BEYOND THE TREES THE MAJESTY OF THE FOREST

BEYOND THE TREES: THE MAJESTY OF THE FOREST PLOT DISCOVERY SUMMIT EUROPE, MARCH 2017 Richard C. Zink, Ph. D. Principal Research Statistician Developer JMP Life Sciences SAS Institute, Inc. Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOT INTRODUCTION • The idiom unable to see the forest for the trees means that one fails to grasp the main issue because of excessive attention to details [1] • Data visualization helps us to see such forests Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOTS Forest plots have been around since 1970 s [2] • First displayed results from several studies (1978) • First meta-analysis (1982) • Adjust symbol size (1998) • Figure from [2]. Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOTS • This plot represents a more modern update of the previous figure • Figure from [2] • Tables have become part of the figure Copyright © 2012, SAS Institute Inc. All rights reserved.
![THE MAJESTY OF THE FOREST PLOT META-ANALYSIS Figure from [3] using data from [4, THE MAJESTY OF THE FOREST PLOT META-ANALYSIS Figure from [3] using data from [4,](http://slidetodoc.com/presentation_image/29f90d32b11fd01718a9d3e8d7f1a9ca/image-5.jpg)
THE MAJESTY OF THE FOREST PLOT META-ANALYSIS Figure from [3] using data from [4, 5] • Uses CI for overall results • Should use proper metaanalysis techniques • Copyright © 2012, SAS Institute Inc. All rights reserved.
![THE MAJESTY OF THE FOREST PLOT SUBGROUPS Figure from [6] using suggestions for summarizing THE MAJESTY OF THE FOREST PLOT SUBGROUPS Figure from [6] using suggestions for summarizing](http://slidetodoc.com/presentation_image/29f90d32b11fd01718a9d3e8d7f1a9ca/image-6.jpg)
THE MAJESTY OF THE FOREST PLOT SUBGROUPS Figure from [6] using suggestions for summarizing interactions from [7]. Copyright © 2012, SAS Institute Inc. All rights reserved.
![THE MAJESTY OF THE FOREST PLOT REPLACEMENT FOR BAR CHARTS Figures from [8]. Can THE MAJESTY OF THE FOREST PLOT REPLACEMENT FOR BAR CHARTS Figures from [8]. Can](http://slidetodoc.com/presentation_image/29f90d32b11fd01718a9d3e8d7f1a9ca/image-7.jpg)
THE MAJESTY OF THE FOREST PLOT REPLACEMENT FOR BAR CHARTS Figures from [8]. Can summarize baseline fraud as suggested in [9]. Copyright © 2012, SAS Institute Inc. All rights reserved.
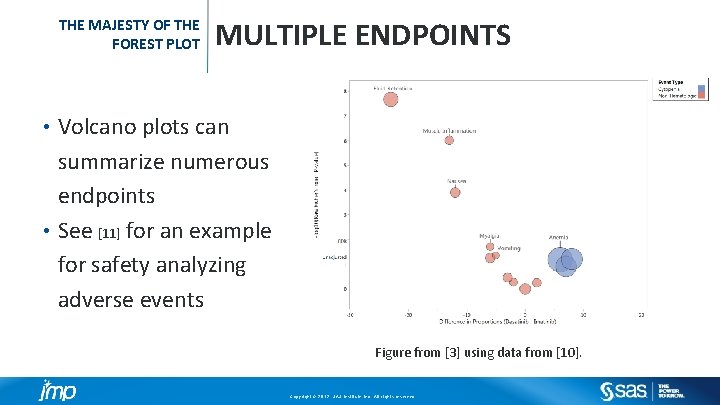
THE MAJESTY OF THE FOREST PLOT MULTIPLE ENDPOINTS Volcano plots can summarize numerous endpoints • See [11] for an example for safety analyzing adverse events • Figure from [3] using data from [10]. Copyright © 2012, SAS Institute Inc. All rights reserved.
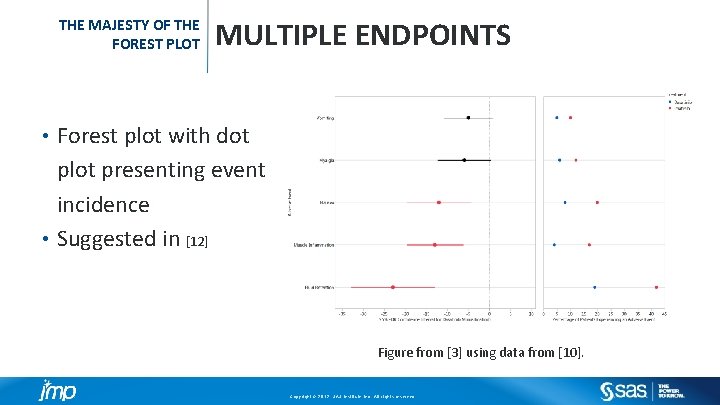
THE MAJESTY OF THE FOREST PLOT MULTIPLE ENDPOINTS Forest plot with dot plot presenting event incidence • Suggested in [12] • Figure from [3] using data from [10]. Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOT BAYESIAN ANALYSES Bayesian hierarchical models for adverse events [13] Yse ~ Bin(Nt, tse) and Xse ~ Bin(Nc, cse) and define logit(cse) = γse and logit(tse) = γse+ θse. Nt subjects and the Control group of Nc subjects, respectively. Assume the following priors: Stage 1 Priors: γse ~ N(μγs, σ2γs) θse ~ πs δ(0) + (1 - πs) N(μθs, σ2θs) Stage 2 Priors: μγs ~ N(μγ 0, τ2γ 0) σ2γs ~ IG(∝γ, βγ) μθs ~ N(μθ 0, τ2θ 0) σ2θs ~ IG(∝θ, βθ) Stage 3 Priors: μγ 0 ~ N(μγ 00, τ2γ 00) τ2γ 0 ~ IG(∝γ 00, βγ 00) μθ 0 ~ N(μθ 00, τ2θ 00) τ2θ 0 ~ IG(∝θ 00, βθ 00) Further assume that πs ~ Beta(∝π, βπ), ∝π ~ Exp(λα) I[∝π > 1] and βπ ~Exp(λβ) I[βπ > 1], and set μγ 00 = μθ 00 = 0, τ2γ 00 = τ2θ 00 = 10, ∝γ = ∝θ = ∝γ 00 = ∝θ 00 = 3, βγ = βθ = βγ 00 = βθ 00 = 1 and λα = λβ = 0. 1. Copyright © 2012, SAS Institute Inc. All rights reserved.
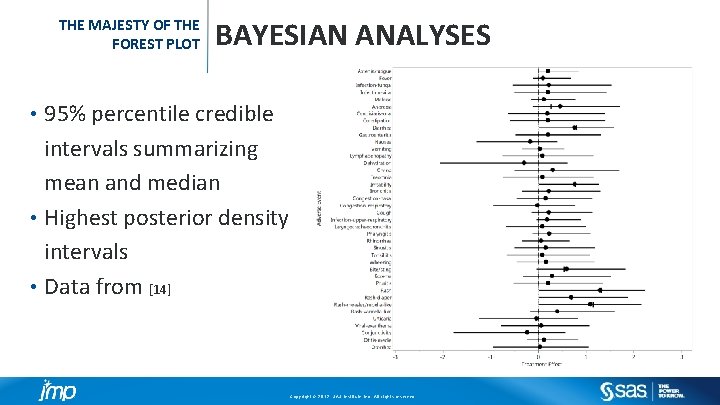
THE MAJESTY OF THE FOREST PLOT BAYESIAN ANALYSES 95% percentile credible intervals summarizing mean and median • Highest posterior density intervals • Data from [14] • Copyright © 2012, SAS Institute Inc. All rights reserved.
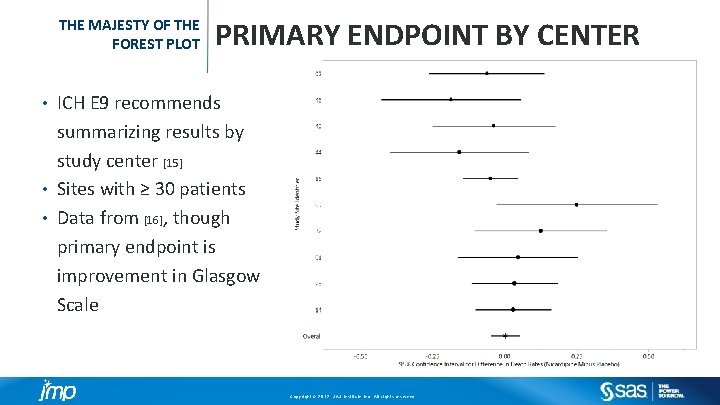
THE MAJESTY OF THE FOREST PLOT PRIMARY ENDPOINT BY CENTER ICH E 9 recommends summarizing results by study center [15] • Sites with ≥ 30 patients • Data from [16], though primary endpoint is improvement in Glasgow Scale • Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOT • • • ESTIMANDS ICH E 9 revision to address analysis populations [17, 18] National research council report on missing data [19] Inconsistent definitions of ITT and PP [17, 20] ITT and PP are ways of getting an estimate, all have flaws Estimates of interest may vary according to sponsor, regulator, patient, payer Greater emphasis on multiple sensitivity analyses Copyright © 2012, SAS Institute Inc. All rights reserved.
![THE MAJESTY OF THE FOREST PLOT ESTIMANDS • Example from [21] • Comparing progression THE MAJESTY OF THE FOREST PLOT ESTIMANDS • Example from [21] • Comparing progression](http://slidetodoc.com/presentation_image/29f90d32b11fd01718a9d3e8d7f1a9ca/image-14.jpg)
THE MAJESTY OF THE FOREST PLOT ESTIMANDS • Example from [21] • Comparing progression free survival (PFS) between drug + chemo vs chemo alone in patients with metastatic colorectal cancer • Would likely also specify mechanism to account for missing data (if needed) Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOT JMP FOREST PLOT ADD-IN • Available for free download from the JMP User Community [22] • Plots Meta-analysis and subgroup plots • Size bubbles, color significant intervals • Add confidence interval text • Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOT 1. 2. 3. 4. 5. 6. 7. 8. REFERENCES Google. Lewis S & Clarke M. (2001). Forest plots: trying to see the wood and the trees. British Medical Journal 322: 1479 -1480. Ivanova A, Marchenko O, Jiang Q & Zink RC. (2017). Safety monitoring and analysis in oncology trials. In: Roychoudhury S & Lahiri S, eds. Statistical Challenges in Oncology Clinical Development. Boca Raton, Florida: CRC Press. (Forthcoming) Novartis Pharmaceuticals Corporation. (2015, Jan). Prescribing Information for Gleevec (imatinib mesylate) tablets for oral use. Available at: http: //www. pharma. us. novartis. com/product/pi/pdf/gleevec_tabs. pdf. Gastrointestinal Stromal Tumor Meta-Analysis Group (Meta. GIST). (2010). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: A meta-analysis of 1, 640 patients. Journal of Clinical Oncology 28: 1247 -1253. Home PD, Pocock SJ, Beck-Nielsen H, Curtis PS, Gomis R, Hanefeld M, Jones NP, Komajda M & JJV Mc. Murray. (2009). Rosiglitazone evaluated for cardiovascular outcomes in oral agent combination therapy for type 2 diabetes (RECORD): A multicentre, randomised, open-label trial. Lancet 373: 2125– 2135. Committee for Medicinal Products for Human Use (CHMP). (2014). Guideline on the investigation of subgroups in confirmatory clinical trials (draft). European Medicines Agency. Available at: http: //www. ema. europa. eu/docs/en_GB/document_library/Scientific_guideline/2014/02/WC 500160523. pdf. Duke SP, Bancken F, Crowe B, Soukup M, Botsis T & Forshee R. (2005). Seeing is believing: good graphic design principles for medical research. Statistics in Medicine 34: 3040 -3059. Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOT 9. 10. 11. 12. 13. 14. 15. 16. REFERENCES Al-Marzouki S, Evans S, Marshall T & Roberts I. (2005). Are these data real? Statistical methods for the detection of data fabrication in clinical trials. British Medical Journal 331: 267– 270. Kantarjian H, Shah NP, Hochhaus A, et. al. (2010). Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. New England Journal of Medicine 362: 2260 -2270. Zink RC, Wolfinger RD and Mann G. (2013). Summarizing the incidence of adverse events using volcano plots and time windows. Clinical Trials 10: 398 -406. Amit O, Heiberger RM & Lane PW. (2008). Graphical approaches to the analysis of safety data from clinical trials. Pharmaceutical Statistics 7: 20 -35. Berry SM & Berry DA. (2004). Accounting for multiplicities in assessing drug safety: A three-level hierarchical mixture model. Biometrics 60: 418 -426. Mehrotra DV & Heyse JF. (2004). Use of the false discovery rate for evaluating clinical safety data. Statistical Methods in Medical Research 13: 227 -238. International Conference on Harmonization. (1998). Guidline E 9: Statistical Principles for Clinical Trials. Haley EC, Kassell NF & Torner JC. (1993). A randomized controlled trial of high-dose intravenous nicardipine in aneurysmal subarachnoid hemorrhage. Journal of Neurosurgery 78: 537 -547. Copyright © 2012, SAS Institute Inc. All rights reserved.

THE MAJESTY OF THE FOREST PLOT REFERENCES 17. International Conference of Harmonisation. (2014). Final concept paper for E 9(R 1): Addendum to statistical principles for 18. 19. 20. 21. 22. clinical trials on choosing appropriate estimands and defining sensitivity analyses in clinical trials. Akacha M, Bretz F & Ruberg S. (2016). Estimands in clinical trials – broadening the perspective. Statistics in Medicine 36: 5 -19. National Research Council. (2010). The Prevention and Treatment of Missing Data in Clinical Trials. Washington, DC: National Academies Press. Fletcher C, Tsuchiya S & Mehrotra DV. (2017). Current practices in choosing estimands and sensitivity analyses in clinical trials: Results of the ICH E 9 survey. Therapeutic Innovation and Regulatory Science 51: 69 -76. Fletcher C. (2016). Incorporating estimands in the clinical trial protocol. EFSPI Workshop on Regulatory Statistics. Zink RC. (2012). JMP forest plot add-in. Available at: https: //community. jmp. com/t 5/JMP-Add-Ins/Forest-Plot-Add-In/tap/23977. Copyright © 2012, SAS Institute Inc. All rights reserved.
- Slides: 18