Bi CNS 150 Lecture 10 Synaptic inhibition cable

Bi / CNS 150 Lecture 10 Synaptic inhibition; cable properties of neurons; electrical integration in cerebellum Monday, October 19, 2015 Henry Lester Chapter 2 (p. 22 -30); Chapter 10 (223 -234) 1
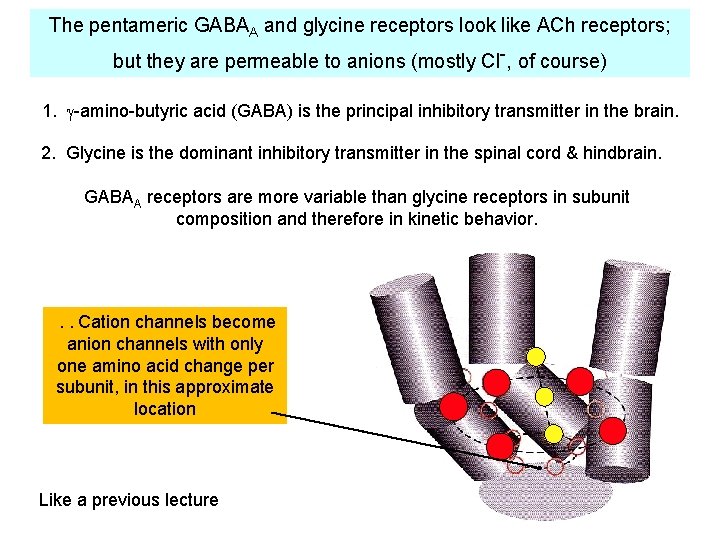
The pentameric GABAA and glycine receptors look like ACh receptors; but they are permeable to anions (mostly Cl-, of course) 1. -amino-butyric acid (GABA) is the principal inhibitory transmitter in the brain. 2. Glycine is the dominant inhibitory transmitter in the spinal cord & hindbrain. GABAA receptors are more variable than glycine receptors in subunit composition and therefore in kinetic behavior. . . Cation channels become anion channels with only one amino acid change per subunit, in this approximate location Like a previous lecture
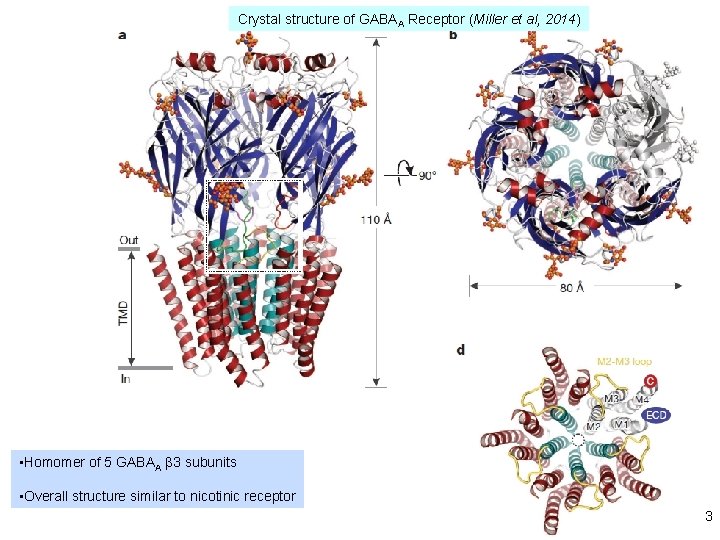
Crystal structure of GABAA Receptor (Miller et al, 2014) • Homomer of 5 GABAA β 3 subunits • Overall structure similar to nicotinic receptor 3
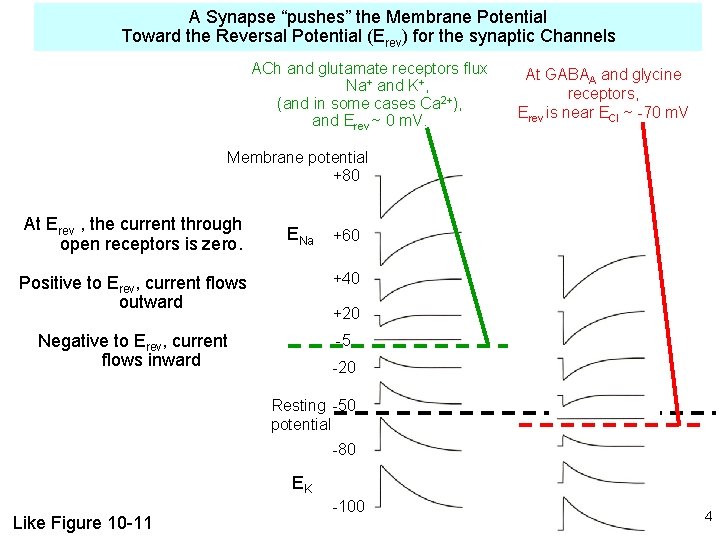
A Synapse “pushes” the Membrane Potential Toward the Reversal Potential (Erev) for the synaptic Channels ACh and glutamate receptors flux Na+ and K+, (and in some cases Ca 2+), and Erev ~ 0 m. V. At GABAA and glycine receptors, Erev is near ECl ~ -70 m. V Membrane potential +80 At Erev , the current through open receptors is zero. ENa +60 Positive to Erev, current flows outward +40 Negative to Erev, current flows inward -5 +20 -20 Resting -50 potential -80 EK Like Figure 10 -11 -100 4
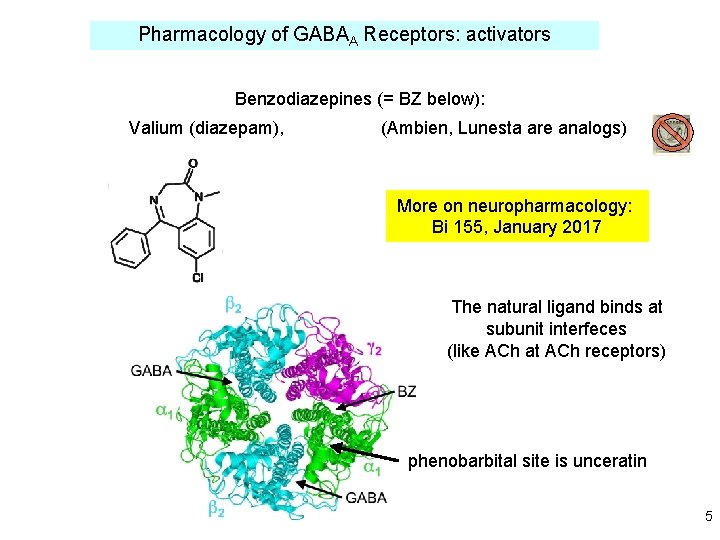
Pharmacology of GABAA Receptors: activators Benzodiazepines (= BZ below): Valium (diazepam), (Ambien, Lunesta are analogs) More on neuropharmacology: Bi 155, January 2017 The natural ligand binds at subunit interfeces (like ACh at ACh receptors) phenobarbital site is unceratin 5
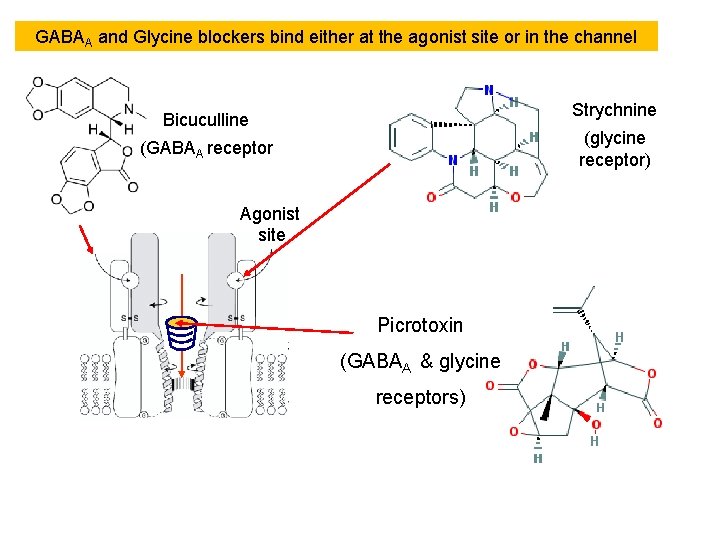
GABAA and Glycine blockers bind either at the agonist site or in the channel Strychnine Bicuculline (glycine receptor) (GABAA receptor Agonist site Picrotoxin (GABAA & glycine receptors)
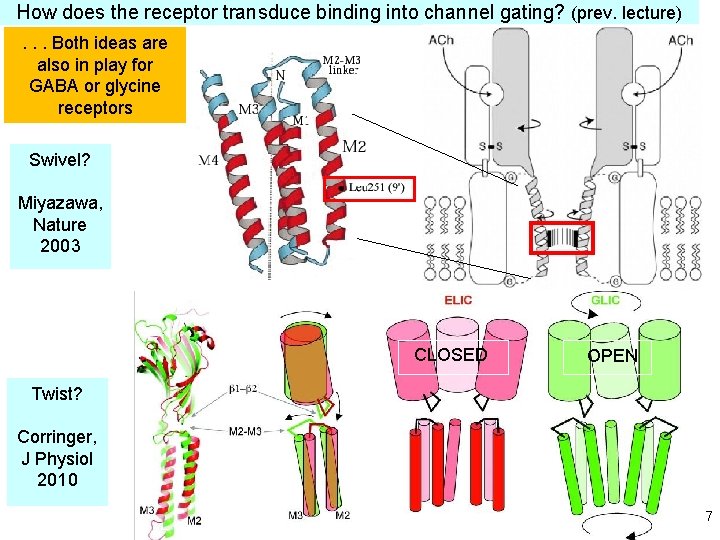
How does the receptor transduce binding into channel gating? (prev. lecture). . . Both ideas are also in play for GABA or glycine receptors Swivel? Miyazawa, Nature 2003 CLOSED OPEN Twist? Corringer, J Physiol 2010 7
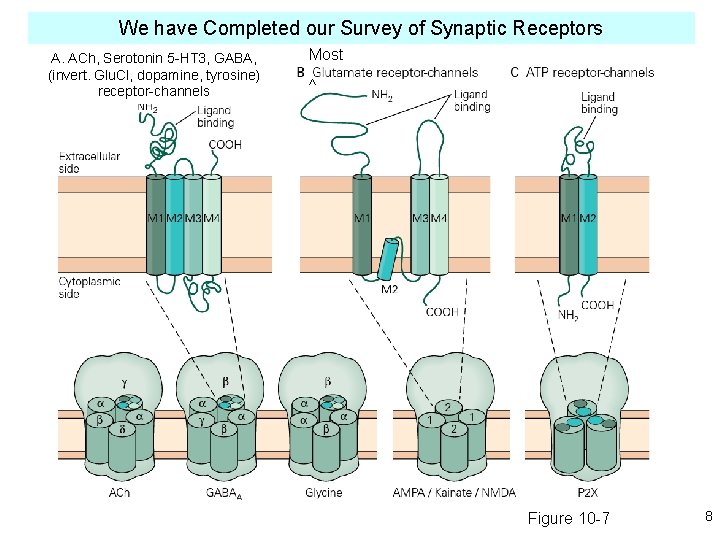
We have Completed our Survey of Synaptic Receptors A. ACh, Serotonin 5 -HT 3, GABA, (invert. Glu. Cl, dopamine, tyrosine) receptor-channels Most ^ Figure 10 -7 8
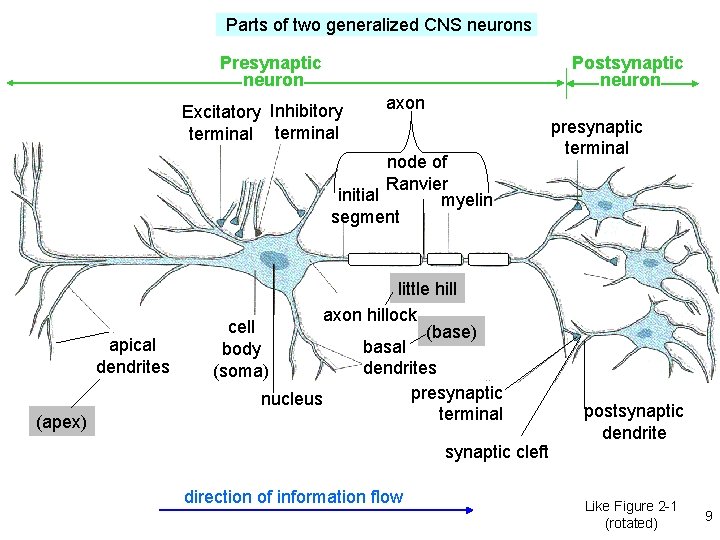
Parts of two generalized CNS neurons Postsynaptic neuron Presynaptic neuron Excitatory Inhibitory terminal axon node of Ranvier initial myelin segment apical dendrites (apex) little hill axon hillock cell (base) basal body dendrites (soma) presynaptic nucleus terminal synaptic cleft direction of information flow presynaptic terminal postsynaptic dendrite Like Figure 2 -1 (rotated) 9
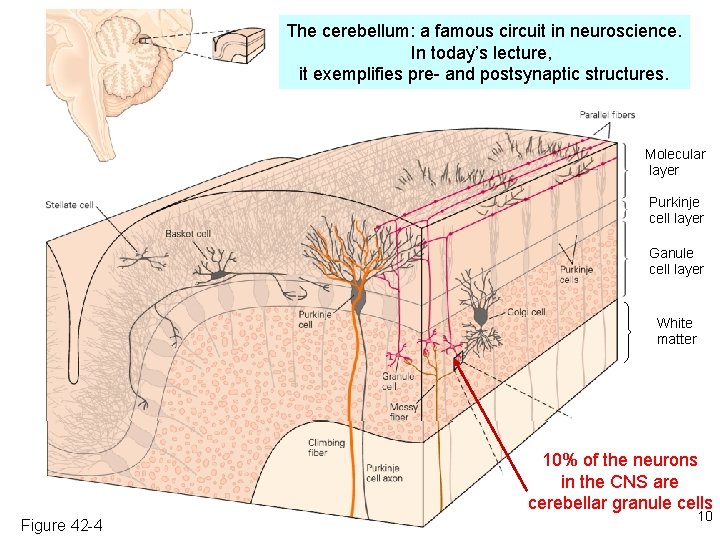
The cerebellum: a famous circuit in neuroscience. In today’s lecture, it exemplifies pre- and postsynaptic structures. Molecular layer Purkinje cell layer Ganule cell layer White matter 10% of the neurons in the CNS are cerebellar granule cells Figure 42 -4 10
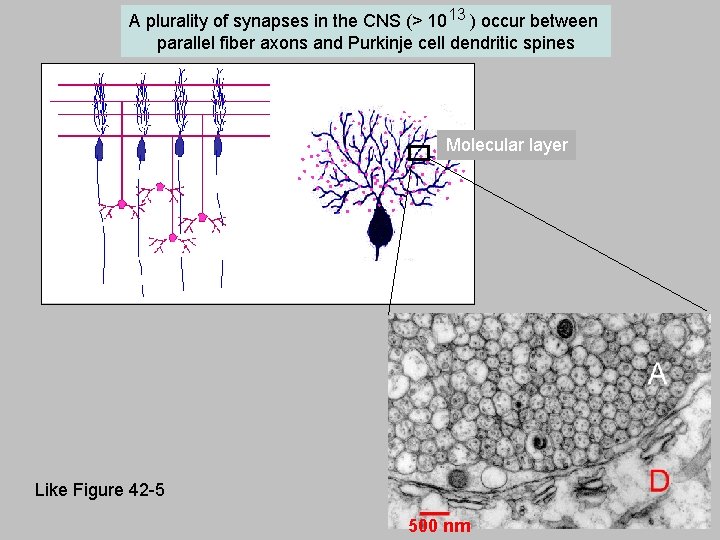
A plurality of synapses in the CNS (> 1013 ) occur between parallel fiber axons and Purkinje cell dendritic spines Molecular layer Like Figure 42 -5 500 nm 11
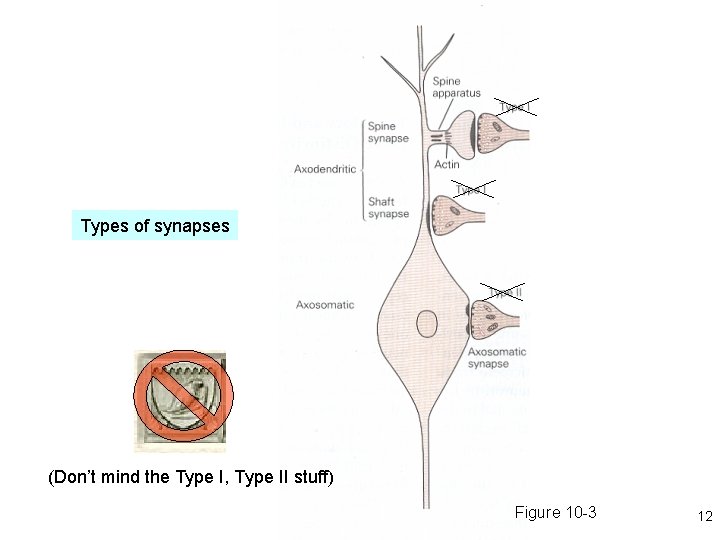
Types of synapses (Don’t mind the Type I, Type II stuff) Figure 10 -3 12

Types of synaptic integration 1. Temporal A. Molecular lifetimes B. Capacitive filtering 2. Spatial 3. Excitatory-inhibitory 13
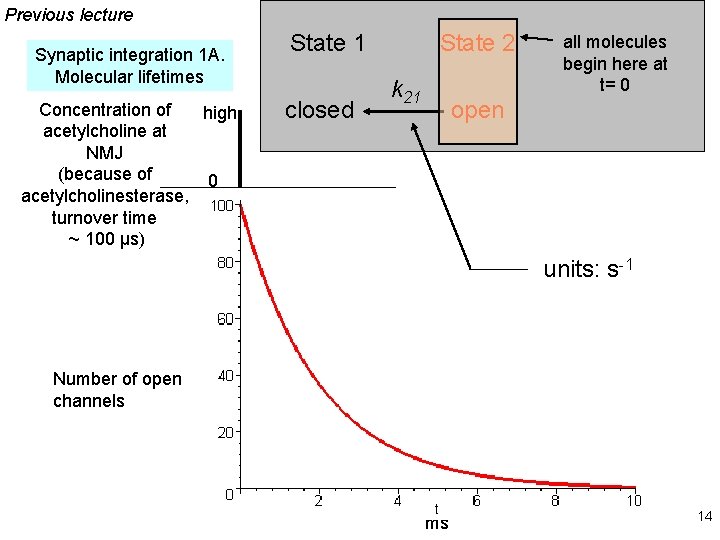
Previous lecture Synaptic integration 1 A. Molecular lifetimes Concentration of high acetylcholine at NMJ (because of 0 acetylcholinesterase, turnover time ~ 100 μs) State 1 closed State 2 k 21 all molecules begin here at t= 0 open units: s-1 Number of open channels ms 14
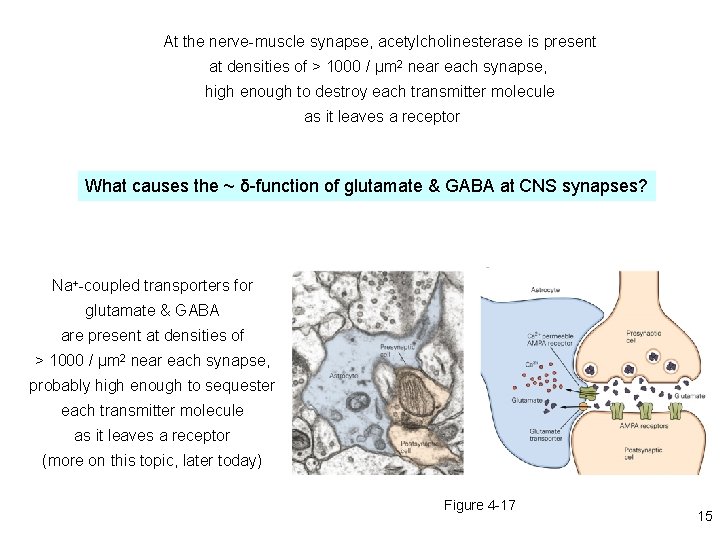
At the nerve-muscle synapse, acetylcholinesterase is present at densities of > 1000 / μm 2 near each synapse, high enough to destroy each transmitter molecule as it leaves a receptor What causes the ~ δ-function of glutamate & GABA at CNS synapses? Na+-coupled transporters for glutamate & GABA are present at densities of > 1000 / μm 2 near each synapse, probably high enough to sequester each transmitter molecule as it leaves a receptor (more on this topic, later today) Figure 4 -17 15
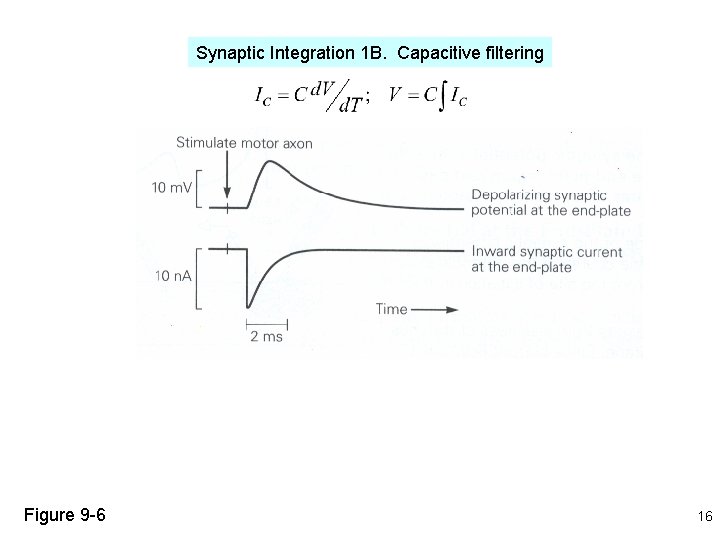
Synaptic Integration 1 B. Capacitive filtering Figure 9 -6 16
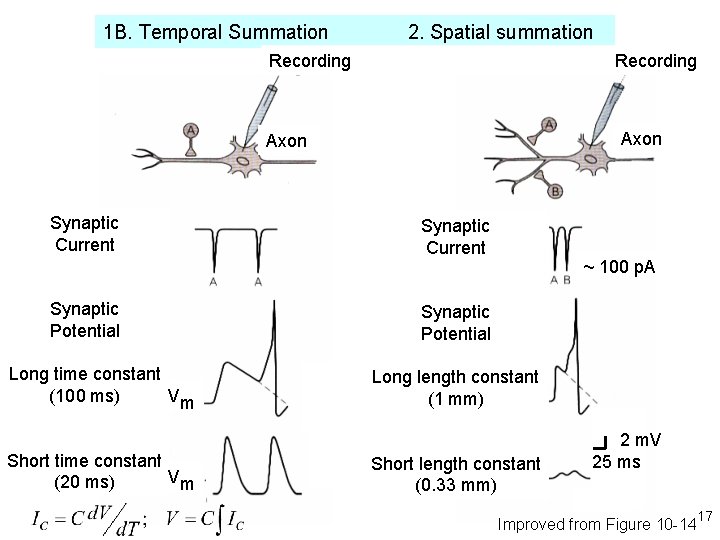
1 B. Temporal Summation 2. Spatial summation Recording Axon Synaptic Current Synaptic Potential Long time constant (100 ms) Vm Short time constant Vm (20 ms) Synaptic Current ~ 100 p. A Synaptic Potential Long length constant (1 mm) Short length constant (0. 33 mm) 2 m. V 25 ms Improved from Figure 10 -14 17
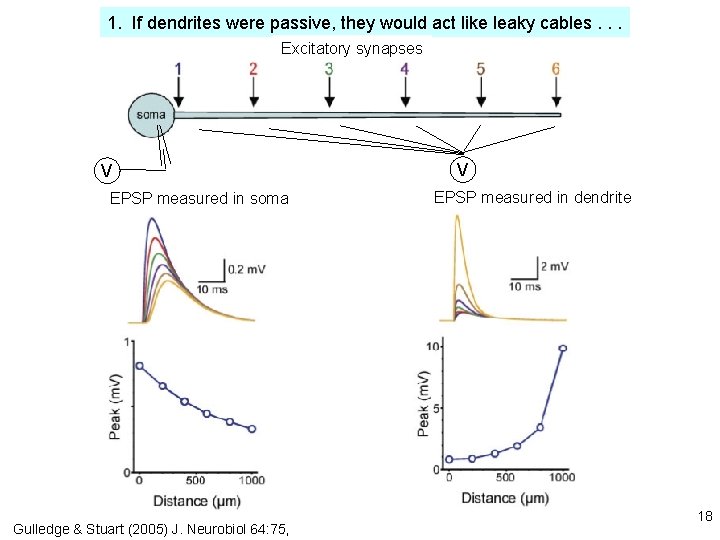
1. If dendrites were passive, they would act like leaky cables. . . Excitatory synapses V EPSP measured in soma Gulledge & Stuart (2005) J. Neurobiol 64: 75, V EPSP measured in dendrite 18
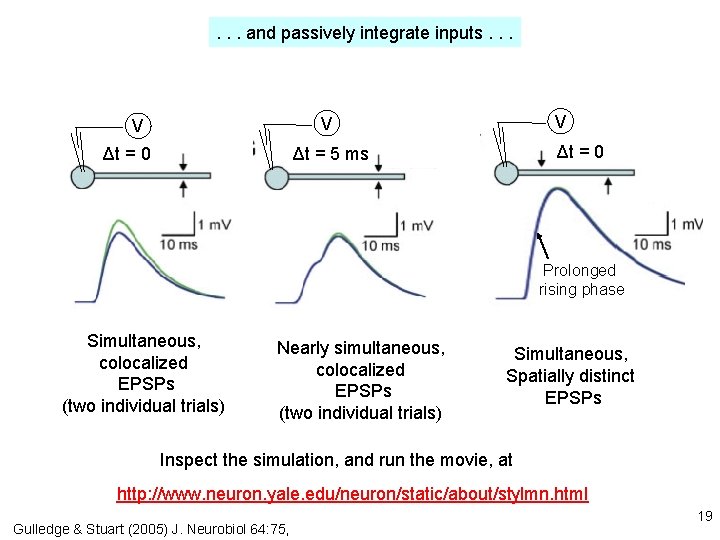
. . . and passively integrate inputs. . . V V V Δt = 0 Δt = 5 ms Prolonged rising phase Simultaneous, colocalized EPSPs (two individual trials) Nearly simultaneous, colocalized EPSPs (two individual trials) Simultaneous, Spatially distinct EPSPs Inspect the simulation, and run the movie, at http: //www. neuron. yale. edu/neuron/static/about/stylmn. html Gulledge & Stuart (2005) J. Neurobiol 64: 75, 19
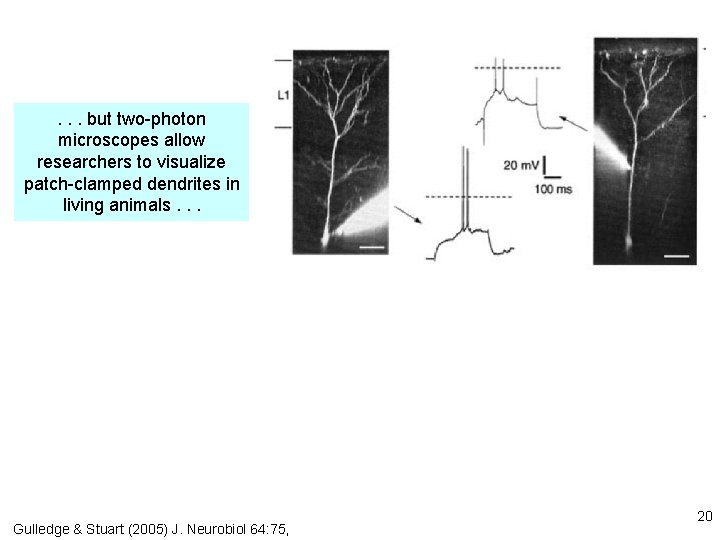
. . . but two-photon microscopes allow researchers to visualize patch-clamped dendrites in living animals. . . Gulledge & Stuart (2005) J. Neurobiol 64: 75, 20
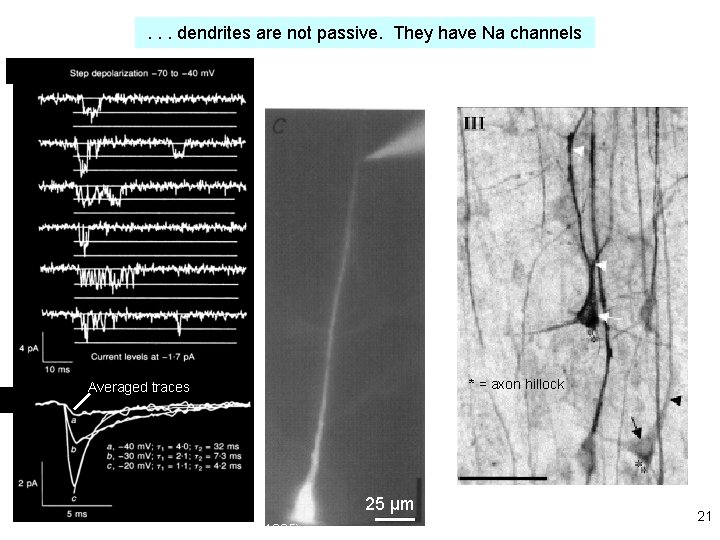
. . . dendrites are not passive. They have Na channels Now break the patch, to fill the cell with dye: * = axon hillock Averaged traces 25 μm Magee & Johnston, J Physiol (1995) immunocytochemistry Whitaker, Brain Res, 2001 21

brain slice . . . voltage-gated Na+ and Ca 2+ channels in dendrites lead to partial “backpropagation” of action potentials, implying that parts of cells can process signals semi-independently. Stay tuned! Gulledge & Stuart (2005) J. Neurobiol 64: 75, 22

3. Excitatory-inhibitory integration: The “veto principle” of inhibitory transmission Inhibitory synapses work best when they are “near“ the excitatory event they will inhibit. “Near” means < one cable length. A. Inhibitory synapses on dendrites do a good job of inhibiting EPSPs on nearby spines B. Inhibitory synapses on cell bodies and initial segments do a good job of inhibiting spikes 23
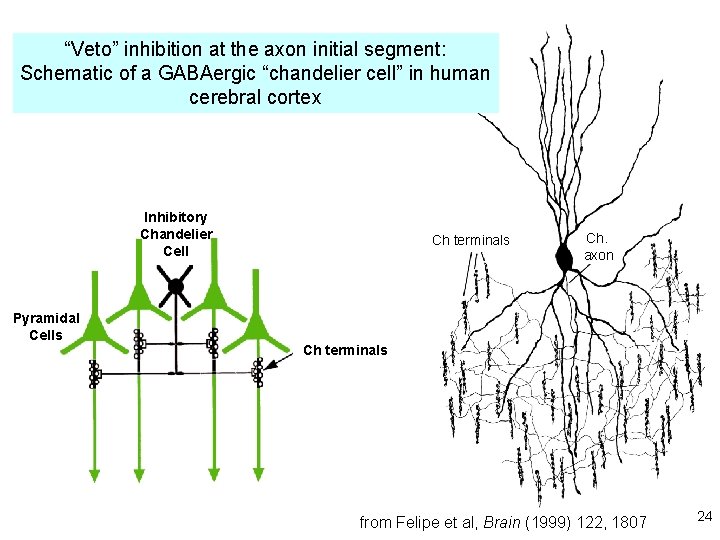
“Veto” inhibition at the axon initial segment: Schematic of a GABAergic “chandelier cell” in human cerebral cortex Inhibitory Chandelier Cell Pyramidal Cells Ch terminals Ch. axon Ch terminals from Felipe et al, Brain (1999) 122, 1807 24
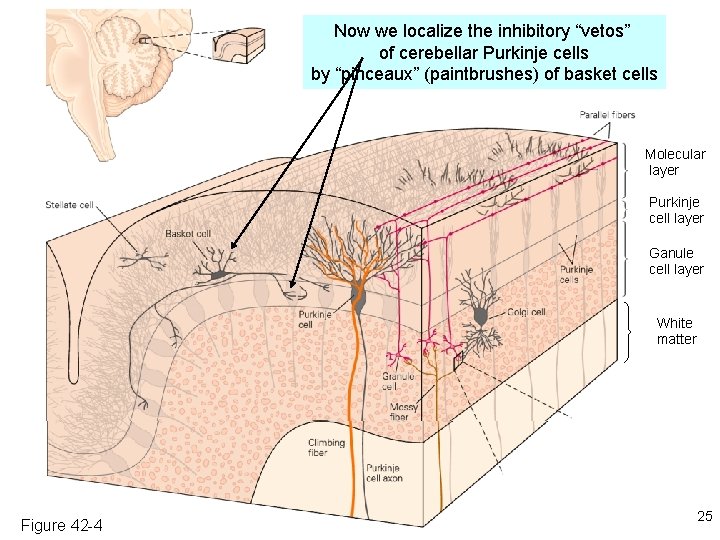
Now we localize the inhibitory “vetos” of cerebellar Purkinje cells by “pinceaux” (paintbrushes) of basket cells Molecular layer Purkinje cell layer Ganule cell layer White matter Figure 42 -4 25
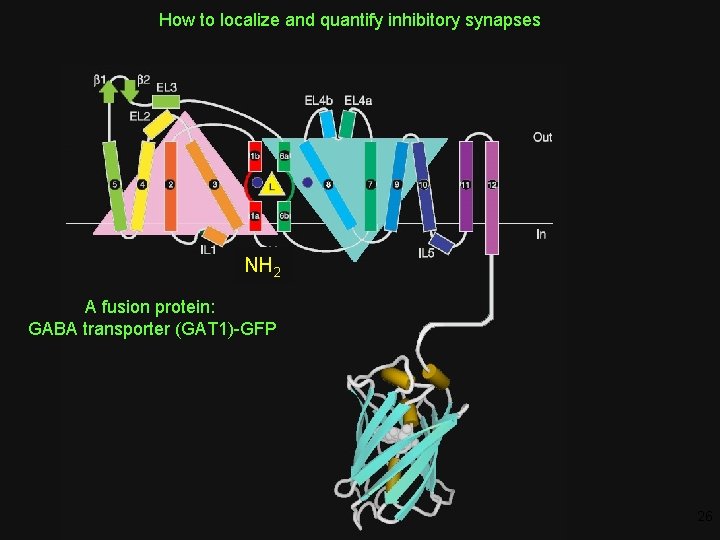
How to localize and quantify inhibitory synapses NH 2 A fusion protein: GABA transporter (GAT 1)-GFP 26
cerebellum 27
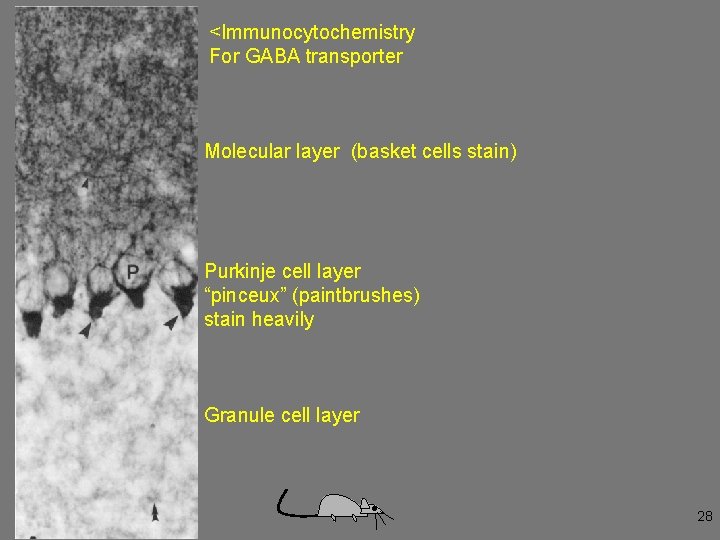
<Immunocytochemistry For GABA transporter Molecular layer (basket cells stain) Purkinje cell layer “pinceux” (paintbrushes) stain heavily Granule cell layer 28
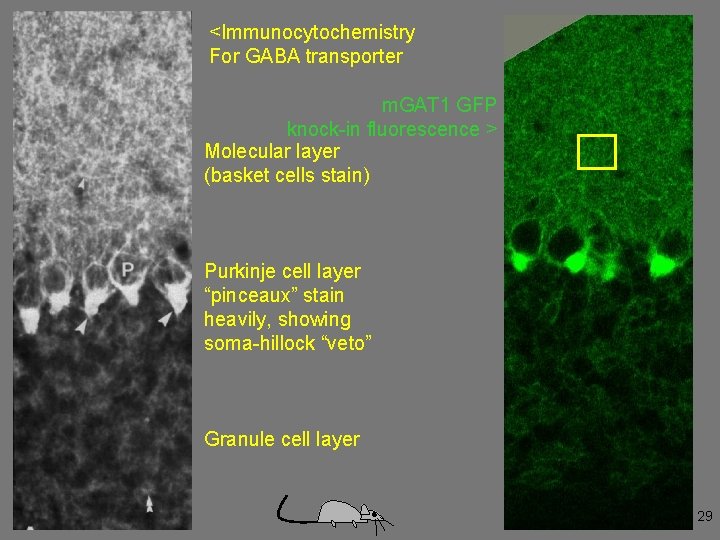
<Immunocytochemistry For GABA transporter m. GAT 1 GFP knock-in fluorescence > Molecular layer (basket cells stain) Purkinje cell layer “pinceaux” stain heavily, showing soma-hillock “veto” Granule cell layer 29
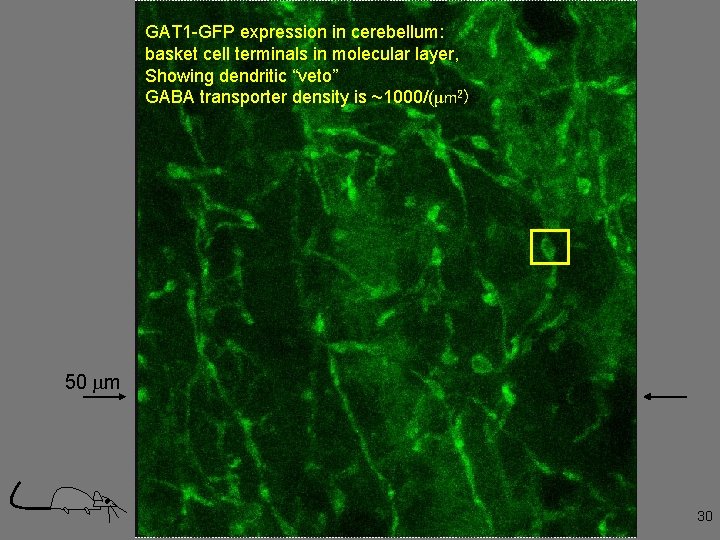
GAT 1 -GFP expression in cerebellum: basket cell terminals in molecular layer, Showing dendritic “veto” GABA transporter density is ~1000/(μm 2) 50 mm 30

HAL’s Office Hours, as usual Mon 1: 15 -2 Red Door End of Lecture 10 31
- Slides: 31