BIG BANG THEORY Universe Space all the matter

BIG BANG THEORY

Universe • Space & all the matter and energy in it • Formed ~ 13. 7 billion years ago a n y

Components of Universe 1. 2. 3. 4. Galaxies Solar systems Stars Nothing (matter)

What is this?

What is it made of?


MANY STARS
MANY STARS SOME SUN-LIKE

Galaxy • Large system of stars • At the center of most galaxies is a massive black hole! • There are billions of galaxies in the universe • Ex) Milky Way

Milky Way • The galaxy our sun and solar system is apart of • Our sun is 1 of billions of stars in the Milky Way

You are here!


Our Galaxy from the Earth

Galaxies: Hubble Space telescope image
Solar System • the sun (star) and all things orbiting around it • Includes planets, their moons, asteroids and comets • Our S. S. formed around 4. 6 billion years ago What is incorrect or misleading about this visual? Image taken from: http: //asm. arc. nasa. gov/Gallery/images/generic/LG_Capableofeverything. jpg

Planet • a major object which orbits around a star • 8 in our solar system

Q: What is a STAR?


Our Sun is a star!

Star • A star is a big ball of burning gas • Ex) Our Sun

IN THE BEGINNING…….
EVERYTHING!
EVERYTHING!
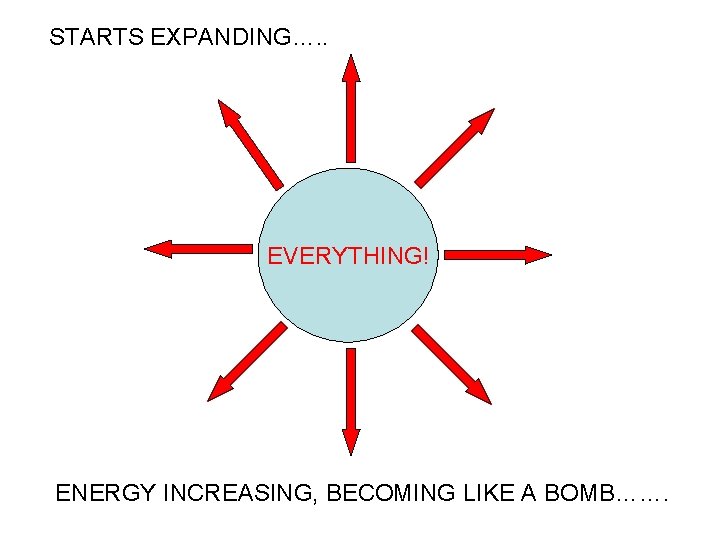
STARTS EXPANDING…. . EVERYTHING! ENERGY INCREASING, BECOMING LIKE A BOMB…….

Tick tick……

BOOM!

The Big Bang • Theory about how the universe formed

Big Bang Facts • Not really an ‘explosion’ • Universe expanded rapidly as a whole • (Universe is still expanding today as a result of the Big Bang) • Matter was created in the form of tiny particles (protons, neutrons, electrons) • Too hot for normal ‘stuff’ to form ( atoms, molecules)


COSMIC ‘SOUP’
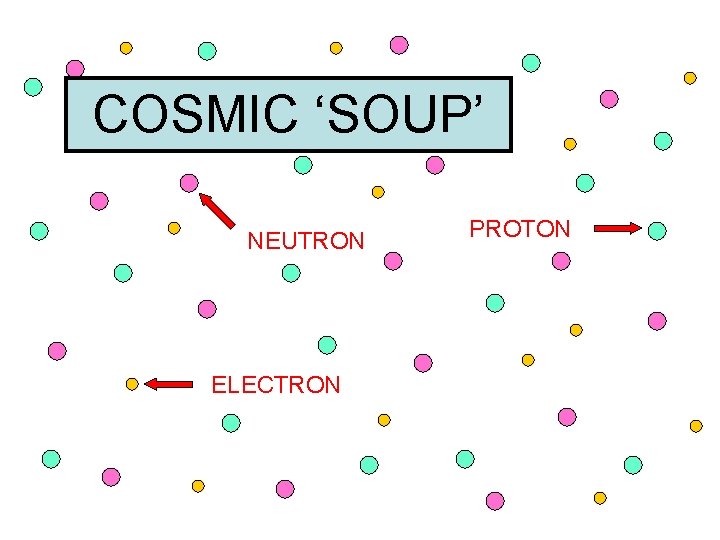
COSMIC ‘SOUP’ NEUTRON ELECTRON PROTON

300, 000 Years Later…. . • Universe much cooler, atoms start to form…. . • Hydrogen, Helium, normal ‘stuff’
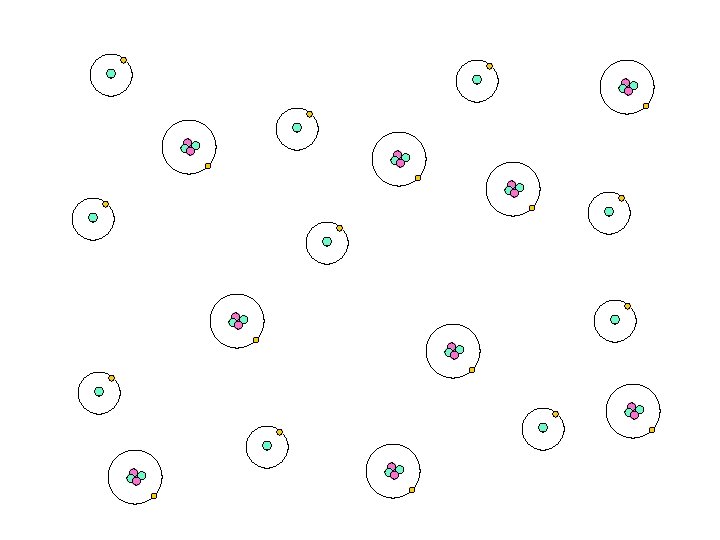
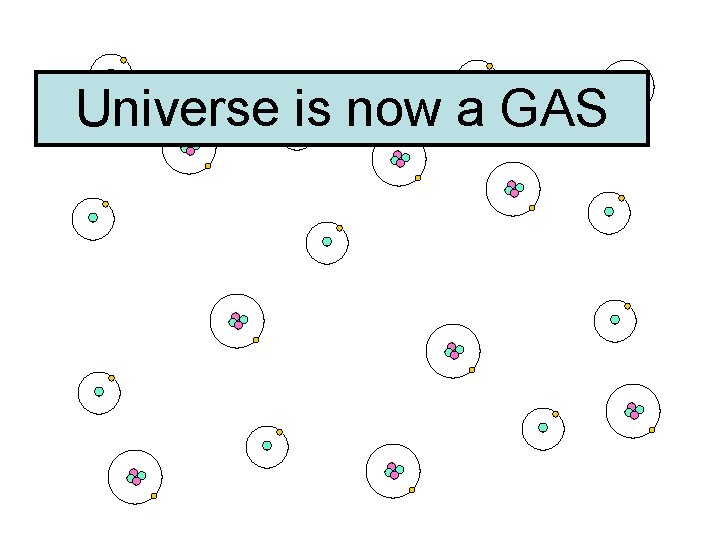
Universe is now a GAS
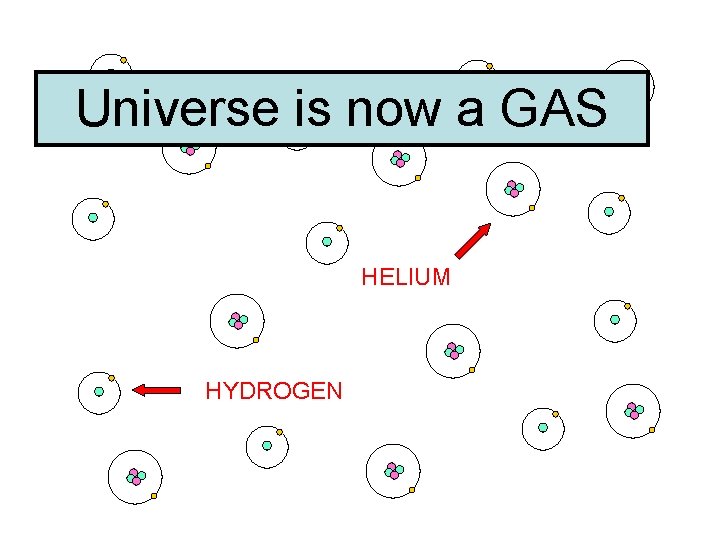
Universe is now a GAS HELIUM HYDROGEN
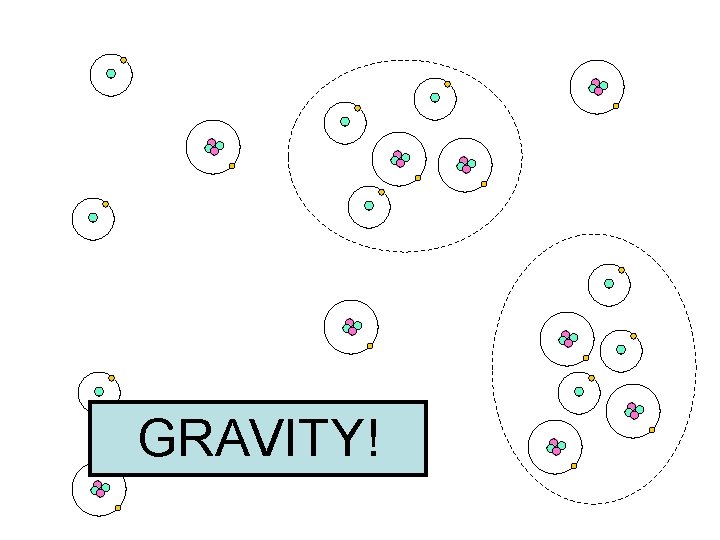
GRAVITY!

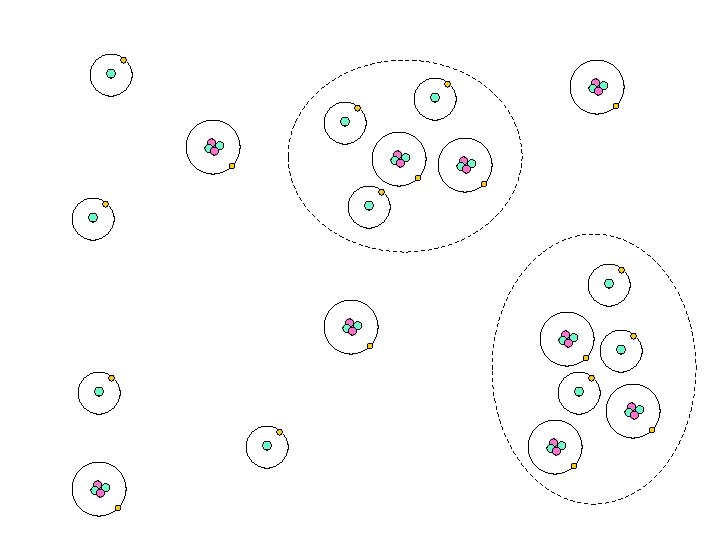
Universe Progression 1. started w/ a uniform gas 2. Small regions became more dense 3. The denser regions had larger gravity & started to pull other particles towards them 4. The gaseous Universe began to look ‘clumpy’

Universe Progression (cont) 5. Regions w/ more particles (higher density) become gas clouds 6. Gas clouds become stars & galaxies 7. Galaxies group together

The Gemini Telescope



Future? • The Universe is still expanding • All galaxies are moving apart • The ‘explosion’ of the big bang is still happening! • It may contine expanding forever • Or is may stop expanding, and fall inwards in a big CRUNCH!

What have we learnt? (don’t write) • The Universe started with a BIG BANG • Gradually the particles were drawn together under GRAVITY to form STARS and GALAXIES • Many stars have planets around them • The fate of the Universe is uncertain…

• We live on the Earth • The Earth is a planet in the Solar System, orbiting our star which we call the Sun • The Sun is part of a galaxy • When we look out into the sky at night, all of the stars we see are in our galaxy • But our galaxy is just one of many…. .

THE END
Lesson Overview 2. 3 Carbon Compounds

The Chemistry of Carbon – Carbon atoms have four valence electrons, allowing them to form strong covalent bonds with many other elements, including hydrogen, oxygen, phosphorus, sulfur, and nitrogen. – Living organisms are made up of molecules that consist of carbon and these other elements.
The Chemistry of Carbon – Carbon atoms can also bond to each other, which gives carbon the ability to form millions of different large and complex structures. – Carbon-carbon bonds can be single, double, or triple covalent bonds. – Chains of carbon atoms can even close up on themselves to form rings.

Macromolecules – Many of the organic compounds in living cells are macromolecules, or “giant molecules, ” made from thousands or even hundreds of thousands of smaller molecules. – Most macromolecules are formed by a process known as polymerization, in which large compounds are built by joining smaller ones together.
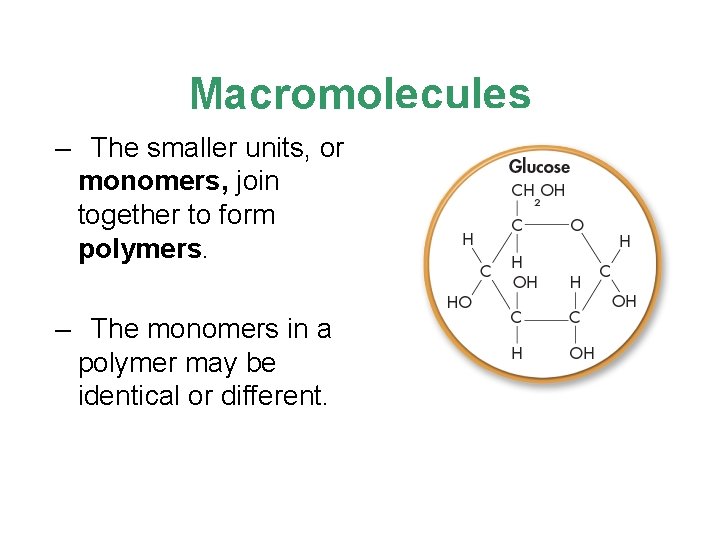
Macromolecules – The smaller units, or monomers, join together to form polymers. – The monomers in a polymer may be identical or different.

Macromolecules – Biochemists sort the macromolecules found in living things into groups based on their chemical composition. – The four major groups of macromolecules found in living things are carbohydrates, lipids, nucleic acids, and proteins.

Carbohydrates – Carbohydrates are compounds made up of carbon, hydrogen, and oxygen atoms, usually in a ratio of 1 : 2 : 1. – Living things use carbohydrates as their main source of energy. The breakdown of sugars, such as glucose, supplies immediate energy for cell activities. – Plants, some animals, and other organisms also use carbohydrates for structural purposes.
Carbohydrates – Many organisms store extra sugar as complex carbohydrates known as starches. The monomers in starch polymers are sugar molecules, such as glucose.

Simple Sugars – Single sugar molecules are also known as monosaccharides. – Besides glucose, monosaccharides include galactose, which is a component of milk, and fructose, which is found in many fruits. – Ordinary table sugar, sucrose, is a disaccharide, a compound made by joining glucose and fructose together.

Complex Carbohydrates – The large macromolecules formed from monosaccharides are known as polysaccharides.

Complex Carbohydrates – Many animals store excess sugar in a polysaccharide called glycogen. – When the level of glucose in your blood runs low, glycogen is broken down into glucose, which is then released into the blood. – The glycogen stored in your muscles supplies the energy for muscle contraction.

Complex Carbohydrates – Plants use a slightly different polysaccharide, called starch, to store excess sugar. – Plants also make another important polysaccharide called cellulose, which gives plants much of their strength and rigidity.

Lipids – – Lipids are a large and varied group of biological molecules. Lipids are made mostly from carbon and hydrogen atoms and are generally not soluble in water. The common categories of lipids are fats, oils, and waxes. – Lipids can be used to store energy. Some lipids are important parts of biological membranes and waterproof coverings. – Steroids synthesized by the body are lipids as well. Many steroids, such as hormones, serve as chemical messengers.

Lipids – Many lipids are formed when a glycerol molecule combines with compounds called fatty acids.

Lipids – If each carbon atom in a lipid’s fatty acid chains is joined to another carbon atom by a single bond, the lipid is said to be saturated. – If there is at least one carbon-carbon double bond in a fatty acid, the fatty acid is said to be unsaturated. – Lipids whose fatty acids contain more than one double bond are said to be polyunsaturated.
Lipids – Lipids that contain unsaturated fatty acids, such as olive oil, tend to be liquid at room temperature. – The data in the table illustrate how melting point decreases as the degree of unsaturation (number of double bonds) increases.

Nucleic Acids – Nucleic acids store and transmit hereditary, or genetic, information. – – Nucleic acids are macromolecules containing hydrogen, oxygen, nitrogen, carbon, and phosphorus. – Nucleic acids are polymers assembled from individual monomers known as nucleotides.

Nucleic Acids – Nucleotides consist of three parts: a 5 -carbon sugar, a phosphate group (–PO 4), and a nitrogenous base. – Some nucleotides, including adenosine triphosphate (ATP), play important roles in capturing and transferring chemical energy.
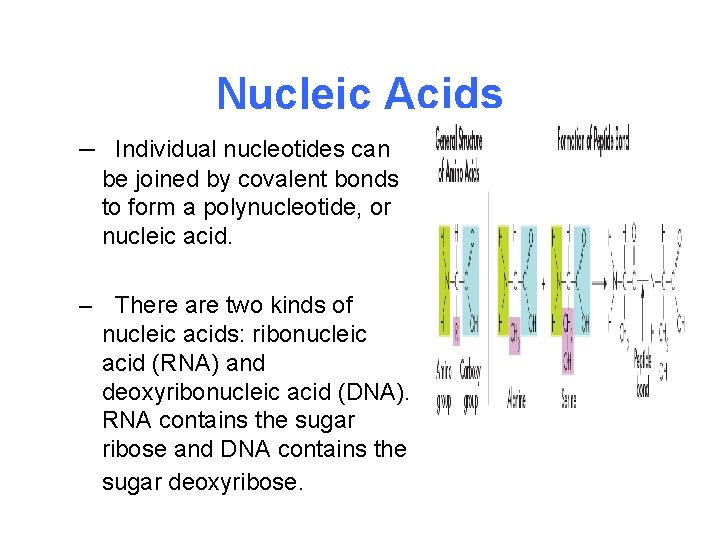
Nucleic Acids – Individual nucleotides can be joined by covalent bonds to form a polynucleotide, or nucleic acid. – There are two kinds of nucleic acids: ribonucleic acid (RNA) and deoxyribonucleic acid (DNA). RNA contains the sugar ribose and DNA contains the sugar deoxyribose.

Protein – Proteins are macromolecules that contain nitrogen as well as carbon, hydrogen, and oxygen. – Proteins are polymers of molecules called amino acids. – Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting substances into or out of cells, and helping to fight disease.

Protein – Amino acids are compounds with an amino group (–NH 2) on one end a carboxyl group (–COOH) on the other end. – Covalent bonds called peptide bonds link amino acids together to form a polypeptide. – A protein is a functional molecule built from one or more polypeptides.

Structure and Function – All amino acids are identical in the amino and carboxyl groups. Any amino acid can be joined to any other amino acid by a peptide bond formed between these amino and carboxyl groups.
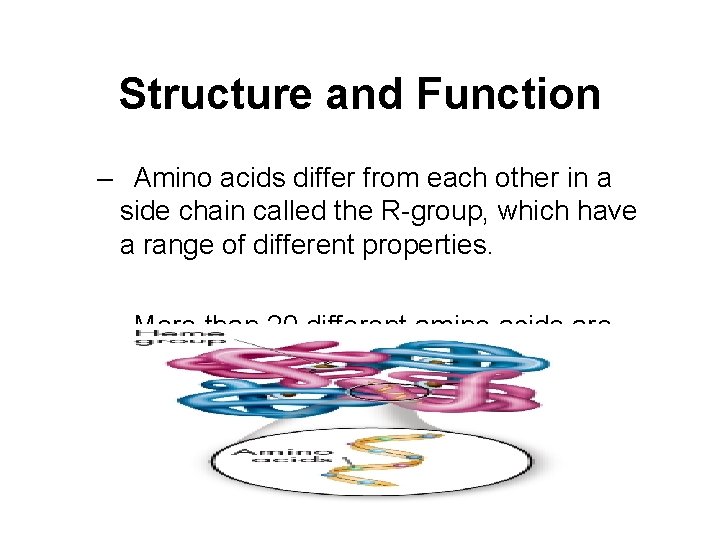
Structure and Function – Amino acids differ from each other in a side chain called the R-group, which have a range of different properties. – More than 20 different amino acids are found in nature. – This variety results in proteins being among the most diverse macromolecules.
Levels of Organization – Proteins have four levels of structure. – A protein’s primary structure is the sequence of its amino acids. – Secondary structure is the folding or coiling of the polypeptide chain.

Levels of Organization – Tertiary structure is the complete, threedimensional arrangement of a polypeptide chain. – Proteins with more than one chain have a fourth level of structure, which describes the way in which the different polypeptide chains are arranged with respect to each other. For example, the protein shown, hemoglobin, consists of four subunits .

Scientific Methodology: The Heart of Science – – What procedures are at the core of scientific methodology? – Scientific methodology involves observing and asking questions, making inferences and forming hypotheses, conducting controlled experiments, collecting and analyzing data, and drawing conclusions.

Observing and Asking Questions – Scientific investigations begin with observation, the act of noticing and describing events or processes in a careful, orderly way. – For example, researchers observed that marsh grass grows taller in some places than others. This observation led to a question: Why do marsh grasses grow to different heights in different places?

Inferring and Forming a Hypothesis – After posing questions, scientists use further observations to make inferences, or logical interpretations based on what is already known. – Inference can lead to a hypothesis, or a scientific explanation for a set of observations that can be tested in ways that support or reject it. –

Designing Controlled Experiments – Testing a scientific hypothesis often involves designing an experiment that keeps track of various factors that can change, or variables. Examples of variables include temperature, light, time, and availability of nutrients. – Whenever possible, a hypothesis should be tested by an experiment in which only one variable is changed. All other variables should be kept unchanged, or controlled. This type of experiment is called a controlled experiment.

Controlling Variables – It is important to control variables because if several variables are changed in the experiment, researchers can’t easily tell which variable is responsible for any results they observe. – The variable that is deliberately changed is called the independent variable (also called the manipulated variable). – The variable that is observed and that changes in response to the independent variable is called the dependent variable (also called the responding variable).

Control and Experimental Groups – Typically, an experiment is divided into control and experimental groups. – A control group is exposed to the same conditions as the experimental group except for one independent variable. – Scientists set up several sets of control and experimental groups to try to reproduce or replicate their observations.



- Slides: 80