Beta and Gamma Decay Series Bell Ringer Write

Beta and Gamma Decay Series

Bell Ringer • Write a nuclear decay equation for the alpha decay of Einsteinium-255. • 255 Es • 99 α 251 97 Bk + 4α 2

• I can complete beta and gamma decay equations and write a decay series with a net equation.

What happens in BETA DECAY? • During BETA DECAY: – A NEUTRON splits to form a PROTON and an ELECTRON. – The PROTON is added to the NUCLEUS. – The ELECTRON is lost as a BETA PARTICLE.

How does Beta Decay change the nucleus? • What really happened? – 1 – Lost a NEUTRON – decreases NUCLEON NUMBER by 1. – 2. – Gained a PROTON, increases BOTH the NUCLEON NUMBER and the AN by 1. – NET CHANGE – NUCLEON NUMBER stays the same, AN increases by 1…. the atom is transmuted into an isotope of the next higher element.

What does a BETA PARTICLE consist of? What is its SYMBOL? • A BETA PARTICLE consists of a SINGLE ELECTRON. • The SYMBOL is: • 0 -1 β

Beta Decay Equation • Show a Beta decay for I-128. • Follows same pattern as Alpha Decay Equation • • 128 I 53 β 128 Xe 54 + 0 -1 β Write Beta Decay equations for: A. Po-210 B. N-16 C. Co-60
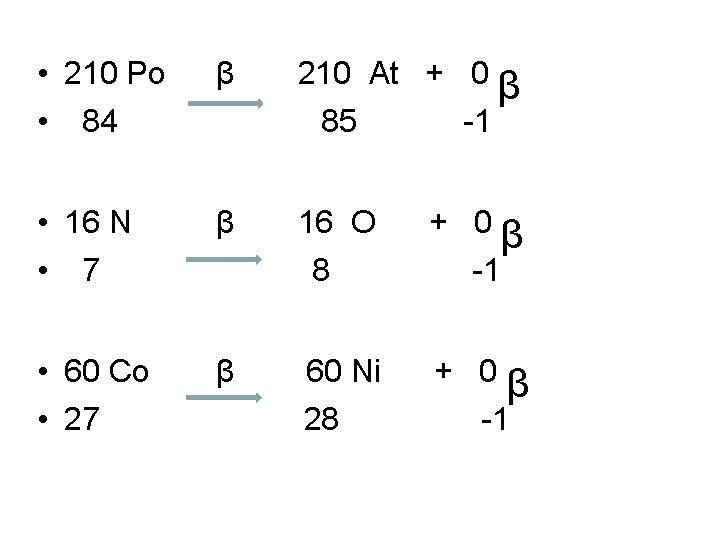
• 210 Po • 84 β 210 At + 0 β 85 -1 • 16 N • 7 β 16 O 8 + 0β -1 • 60 Co • 27 β 60 Ni 28 + 0β -1

GAMMA DECAY • GAMMA DECAY differs from Alpha and Beta in the fact that NO PARTICLES are lost during GAMMA DECAY. • GAMMA DECAY involves the emission of HIGH FREQUENCY RADIATION [Energy]. • Usually occurs along side alpha and beta decays. o • Symbol for Gamma is γ o •

• A Gamma Decay is written in the same manner as an Alpha or Beta Decay, except there are NO CHANGES to the numbers: it is simply a RELEASE OF ENERGY. • Example: 238 • 92 U γ E + 238 U + 0 Y 92 0

DECAY SERIES • A DECAY SERIES is all the ALPHA, BETA and GAMMA DECAYS an element undergoes to reach a STABLE FORM. – Below AN 83 and a P+ to No ratio that is stable!

Writing a Decay Series • Write the decay series for Radon-224 which undergoes this series of decays: α, β, γ, β, α, γ • Write a NET EQUATION to summarize this series.
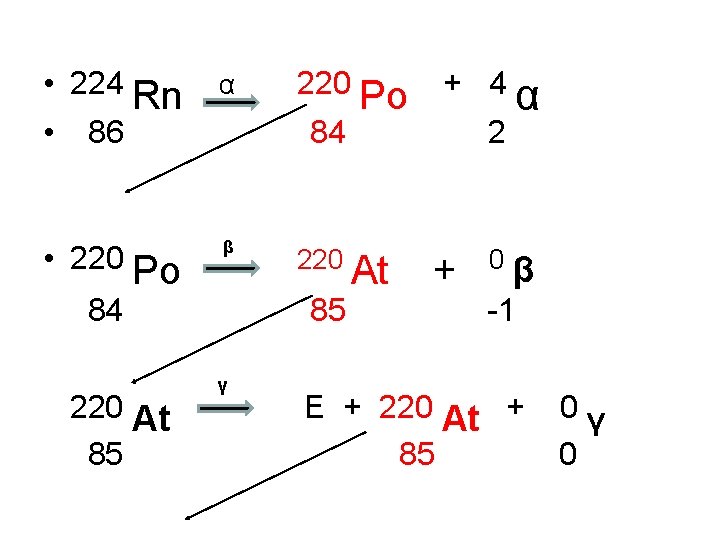
• 224 Rn • 86 α • 220 Po β + 4 84 84 220 At 85 220 Po 220 85 γ At 2 + α 0β -1 E + 220 At + 85 0γ 0
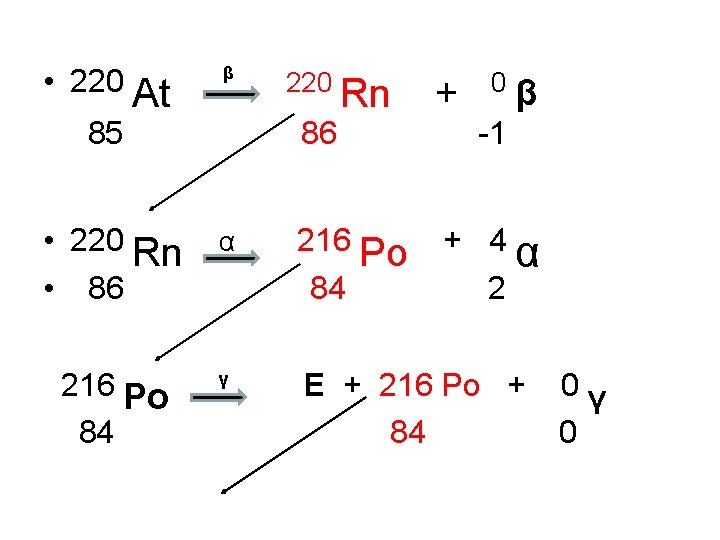
• 220 At β 85 220 Rn 86 • 220 Rn • 86 α 216 Po 84 γ 216 Po 84 + 0β -1 + 4 2 α E + 216 Po + 84 0γ 0
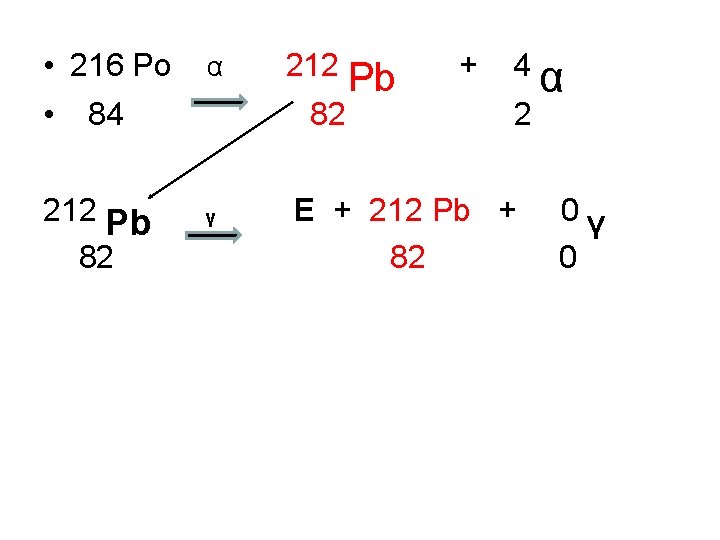
• 216 Po • 84 212 Pb 82 α 212 Pb 82 γ + 4 2 E + 212 Pb + 82 α 0γ 0

How is the NET EQUATION determined from a DECAY SERIES? • A NET EQUATION CONTAINS: • Starting Ending + Total # Isotope Alpha Beta Gamma Particles Releases

Net Equation • 224 Rn • 86 • Starting Isotope net + 34α + 2 0β + 3 0γ 212 Pb 82 Ending Isotope 2 + Total # + Alpha Particles -1 Total # + Beta Particles 0 Total # Gamma Releases

EXIT SLIP Write an alpha decay equation for Lawrencium-262. Write a beta decay equation for Oxygen-13. 262 La α 103 13 8 O 258 101 β 13 9 Md + F + 4 2 α 0β -1
- Slides: 18