Binary compound naming 1 NonMetal NonMetal 2 Metal

Binary compound naming 1. Non-Metal + Non-Metal 2. Metal + Non-Metal 3. Transition Metal + Non-Metal + NM = NM+NM = M + NM = TM The key mistake students make when naming binaries is to misidentify the class of binary being named. Classify…then name !

What is the formula and name of the binary formed from Ca and N ? A. Ca. N Nitrogen calcide B. Ca 3 N 2 tricalcium dinitride C. Ca 3 N 2 calcium nitrogen D. Ca 3 N 2 calcium nitride
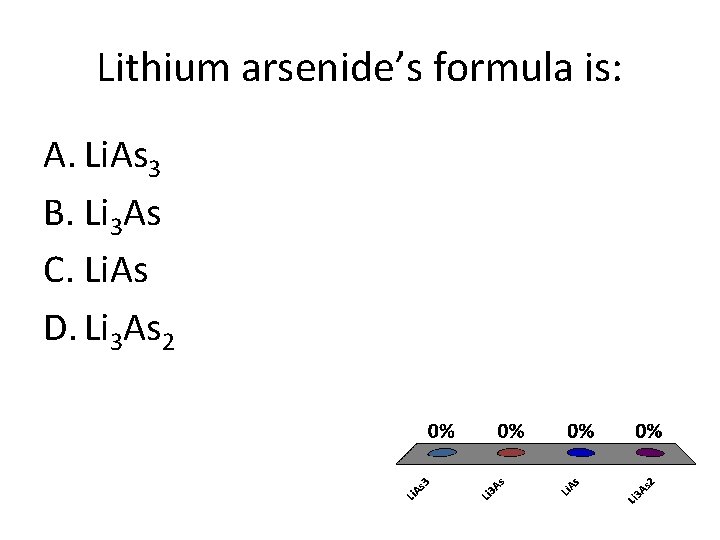
Lithium arsenide’s formula is: A. Li. As 3 B. Li 3 As C. Li. As D. Li 3 As 2

Given Mn is +5, what is the correct formula and name when it is combined with N? A. B. C. D. Mn 3 N 5, magnesium nitride Mn 5 N 3. manganese(5) nitride Mn 3 N 5 manganese (V) nitride Mn 3 N 5 trimanganese pentanitride
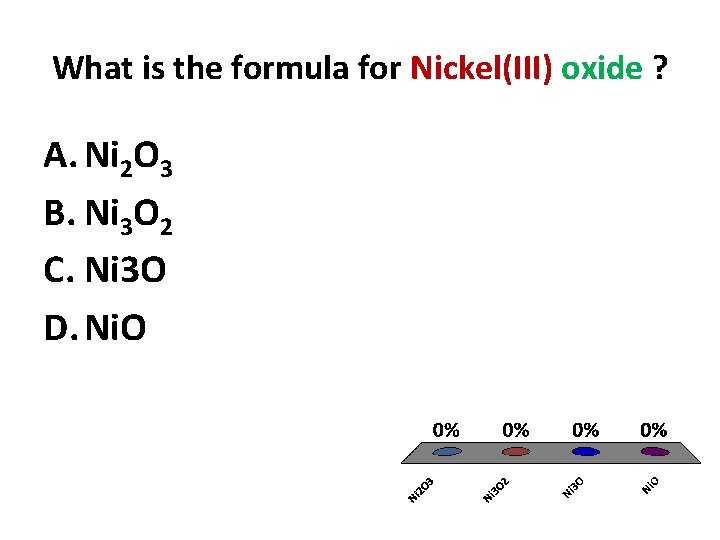
What is the formula for Nickel(III) oxide ? A. Ni 2 O 3 B. Ni 3 O 2 C. Ni 3 O D. Ni. O

What is the name for N 3 O 5 ? A. B. C. D. Nitrogen(III) oxide Trinitrogen pentoxide Nitrogen(V) Oxide

Naming `Oxyanionic’ Compounds Binary ionic compounds (M or TM) + NM Na. Cl Sodium chloride Fe. Cl 3 Iron(III) chloride Oxyanionic Compounds (M or TM) + Oxyanions Na. Cl. O 3 Sodium chlorate Fe(Cl. O 3)3 Iron(III) chlorate

An Oxyanion (abbreviated Oxy) is simply a negatively charged chemical species with several atoms glued together in a stable group. Most have lots of oxygens. Examples: NO 3 - = nitrate SO 4 2 - = sulfate PO 4 3 - = phosphate

• The naming scheme for M + Oxy and TM + Oxy is the same as M + NM and TM + NM • You will always be given the charge, formula and name for any Oxyanion (no need to memorize). (See course website: under Handouts Feb 11 )

In-class examples (from homework): name formula Lithium cyanide = ? given: cyanide = (CN)-1 M + Oxy +1 -1 Lithium cyanide = Li (CN) =Li(CN)=Li. CN 1. 2. 3. 4. Kind of name needed ? M+Oxy or TM +Oxy ? Charges are ? ? ? Cross charges to balance Remove signs (and if 1, remove 1)

Homework examples (cont. ) formula-> name: Fe(Mn. O 4)2= ? given: permanganate = Mn. O 4 -1 TM + Oxy Fe(Mn. O 4) 2 = 1 Fe + 2(Mn. O 4)-1 Iron (II) permanganate 1. Kind of name needed ? M+Oxy or TM +Oxy ? 2. Charge on Fe ? ? ? Uncross formula to find Fe charge 3. Write name with Roman Numeral for Fe charge and use given oxyanion name

Homework examples (cont. ) formula-> name: Na 3(PO 4)= ? given: phosphate=PO 43 - Guided practice… U-Do-It 1. Kind of name needed ? M+Oxy or TM +Oxy ? 2. Charge on Na ? ? ? Uncross formula to find Na charge (or use Periodic Table here) 3. Write name using M + Oxy format

Homework examples (cont. ) formula-> name: Mg(OH)2= ? given: hydroxide=OH-1 Guided practice… U-Do-It (again) 1. Kind of name needed ? M+Oxy or TM +Oxy ? 2. Charge on Mg ? ? ? Uncross formula to find Mg charge (or use Periodic Table here) 3. Write name using M + Oxy format

Homework examples (cont. ) name formula: copper(II) bisulfite given: bisulfite = HSO 3 -1 Guided practice… U-Do-It (one more time) 1. 2. 3. 4. 5. Kind of compound? M+Oxy or TM +Oxy ? Charge on Cu ? ? (given…) Charge on bisulfite (givern) Write charges over Cu and Oxy Cross….

Skip ammonium sulfate and stannous fluoride on homework…free points

The chemistry road trip so far: • Pictures and pieces of that atom • Elements : their names and symbols • Periodic Table: Why it’s cool & its’ geography • Using the Table to build and name simple compounds Next stop: Chemistry by the numbers: Units, conversions and scientific notation (read sections 1. 3 and 1. 5)

Fundamental SI (le Systeme International) Units (see Table 1. 1, p. 9) Basic biggies to know • • +Chemistry add-on Length (meters, m) Mass (kilograms, kg) time (seconds, s) Temperature (Kelvin, K) `mks’ • count of stuff (moles, mol)

The need for something besides decimal numbers • Width of a human blood cell ~0. 000002 m • time for computer to do a single operation ~ 0. 00003 s • Nimitz class aircraft carrier weighs ~ 100, 000 kg • Distance to Sun~13, 000, 000 m Are there easier ways to keep track of the zeroes and the decimal place?
- Slides: 18