Billion The British say that a billion is

Billion: The British say that a billion is a million (1, 000, 000). American say that a billion is a thousand million (1, 000, 000) and insist that a million is actually a trillion. The Canadian Press agrees with the Americans http: //www. scit. wlv. ac. uk/~jphb/american. html Please note that "tonne" is not a British spelling of "ton" but a quite separate metric unit equal to 1000 kg as distinct from the British ton of 2240 lbs (= 1016. 96 kg). Billion: thousand million The old British usage in which a billion was a million 2 is now largely obsolete and most British speakers would assume the American meaning. Careful users avoid the words altogether and use exponent notation. The usage continued ·trillion = tri+(m)illion = million 3 = 1018 ·quadrillion = quad+(m)illion = million 4 = 1024 ·centillion = cent+(m)illion = million 100 = 10600 CHBI 201 The American naming seems to work on the principle 10 3+(number× 3) 1

CHAPTER 4 MATERIAL BALANCES CHBI 201 2
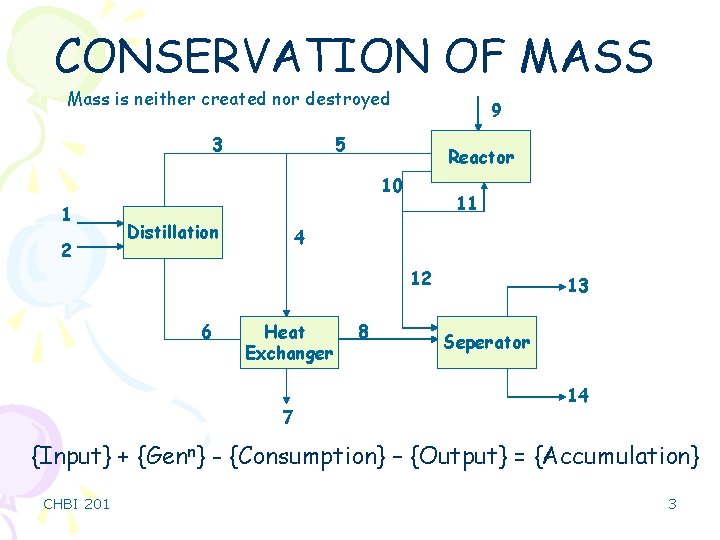
CONSERVATION OF MASS Mass is neither created nor destroyed 3 9 5 Reactor 10 1 2 Distillation 11 4 12 6 Heat Exchanger 7 8 13 Seperator 14 {Input} + {Genn} - {Consumption} – {Output} = {Accumulation} CHBI 201 3

SYSTEMS q Systems ü OPEN or CLOSED ü Any arbitrary portion of or a whole process that you want to consider for analysis ü Reactor, the cell, mitochondria, human body, section of a pipe q Closed System ü Material neither enters nor leaves the system ü Changes can take place inside the system q Open System ü Material can enter through the boundaries CHBI 201 4

STEADY-STATE q Steady-State ü Nothing is changing with time ü @ steady-state accumulation = 0 100 kg/min H 2 O 500 kg H 2 O 100 kg/min H 2 O Rate of addition = Rate of removal q Unsteady-State (transient system) ü {Input} ≠ {Output} CHBI 201 5
PROCESSES q Batch Process ü Feed is fed at the beginning of the process q Continuous Process ü The input and outputs flow continuously throughout the duration of proces q Semibatch Process ü Any process neither batch nor continuous CHBI 201 6
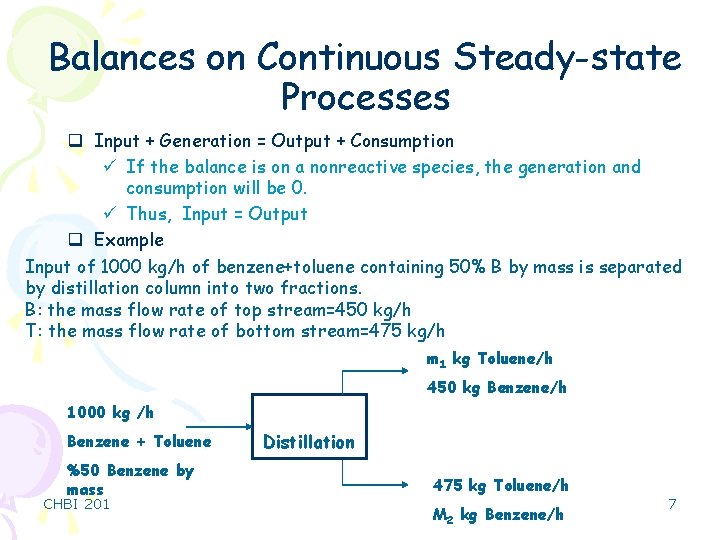
Balances on Continuous Steady-state Processes q Input + Generation = Output + Consumption ü If the balance is on a nonreactive species, the generation and consumption will be 0. ü Thus, Input = Output q Example Input of 1000 kg/h of benzene+toluene containing 50% B by mass is separated by distillation column into two fractions. B: the mass flow rate of top stream=450 kg/h T: the mass flow rate of bottom stream=475 kg/h m 1 kg Toluene/h 450 kg Benzene/h 1000 kg /h Benzene + Toluene %50 Benzene by mass CHBI 201 Distillation 475 kg Toluene/h M 2 kg Benzene/h 7

Balances on Continuous Steady-state Processes q Solution of the example q Benzene balance Input = Output . 1000 kg/h · 0. 5 = 450 kg/h + m 2 = 50 kg/h Benzene q Toluene balance . 1000 kg/h · 0. 5 = 475 kg/h + m 1 = 25 kg/h Toluene CHBI 201 8
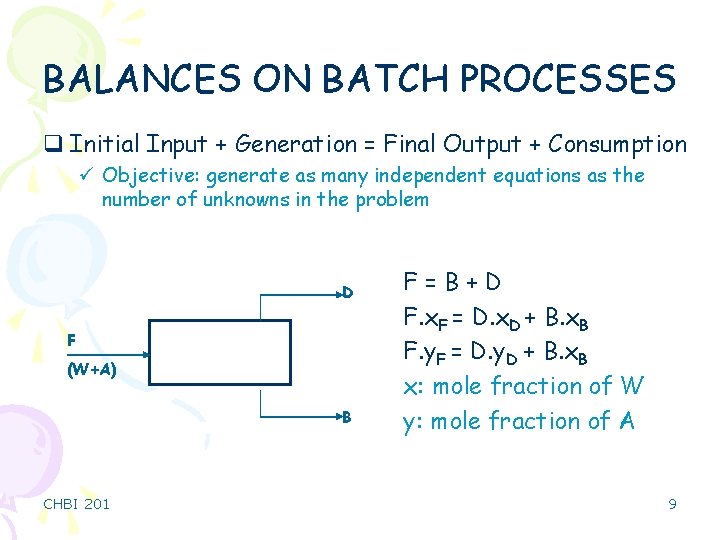
BALANCES ON BATCH PROCESSES q Initial Input + Generation = Final Output + Consumption ü Objective: generate as many independent equations as the number of unknowns in the problem D F (W+A) B CHBI 201 F=B+D F. x. F = D. x. D + B. x. B F. y. F = D. y. D + B. x. B x: mole fraction of W y: mole fraction of A 9

EXAMPLE (Batch Process) q Centrifuges are used to seperate particles in the range of 0. 1 to 100 µm in diameter from a liquid using centrifugal force. Yeast cells are recovered from a broth ( a mix with cells) using tubular centrifuge. Determine the amount of the cell-free discharge per hour if 1000 L/hr is fed to the centrifuge, the feed contains 500 mg cells/L, and the product stream contains 50 wt% cells. Assume that the feed has a density of 1 g/cm 3. Feed (broth) 1000 L/hr 500 mg cells/L feed ( d= 1 g/cm 3) CHBI 201 Centrifuge Concantrated cells P(g/hr) 50 % by weight cells Cell-free discahrge D(g/hr) 10

EXAMPLE (Batch Process) q Centrifuges are used to seperate particles in the range of 0. 1 to 100 µm in diameter from a liquid using centrifugal force. Yeast cells are recovered from a broth ( a mix with cells) using tubular centrifuge. Determine the amount of the cell-free discharge per hour if 1000 L/hr is fed to the centrifuge, the feed contains 500 mg cells/L, and the product stream contains 50 wt% cells. Assume that the feed has a density of 1 g/cm 3. Feed (broth) 1000 L/hr 500 mg cells/L feed (d= 1 g/cm 3) Centrifuge Concantrated cells P(g/hr) 50 % by weight cells Cell-free discharge D(g/hr) q Cell balance q Fluid balance Input: (106 – 500) g/h fluid Output 1: 1000 g/h. 0. 5 = 500 g/h fluid Output 2: D(g/h) = (106 – 500)g/h – 500 g/h = (106 -103)g/h fluid CHBI 201 11

FLOW CHARTS q Boxes and other symbols are used to represent process units. ü Write the values and units of all known streams ü Assign algebraic symbols to unknown stream variables 100 mol C 3 H 8 Combustion Chamber Condenser 50 mol C 3 H 8 750 mol O 2 1000 mol O 2 3760 mol N 2 150 mol CO 2 200 mol H 2 O CHBI 201 12

EXAMPLE (Flow charts) q Humidification and Oxygenation Process in the Body: An exp. on the growth rate of certain organisms requires an environemnt of humid air enriched in oxygen. Three input streams are fed into an evaporator to produce an output stream with the desired composition. A: liquid water, fed at a rate of 20 cm 3/min, B: Air, C: Pure oxygen (with a molar flow rate one-fifth of the molar flow rate of stream B) . n . 0. 2 n mol O /min 1 3 2 0. 015 mol H 2 O/mol C . n 1 mol air/min CHBI 201 mol/min y mol O 2/mol (0. 985 – y ) mol N 2/mol B A 0. 21 mol O 2/mol 20 cm 3 H 2 O /min 0. 79 mol N 2/mol n 2 mol H 2 O/min . 13

EXAMPLE (Flow chart) n 2 = 20 cm 3 H 2 O/min. 1 g H 2 O/cm 3. 1 mol/18. 02 g n 2 = 1. 11 mol H 2 O/min q H 2 O Balance n 2 mol H 2 O/min = n 3 mol/min. 0. 015 mol H 2 O/mol n 3 = 74. 1 mol/min q Total Mole Balance 0. 2 n 1 + n 2 = n 3 n 1 = 60. 8 mol/min q N 2 Balance n 1 mol/min. 0. 79 mol N 2/mol = n 3 mol/min. (0. 985 -y) mol N 2/mol y = 0. 337 mol O 2/mol CHBI 201 14
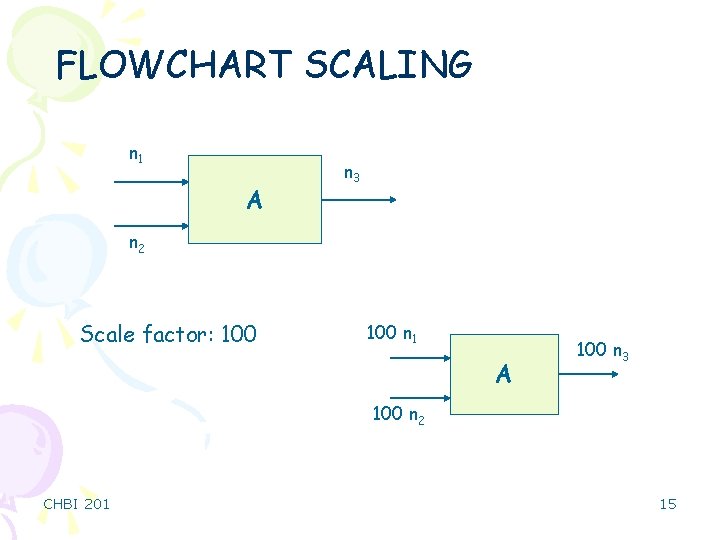
FLOWCHART SCALING n 1 A n 3 n 2 Scale factor: 100 n 1 A 100 n 3 100 n 2 CHBI 201 15

DEGREE OF FREEDOM ANALYSIS (df) q ndf = nunknowns – nindep. eqn’s q If ndf = 0 ü Problem can be solved (determined) q If ndf > 0 ü Unknowns > knowns (underspecified) q If ndf < 0 ü overspecified (no solution) q q q CHBI 201 Material balances, Energy balances, Process specificaitons, Physical props&laws, Physical constraints 16
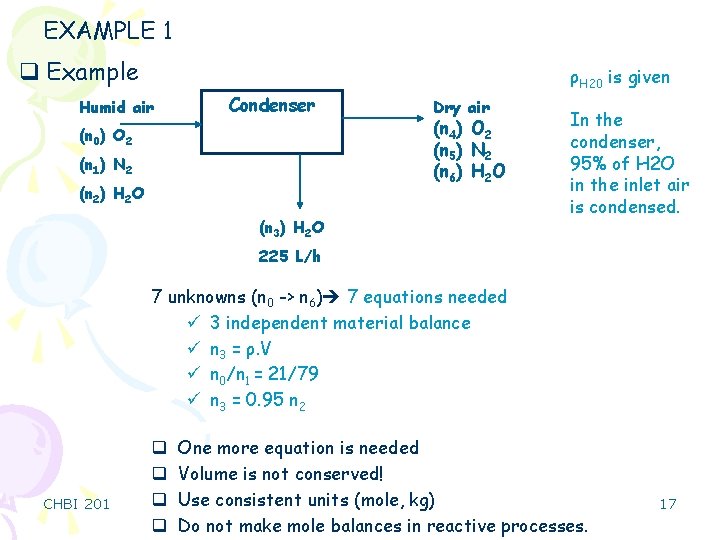
EXAMPLE 1 q Example Humid air Condenser (n 0) O 2 (n 1) N 2 (n 2) H 2 O ρH 20 is given Dry air (n 4) O 2 (n 5) N 2 (n 6) H 2 O (n 3) H 2 O In the condenser, 95% of H 2 O in the inlet air is condensed. 225 L/h 7 unknowns (n 0 -> n 6) 7 equations needed ü 3 independent material balance ü n 3 = ρ. V ü n 0/n 1 = 21/79 ü n 3 = 0. 95 n 2 CHBI 201 q q One more equation is needed Volume is not conserved! Use consistent units (mole, kg) Do not make mole balances in reactive processes. 17

EXAMPLE 2 q A continuous mixer mixes Na. OH with H 2 O to produce an aqueous solution of Na. OH. Determine the composition and flow rate of the product, if the flow rate of Na. OH is 1000 kg/hr and the ratio of the flow rate of H 2 O to the product solution is 0. 9. Nsp = number of species Ns = number of streams Nu = total number of variables System boundary H 2 O M Na. OH Product CHBI 201 18

EXAMPLE 2 - continue Streams FEED WATER PRODUCT Species Na. OH FNa. OH WNa. OH PNa. OH H 2 O FH 2 O WH 2 O PH 2 O Total F W P Nu = 3(2+1) = 9 Last row in the table Specifications: ratio of two streams the % conversion in a reaction the value of each concentration, flow rate, T, P, ρ, V, etc. CHBI 201 a variable is not present in a stream, hence , it is 0 19

EXAMPLE 3 q A cylinder containing CH 4, C 2 H 6, and N 2 has to be prepared containing a CH 4 to C 2 H 6 mole ratio of 1. 5 to 1. Avaliable to prepare the mixture are 1) a cylinder containing a mixture of 80% N 2 and 20% CH 4 2) a cylinder containing a mixture of 90% N 2 and 10% C 2 H 6 3) a cylinder containing a mixture of pure N 2 What is the number of degrees of freedom? CHBI 201 20
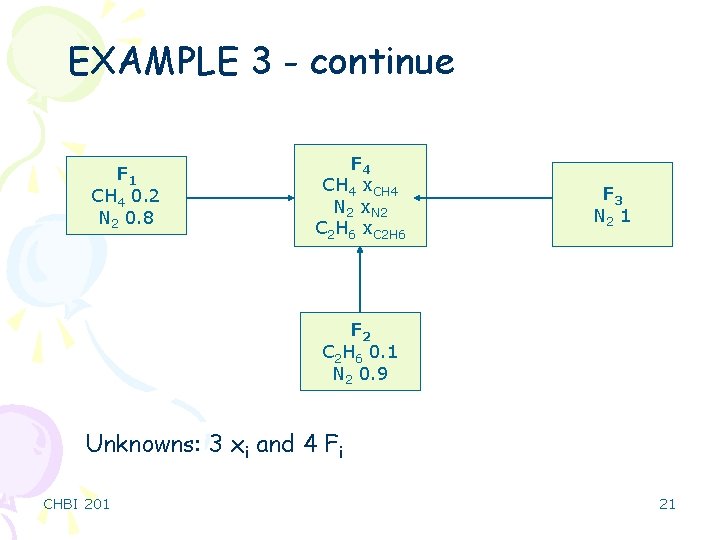
EXAMPLE 3 - continue F 1 CH 4 0. 2 N 2 0. 8 F 4 CH 4 x. CH 4 N 2 x. N 2 C 2 H 6 x. C 2 H 6 F 3 N 2 1 F 2 C 2 H 6 0. 1 N 2 0. 9 Unknowns: 3 xi and 4 Fi CHBI 201 21

EXAMPLE 3 - continue Equations: ü Material balance (CH 4, C 2 H 6, N 2) ü One specified ratio x. CH 4/x. C 2 H 6 = 1. 5 ü One summation of mole fractions ü 5 independent equations Ndf = 7 – 5 = 2 If you pick a basis as F 4=1, one other value has to be specified in order to solve the problem. CHBI 201 22
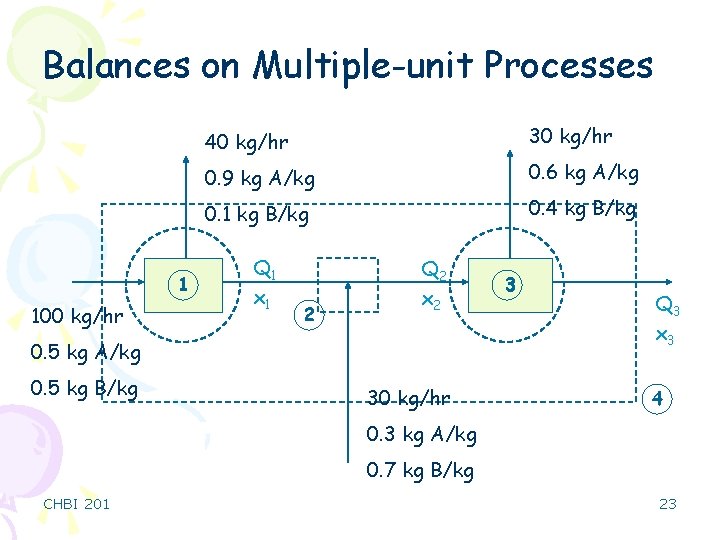
Balances on Multiple-unit Processes 1 100 kg/hr 40 kg/hr 30 kg/hr 0. 9 kg A/kg 0. 6 kg A/kg 0. 1 kg B/kg 0. 4 kg B/kg Q 1 x 1 2 Q 2 x 2 0. 5 kg A/kg 0. 5 kg B/kg 30 kg/hr 3 Q 3 x 3 4 0. 3 kg A/kg 0. 7 kg B/kg CHBI 201 23
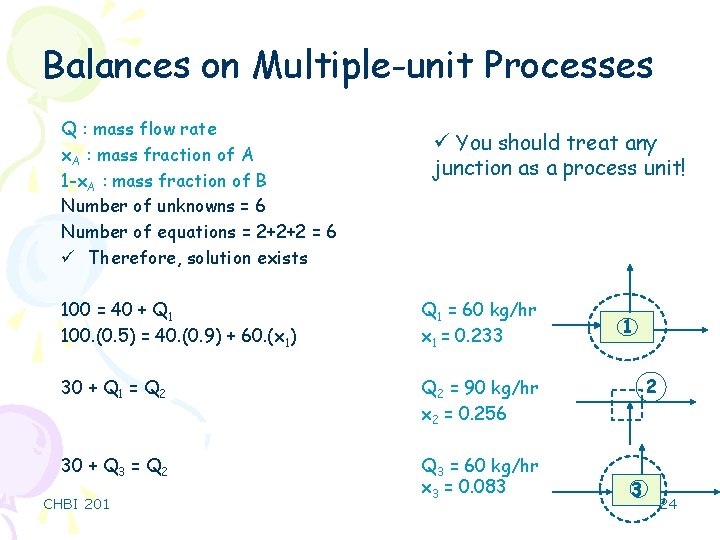
Balances on Multiple-unit Processes Q : mass flow rate x. A : mass fraction of A 1 -x. A : mass fraction of B Number of unknowns = 6 Number of equations = 2+2+2 = 6 ü Therefore, solution exists ü You should treat any junction as a process unit! 100 = 40 + Q 1 100. (0. 5) = 40. (0. 9) + 60. (x 1) Q 1 = 60 kg/hr x 1 = 0. 233 30 + Q 1 = Q 2 = 90 kg/hr x 2 = 0. 256 30 + Q 3 = Q 2 Q 3 = 60 kg/hr x 3 = 0. 083 CHBI 201 1 2 3 24

CHBI 201 25
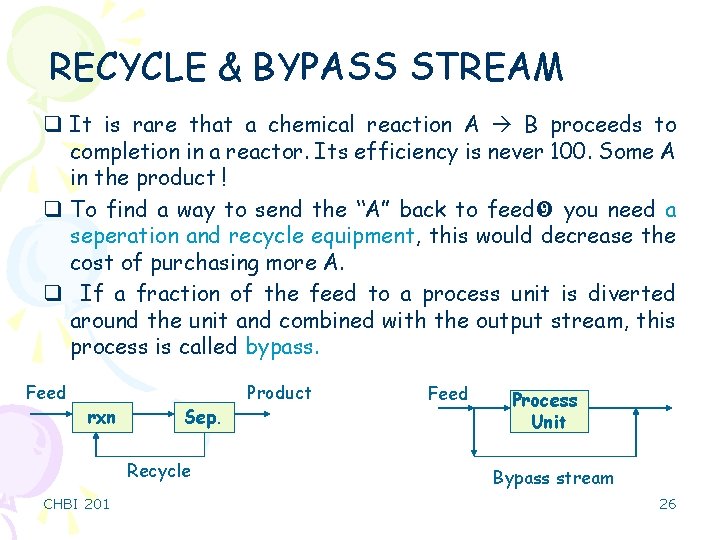
RECYCLE & BYPASS STREAM q It is rare that a chemical reaction A B proceeds to completion in a reactor. Its efficiency is never 100. Some A in the product ! q To find a way to send the “A” back to feed you need a seperation and recycle equipment, this would decrease the cost of purchasing more A. q If a fraction of the feed to a process unit is diverted around the unit and combined with the output stream, this process is called bypass. Feed rxn Sep. Recycle CHBI 201 Product Feed Process Unit Bypass stream 26

EXAMPLE (pg 110) q Feed: Fresh air with 4 mole% H 2 O(v) is “cooled” and “dehumidified” to a water content of 1. 7 mole% H 2 O. Fresh air is combined with a recycle stream of dehumidified air. The blended stream entering unit contains 2. 3 mole% H 2 O. In the air conditioner some of the water is removed as liquid. Take 100 mole of dehumidified air delivered to the room, calculate moles of feed, water condensed, dehumidified air recycled. CHBI 201 27
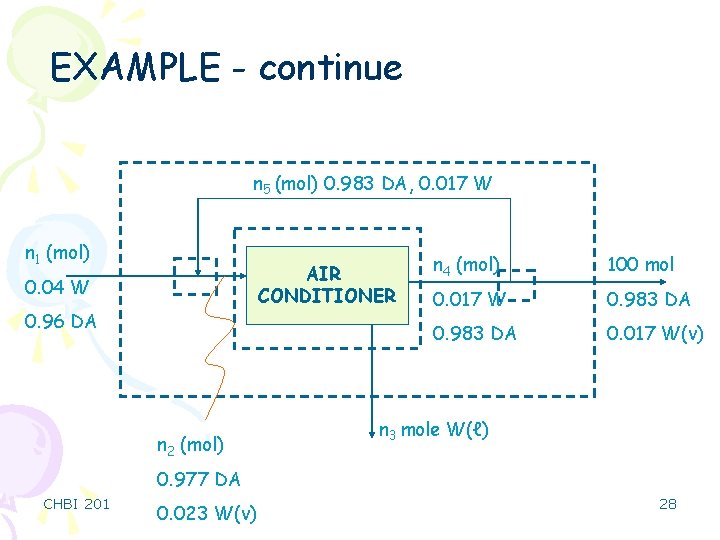
EXAMPLE - continue n 5 (mol) 0. 983 DA, 0. 017 W n 1 (mol) AIR CONDITIONER 0. 04 W 0. 96 DA n 2 (mol) n 4 (mol) 100 mol 0. 017 W 0. 983 DA 0. 017 W(v) n 3 mole W(ℓ) 0. 977 DA CHBI 201 0. 023 W(v) 28

EXAMPLE - continue q Overall system: 2 variables (n 1, n 3) 2 balance equations (two species) Degree of freedom = 0 (n 1, n 3) are determined!!! q Mixing point: 2 variables (n 2, n 5) 2 balance equations (two species) Degree of freedom = 0 q Cooler: 2 variables (n 2, n 4) 2 balance equations (two species) Degree of freedom = 0 q Splitting point: 2 variables (n 4, n 5) Donot use SP in the 1 balance equation Degree of freedom = 1 CHBI 201 solution 29

EXAMPLE - continue Overall DA balance: 0. 96 n 1 = 0. 983 (100) n 1 = 102. 4 mol fresh feed Overall mole balance: n 1 = n 3 + 100 n 3 = 2. 4 mol H 2 O condensed Mole balance on Mixing point: n 1 + n 5 = n 2 Water blance on Mixing point: 0. 04 n 1 + 0. 017 n 5 = 0. 023 n 2 = 392. 5 mol n 5 = 290 mol recycled CHBI 201 30

CHEMICAL REACTION STOICHIOMETRY q If there is a chemical reaction in a process More complications q The stoichiometric ratios of the chemical reactions Constraints q The stoichiometric equation 2 SO 2 + O 2 2 SO 3 2 molecules of SO 2 reacts with 1 molecule of O 2 and yields 2 molecules of SO 3 q 2, 1 and 2 are stoichiometric coefficients of a reaction CHBI 201 31

LIMITING & EXCESS REACTANTS q If the reactants are not in stoichiometric proportion one of them will be excess, the other will be limiting CHBI 201 32
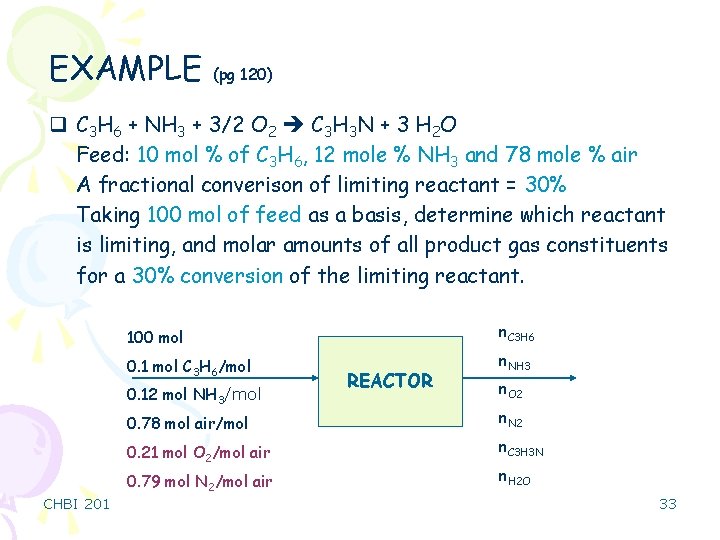
EXAMPLE (pg 120) q C 3 H 6 + NH 3 + 3/2 O 2 C 3 H 3 N + 3 H 2 O Feed: 10 mol % of C 3 H 6, 12 mole % NH 3 and 78 mole % air A fractional converison of limiting reactant = 30% Taking 100 mol of feed as a basis, determine which reactant is limiting, and molar amounts of all product gas constituents for a 30% conversion of the limiting reactant. 100 mol n. C 3 H 6 0. 1 mol C 3 H 6/mol n. NH 3 0. 12 mol NH 3/mol n. O 2 0. 78 mol air/mol n. N 2 0. 21 mol O 2/mol air n. C 3 H 3 N 0. 79 mol N 2/mol air CHBI 201 REACTOR n. H 2 O 33

EXAMPLE – continue Feed: n. C 3 H 6= 10 mole n. NH 3=12 mole n. O 2= 78. (0. 21) =16. 4 mole n. NH 3/n. C 3 H 6= 12/10 = 1. 2 NH 3 is excess (stoich. 1) n. O 2/n. C 3 H 6= 16. 4/10 = 1. 64 O 2 is excess (stoich. 1. 5) (n. NH 3)stoich. = 10 mole (n. O 2)stoich. = 15 mole (% excess)NH 3 = (12 -10) /10 x 100 = 20% excess NH 3 (% excess)O 2 = (16. 4 -15) /15 x 100 = 9. 3% excess O 2 Moles reacted Moles fed (n. C 3 H 6)out=0. 7 x (n. C 3 H 6)0= 7 mole C 3 H 6 (since the fractional conversion of C 3 H 6 is 30%) Extent of reaction = ζ = 3 mole (since ni = ni 0 + ni ξ => 7= 10 - 1. ξ) n. NH 3 = 12 - ζ =9 mole n. O 2=16. 4 – 1. 5. (ζ)= 11. 9 n. C 3 H 3 N= ζ = 3 mole n. H 2 O=3. (ζ) = 9 mole n. N 2= (n. N 2)0=61. 6 mole CHBI 201 34

CHEMICAL EQUILIBRIUM q If you are given a set of reactive species and reaction conditions; a) What will be the final (equilibrium) composition of the reaction mixture? b) How long will the system take to reach a specified state short of equilibrium? q Chemical equilibrium thermodynamics & Chemical Kinetics q A reaction can be ü Reversible ü Irreversible CHBI 201 35

EXAMPLE CO (g) + H 2 O (g) CO 2 (g) + H 2 (g) Given @ T=1105 K, K=1 n. CO= 1 mol, n. H 2 O= 2 mol, initially no CO 2 and H 2 Calculate the equilibrium composition and the fractional converison of the limiting reactant. Equilibrium constant; K(T) = CHBI 201 36

EXAMPLE – continue n. CO = 1 -ζe , n. H 2 O = 2 -ζe , n. CO 2 = ζe , n. H 2 = ζe y. CO = (1 -ζe)/3 y. H 2 O = (2 -ζe)/3 y. CO 2 = ζe /3 y. H 2 = ζe /3 K(T) = (ζe)2 / (1 -ζe) (2 -ζe) = 1 ζe = 0. 667 mole y. CO = 0. 111 y. H 2 O = 0. 444 y. CO 2 = 0. 222 y. H 2 = 0. 222 Limiting reactant is CO. n. CO = 1 -0. 667 = 0. 333 Fractional conversion = (1 -0. 333) / 1 mol feed = 0. 667 CHBI 201 37
- Slides: 37