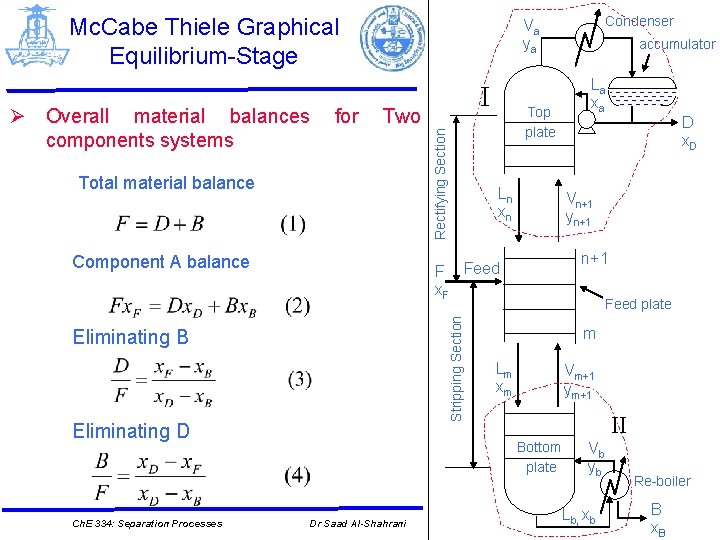
Mc. Cabe Thiele Graphical Equilibrium-Stage for Two Total material balance Component A balance Top plate Ln xn Stripping Section Eliminating D La xa D x. D n+1 Feed plate m Lm xm Vm+1 ym+1 II Bottom plate Dr Saad Al-Shahrani accumulator Vn+1 yn+1 F Feed x. F Eliminating B Ch. E 334: Separation Processes I Rectifying Section Ø Overall material balances components systems Condenser Va ya Vb yb Lb, xb Re-boiler B x. B
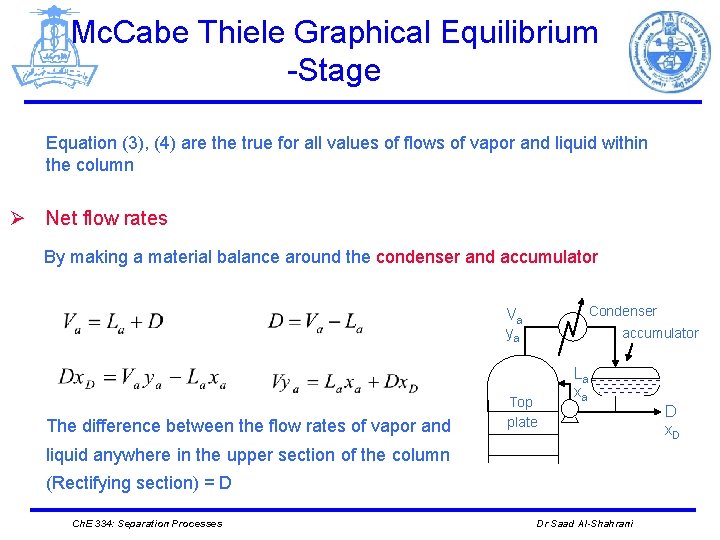
Mc. Cabe Thiele Graphical Equilibrium -Stage Equation (3), (4) are the true for all values of flows of vapor and liquid within the column Ø Net flow rates By making a material balance around the condenser and accumulator Condenser Va ya The difference between the flow rates of vapor and accumulator Top plate La xa liquid anywhere in the upper section of the column (Rectifying section) = D Ch. E 334: Separation Processes Dr Saad Al-Shahrani D x. D
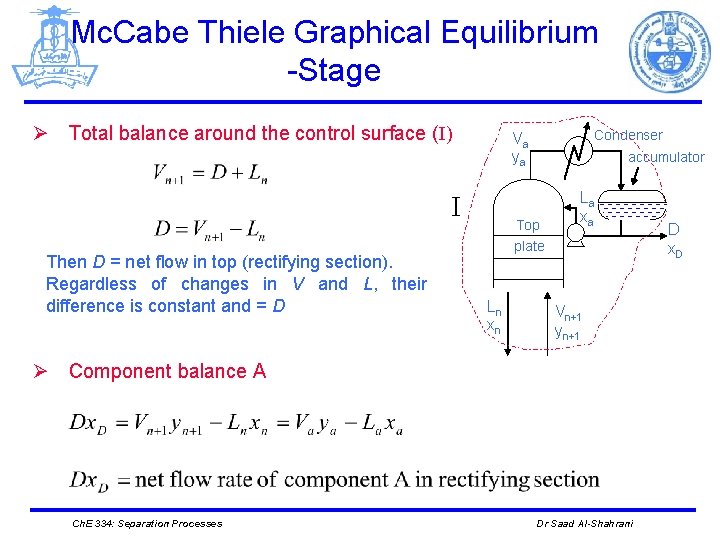
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø Total balance around the control surface (I) I Then D = net flow in top (rectifying section). Regardless of changes in V and L, their difference is constant and = D Condenser Va ya accumulator Top plate Ln xn La xa Vn+1 yn+1 Ø Component balance A Ch. E 334: Separation Processes Dr Saad Al-Shahrani D x. D
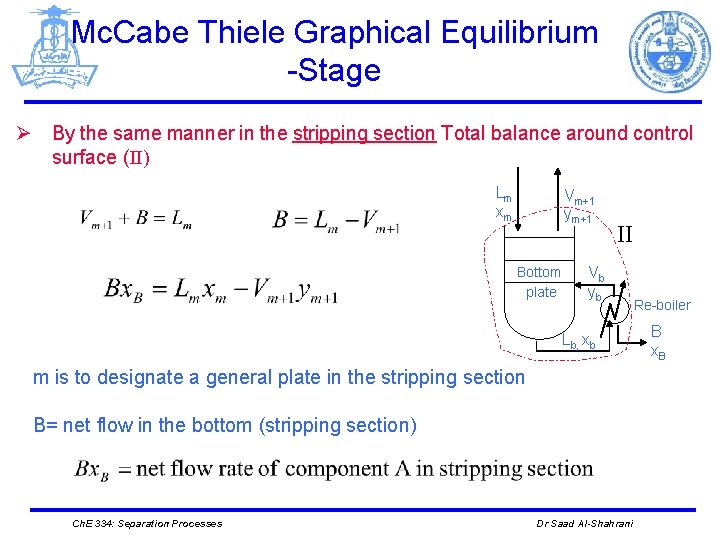
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø By the same manner in the stripping section Total balance around control surface (II) Lm xm Vm+1 ym+1 Bottom plate Vb yb II Re-boiler Lb, xb m is to designate a general plate in the stripping section B= net flow in the bottom (stripping section) Ch. E 334: Separation Processes Dr Saad Al-Shahrani B x. B
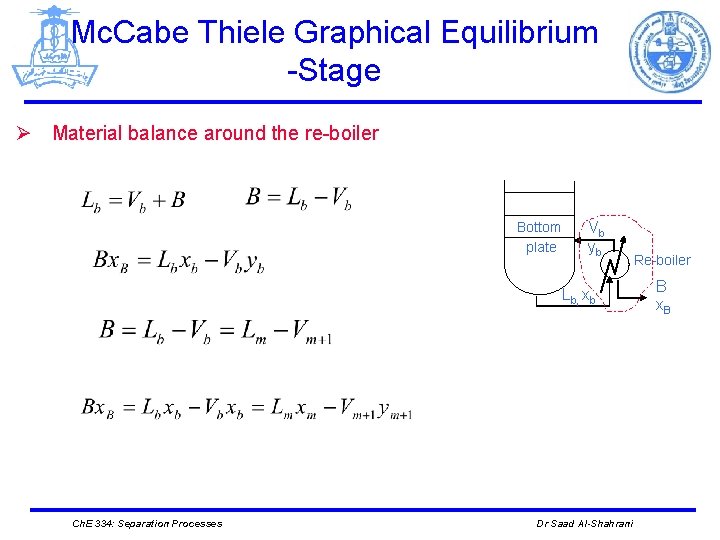
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø Material balance around the re-boiler Bottom plate Vb yb Re-boiler Lb, xb Ch. E 334: Separation Processes Dr Saad Al-Shahrani B x. B

Mc. Cabe Thiele Graphical Equilibrium-Stage Condenser Va ya Ø Operating lines I Because there are two sections in the column, Top plate there are two operating lines accumulato La xa D x. D 1. For rectifying section (I) For control surface (I) ( Ln xn ) Vn+1 yn+1 n Ln xn n+1 Ch. E 334: Separation Processes Dr Saad Al-Shahrani Operation relationship Vn+1 yn+1
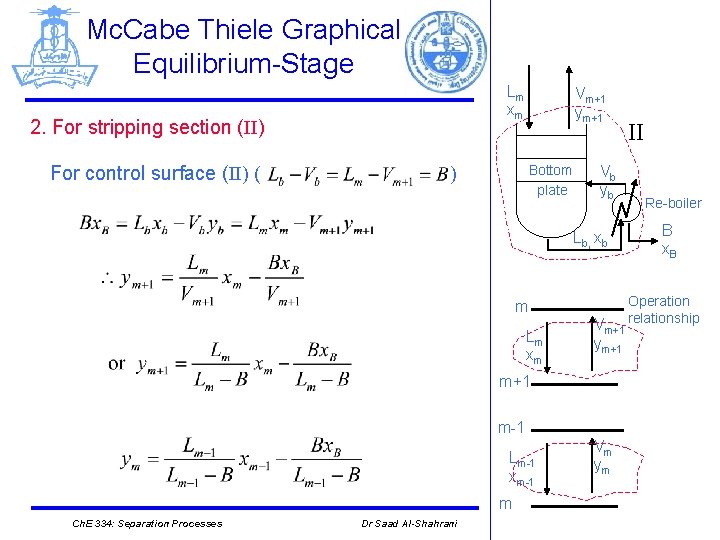
Mc. Cabe Thiele Graphical Equilibrium-Stage Lm xm 2. For stripping section (II) For control surface (II) ( Vm+1 ym+1 ) Bottom plate Vb yb Lb, xb m Lm xm Vm+1 ym+1 m-1 Lm-1 xm-1 m Ch. E 334: Separation Processes Dr Saad Al-Shahrani Vm ym II Re-boiler B x. B Operation relationship

Mc. Cabe Thiele Graphical Equilibrium -Stage Ø Number of ideal plates, Mc. Cape Thiele Method When the operating lines represented by: For rectifying section For stripping section are plotted with xy equilibrium diagram, the Mc. Cabe Thiele step by step construction can be used to compute the number of ideal plates needed to accomplish a definite concentration difference in either rectifying or the stripping section Ch. E 334: Separation Processes Dr Saad Al-Shahrani
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø Constant Molal overflow For most distillation, the molar flow rates of vapor and liquid are nearly constant in each section of the column (rectifying and stripping) and the operating lines are almost straight. (note: V-L=D, L-V=B) Ø Reflux Ratio V Reflux to overhead product L Reflux to the vapor Ch. E 334: Separation Processes Top plate Dr Saad Al-Shahrani D x. D

Mc. Cabe Thiele Graphical Equilibrium -Stage In both numerator and denominator of the terms on the right hand side of the last two equations are divided by D, the result is for constant molal over flow. slope intercept This equation is the operating line of the rectifying section. The y axis intercept of this line is x. D/(RD+1) Ch. E 334: Separation Processes Dr Saad Al-Shahrani

Mc. Cabe Thiele Graphical Equilibrium -Stage x. D is set by the conditions of design RD , the reflux ratio, is an operating variable that can be controlled by adjusting the split between reflux and overhead product or by changing the amount of vapor formed in the re-boiler for a given flow rate of the over head product. Put xn= x. D xn=x. D Ln Vn+1 yn+1 Top plate L Reflux n+1 yn+1= x. D So the operating line for rectifying section intersect the diagonal at point x. D yn+1 (x. D, x. D). This is true for both partial and total condenser Ch. E 334: Separation Processes Dr Saad Al-Shahrani
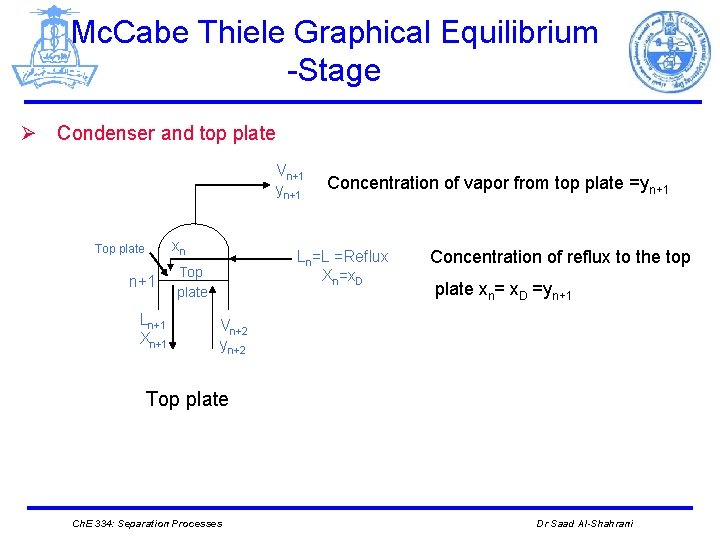
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø Condenser and top plate Vn+1 yn+1 xn Top plate n+1 Ln+1 Xn+1 Concentration of vapor from top plate =yn+1 Ln=L =Reflux Xn=x. D Top plate Concentration of reflux to the top plate xn= x. D =yn+1 Vn+2 yn+2 Top plate Ch. E 334: Separation Processes Dr Saad Al-Shahrani
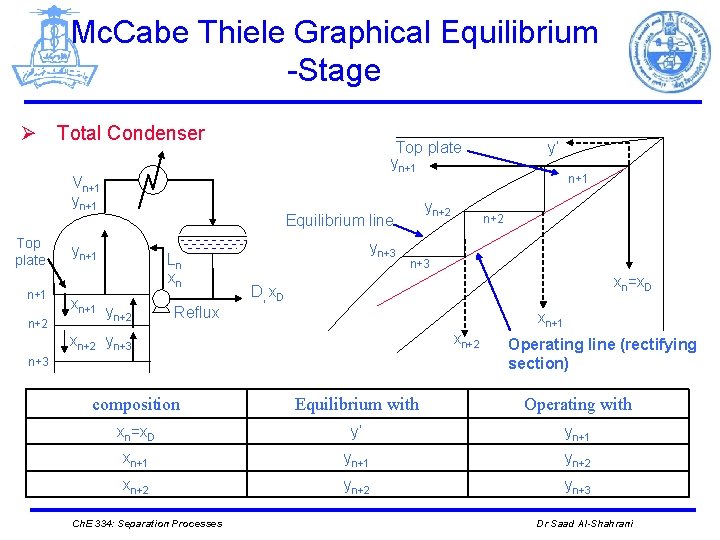
Mc. Cabe Thiele Graphical Equilibrium -Stage Ø Total Condenser Top plate yn+1 Vn+1 yn+1 Top plate n+1 n+2 yn+2 Equilibrium line yn+1 Ln xn xn+1 y n+2 Reflux yn+3 y’ n+1 n+2 n+3 xn=x. D D, x. D xn+1 xn+2 yn+3 Operating line (rectifying section) composition Equilibrium with Operating with xn=x. D y’ yn+1 xn+1 yn+2 xn+2 yn+3 Ch. E 334: Separation Processes Dr Saad Al-Shahrani
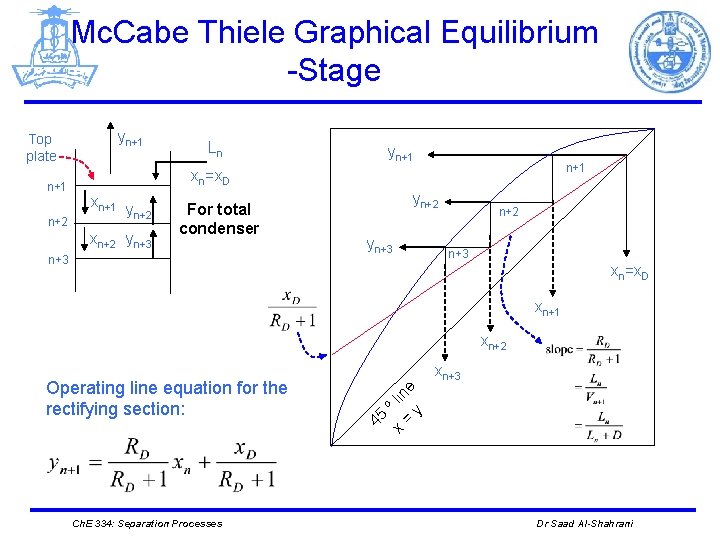
Mc. Cabe Thiele Graphical Equilibrium -Stage Top plate n+1 n+2 yn+1 Ln yn+1 xn=x. D xn+1 y n+2 xn+2 yn+3 For total condenser n+3 yn+2 yn+3 n+2 n+3 xn=x. D xn+1 xn+2 e lin = y o x 45 Operating line equation for the rectifying section: xn+3 Ch. E 334: Separation Processes Dr Saad Al-Shahrani
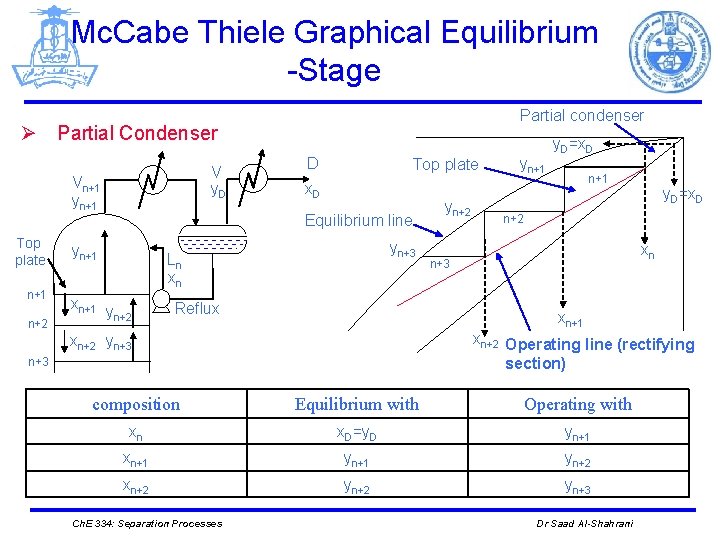
Mc. Cabe Thiele Graphical Equilibrium -Stage Partial condenser Ø Partial Condenser V y. D Vn+1 yn+1 Top plate n+1 n+2 D Top plate x. D Equilibrium line yn+1 yn+3 Ln xn xn+1 y n+2 Reflux yn+2 yn+1 y. D=x. D n+1 n+2 xn n+3 xn+1 xn+2 Operating line (rectifying section) xn+2 yn+3 composition Equilibrium with Operating with xn x. D=y. D yn+1 xn+1 yn+2 xn+2 yn+3 Ch. E 334: Separation Processes y. D=x. D Dr Saad Al-Shahrani
- Slides: 27