BIG IDEA II Biological systems utilize free energy

BIG IDEA II Biological systems utilize free energy and molecular building blocks to grow, to reproduce and to maintain dynamic homeostasis. Enduring Understanding 2. A Growth, reproduction and maintenance of the organization of living systems require free energy and matter. Essential Knowledge 2. A. 2 Organisms capture and store free energy for use in biological processes. Power. Point® Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Essential Knowledge 2. A. 2: Organisms capture and store free energy for use in biological processes. • Learning Objectives: – (2. 4) The student is able to use representations to pose scientific questions about what mechanisms and structural features allow organisms to capture, store and use free energy. – (2. 5) The student is able to construct explanations of the mechanisms and structural features of cells that allow organisms to capture, store or use free energy. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Autotrophs capture free energy from physical sources in the environment. • Photosynthetic organisms capture free energy present in sunlight. – 6 CO 2 + 6 H 2 O + light energy C 6 H 12 O 6 + 6 O 2 + 6 H 2 O – carbon dioxide + water + light energy sugar + oxygen + water • Chemosynthetic organisms capture free energy from small inorganic molecules present in their environment, and this process can occur in the absence of oxygen. – 6 H 2 S + 6 H 2 O + 6 CO 2 + 6 O 2 C 6 H 12 O 6 + 6 H 2 SO 4 – hydrogen sulfide + water + carbon dioxide + oxygen sugar + sulfuric acid Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Autotrophs capture free energy from physical sources in the environment. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Heterotrophs capture free energy present in carbon compounds produced by other organisms. • Heterotrophs may metabolize carbohydrates, lipids and proteins by hydrolysis as sources of free energy. – C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy (ATP + heat) – organic compounds + oxygen carbon dioxide + water + energy • Fermentation produces organic molecules, including alcohol and lactic acid, and it occurs in the absence of oxygen. – C 6 H 12 O 6 yeast 2 CH 3 CH 2 OH + 2 CO 2 + heat – sugar yeast ethanol + carbon dioxide + heat Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
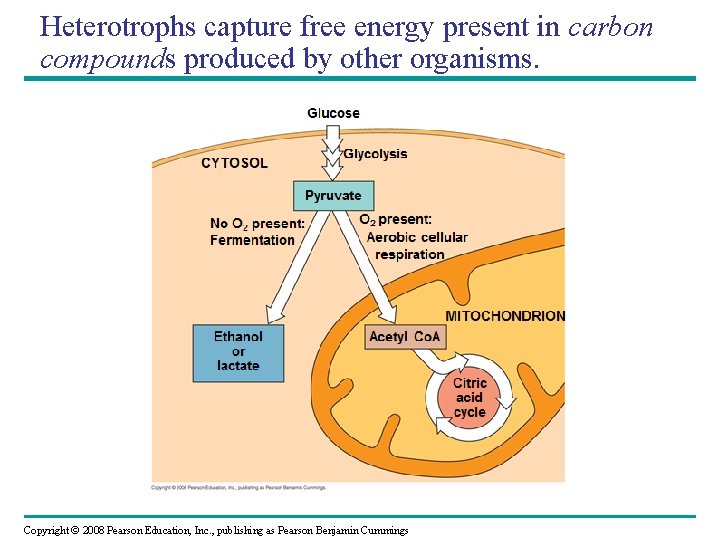
Heterotrophs capture free energy present in carbon compounds produced by other organisms. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
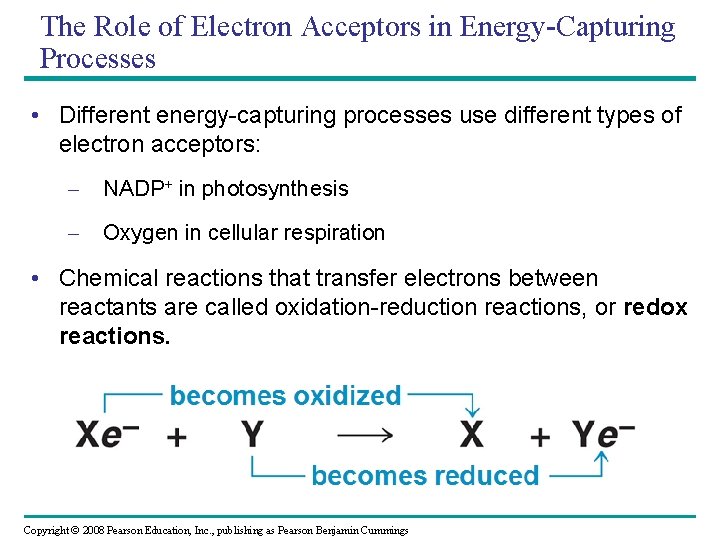
The Role of Electron Acceptors in Energy-Capturing Processes • Different energy-capturing processes use different types of electron acceptors: – NADP+ in photosynthesis – Oxygen in cellular respiration • Chemical reactions that transfer electrons between reactants are called oxidation-reduction reactions, or redox reactions. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Catabolic Pathways & ATP Production • Catabolic Pathways yield energy by oxidizing organic fuels. • Several processes are central to cellular respiration and related pathways. • The breakdown of organic molecules is exergonic: – Fermentation is a partial degradation of sugars that occurs without O 2. – Aerobic respiration consumes organic molecules and O 2 and yields ATP. – Anaerobic respiration is similar to aerobic respiration but consumes compounds other than O 2. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Cellular Respiration • Cellular respiration includes both aerobic and anaerobic respiration but is often used to refer to aerobic respiration • Although carbohydrates, fats, and proteins are all consumed as fuel, it is helpful to trace cellular respiration with the sugar glucose: • C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + Energy (ATP + heat) – The transfer of electrons during chemical reactions releases energy stored in organic molecules – This released energy is ultimately used to synthesize ATP Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
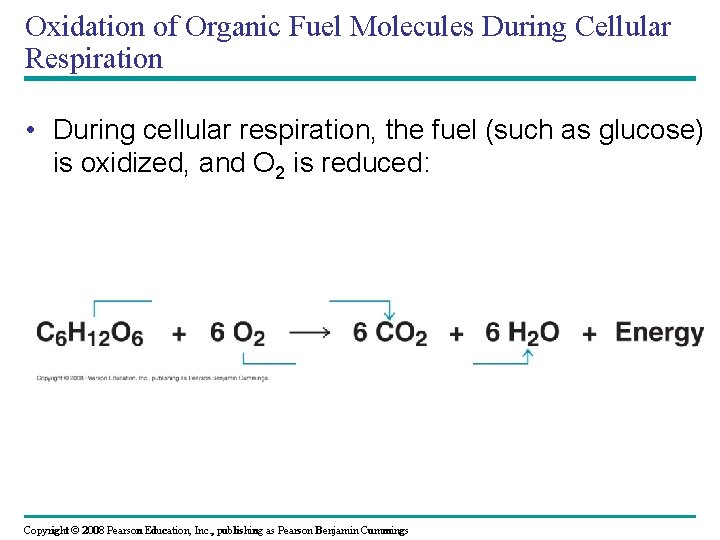
Oxidation of Organic Fuel Molecules During Cellular Respiration • During cellular respiration, the fuel (such as glucose) is oxidized, and O 2 is reduced: Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Stepwise Energy Harvest via NAD+ and the Electron Transport Chain • In cellular respiration, glucose and other organic molecules are broken down in a series of steps. • Cellular respiration in eukaryotes involves a series of coordinated enzyme-catalyzed reactions that harvest free energy from simple carbohydrates. • Electrons from organic compounds are usually first transferred to NAD+, a coenzyme. • As an electron acceptor, NAD+ functions as an oxidizing agent during cellular respiration. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

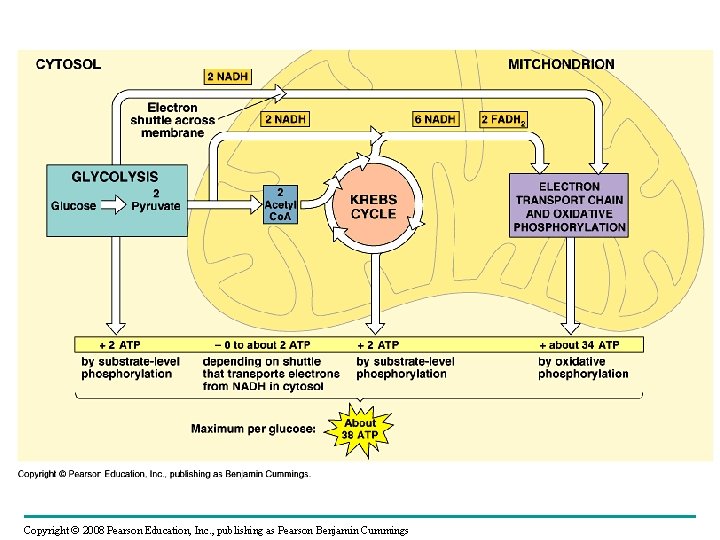
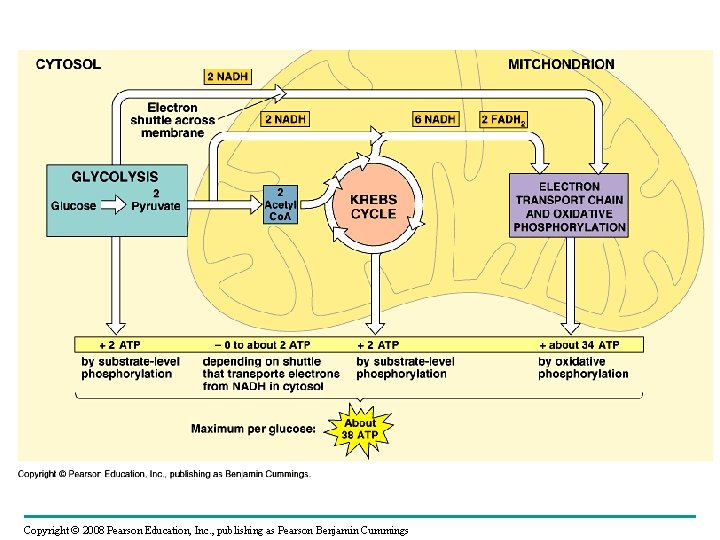
The Stages of Cellular Respiration: A Preview • Cellular respiration has three stages: – Glycolysis (breaks down glucose into two molecules of pyruvate) – occurs in cytosol – The citric acid cycle (completes the breakdown of glucose) – occurs in mitochondrial matrix – Electron Transport/Oxidative Phosphorylation (accounts for most of the ATP synthesis) – occurs across inner membrane of mitochondria http: //www. sumanasinc. com/webcontent/animations/content/cellularrespiration. html Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
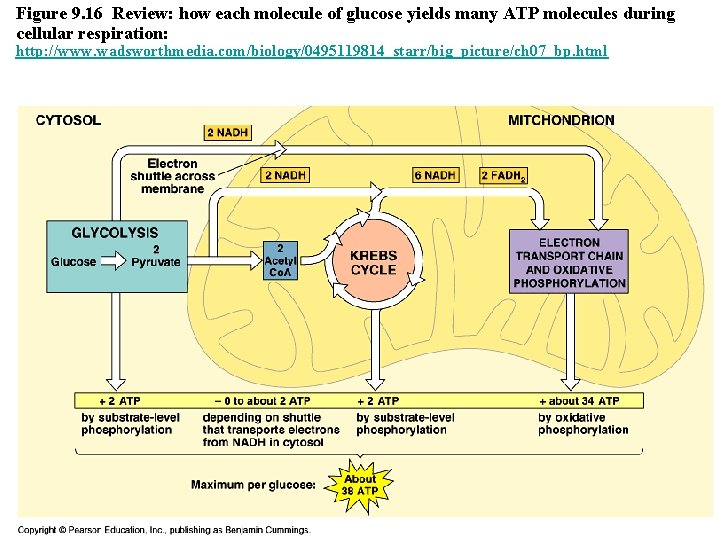
Figure 9. 16 Review: how each molecule of glucose yields many ATP molecules during cellular respiration: http: //www. wadsworthmedia. com/biology/0495119814_starr/big_picture/ch 07_bp. html

Oxidative Phosphorylation • The process that generates most of the ATP during cellular respiration is called oxidative phosphorylation because it is powered by redox reactions of an electron transport chain. • Oxidative phosphorylation accounts for almost 90% of the ATP generated by cellular respiration. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
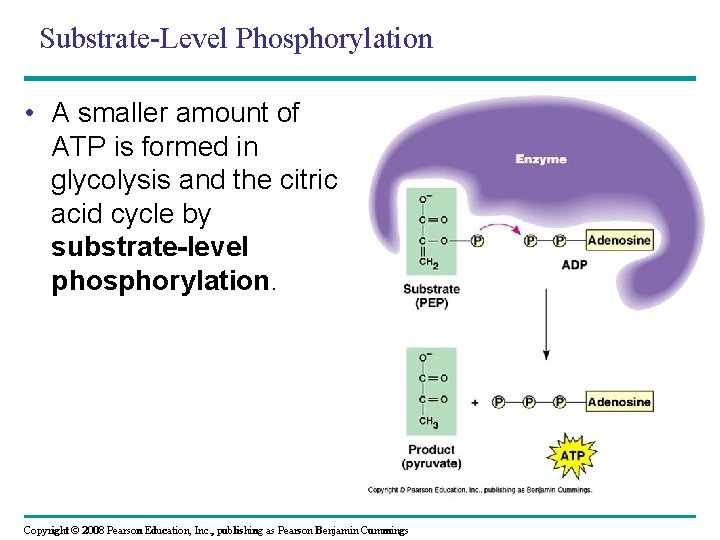
Substrate-Level Phosphorylation • A smaller amount of ATP is formed in glycolysis and the citric acid cycle by substrate-level phosphorylation. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
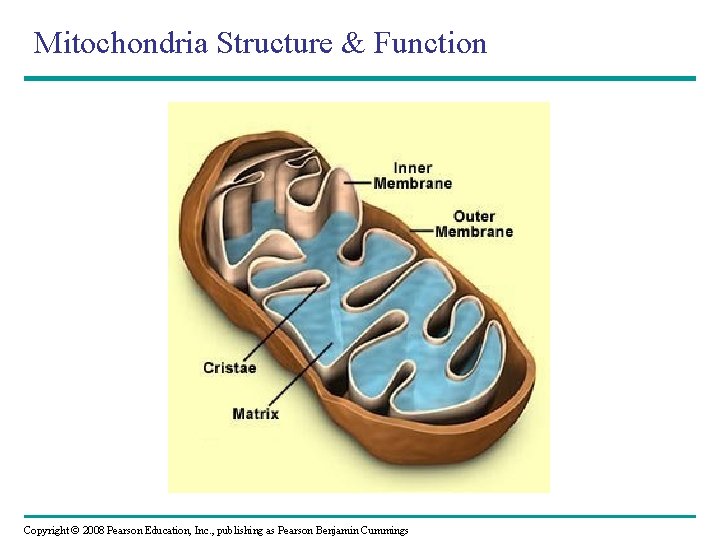
Mitochondria Structure & Function Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
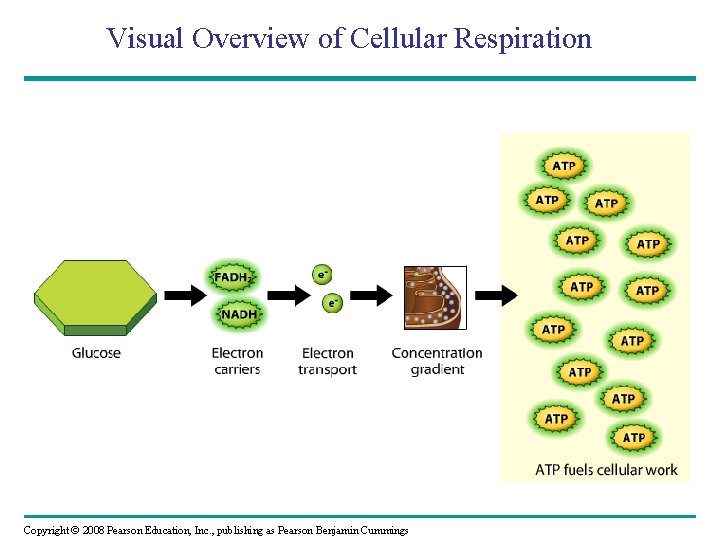
Visual Overview of Cellular Respiration Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Glycolysis http: //highered. mcgrawhill. com/sites/0072507470/student_view 0/chapter 25/animation__how_glycolysis_works. html • Glycolysis rearranges the bonds in glucose molecules, releasing free energy to form ATP from ADP and inorganic phosphate, and resulting in the production of pyruvate. • Glycolysis harvests chemical energy by oxidizing glucose to pyruvate – it is the first stage of cellular respiration. • Glycolysis (“splitting of sugar”) breaks down glucose into two molecules of pyruvate. • Glycolysis occurs in the cytoplasm and has two major phases: – Energy investment phase – Energy payoff phase Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Glycolysis • Glycolysis occurs WITH or WITHOUT oxygen. • The first step is the phosphorylation of glucose (glucose molecule gains 2 inorganic phosphates) – this ACTIVATES the glucose to split. • The second step is the splitting of glucose – breaking it down into (2) 3 -carbon molecules called pyruvic acid. – This process is achieved by stripping electrons and hydrogens from the unstable 3 -C molecules (as well as the borrowed phosphates). • 2 ATPs are needed to produce 4 ATPs (energy investment and energy payoff phases). • A second product in glycolysis is 2 NADH, which results from the transfer of e- and H+ to the coenzyme NAD+. – Occurs in the cytoplasm – Net of 2 ATPs produced – 2 pyruvic acids formed – 2 NADH produced Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
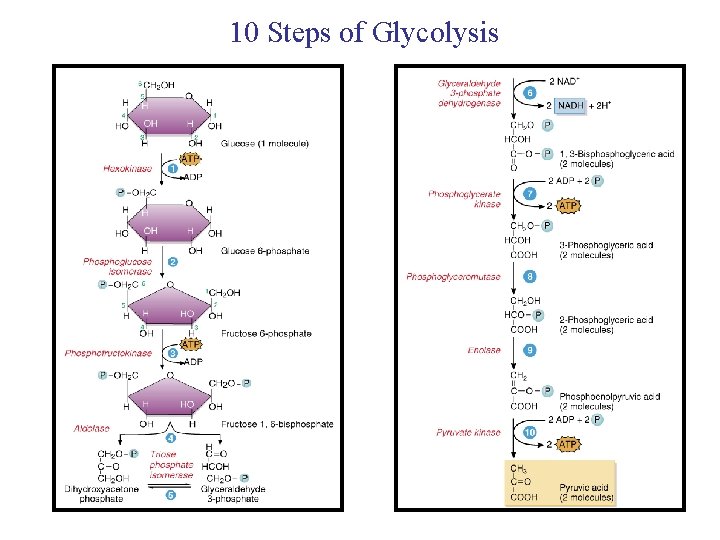
10 Steps of Glycolysis
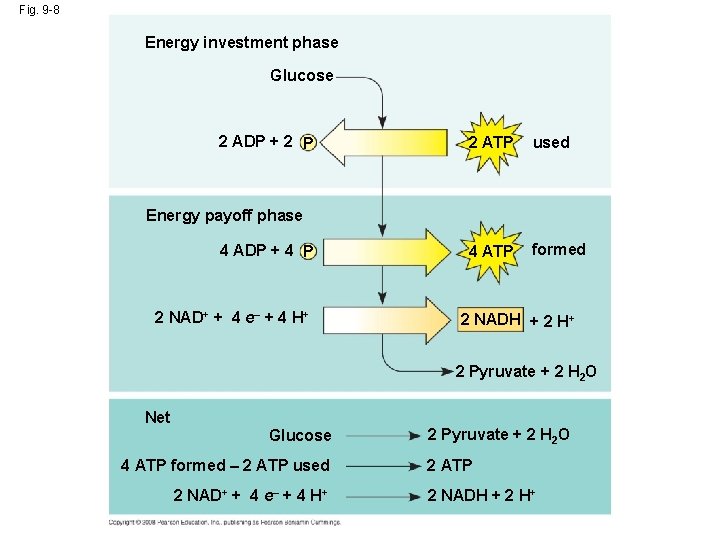
Fig. 9 -8 Energy investment phase Glucose 2 ADP + 2 P 2 ATP used 4 ATP formed Energy payoff phase 4 ADP + 4 P 2 NAD+ + 4 e– + 4 H+ 2 NADH + 2 H+ 2 Pyruvate + 2 H 2 O Net Glucose 4 ATP formed – 2 ATP used 2 NAD+ + 4 e– + 4 H+ 2 Pyruvate + 2 H 2 O 2 ATP 2 NADH + 2 H+
Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
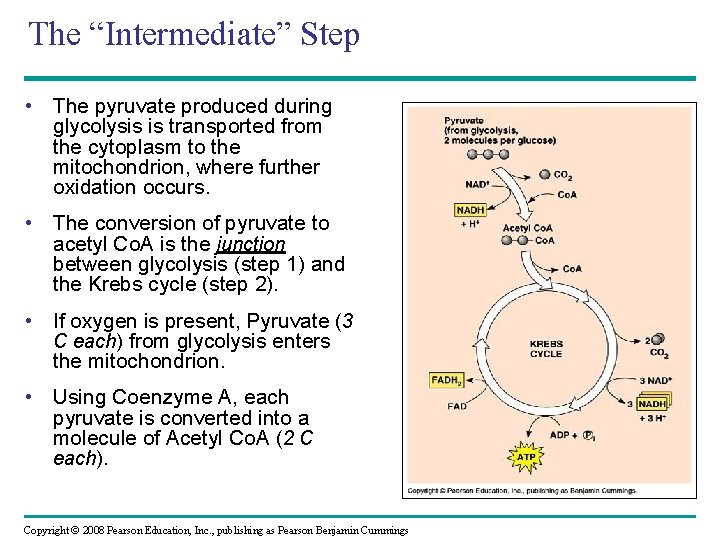
The “Intermediate” Step • The pyruvate produced during glycolysis is transported from the cytoplasm to the mitochondrion, where further oxidation occurs. • The conversion of pyruvate to acetyl Co. A is the junction between glycolysis (step 1) and the Krebs cycle (step 2). • If oxygen is present, Pyruvate (3 C each) from glycolysis enters the mitochondrion. • Using Coenzyme A, each pyruvate is converted into a molecule of Acetyl Co. A (2 C each). Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
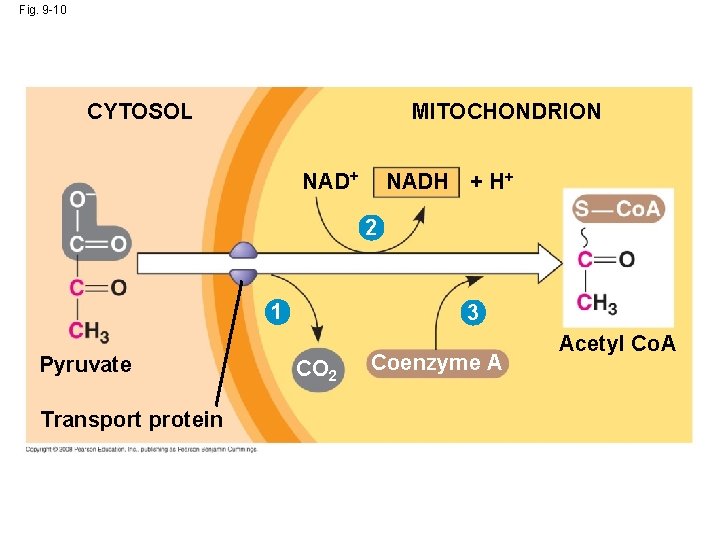
Fig. 9 -10 CYTOSOL MITOCHONDRION NAD+ NADH + H+ 2 1 Pyruvate Transport protein 3 CO 2 Coenzyme A Acetyl Co. A

The Citric Acid Cycle http: //highered. mcgrawhill. com/sites/0072507470/student_view 0/chapter 25/animation__how_the_krebs_cycle_works__quiz_1_. html • In the Krebs cycle, carbon dioxide is released from organic intermediations ATP is synthesized from ADP and inorganic phosphate via substrate level phosphorylation and electrons are captured by coenzymes. • The citric acid (Krebs) cycle completes the energyyielding oxidation of organic molecules – and its events take place within the mitochondrial matrix. • The cycle oxidizes organic fuel derived from pyruvate, generating 1 ATP, 3 NADH, and 1 FADH 2 per turn. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
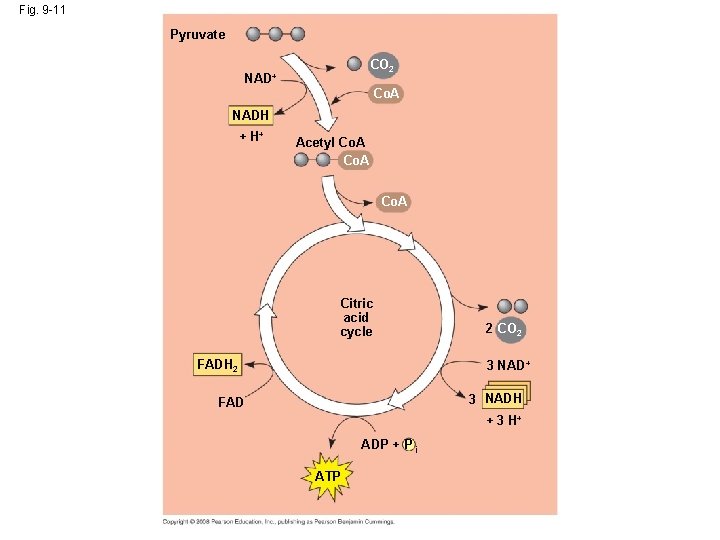
Fig. 9 -11 Pyruvate CO 2 NAD+ Co. A NADH + H+ Acetyl Co. A Citric acid cycle FADH 2 2 CO 2 3 NAD+ 3 NADH FAD + 3 H+ ADP + P i ATP
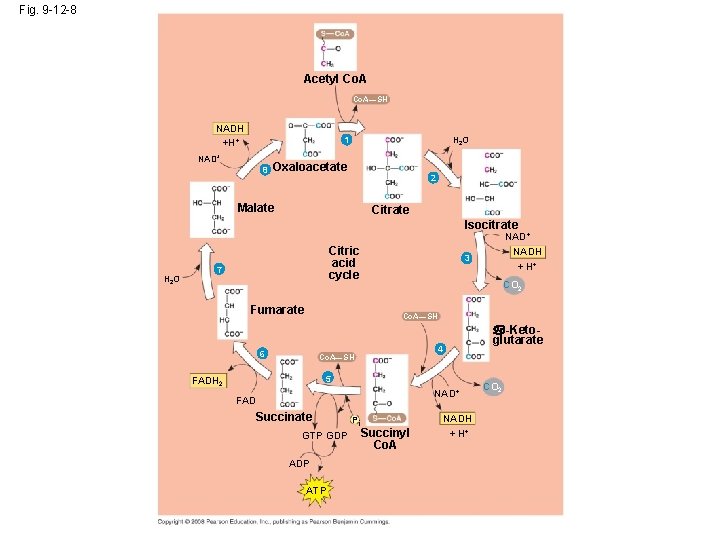
Fig. 9 -12 -8 Acetyl Co. A—SH NADH +H+ H 2 O 1 NAD+ 8 Oxaloacetate 2 Malate Citrate Isocitrate NAD+ H 2 O Citric acid cycle 7 Fumarate NADH + H+ 3 CO 2 Co. A—SH 6 -Ketoglutarate 4 Co. A—SH 5 FADH 2 NAD+ FAD Succinate GTP GDP ATP Pi Succinyl Co. A NADH + H+ CO 2
Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

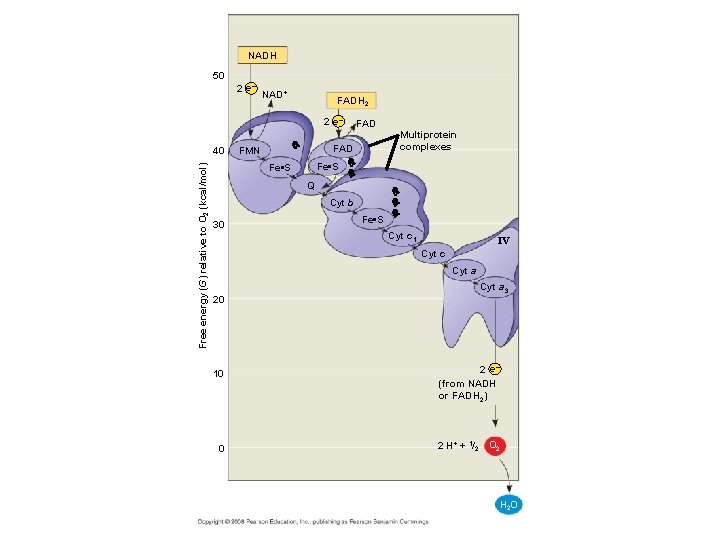
Chemiosmosis & Electron Transport http: //highered. mcgrawhill. com/sites/0072507470/student_view 0/chapter 25/animation__electron_transport_system_and_atp_synthesis__quiz_1_. html • Following the Krebs cycle, the electrons captured by NADH and FADH 2 are passed to the electron transport chain: – The electron transport chain uses the high-energy electrons from the Krebs cycle to convert ADP to ATP. – Every time 2 high energy electrons transport down the ETC, their energy is used to transport H+ across the inner membrane of the mitochondria…this creates a + charge on the inside of the membrane and a – charge in the matrix of the mitochondria. – As a result of this charge difference, H+ ions escape through channel proteins called ATP synthase causing it to rotate. – Each time it rotates, the enzyme ATP synthase grabs a low energy ADP and attaches a phosphate, forming high-energy ATP. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
NADH 50 2 e– NAD+ FADH 2 2 e– Free energy (G) relative to O 2 (kcal/mol) 40 FMN FAD Multiprotein complexes FAD Fe • S Q Cyt b 30 Fe • S Cyt c 1 IV Cyt c Cyt a 20 10 0 Cyt a 3 2 e– (from NADH or FADH 2) 2 H+ + 1/2 O 2 H 2 O
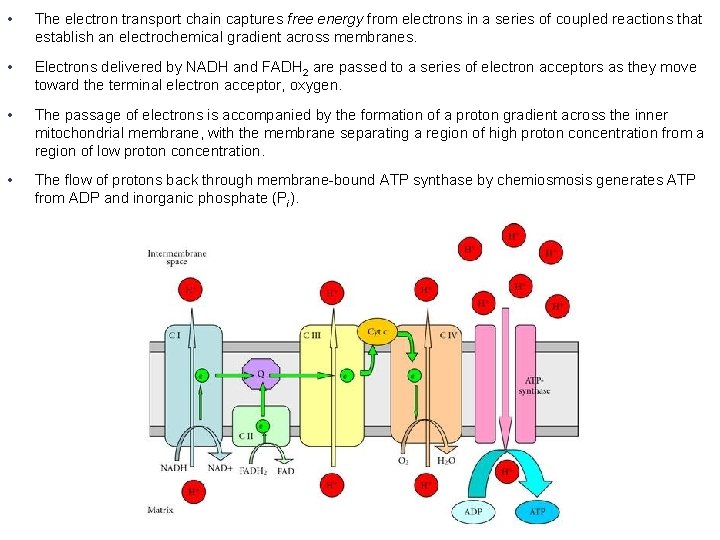
• The electron transport chain captures free energy from electrons in a series of coupled reactions that establish an electrochemical gradient across membranes. • Electrons delivered by NADH and FADH 2 are passed to a series of electron acceptors as they move toward the terminal electron acceptor, oxygen. • The passage of electrons is accompanied by the formation of a proton gradient across the inner mitochondrial membrane, with the membrane separating a region of high proton concentration from a region of low proton concentration. • The flow of protons back through membrane-bound ATP synthase by chemiosmosis generates ATP from ADP and inorganic phosphate (Pi).
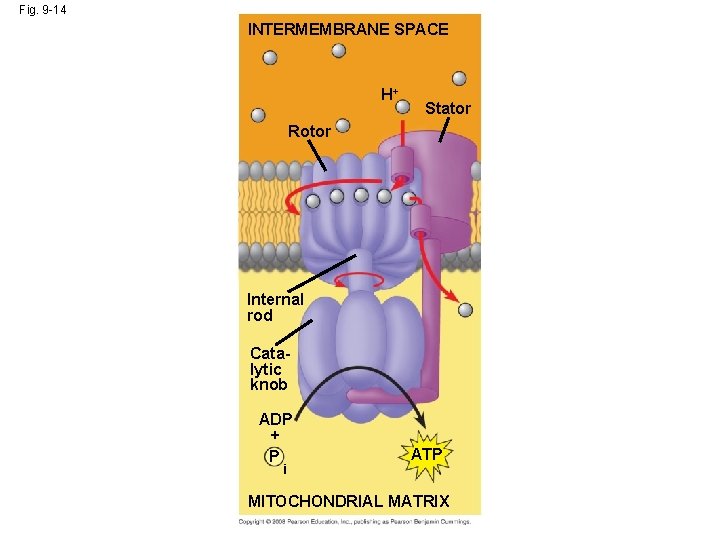
Fig. 9 -14 INTERMEMBRANE SPACE H+ Stator Rotor Internal rod Catalytic knob ADP + P i ATP MITOCHONDRIAL MATRIX
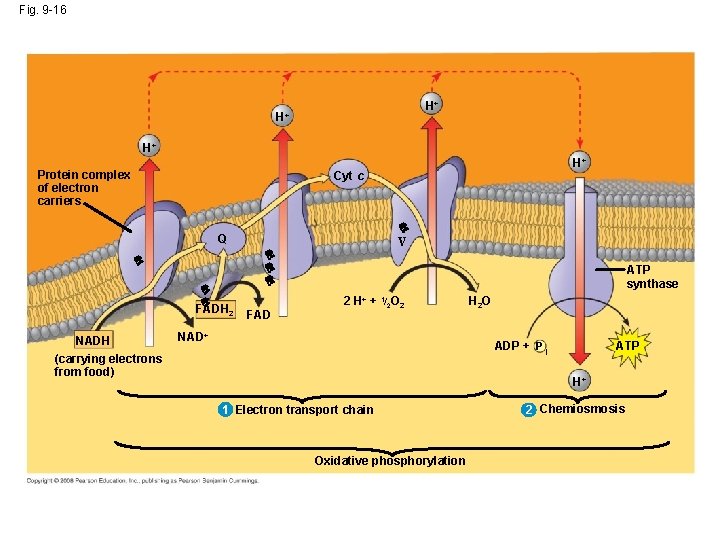
Fig. 9 -16 H+ H+ H+ Protein complex of electron carriers H+ Cyt c V Q FADH 2 NADH FAD ATP synthase 2 H+ + 1/2 O 2 NAD+ H 2 O ADP + P i (carrying electrons from food) ATP H+ 1 Electron transport chain Oxidative phosphorylation 2 Chemiosmosis
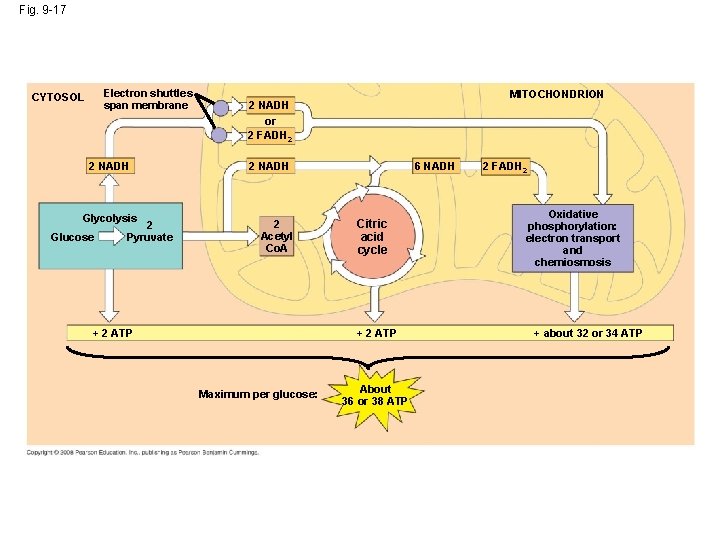
Fig. 9 -17 Electron shuttles span membrane CYTOSOL 2 NADH Glycolysis Glucose 2 Pyruvate MITOCHONDRION 2 NADH or 2 FADH 2 6 NADH 2 Acetyl Co. A + 2 ATP Citric acid cycle + 2 ATP Maximum per glucose: About 36 or 38 ATP 2 FADH 2 Oxidative phosphorylation: electron transport and chemiosmosis + about 32 or 34 ATP

Fermentation/Anaerobic Respiration • Fermentation and anaerobic respiration enable cells to produce ATP without the use of oxygen • Most cellular respiration requires O 2 to produce ATP • Glycolysis can produce ATP with or without O 2 (in aerobic or anaerobic conditions) • In the absence of O 2, glycolysis couples with fermentation or anaerobic respiration to produce ATP – Anaerobic respiration uses an electron transport chain with an electron acceptor other than O 2, for example sulfate – Fermentation uses phosphorylation instead of an electron transport chain to generate ATP Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Types of Fermentation • Fermentation consists of glycolysis plus reactions that regenerate NAD+, which can be reused by glycolysis – Two common types are alcohol fermentation and lactic acid fermentation Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
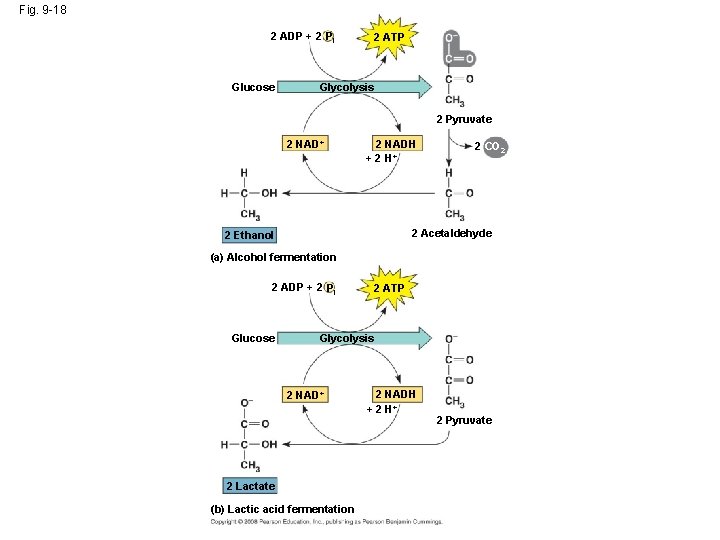
Fig. 9 -18 2 ADP + 2 Pi Glucose 2 ATP Glycolysis 2 Pyruvate 2 NAD+ 2 NADH + 2 H+ 2 CO 2 2 Acetaldehyde 2 Ethanol (a) Alcohol fermentation 2 ADP + 2 Pi Glucose 2 ATP Glycolysis 2 NAD+ 2 Lactate (b) Lactic acid fermentation 2 NADH + 2 H+ 2 Pyruvate

Fermentation and Aerobic Respiration Compared • Both processes use glycolysis to oxidize glucose and other organic fuels to pyruvate • The processes have different final electron acceptors: an organic molecule (such as pyruvate or acetaldehyde) in fermentation and O 2 in cellular respiration • Cellular respiration produces 38 ATP per glucose molecule; fermentation produces 2 ATP per glucose molecule Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

The Anaerobes • Obligate anaerobes carry out fermentation or anaerobic respiration and cannot survive in the presence of O 2 • Yeast and many bacteria are facultative anaerobes, meaning that they can survive using either fermentation or cellular respiration – In a facultative anaerobe, pyruvate is a fork in the metabolic road that leads to two alternative catabolic routes Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
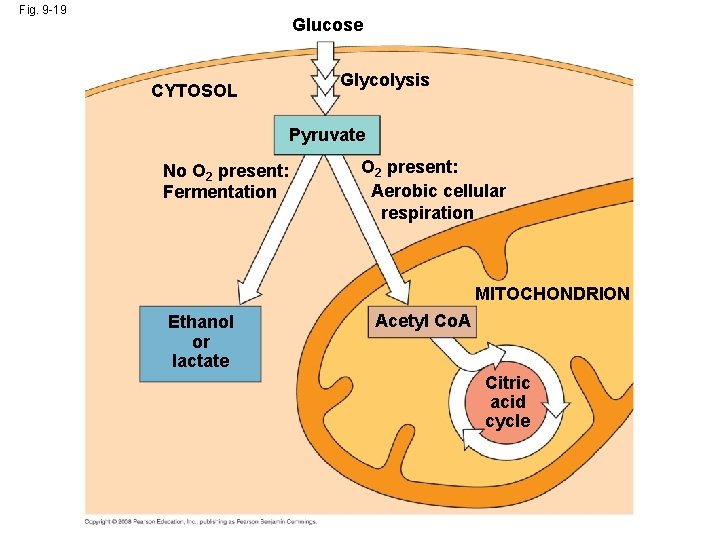
Fig. 9 -19 Glucose CYTOSOL Glycolysis Pyruvate No O 2 present: Fermentation O 2 present: Aerobic cellular respiration MITOCHONDRION Ethanol or lactate Acetyl Co. A Citric acid cycle

The Versatility of Catabolism • Glycolysis and the citric acid cycle are major intersections to various catabolic and anabolic pathways. • Catabolic pathways funnel electrons from many kinds of organic molecules into cellular respiration. • Glycolysis accepts a wide range of carbohydrates. • In addition to carbohydrates, heterotrophs may metabolize lipids and proteins by hydrolysis as sources of free energy. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
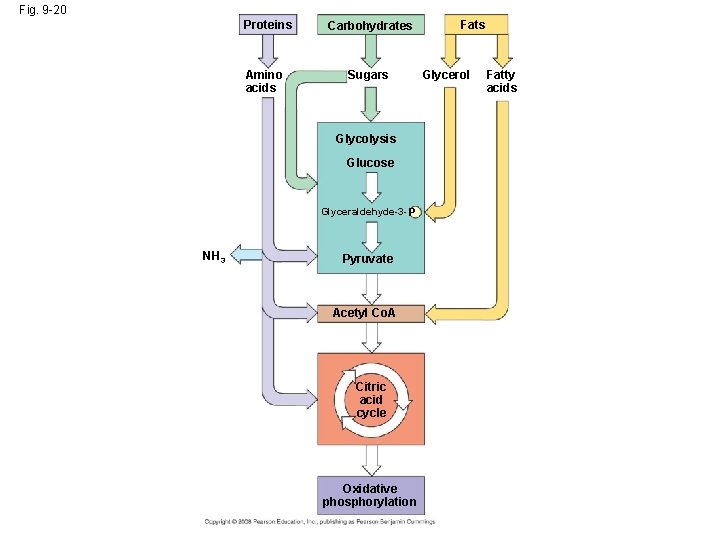
Fig. 9 -20 Proteins Amino acids Carbohydrates Sugars Glycolysis Glucose Glyceraldehyde-3 - P NH 3 Pyruvate Acetyl Co. A Citric acid cycle Oxidative phosphorylation Fats Glycerol Fatty acids
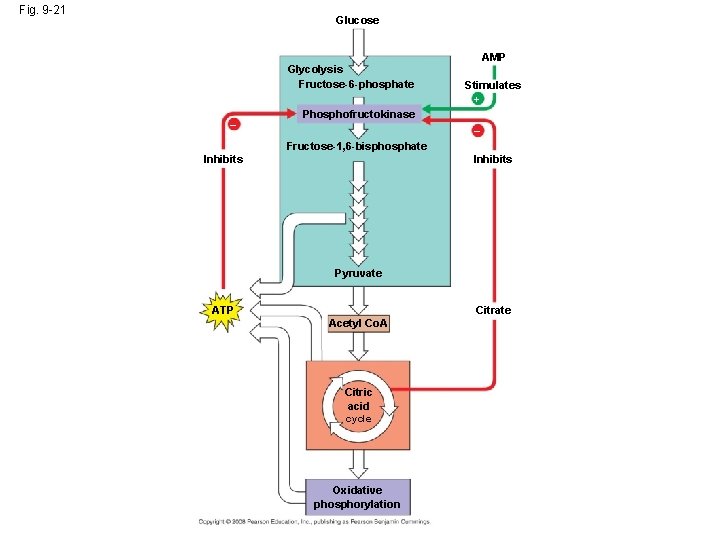
Fig. 9 -21 Glucose Glycolysis Fructose-6 -phosphate – AMP Stimulates + Phosphofructokinase – Fructose-1, 6 -bisphosphate Inhibits Pyruvate ATP Citrate Acetyl Co. A Citric acid cycle Oxidative phosphorylation

What do you know…. • Glycolysis • Kreb Cycle • ETC • Fermentation Pathways Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Overview of Cellular Respiration A. Glycolysis – *takes place in cytosol *breaks glucose into 2 molecules of pyruvate *produces a net of 2 ATP’s B. Krebs cycle - *takes place in mitochondrial matrix *makes a derivative of pyruvate into carbon dioxide *produces a net of 2 ATP’s C. ETC and Oxidative Phosphorylation – *takes place in inner membrane of mitochondrion *accepts e-’s from A and B via NADH *at end, e-’s are combined with H+ and oxygen to form water *forms a net of 34 ATP’s D. Fermentation Pathways *takes place in cytosol *2 types: Alcoholic and Lactic Acid Fermentation *stores most of the energy in chemical bonds *no production of ATP
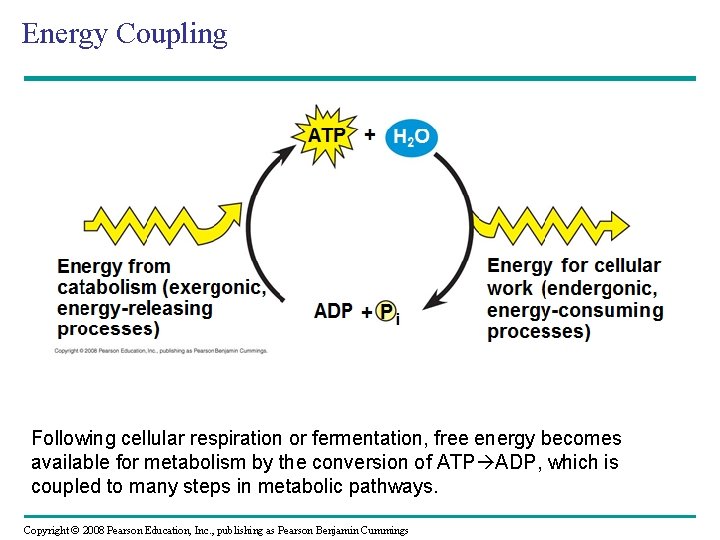
Energy Coupling H 2 O Following cellular respiration or fermentation, free energy becomes available for metabolism by the conversion of ATP ADP, which is coupled to many steps in metabolic pathways. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Overview of Photosynthesis • Photosynthesis is the process whereby light energy is converted to chemical energy and carbon is fixed into organic compounds. • In the presence of light, plants transform carbon dioxide and water into carbohydrates and release oxygen: – Photosynthesis uses the energy of sunlight to convert water and CO 2 into O 2 and high energy sugars – 6 CO 2 + 6 H 2 O + light → C 6 H 12 O 6 + 6 O 2 – carbon dioxide + water + light → sugar + oxygen • Plants then use the sugars to produce complex carbohydrates such as starches: – Plants obtain carbon dioxide from the air or water in which they grow. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
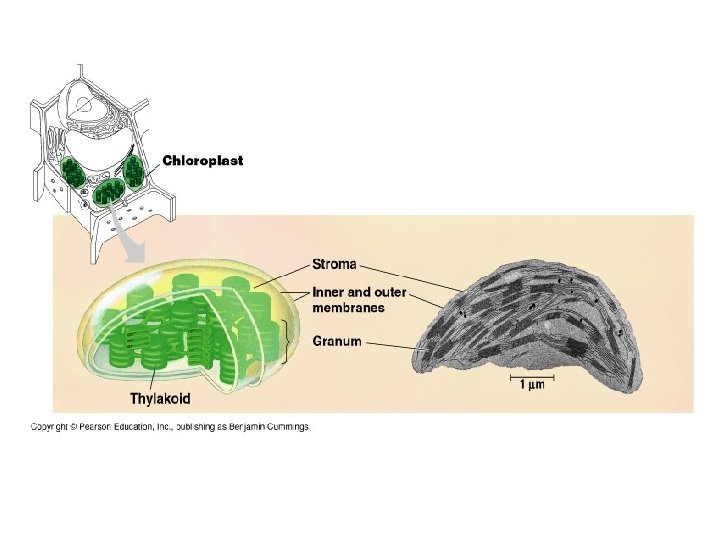
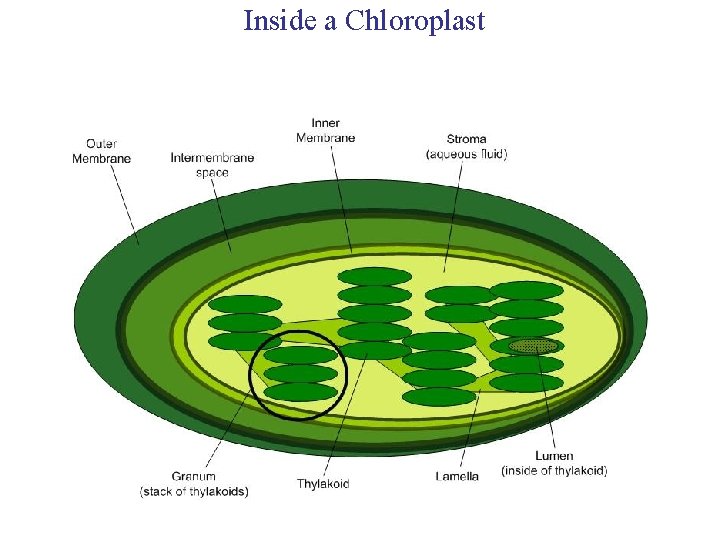
Inside a Chloroplast

Stages of Photosynthesis • The reaction that occurs during photosynthesis can be broken into 2 stages: 1. 2. Light Dependent Reactions • Take place within the thylakoid membranes inside a chloroplast • “PHOTO” phase – make ATP & NADPH…USE LIGHT ENERGY TO PRODUCE ATP & NADPH Light Independent Reactions (Calvin Cycle) • Take place in the stroma of the chloroplast • “SYNTHESIS” phase – coverts CO 2 to sugar…PRODUCE SUGAR Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Photosynthetic Pigments • Photosynthetic pigments absorb light energy and use it to provide energy to carry out photosynthesis. – – Chlorophylls (absorb light in the red, blue, and violet range): • Chlorophyll a - directly involved in transformation of photons to chemical energy • Chlorophyll b - helps trap other wavelengths and transfers it to chlorophyll a Carotenoids (absorb light in the blue, green, and violet range): • xanthophyll - Yellow • beta carotene - Orange • Phycobilins – Red – Chlorophyll b, the carotenoids, and the phycobilins are known as ANTENNA PIGMENTS – they capture light in other wavelengths and pass the energy along to chlorphyll a. – Chlorophyll a is the pigment that participates directly in the light reactions of photosynthesis! Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
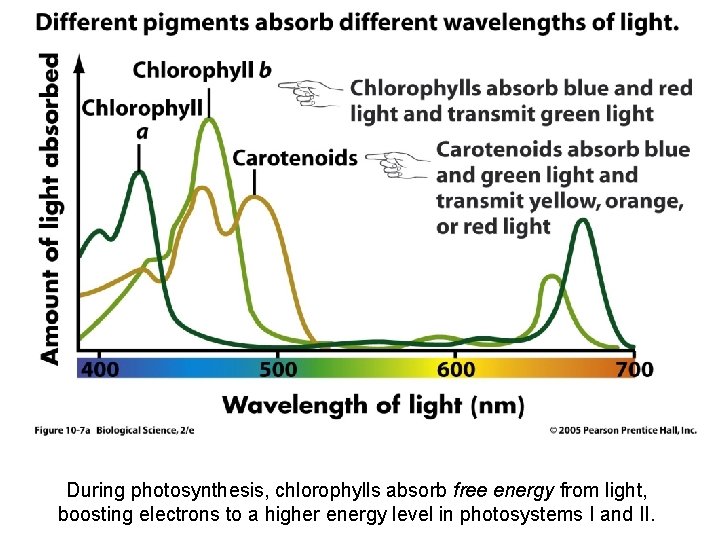
During photosynthesis, chlorophylls absorb free energy from light, boosting electrons to a higher energy level in photosystems I and II.
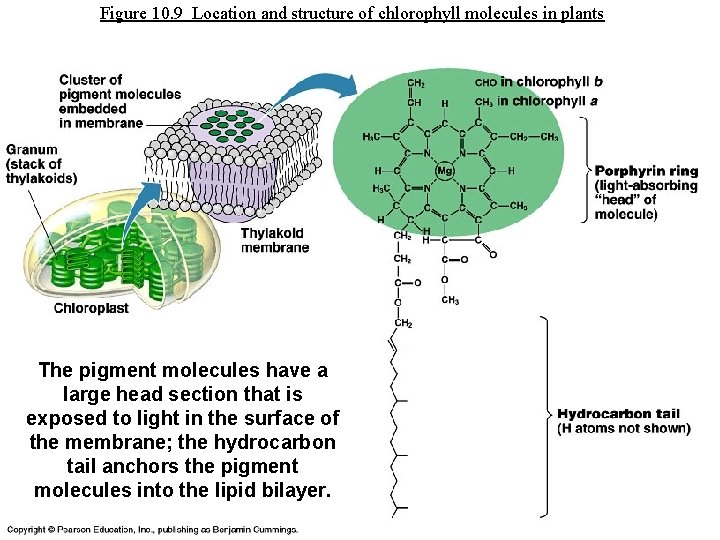
Figure 10. 9 Location and structure of chlorophyll molecules in plants The pigment molecules have a large head section that is exposed to light in the surface of the membrane; the hydrocarbon tail anchors the pigment molecules into the lipid bilayer.
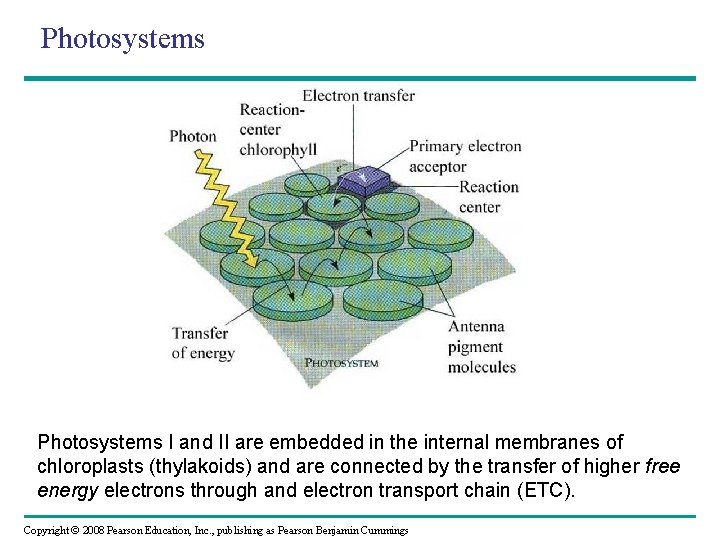
Photosystems I and II are embedded in the internal membranes of chloroplasts (thylakoids) and are connected by the transfer of higher free energy electrons through and electron transport chain (ETC). Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

PS I and PS II • Named in the order they were discovered – however, PS II occurs first, followed by PS I. – PS I absorbs light best in the 700 nm range (so called P 700). – PS II absorbs light best in the 680 nm range (so called P 680). Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Stages of Photosynthesis • There are 2 stages in Photosynthesis: 1. Light dependent reactions 2. Light independent reactions (Calvin Cycle) • BOTH REQUIRE LIGHT (SOMEWHAT): – Even the dark reactions in most plants occurs during daylight – because that is the only time the light reactions can operate AND the dark reactions depend on the light reactions!!! Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
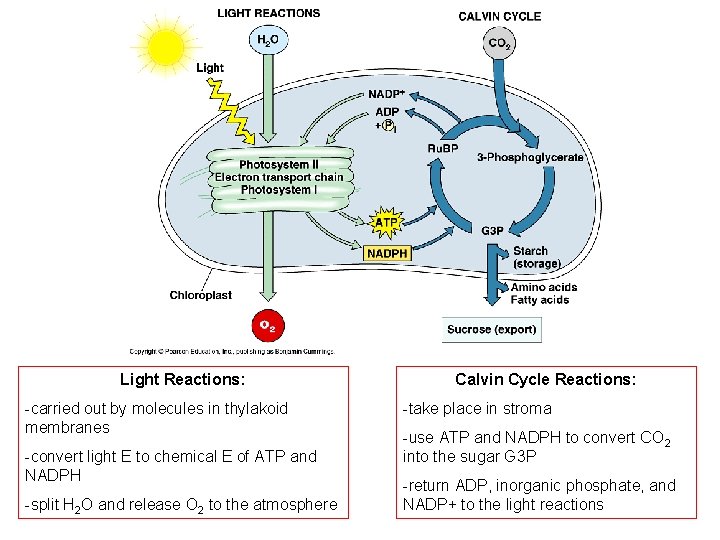
Light Reactions: -carried out by molecules in thylakoid membranes -convert light E to chemical E of ATP and NADPH -split H 2 O and release O 2 to the atmosphere Calvin Cycle Reactions: -take place in stroma -use ATP and NADPH to convert CO 2 into the sugar G 3 P -return ADP, inorganic phosphate, and NADP+ to the light reactions

Light Dependent Reactions - Overview • require presence of light • occur in thylakoids of chloroplasts • use energy from light to produce ATP and NADPH (a temporary, mobile energy source that helps store even more energy) • water is split during the process to replace electrons lost from excited chlorophyll • oxygen gas is produced as a by-product Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Light Independent Reactions - Overview • do not require light directly – so also known as the Dark Reactions or the Calvin Cycle • take place in the stroma of chloroplasts • ATP and NADPH produced during light dependent reactions are used to make glucose Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

The Light Reactions • Light is absorbed by PS II and PS I in the thylakoid membranes and electrons flow through TWO electron transport chains. – There are 2 possible routes for electron flow: 1. Noncylic photophosphorylation 2. Cyclic photophosphorylation • Photophosphorylation is a method of generating ATP by using light to add P to ADP Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Cyclic vs. Noncyclic Electron Flow http: //highered. mcgraw-hill. com/olc/dl/120072/bio 12. swf • Noncyclic Electron Flow – uses Photosystem II, and ETC (with the electron carriers B 6 f comlex and Pq) , Photosystem I, and another ETC using an ironcontaining protein called ferredoxin & NADP reductase – produces ATP and NADPH • Cyclic Electron Flow – uses only Photosystem II and the first ETC – no production of NADPH and no release of Oxygen – DOES produce ATP to be used to make up the difference needed due to Calvin cycle demands. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
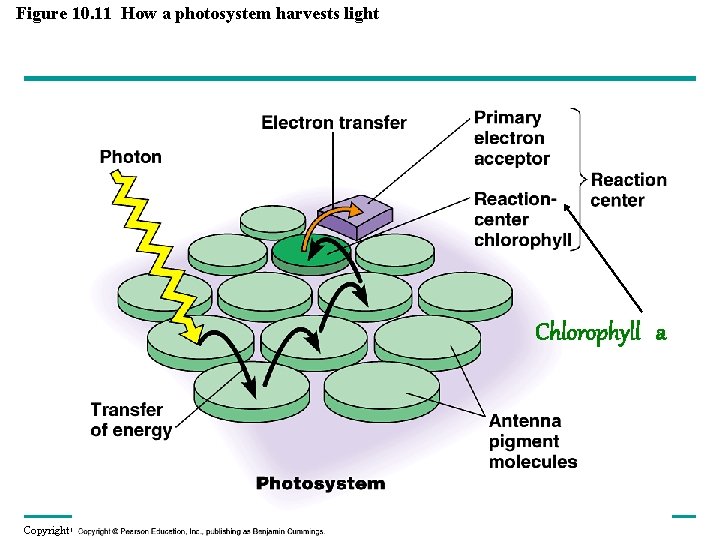
Figure 10. 11 How a photosystem harvests light Chlorophyll a Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
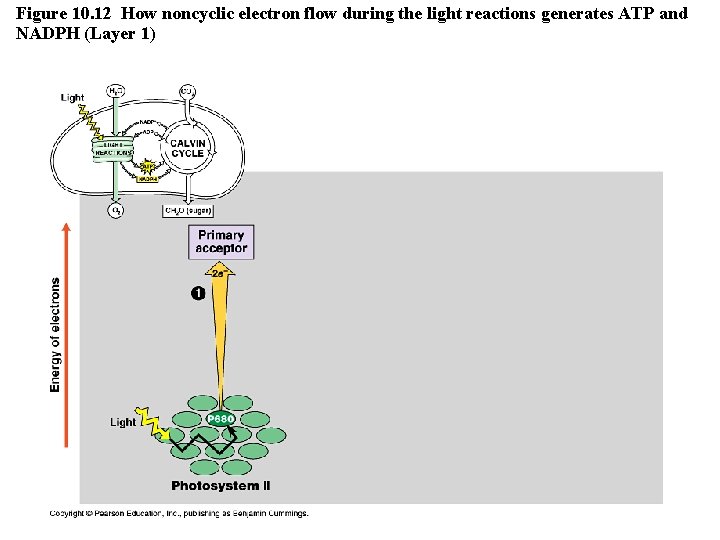
Figure 10. 12 How noncyclic electron flow during the light reactions generates ATP and NADPH (Layer 1)
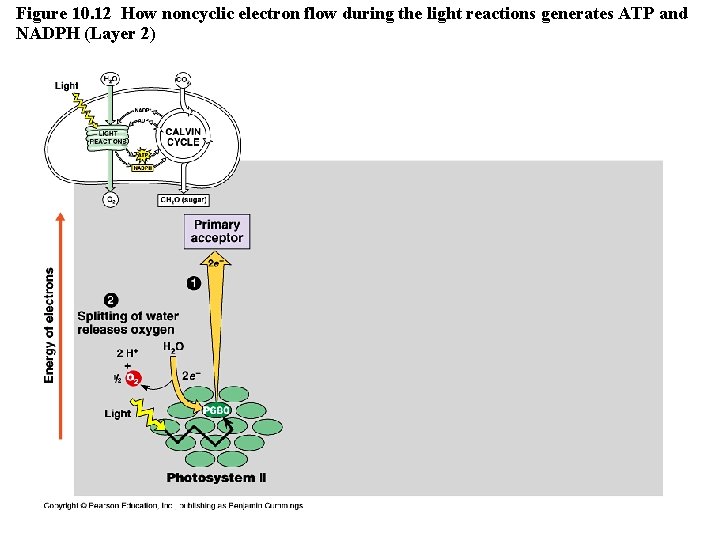
Figure 10. 12 How noncyclic electron flow during the light reactions generates ATP and NADPH (Layer 2)
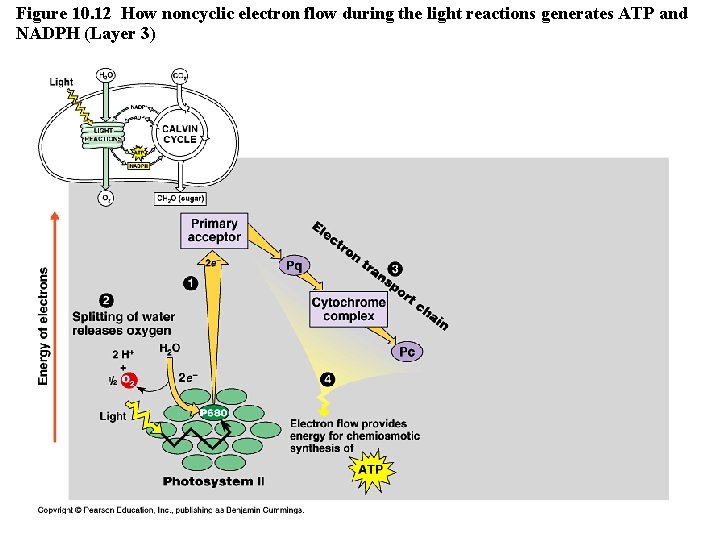
Figure 10. 12 How noncyclic electron flow during the light reactions generates ATP and NADPH (Layer 3)
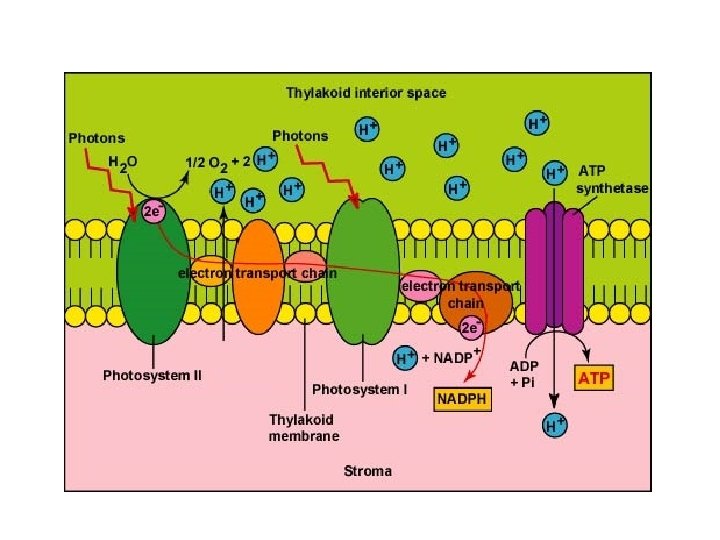
Figure 8 -10 Light-Dependent Reactions Section 8 -3 Go to Section:
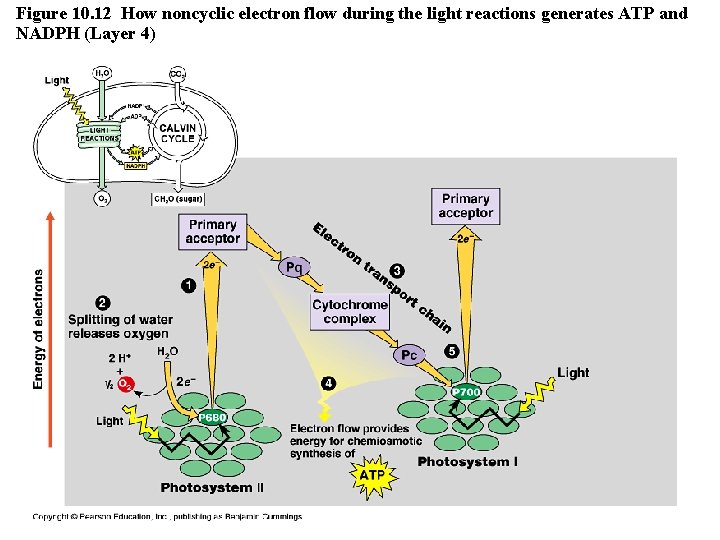
Figure 10. 12 How noncyclic electron flow during the light reactions generates ATP and NADPH (Layer 4)
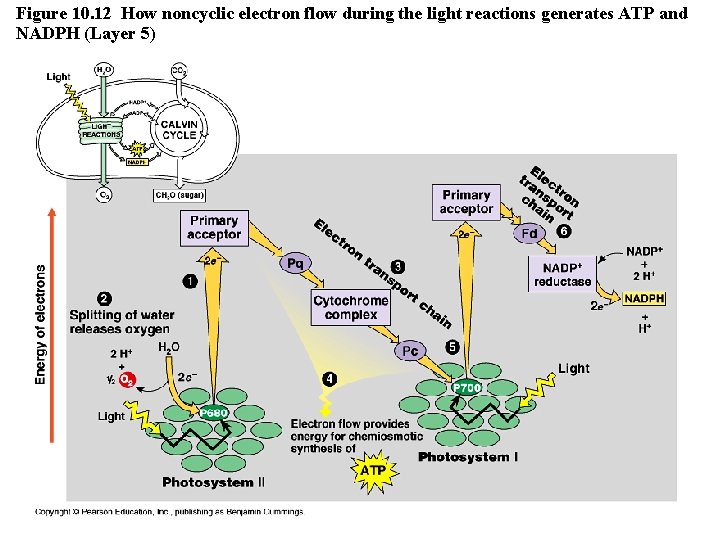
Figure 10. 12 How noncyclic electron flow during the light reactions generates ATP and NADPH (Layer 5)
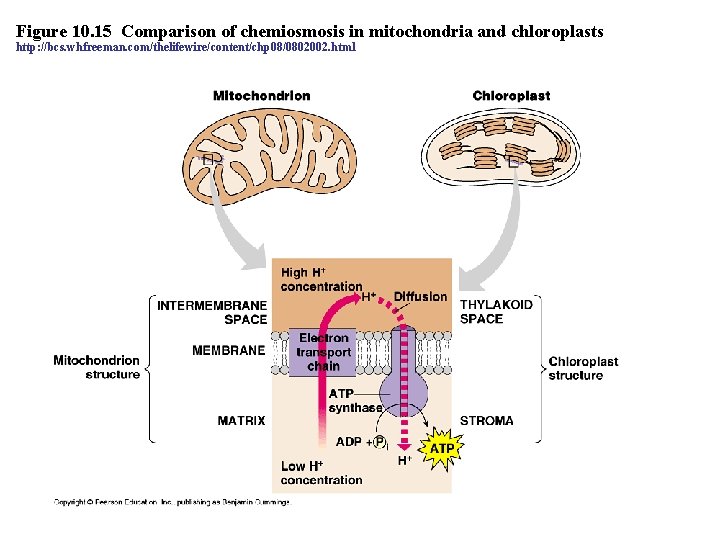
Figure 10. 15 Comparison of chemiosmosis in mitochondria and chloroplasts http: //bcs. whfreeman. com/thelifewire/content/chp 08/0802002. html

The Dark Reactions (Calvin cycle) • Calvin cycle can be divided into 3 phases: – Phase 1: Carbon Fixation – Phase 2: Reduction – Phase 3: Regeneration of CO 2 Acceptor (Ru. BP) • REMEMBER: The Calvin cycle is an ANABOLIC process – and therefore requires ENERGY – this energy is provided by the ATP and NADPH made during the light reactions!!! Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
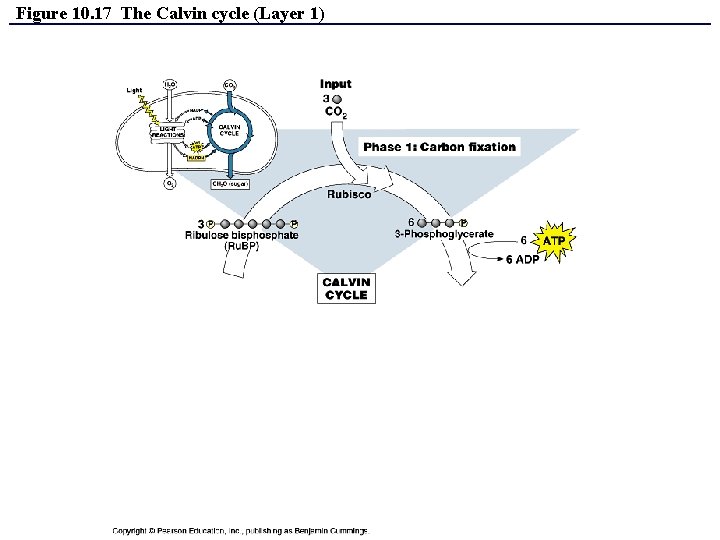
Figure 10. 17 The Calvin cycle (Layer 1)
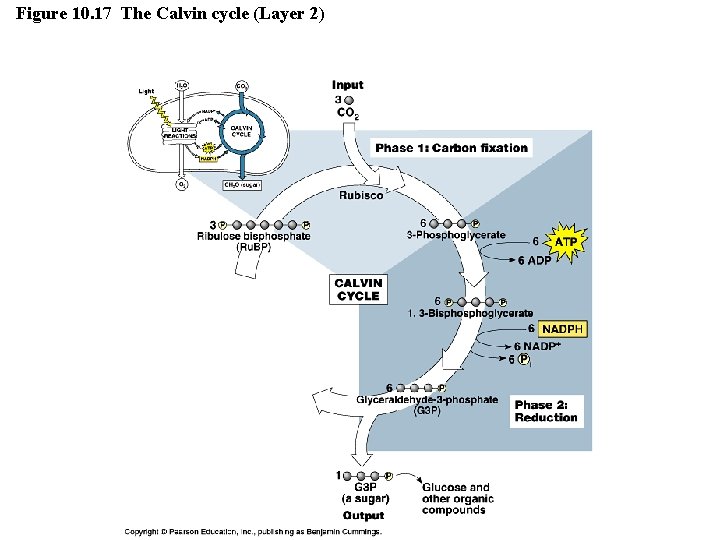
Figure 10. 17 The Calvin cycle (Layer 2)
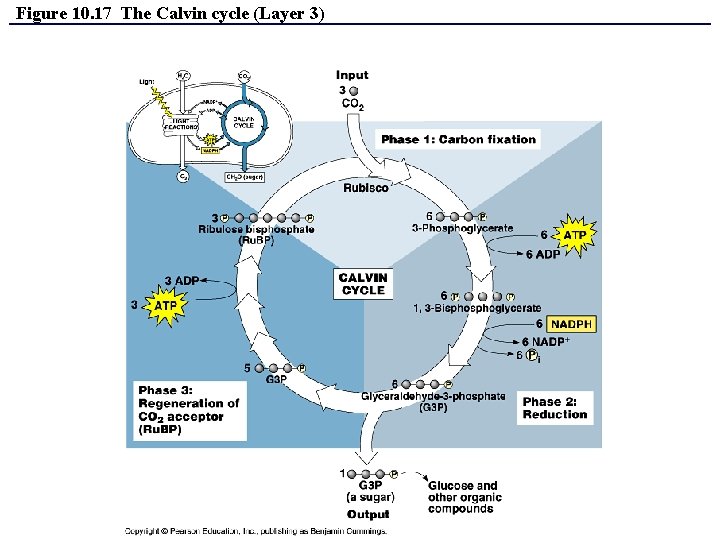
Figure 10. 17 The Calvin cycle (Layer 3)

The Dark Reactions (Calvin cycle) • Calvin cycle can be divided into 3 phases: – – – Phase 1: Carbon Fixation • CO 2 is incorporated and attached to Ru. BP (catalyzed by enzyme rubisco). • Product of reaction is 6 -carbon intermediate so unstable that it splits in half to form two molecules of 3 -phosphoglycerate. Phase 2: Reduction • Each molecule of 3 -phosphoglycerate receives additional phosphate group from ATP to become 1, 3 bisphoglycerate. • A pair of electrons donated from NADPH reduces 1, 3 bisphoglycerate into Glyceraldehide-3 -phosphate (a sugar). • One of the G 3 P molecules is exported and used to build glucose. Phase 3: Regeneration of CO 2 Acceptor (Ru. BP) • In a series of complex reactions, the carbon skeletons of 5 molecules of G 3 P are rearranged by the last steps of the Calvin cycle into three molecules of Ru. BP. • The Ru. BP is now prepared again to receive CO 2…and the cycle continues. • The regeneration phase requires ATP. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings

Conserved Core Processes • Photosynthesis first evolved in prokaryotic organisms; • Scientific evidence supports that prokaryotic (bacterial) photosynthesis was responsible for the production of an oxygenated atmosphere; • Prokaryotic photosynthetic pathways were the foundation of eukaryotic photosynthesis. Copyright © 2008 Pearson Education, Inc. , publishing as Pearson Benjamin Cummings
- Slides: 75