Big Idea 5 Thermodynamics The laws of thermodynamics

Big Idea #5 Thermodynamics: “The laws of thermodynamics describe the essential role of energy and explain and predict the direction of changes in matter

Basic concepts in Big Idea #5 • • Law of conservation of energy Direction of heat transfer (-)(+) Specific heat calculations and calorimetry Hess’s law & applications Entropy Prediction of Spontaneity Gibb’s free energy Gibbs equation (linking temp, enthalpy and entropy)
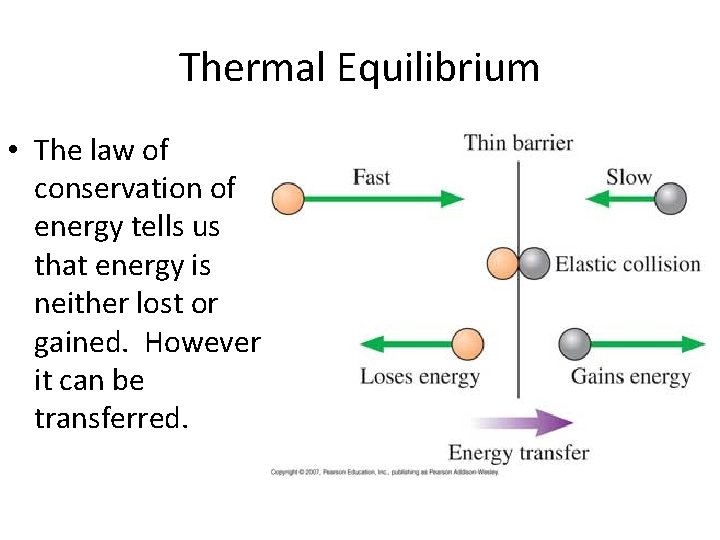
Thermal Equilibrium • The law of conservation of energy tells us that energy is neither lost or gained. However it can be transferred.
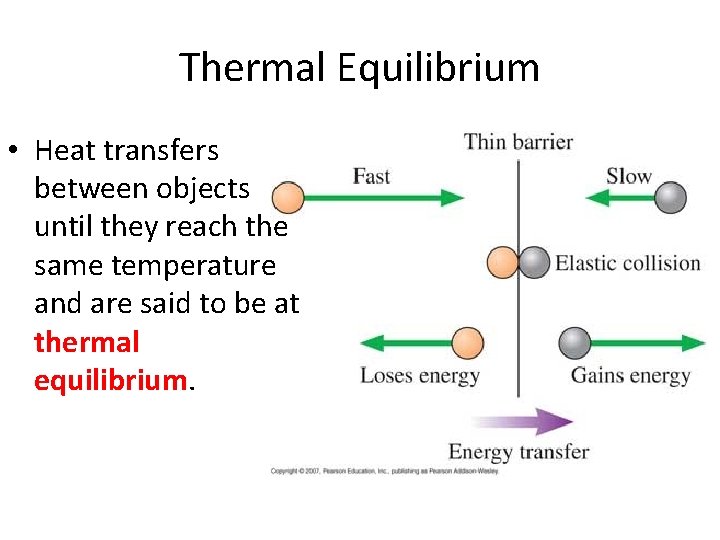
Thermal Equilibrium • Heat transfers between objects until they reach the same temperature and are said to be at thermal equilibrium.

Various Specific Heat Capacities Substance Specific heat capacity (J/g K) Gold 0. 129 Silver 0. 235 Copper 0. 385 Iron 0. 449 Aluminum 0. 897 H 2 O(l) 4. 184 H 2 O(s) 2. 03 H 2 O(g) 1. 998 Metals do not generally require much energy to heat them up (i. e. they heat up easily) Water requires much more energy to heat up
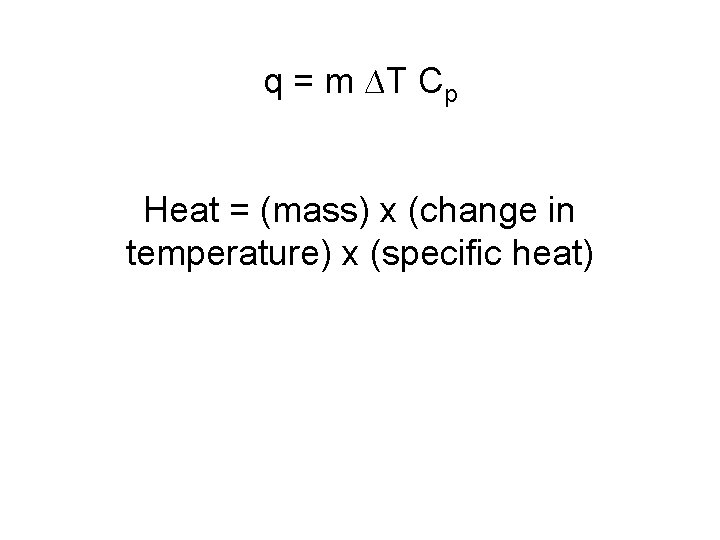
q = m ∆T Cp Heat = (mass) x (change in temperature) x (specific heat)

We can find the heat a substance loses or gains using: where q = heat (J) q = m TCp m = mass of substance (g) (used within a given state of matter) C = specific heat (J/go. C) T = temperature change (o. C) H = heat of vap/fus (k. J/mol) AND q = mol ΔH (used between two states of matter or during a phase change)

Heating Curves • Temperature Change within phase • change in KE (molecular motion) • depends on the specific heat capacity of phase Cp of H 2 O (l) = 4. 184 J/go. C Cp of H 2 O (s) = 2. 077 J/go. C Cp of H 2 O (g) = 2. 042 J/go. C • Phase Changes (s ↔ l ↔ g) • change in PE (The extra energy required to cause a phase transition is actually potential energy. It is the energy required to overcome the bonds/intermolecular forces of adjacent particles to the point that a phase transition can occur. So it really isn't a change in kinetic energy) • temperature remains constant. ΔHfus = +6. 01 k. J/mol (s ↔ l) ΔHvap = +40. 65 k. J/mol (l ↔ g) Why is this so much larger?
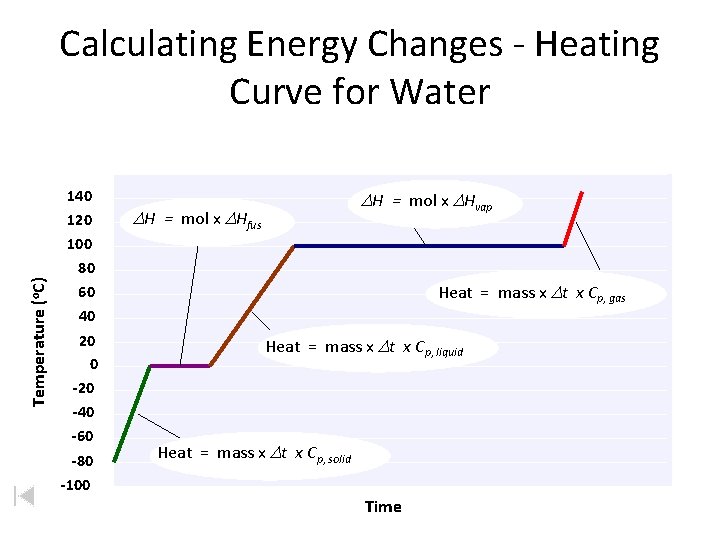
Calculating Energy Changes - Heating Curve for Water Temperature (o. C) 140 120 100 DH = mol x DHvap DH = mol x DHfus 80 60 40 20 0 -20 -40 -60 -80 -100 Heat = mass x Dt x Cp, gas Heat = mass x Dt x Cp, liquid Heat = mass x Dt x Cp, solid Time

Much calorimetry is carried out using a coffee-cup calorimeter, under constant pressure (i. e. atmospheric pressure) • If we assume that no heat is lost to the surroundings, then the energy absorbed inside the calorimeter must be equal to the energy released inside the calorimeter. i. e. , q absorbed = q released Thermometer Styrofoam cover Styrofoam cups Stirrer

240. g of water at 20. 0 o. C are mixed with an unknown mass of iron initially at 500. 0 o. C (CFe = 0. 4495 J/go. C). When thermal equilibrium is reached, the mixture has a temperature of 42. 0 o. C. Find the mass of the iron. Fe T = 500 o. C mass = ? grams T = 20 o. C mass = 240 g Fe heat loss = Water heat gain q 1 = q 2 [ (mass) (CFe ) ( T)] [ (X g) (0. 4495 J/go. C) (42 o. C - 500 o. C)] [ (X) (0. 4495 J/go. C) (-458 o. C)] 205. 9 J/g (X) = (mass) (CH 2 O) ( T) = (240 g) (4. 184 J/go. C) (42 o. C - 20 o. C)] = (240 g) (4. 184 J/go. C) (22 o. C) = 22091 J X = 107 g Fe

The reaction of 0. 302 g of Mg with excess HCl was carried out in a calorimeter. This reaction caused the temperature of 27. 0 grams of water (Cp = 4. 184 J/g °C), within the calorimeter, to raise from 25. 0 C to 76. 0 C. Calculate heat of reaction in k. J/mol. qwter = (27. 0 g)(76. 0 °C – 25. 0°C)(4. 184 J/g °C) = 5760 J = 5. 76 k. J qwater = qreaction mol Mg = 0. 302 g / 24. 3 g/mol = 0. 0124 mol Mg ∆H = 5. 76 k. J / 0. 0124 mol = - 465 k. J/mol

∆H (enthalpy of or heat of reaction) • k. J/mol • Can be used to convert mol to k. J • Can be used to convert k. J to mol

Sample Problem The molar heat of fusion of water is 6. 009 k. J/mol. How much energy is needed to convert 60. grams of ice at 0 C to liquid water at 0 C? =20. k. J

Determine the heat of reaction for the reaction: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) Using the following sets of reactions: N 2(g) + O 2(g) 2 NO(g) H = 180. 6 k. J/mol N 2(g) + 3 H 2(g) 2 NH 3(g) H = -91. 8 k. J /mol 2 H 2(g) + O 2(g) 2 H 2 O(g) H = -483. 7 k. J /mol 4 NH 3: Reverse and x 2 4 NH 3 2 N 2 + 6 H 2 H = +183. 6 k. J /mol 5 O 2 : Found in more than one place, so SKIP IT (its confusing). 4 NO: x 2 2 N 2 + 2 O 2 4 NO H = 361. 2 k. J /mol 6 H 2 O: x 3 6 H 2 + 3 O 2 6 H 2 O H = -1451. 1 k. J /mol 15

Goal: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) NH 3: Reverse and x 2 4 NH 3 2 N 2 + 6 H 2 H = +183. 6 k. J /mol O 2 : Found in more than one place, SKIP IT. NO: x 2 2 N 2 + 2 O 2 4 NO H = 361. 2 k. J /mol H 2 O: x 3 6 H 2 + 3 O 2 6 H 2 O H = -1451. 1 k. J /mol Cancel terms and take sum. 4 NH 3 + 5 O 2 4 NO + 6 H 2 O H = -906. 3 k. J /mol Is the reaction endothermic or exothermic? 16
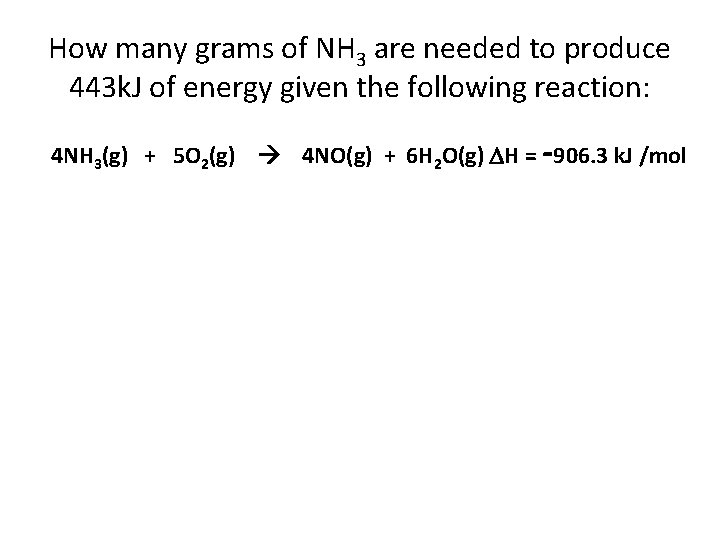
How many grams of NH 3 are needed to produce 443 k. J of energy given the following reaction: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) H = -906. 3 k. J /mol

How many grams of NH 3 are needed to produce 443 k. J of energy given the following reaction: 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g) H = -906. 3 k. J /mol 443 k. J 4 mol NH 3 17. 0 g = 33. 2 g NH 3 906, 3 k. J mol NH 3

Enthalpy of Formation or Heat of Formation (∆Hf) • Heat released or absorbed when one mole of a substance is formed from free elements in their most standard states. ∆H = ∑ ∆ Hf(products) ─ ∑∆Hf(reactants)

Calculate H° for the following reaction: 2 Na(s) + 2 H 2 O(l) → 2 Na. OH(aq) + H 2(g) Given the following information: Hf° (k. J/mol) Na(s) 0 H 2 O(l) – 286 Na. OH(aq) – 470 H 2(g) 0 H° = [2(-470 k. J/mol) + 0 k. J/mol)] – [2(-286 k. J/mol) + 0 k. J/mol] H° = [-940 kj/mol] – [-572 k. J/mol] H° = -368 k. J/mol

Entropy • Entropy is a complicated concept to truly understand. • Entropy can be thought of as a measure of the randomness of a system. • It may be best to think of entropy as the degree of dispersion. – As matter or energy disperses (becomes more free to move or becomes more spread out) entropy will increase.
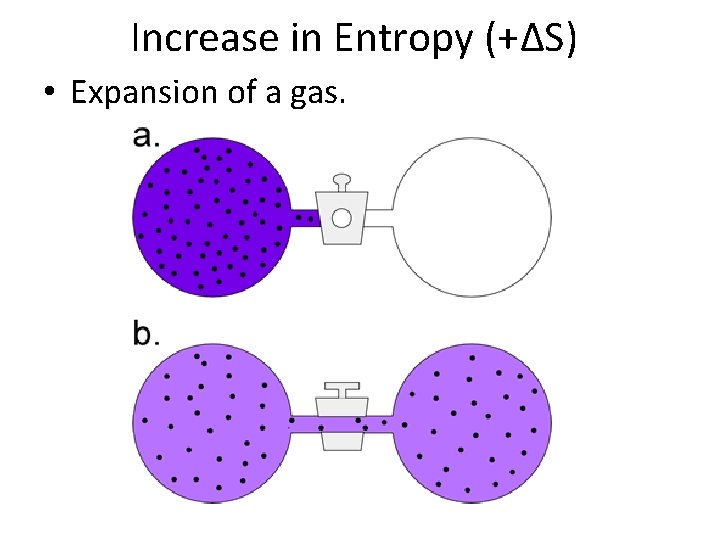
Increase in Entropy (+∆S) • Expansion of a gas.
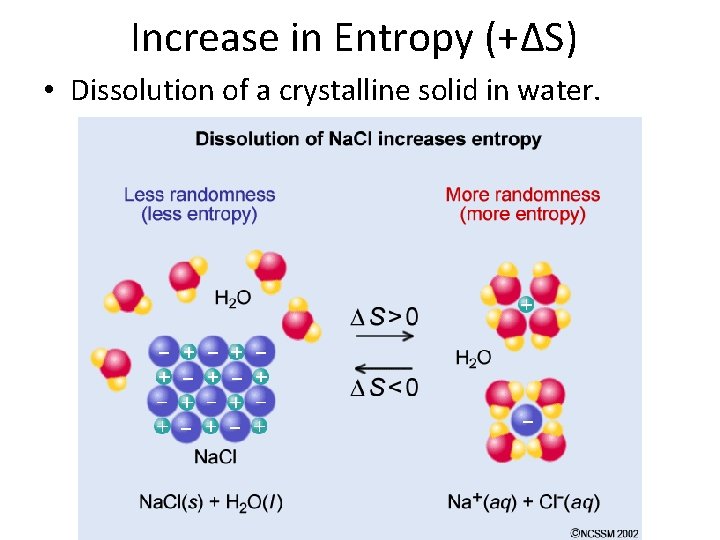
Increase in Entropy (+∆S) • Dissolution of a crystalline solid in water.
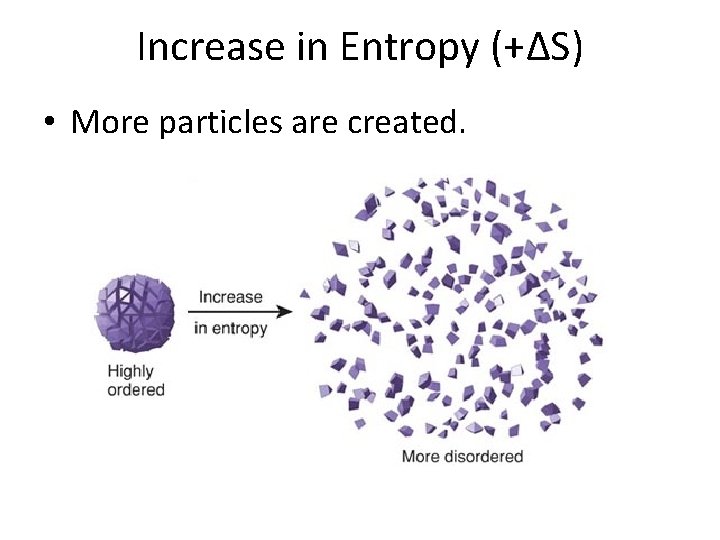
Increase in Entropy (+∆S) • More particles are created.

Increase in Entropy (+∆S) • More moles of particles are created.
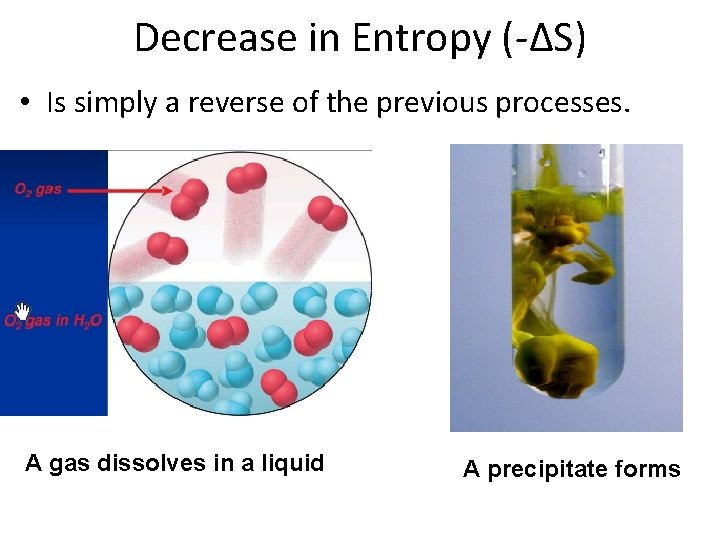
Decrease in Entropy (-∆S) • Is simply a reverse of the previous processes. A gas dissolves in a liquid A precipitate forms
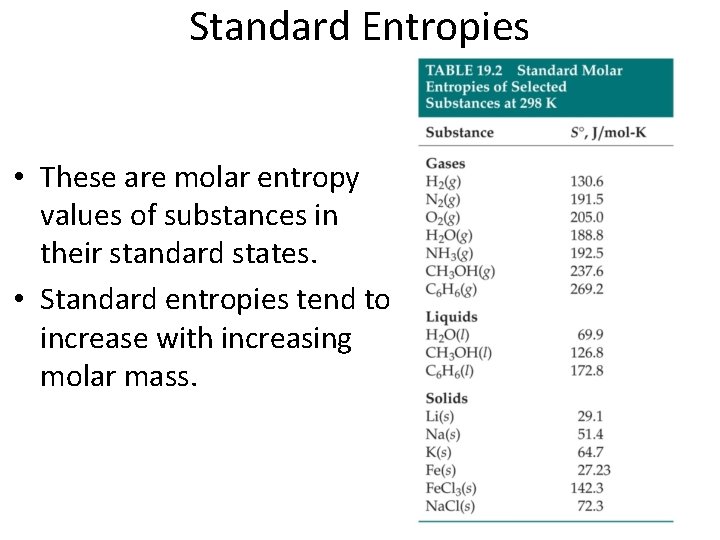
Standard Entropies • These are molar entropy values of substances in their standard states. • Standard entropies tend to increase with increasing molar mass.

Standard Entropies Larger and more complex molecules have greater entropies.

Third Law of Thermodynamics The entropy of any pure substance at 0 K is zero. We can interpret this to mean that as the temperature of a substance increases it’s entropy increases.

ΔS°reaction = ΣS°products – ΣS°reactants Calculate ΔS° for the following reaction: 2 Na(s) + 2 H 2 O(l) → 2 Na. OH(aq) + H 2(g) Given the following information: S° (J/K·mol) Na(s) 51 H 2 O(l) 70. Na. OH(aq) 50. H 2(g) 131 ΔS°= [2(50. ) + (131)] – [2(51) + 2(70. )] ΔS°= [231 J/mol K] – [242 J/mol K] ΔS°= – 11 J/mol K

Free energy (G) • a thermodynamic function that relates entropy and enthalpy to thermodynamic favorability (spontaneity).

“Gibbs” Free Energy • If a process is “thermodynamically favored” means the products are favored at equilibrium. • The term “not thermodynamically favored” means the reactants are favored at equilibrium.

Change in Free Energy (ΔG) • It is the change in free energy (ΔG) that determines whether a reaction is thermodynamically favorable or not.

(-∆G) vs (+∆G) • If -∆G the process is “thermodynamically favored” (spontaneous) and the products are favored at equilibrium. • If +∆G the process is “not thermodynamically favored” (nonspontaneous) and the reactants are favored at equilibrium.

Understanding ΔG • Just because a process is spontaneous or “thermodynamically favored” (-∆G) does not mean that it will proceed at any measurable rate. • A reaction that is thermodynamically favored, (spontaneous), may occur so slowly that in practice it does not occur at all.

Understanding ΔG • Thermodynamically favored reactions that do not occur at any measurable rate are said to be under “kinetic control”. • These reactions often have a very high activation energy. • The fact that a process does not proceed at a noticeable rate does not mean that the reaction is at equilibrium. • We would conclude that such a reaction is simply under kinetic control.

ΔG°reaction = ΣG°products – ΣG°reactants Determine ∆G for the reaction C 2 H 5 OH(g) + 3 O 2(g) → 2 CO 2(g) + 3 H 2 O(g)

ΔG°reaction = ΣG°products – ΣG°reactants C 2 H 5 OH(g) + 3 O 2(g) → 2 CO 2(g) + 3 H 2 O(g) ∆G = -1306 k. J/mol


Effect of H and S on Spontaneity G = H - T S

∆G = ∆H – T∆S C 2 H 5 OH(g) + 3 O 2(g) → 2 CO 2(g) + 3 H 2 O(g) Determine ∆G given: ∆H = -1277. 3 k. J/mol ∆S = +95. 64 J/mol K

∆G = ∆H – T∆S C 2 H 5 OH(g) + 3 O 2(g) → 2 CO 2(g) + 3 H 2 O(g) Determine ∆G given: ∆H = -1277. 3 k. J/mol ∆S = +95. 64 J/mol K ∆G = -1306 k. J/mol

∆G = ∆H – T∆S • Can be used to solve for ∆H, ∆S, or T

Effect of Temperature on Go Go = Ho - T So • For the reaction: N 2(g) + 3 H 2(g) 2 NH 3(g), Ho = -92 k. J and So = -199 J/K = -0. 199 k. J/K • At 25 o. C, T So = 298 K x (-0. 199 k. J/K) = -59. 3 k. J • Go = Ho - T So = -92 k. J – (-59. 3 k. J) = -33 k. J; • reaction is spontaneous at 25 o. C • At 250 o. C, T So = 523 K x (-0. 199 k. J/K) = -104 k. J; • Go = Ho - T So = -92 k. J – (-104 k. J) = +12 k. J; • reaction is nonspontaneous at 250 o. C • Therefore there is a temperature between 25 o. C and 250 o. C where the transition between spontaneous and nonspontaneous occurs. • How can we estimate this temperature?

∆G = 0 Reactants ↔ Products ∆G is 0 = equilibrium

G = H - T S o o o • Setting Go = 0 allows us to estimate the temperature at which a reaction that is spontaneous at one temperature but not at another transitions. • Go = 0 = Ho - T So

Effect of Temperature on Go G = H - T S o o o • For the reaction: N 2(g) + 3 H 2(g) 2 NH 3(g), Ho = -92 k. J and So = -199 J/K = -0. 199 k. J/K • Go = 0 = Ho - T So • T = Ho/ So • T = -92 k. J / -0. 199 k. J/K = 462 K = 189°C

Free Energy and Equilibrium Constant Go < 0, K > 1; reaction favors products formation Go > 0, K < 1; reaction favors reactants formation Go = 0, K = 1; reaction is at equilibrium and favors neither reactants nor products

Relationship of Free energy and equilibrium (reaction is product or reactant favored) • We can now use the following equation: ΔG° = -RT ln. K (Temp = Kelvin, R = 8. 314 J/mole K)

Calculating K from Go Go = -RTln. K • Consider the reaction: N 2(g) + 3 H 2(g) 2 NH 3(g), • ln. K = Go /-RT • At 25 o. C, Go = -33 k. J/mol • ln. K = -33, 000 J/mol/ -(8. 314 J/mol K x 298 K) = 13 • K = e 13 = 4. 4 x 105 (reaction in effect goes to completion) • At 250 o. C, Go = 12 k. J/mol; • ln. K = (12, 000 J/mol/-(8. 314 J/mol K x 523 K) = -2. 8 • K = e-2. 8 = 0. 061 (very little product is formed)
- Slides: 51