Binary Ionic Compounds Ionic Compounds ions of opposite

Binary Ionic Compounds

Ionic Compounds • ions of opposite charge chemically bonded together • Electrons are transferred from one element to the next – bonds form between anions (-) and cations (+) – OR bonds form between metals and nonmetals – bonded together so that the net charge (oxidation number) of the atom is zero
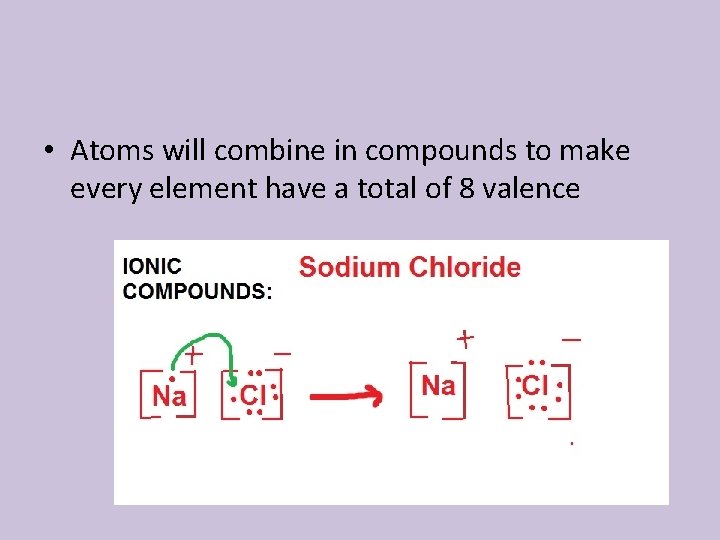
• Atoms will combine in compounds to make every element have a total of 8 valence

Basic Properties of Ionic Compounds • • Electrolytes Conduct electricity in aqueous solution Dissolve well in water Solid crystal at room temperature – Strong interparticle forces

Binary Ionic Compounds • Ionic compound formed from two elements (can contain more than one atom of each element) • Examples: – Na. Cl – Mg. Br 2

Naming Binary Ionic Compounds 1. Name metal first (positive ion) 2. Then add the name of the nonmetal (negative ion) but change the ending to “ide” – Ending is usually the last syllable Example: sodium (Na) and chlorine (Cl) Name: Sodium Chloride Chemical Formula: Na. Cl

Practice Problem 1: Name the ionic compound shown below: Mg. F 2 Magnesium fluoride

Practice Problem 2: Name the ionic compound shown below: Li 2 O Lithium oxide

Writing Chemical Formulas for Binary Ionic Compounds 1. Write the symbol for each element with it’s oxidation number (charge) – Remember: write metal first 2. Chemical formulas of ionic compounds are always neutral (so oxidation numbers must balance) – Add subscripts to the symbols for the ions until the sum of the charges is equal to zero – No subscripts are needed if only one ion is present

Example: Calcium Chloride 1. Ca+2 2. Ca. Cl 2 Cl-1

Practice Problem 3: Write the chemical formula for aluminum bromide.

Practice Problem 4: Write the chemical formula for magnesium nitride.

Practice Problem 5 Write the chemical formula for calcium oxide.
- Slides: 13