Best seen broken into four categories Part 1
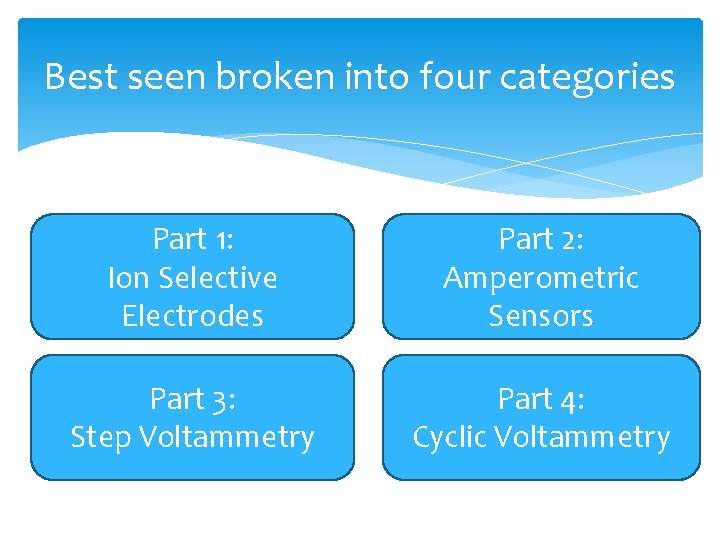
Best seen broken into four categories Part 1: Ion Selective Electrodes Part 2: Amperometric Sensors Part 3: Step Voltammetry Part 4: Cyclic Voltammetry

(3) Step Voltammetry Why Voltammetry at all? Isnt ISE / Amperometrics good enough? According to the Prof – apparently not! Reduction potentials and electron stoichiometry of redox reactions Reversibility of redox reactions undergone by a molecule Diffusion coefficients of species in stable oxidation states
![Step Voltammetry [O] = 5 m. M Can be reduced to R via O Step Voltammetry [O] = 5 m. M Can be reduced to R via O](http://slidetodoc.com/presentation_image_h2/7544ad7c55b8a6f600fd0ee03ac731df/image-3.jpg)
Step Voltammetry [O] = 5 m. M Can be reduced to R via O + e- R E 0 (standard reduction potential) = - 0. 1 V Eelectrode / V 0. 5 0. 1 Solution stirred continuously -0. 3 Time / s
![Step Voltammetry [O] = 5 m. M Can be reduced to R via O Step Voltammetry [O] = 5 m. M Can be reduced to R via O](http://slidetodoc.com/presentation_image_h2/7544ad7c55b8a6f600fd0ee03ac731df/image-4.jpg)
Step Voltammetry [O] = 5 m. M Can be reduced to R via O + e- R I/A E 0 (standard reduction potential) = - 0. 1 V 0 Solution stirred continuously Time / s
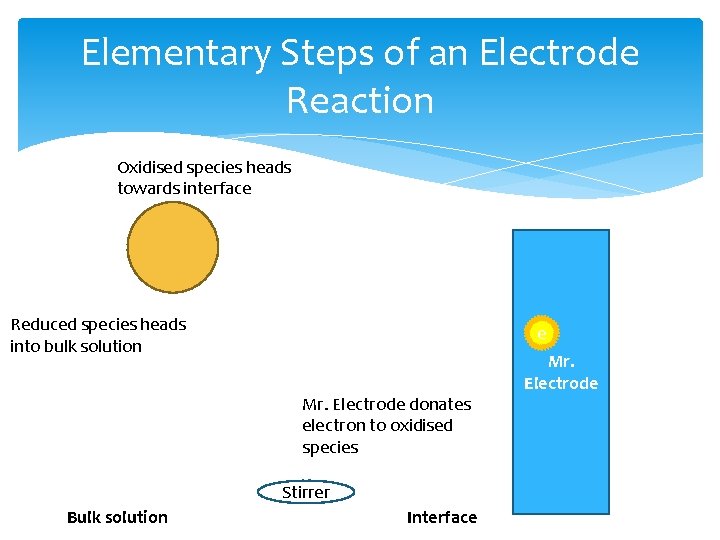
Elementary Steps of an Electrode Reaction Oxidised species heads towards interface Reduced species heads into bulk solution e Mr. Electrode donates electron to oxidised species Stirrer Bulk solution Interface Mr. Electrode
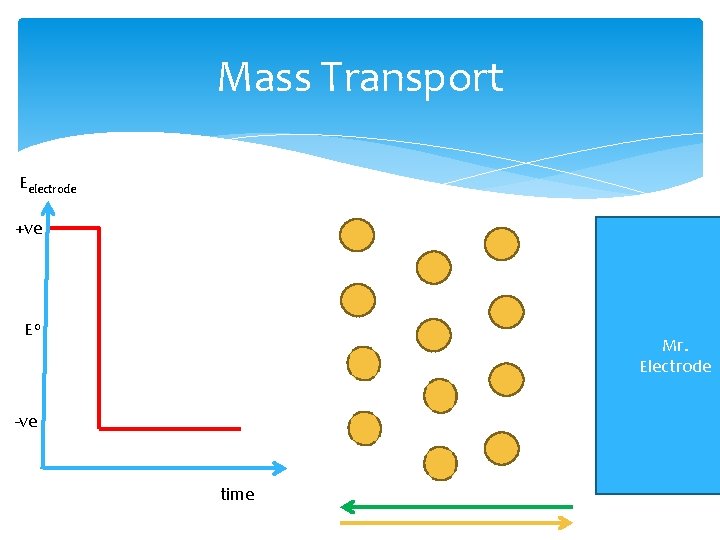
Mass Transport Eelectrode +ve E 0 Mr. Electrode -ve time
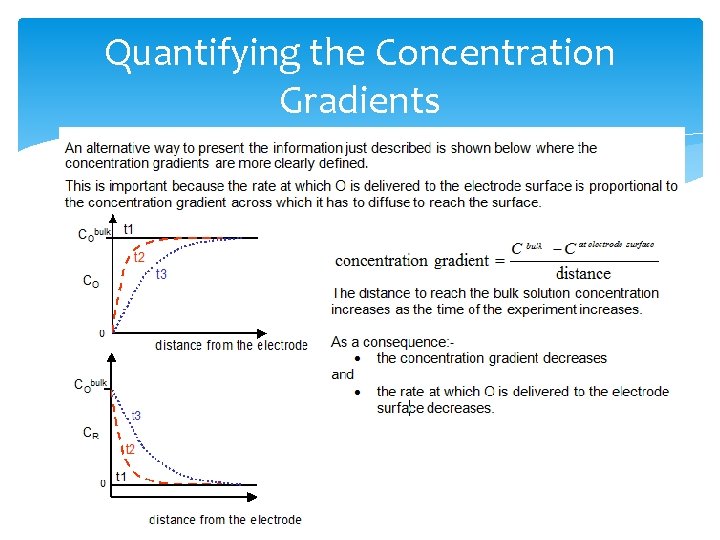
Quantifying the Concentration Gradients

Cottrell Equation
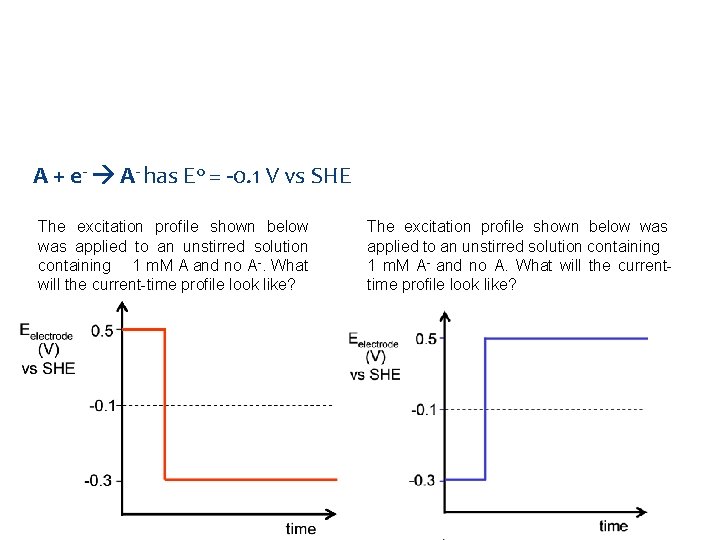
Question A + e- A- has E 0 = -0. 1 V vs SHE The excitation profile shown below was applied to an unstirred solution containing 1 m. M A and no A-. What will the current-time profile look like? The excitation profile shown below was applied to an unstirred solution containing 1 m. M A- and no A. What will the currenttime profile look like?
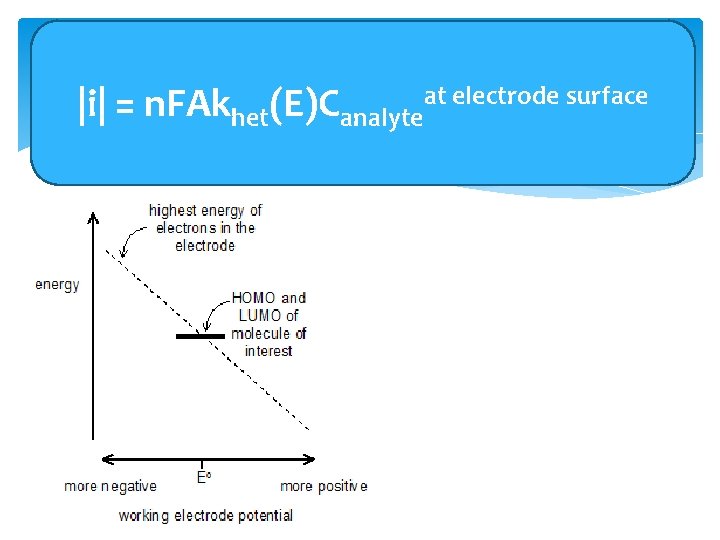
Accounting for Potential Dependence at electrode surface of(E)C theanalyte Current |i| = n. FAkhet
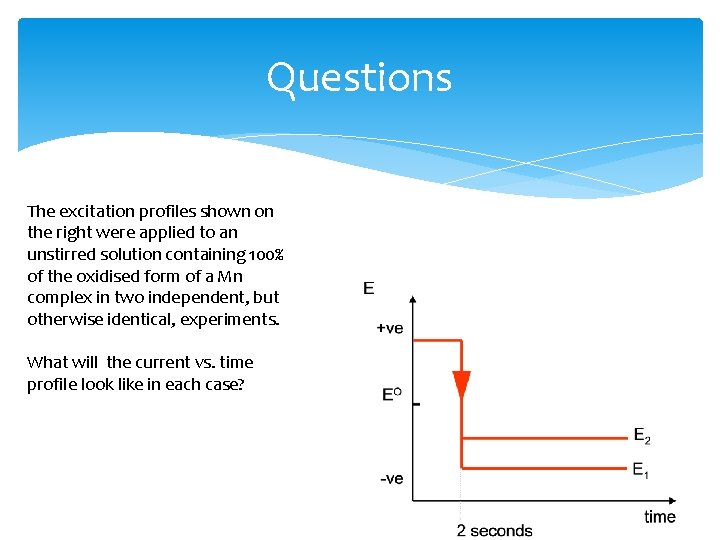
Questions The excitation profiles shown on the right were applied to an unstirred solution containing 100% of the oxidised form of a Mn complex in two independent, but otherwise identical, experiments. What will the current vs. time profile look like in each case?
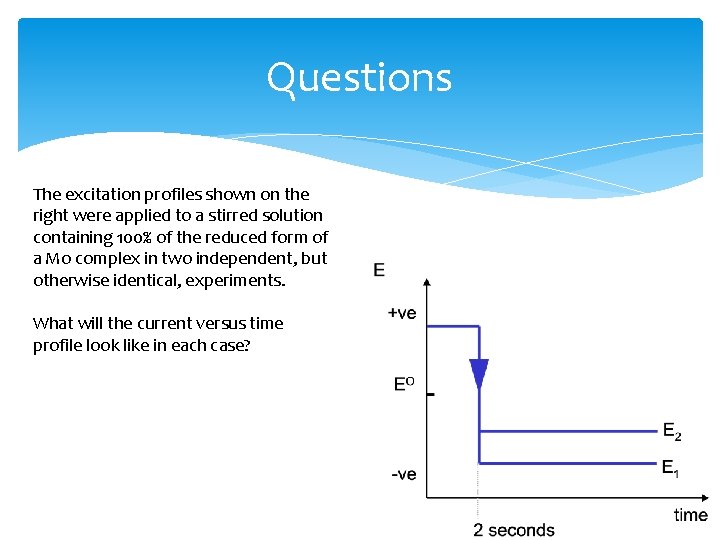
Questions The excitation profiles shown on the right were applied to a stirred solution containing 100% of the reduced form of a Mo complex in two independent, but otherwise identical, experiments. What will the current versus time profile look like in each case?
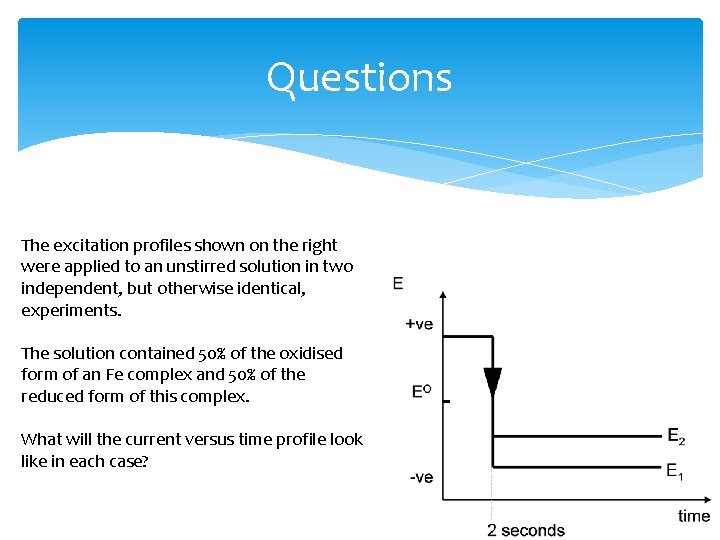
Questions The excitation profiles shown on the right were applied to an unstirred solution in two independent, but otherwise identical, experiments. The solution contained 50% of the oxidised form of an Fe complex and 50% of the reduced form of this complex. What will the current versus time profile look like in each case?
Best seen broken into four categories Part 1: Ion Selective Electrodes Part 2: Amperometric Sensors Part 3: Step Voltammetry Part 4: Cyclic Voltammetry
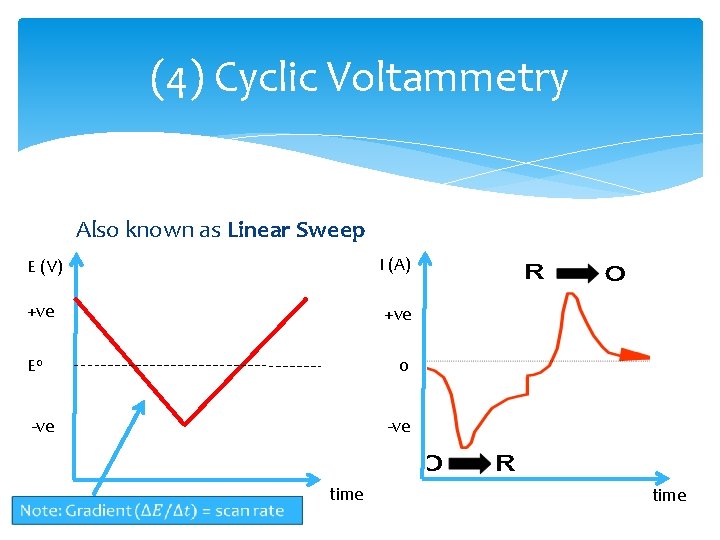
(4) Cyclic Voltammetry Also known as Linear Sweep E (V) I (A) +ve 0 E 0 -ve time
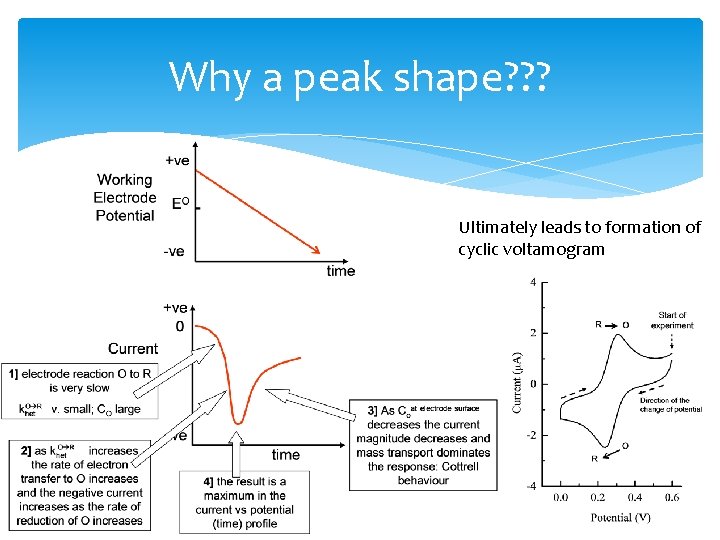
Why a peak shape? ? ? Ultimately leads to formation of cyclic voltamogram
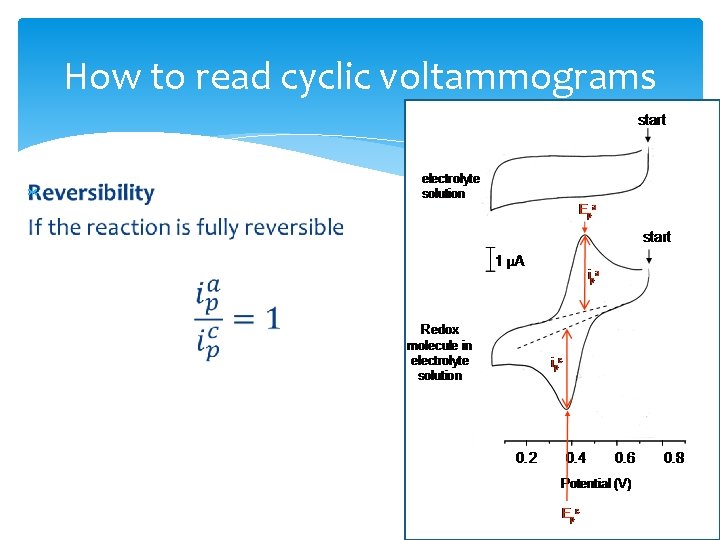
How to read cyclic voltammograms
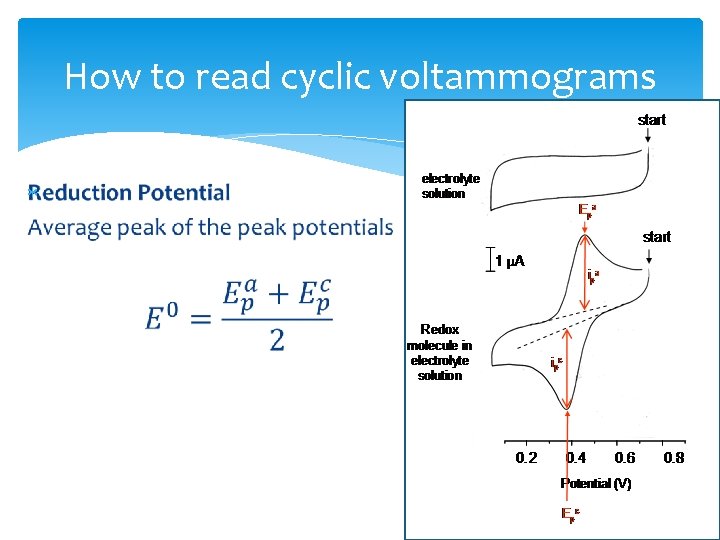
How to read cyclic voltammograms
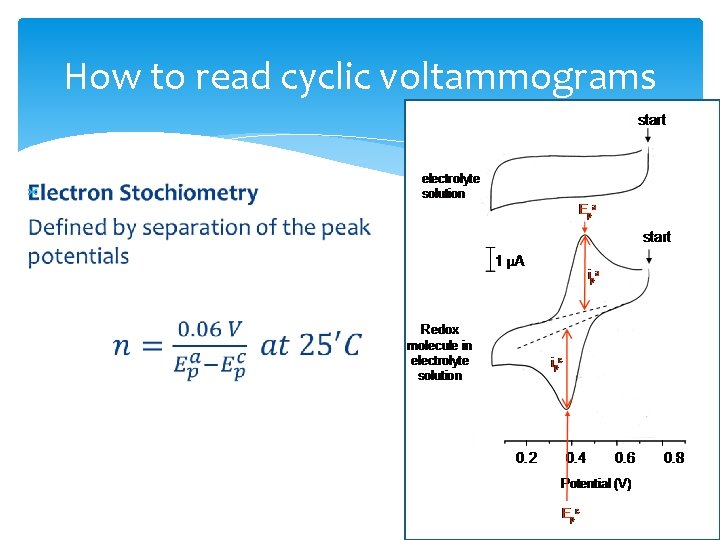
How to read cyclic voltammograms
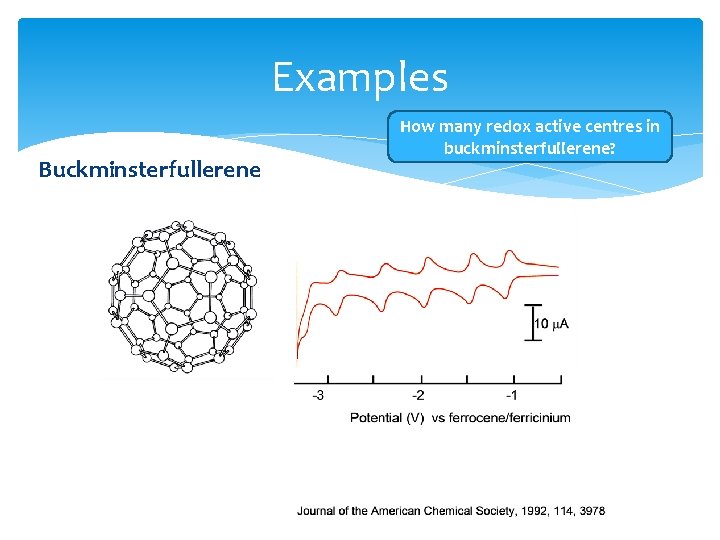
Examples Buckminsterfullerene How many redox active centres in buckminsterfullerene?

More Examples

Effect of Scan Rates
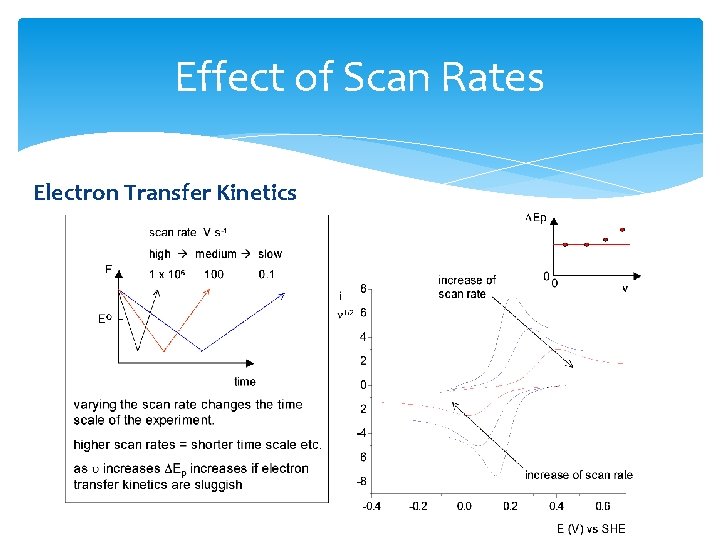
Effect of Scan Rates Electron Transfer Kinetics

Effect of Scan Rates Reaction Mechanism
- Slides: 24