BINARY COVALENT COMPOUNDS DEFINITION Covalent bond atoms share
BINARY COVALENT COMPOUNDS
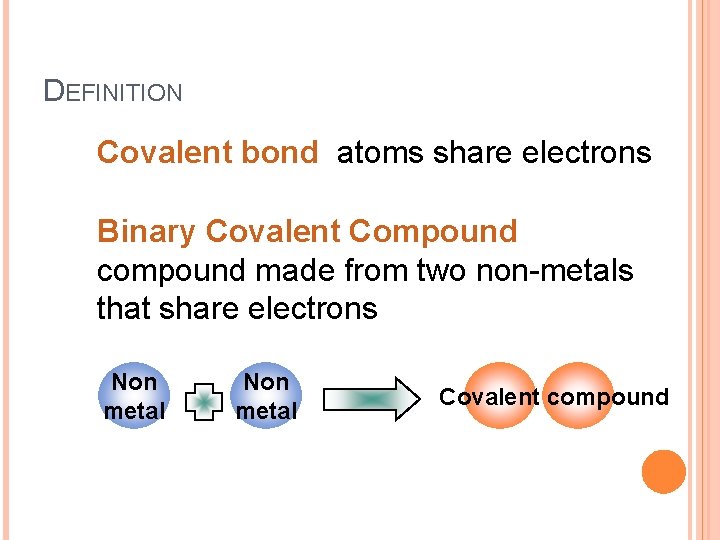
DEFINITION Covalent bond atoms share electrons Binary Covalent Compound compound made from two non-metals that share electrons Non metal Covalent compound

IDENTIFYING BINARY COVALENT COMPOUNDS l These compounds have: ¡ 2 elements (“binary”) ¡Both non-metals (“covalent”)
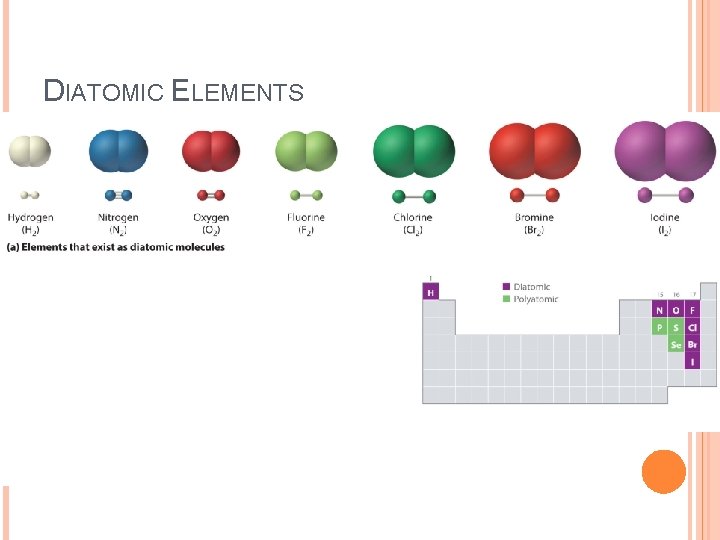
DIATOMIC ELEMENTS


COVALENT PREFIXES The Appendix of your book (Page A-2) has the following chart PREFIXES USED IN MOLECULAR COMPOUNDS 1. mono 2. di 3. tri 4. tetra 5. penta 6. hexa 7. hepta 8. octa 9. nona 10. deca-

NAMING COVALENT COMPOUNDS l Look at the subscript of the first element l If the subscript is an assumed one then just name the element l If the subscript is greater than one then use the prefix to denote the number of atoms present then name the element l Look at the subscript of the second element l Use a prefix to denote the number of atoms present, then name the element with an –ide ending.

EXAMPLE #1 P 2 O 5
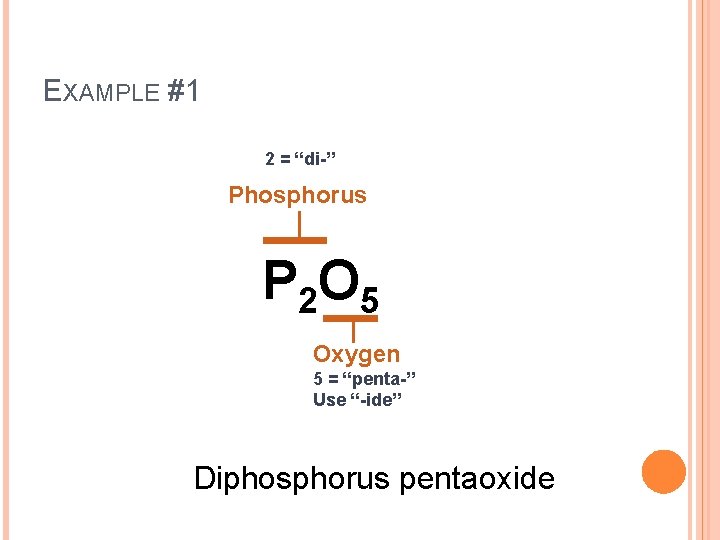
EXAMPLE #1 2 = “di-” Phosphorus P 2 O 5 Oxygen 5 = “penta-” Use “-ide” Diphosphorus pentaoxide

EXAMPLE #2 Si. O 2

EXAMPLE #2 Don’t use “mono-” on first element Silicon Si. O 2 Oxygen 2 = “di-” Use “-ide” Silicon dioxide

LET’S PRACTICE CO 2 Example: Write the name for the following compounds N 2 O 4 P 4 O 10 CO

LET’S PRACTICE Example: Write the name for the following compounds CO 2 Carbon dioxide N 2 O 4 Dinitrogen tetraoxide P 4 O 10 Tetraphosphorus decaoxide CO Carbon monoxide

WRITING FORMULAS FOR BINARY COVALENT l To write these formulas: ¡Write down the symbol of the first element. After that symbol place a subscript. The subscript is determined from the prefix. Assume the first element is “ 1” if there is no prefix. ¡Repeat this for the last element. Atoms do not form charges when bonding covalently…you DO NOT need to worry about balancing charges with this type! THESE ALL END IN -IDE

EXAMPLE #1 Dinitrogen Tetraoxide
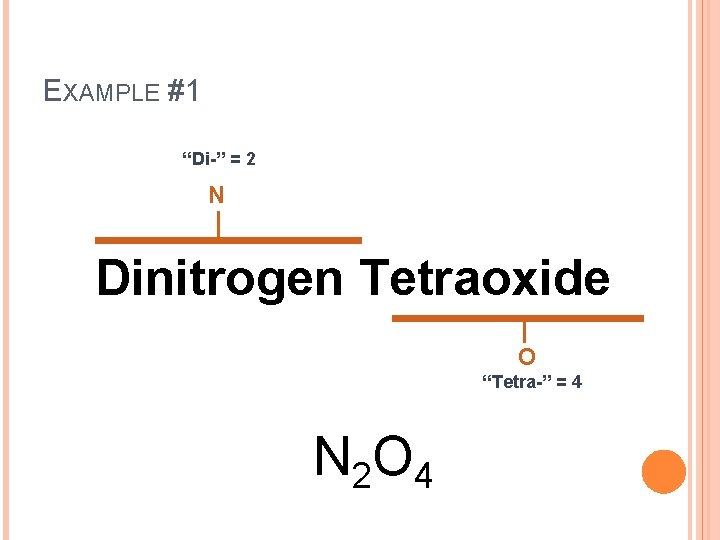
EXAMPLE #1 “Di-” = 2 N Dinitrogen Tetraoxide O “Tetra-” = 4 N 2 O 4

EXAMPLE #2 Silicon dioxide

EXAMPLE #2 “Mono-” is not written for the first element Si Silicon dioxide O “Di-” = 2 Si. O 2

LET’S PRACTICE Example: Write the following chemical formulas Carbon monoxide Nitrogen dioxide Diphosphorus pentaoxide

LET’S PRACTICE Example: Write the following chemical formulas Carbon monoxide CO Nitrogen dioxide NO 2 Diphosphorus pentaoxide P 2 O 5
- Slides: 20