BIHAR ANIMAL SCIENCES UNIVERSITY BIHAR VETERINARY COLLEGE PATNA

BIHAR ANIMAL SCIENCES UNIVERSITY BIHAR VETERINARY COLLEGE, PATNA DEPARTMENT OF ANIMAL NUTRITION PROTEIN) ANN-601 UNIT-I (ANIMAL NUTRITION- ENERGY AND LECTURE ON BASIC TERMINOLOGY AND PROTEIN, AMINO ACID. 05. 10. 20 DR. PRAMOD KUMAR ASSISTANT PROFESSOR-CUM-JR. SCIENTIST BIHAR VETERINARY COLLEGE, BASU, PATNA-14

PROTEIN • The term Protein was given by G. j. Mulder in 1838. • Proteins are complex organic compounds of high molecular weight. In common with carbohydrates and fats they contain carbon, hydrogen and oxygen, but in addition they all contain nitrogen and generally Sulphur. Percent Carbon 51. 0 -55. 0 Hydrogen 6. 5 -7. 3 Oxygen 21. 5 -23. 5 Nitrogen 15. 5 -18. 0 Sulphur 0. 5 -2. 0 Phosphorous 0. 0 -1. 5

AMINO ACID • Proteins are polymers of amino acids which vary in relative amounts and kind from protein to protein. • These amino acids are obtained as hydrolytic end products, when proteins are heated with strong acids or when they are acted upon by certain enzymes. • There are some 20 to 22 different amino acids present in proteins; although in nature over 150 other amino acids exist that never are part of proteins. • The amino acids are derivatives of the short-chain fatty acids and contain a basic amino group (-NH 2) and an acidic carboxyl group (-COOR). • The amino group is attached to the α carbon atom (α -amino acids), and in nature the amino acids generally assume the L configuration, when compared with L. glycerose. • Most are soluble in water and all but glycine are optically active. Because they have both an amino group and a carboxyl group, they are amphoteric, assuming acidic or basic properties depending upon the p. H of the medium. • In acid p. H, the amino acid is a cation; in basic p. H it is an anion; at the p. H at which it is electrically neutral it is dipolar and called a "zwitterion. " At this point the p. H is termed the isoelectric point for that amino acid.
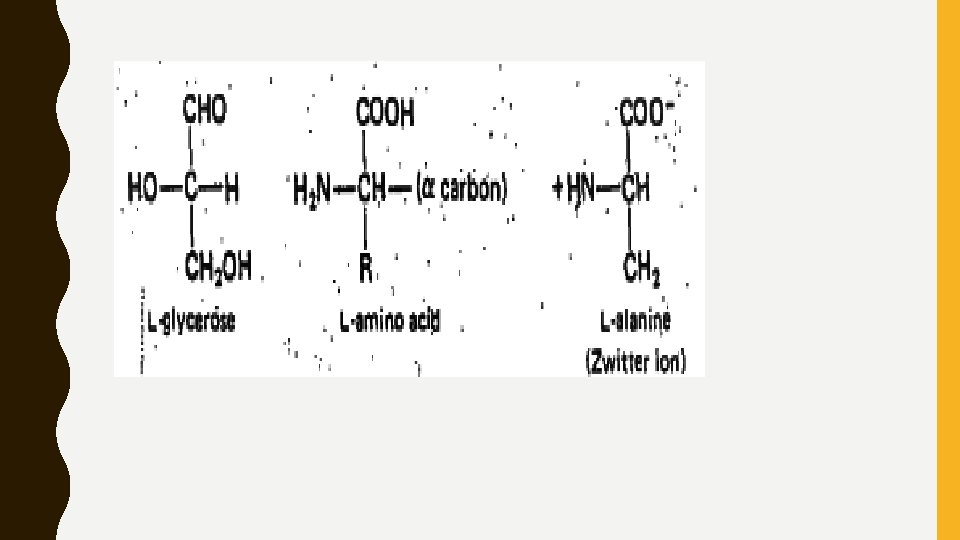

PHYSICAL PROPERTIES OF AMINO ACID • They are colourless, crystalline substances generally soluble in water, slightly soluble in alcohol and insoluble in ether. • The presence of both acidic and basic groups which are readily ionizable, the amino acids, behave as amphoteric electrolytes and give both anions and cations in solution(Zwitterion). • The p. H at which an amino acid exists in solution as a Zwitterion is called Isoelectric p. H. • The isoelectric p. H for Neutral acids 4. 8 to 6. 3 Acidic acids - 2. 8 to 3. 2 Basic amino acid 7. 6 to 10. 8

CHEMICAL PROPERTIES OF AMINO ACID DUE TO –COOH GROUP DUE TO –NH 2 GROUP • They can form esters with alcohols and salts with bases. • They can form salts with acids. • With ammonia, they form the corresponding amides. The amides of aspartic and glutamic acids-asparagine and glutamine are of importance in the transport of ammonia in the body. • Nitrous acid will react with a amino group to liberate nitrogen and form the hydroxyl acid. • Hydrazine will cleave all peptide bonds and convert all the amino acids to the corresponding hydrazides. • The amino group can be methylated. • Two amino acids interact by the amino group of one of them combining with the carboxylic group of the other through peptide bond.

ESSENTIAL AMINO ACIDS • Plants and many microorganisms are able to synthesise proteins from simple nitrogenous compounds such as nitrates. • Animals cannot synthesise the amino group, and in order to build up body proteins they must have a dietary source of amino acids. Certain amino acids can be produced from others by a process known as transamination. • The carbon skeletons of a number of amino acids cannot be synthesised in the animal body; these are referred to as essential or indispensible amino acids. • The chick requires a dietary supply of all ten amino acids but in addition needs a dietary source of glycine. Birds require arginine because their metabolism does not include the urea cycle, which would normally supply this amino acid.
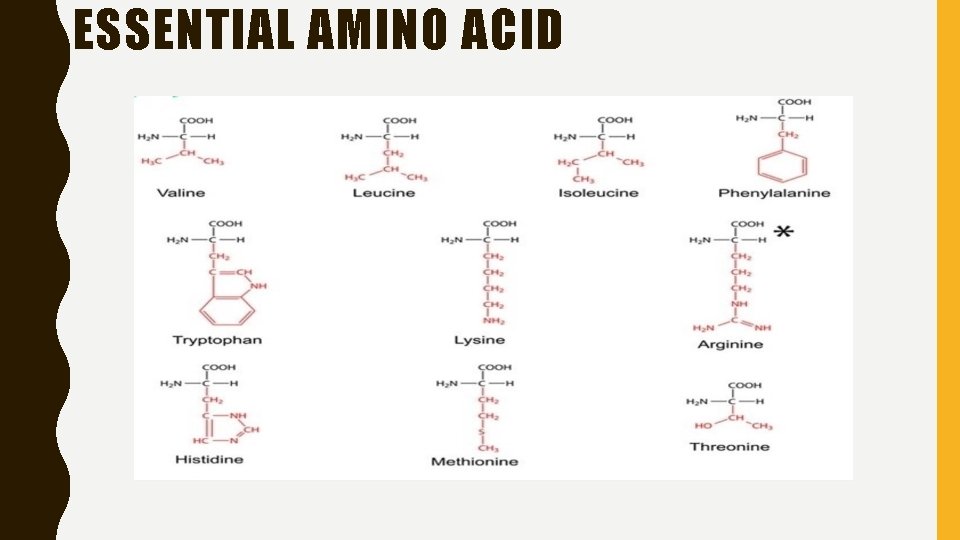
ESSENTIAL AMINO ACID

• The list of essential amino acids required by the pig is similar to that for the rat, with the exception of arginine, which can be synthesised by the pig. • Cats require a dietary supply of arginine, owing to their limited ability to synthesise ornithine from glutamate, and a deficiency of arginine results in the accumulation of ammonia from denatured amino acids in the blood. • Cats also require the β-sulphonic amino acid taurine in their diet as they are Peptides unable to synthesise this from cysteine. Taurine is required for bile acid conjugation. • Poultry have a limited capacity to synthesise proline.

• In the case of the ruminant, all the essential amino acids can be synthesised by the rumen microorganisms, which theoretically makes this class of animal independent of a dietary source once the rumen microorganisms have become established. • The biological value of microbial protein is limited by its content of certain essential amino acids, particularly lysine and methionine. • For maximum productivity the microbial protein must be supplemented with a supply of dietary amino acids, from foods or synthetic amino acids, in a suitable form that is not degraded by the microorganisms in the rumen.

PEPTIDES • Peptides are built up from amino acids by means of a linkage between the α-carboxyl of one amino acid and the α-amino group of another acid, as shown here: • This type of linkage is known as the peptide linkage; in the example shown, a dipeptide has been produced from two amino acids. Large numbers of amino acids can be joined together by this means, with the elimination of one molecule of water at each linkage, to produce polypeptides.

• Besides being important building blocks in the construction of proteins, some peptides possess their own biological activity. Milk, in particular, is a source of many biologically active peptides. • The enzymatic hydrolysis of the milk protein casein releases opioid peptides, which have pharmacological activities such as analgesia and sleep-inducing effects. • Other peptides derived from casein are involved in calcium flow in tissues and modification of the immune system response. • Other milk peptides stimulate growth of desirable bacteria and suppress harmful bacteria, and some act as growth factors for intestinal cells. • Peptides play an important role in the flavour and sensory properties of foods such as yeast extract, cheese and fruit juices.

STRUCTURE OF PROTEINS One the basis of peptide chain structure of protein divided into four type. 1. Primary Structure- The sequence of amino acids along the polypeptide chain of a protein, as described above, is called the primary structure of the protein. 2. Secondary structure - The secondary structure of proteins refers to the conformation of the chain of amino acids resulting from the formation of hydrogen bonds between the imino and carbonyl groups of adjacent amino acids. Ø The secondary structure may be regular, in which case the polypeptide chains exist in the form of an αhelix or a β-pleated sheet, or it may be irregular and exist as, for example, a random coil.
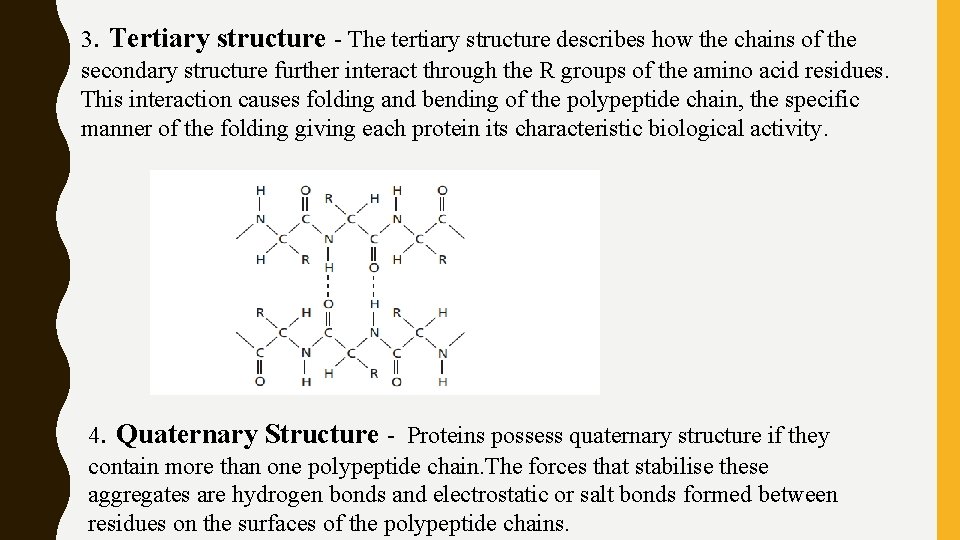
3. Tertiary structure - The tertiary structure describes how the chains of the secondary structure further interact through the R groups of the amino acid residues. This interaction causes folding and bending of the polypeptide chain, the specific manner of the folding giving each protein its characteristic biological activity. 4. Quaternary Structure - Proteins possess quaternary structure if they contain more than one polypeptide chain. The forces that stabilise these aggregates are hydrogen bonds and electrostatic or salt bonds formed between residues on the surfaces of the polypeptide chains.
Thanks
- Slides: 15