Best Practices to Simplify and Optimize Dementia Care

Best Practices to Simplify and Optimize Dementia Care for Health Care Providers Rev 10 -28 -16

Objectives • Understand the value of timely detection and employ simple approaches to identify cognitive impairment in routine practice • Conduct a dementia work-up and communicate a dementia diagnosis effectively to patients and families • Utilize best practices in medication and nonmedication interventions • Help patients and family access services to improve function and promote wellness 2

Where We Have Been 3

Where We Are Going 4

Alzheimer’s Disease: Challenges and Opportunities

Alzheimer’s: A Public Health Crisis • Scope of the problem – 5. 4 M Americans with AD in 2016 – Growing epidemic expected to impact 13. 8 M Americans by 2050 and consume 1. 1 trillion in healthcare spending – Almost 2/3 are women (longer life expectancy) • Some populations at higher risk – Older African Americans (2 x as whites) – Older Hispanics (1. 5 x as whites) Alzheimer’s Association Facts and Figures 2016 6
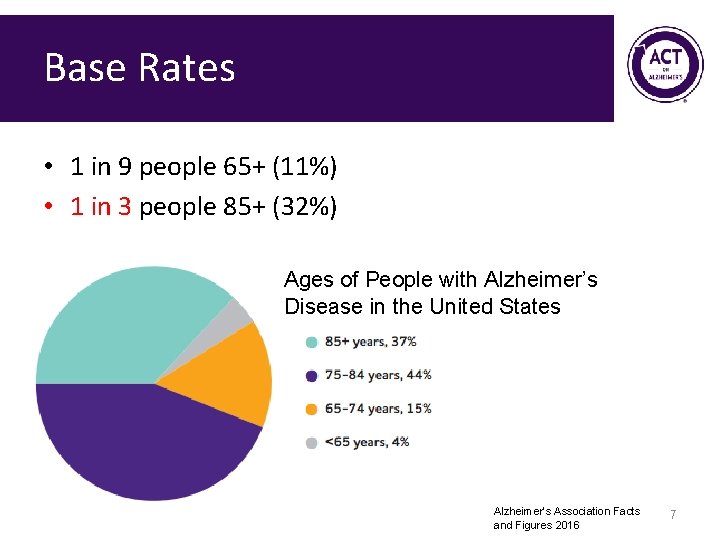
Base Rates • 1 in 9 people 65+ (11%) • 1 in 3 people 85+ (32%) Ages of People with Alzheimer’s Disease in the United States Alzheimer’s Association Facts and Figures 2016 7

Patients with Dementia • A population with complex care needs 2. 5 chronic conditions (average) 5+ medications (average) 3 times more likely to be hospitalized Many admissions from preventable conditions, with higher person costs • Indisputable correlation between chronic conditions and costs Schubert, Boustani, et al. , JAGS, 2006; Alzheimer’s Association Facts & Figures Report 2014. 8

Challenges & Opportunities • AD under-recognized by providers – Fewer than 50% of patients receive formal diagnosis • Millions unaware they have dementia – Diagnosis often delayed on average 2 -5 years after symptom onset – Significant impairment in function by time it is recognized • Poor timing: diagnosis frequently at time of crises, hospitalization, failure to thrive, urgent need for institutionalization Balasa M, et al. 2011; Boise L, et al. 1999; Boise et al. , 2004; Boustani et al. , 2003; Boustani et al. , 2005; Holzer C, Warshaw G. 2000; Silverstein & Maslow, 2006 9

Diagnostic Challenges • Societal – Ageism, lack of understanding re: normal aging – Fear and stigma – Healthcare inequities – Expectation that MD will identify/diagnose health problems • Systemic/Institutional – Low priority – Few incentives – Lack of procedural support – Few dementia specialists available – May lack access to (or awareness of) community resources 10

Diagnostic Challenges • For Providers – Time – Lack of definitive tests – Many patients unaware, do not report symptoms – Limited efficacy of medication treatments – Limited cultural competence – Lack of awareness re: benefits of non-medication interventions – Fear of delivering wrong diagnosis, bad news – Implications for physician/patient relationship 11
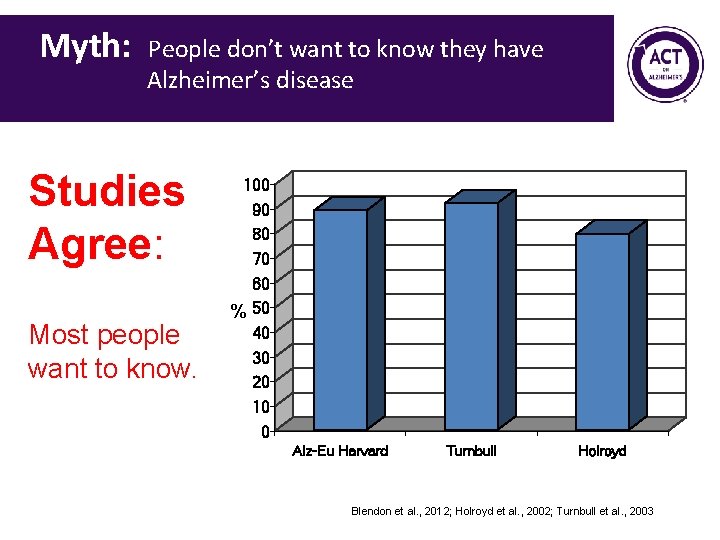
Myth: People don’t want to know they have Alzheimer’s disease Studies Agree: Most people want to know. 100 90 80 70 60 % 50 40 30 20 10 0 Alz-Eu Harvard Turnbull Holroyd Blendon et al. , 2012; Holroyd et al. , 2002; Turnbull et al. , 2003
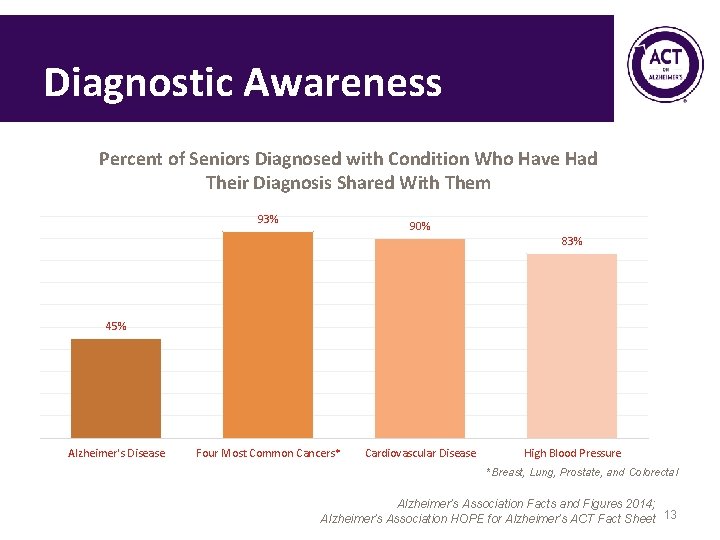
Diagnostic Awareness Percent of Seniors Diagnosed with Condition Who Have Had Their Diagnosis Shared With Them 93% 90% 83% 45% Alzheimer's Disease Four Most Common Cancers* Cardiovascular Disease High Blood Pressure *Breast, Lung, Prostate, and Colorectal Alzheimer’s Association Facts and Figures 2014; Alzheimer’s Association HOPE for Alzheimer’s ACT Fact Sheet 13

Diagnostic Challenges International Physician Survey • Lack of definitive tests (65%, top barrier) • Lack of communication between patients / caregiver and physicians – 75% reported discussion initiated by patients/caregivers – 44% “after they suspected the disease had been present for a while” – 40% said patients/caregivers did not provide enough information to help them make a diagnosis • Patient / Family denial (65%) & social stigma (59%) International Alzheimer’s Disease Physician Survey, 2012

Diagnostic Challenges “Beyond mountains, there are mountains. ” Haitian Proverb

If we don’t diagnose, does it still exist?

Rationale for Timely Detection 1. Patient Care / Outcomes 2. Time 3. Money 17

Patient Outcomes 1. Improve quality of life – – Patients can participate in decisions regarding their future care Decrease burden on family and caregivers 2. Intervene to promote a safe and happy environment that supports independence – – RTC support/counseling intervention Non-pharm intervention reduces NH placement by 30% and delays placement for others by 18+ months The message: You have a bad disease. We can help you make life better for you and your family. Mittelman et al. , 2006 18

Patient Outcomes 3. Improved management of co-morbid conditions – Underlying dementia = risk factor for poor compliance with ALL treatment goals (e. g. , diabetes, hypertension, CHF, anticoagulation) 4. Reduce ineffective, expensive, crisis-driven use of healthcare resources – Unnecessary hospitalization (dehydration/malnourishment, medication mismanagement, accidents and falls, wandering, etc. ) The message: We want to provide you with high quality care that is proactive and cost effective 19

Patient Outcomes 5. Treat reversible causes – NPH, TSH, B 12, hypoglycemia, depression The message: Maybe you don’t really have a bad disease after all! 20

Time • Simple screening tests can be done by rooming nurse – Brain as 6 th vital sign • Recommended tool takes 1. 5 – 3 minutes – • Only conducted annually and in context of signs and symptoms Mini-Cog does not disrupt workflow & increases capture rate of cognitive impairment in primary care Borson et al. , 2007 21

Money • AD most expensive condition in the nation – $203 billion in 2013, $1. 2 trillion in 2050 • Cost effectiveness of early dx/tx? – Large scale studies ongoing • Economic Models – No med known to alter costs of care – Disease education/support interventions increase caregiver capability, save money, and delay NH – Even if assume small # of people benefit (5%), $996 million in potential savings for MN over 15 years Alzheimer’s Association Facts and Figures 2014; Long et al. , 2014 22

Cognitive Impairment Predicts Readmissions Mini-Cog Performance Novel Marker of Post Discharge Risk Among Patients Hospitalized for Heart Failure (Patel, 2015; Cleveland Clinic) • Method: 720 patients screened with Mini. Cog during hospitalization for HF • Results: 23% failed screen (M age 78, 49% men) – Mini. Cog best predictor of readmission over 6 mos. among 55 variables • Stronger than length of stay, cause of HF, and even comorbidity status • Readmission rate 2 times higher among screen fails • Fails discharged to facility (vs. home) had lower readmission rates within first 30 days 23
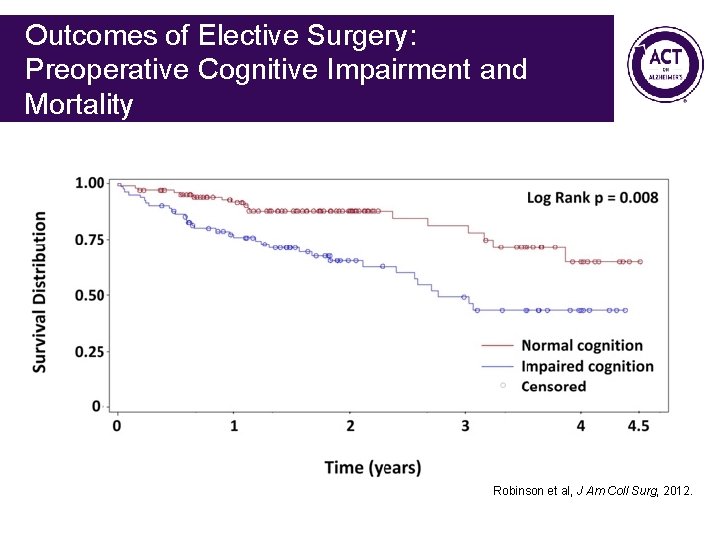
Outcomes of Elective Surgery: Preoperative Cognitive Impairment and Mortality Robinson et al, J Am Coll Surg, 2012.
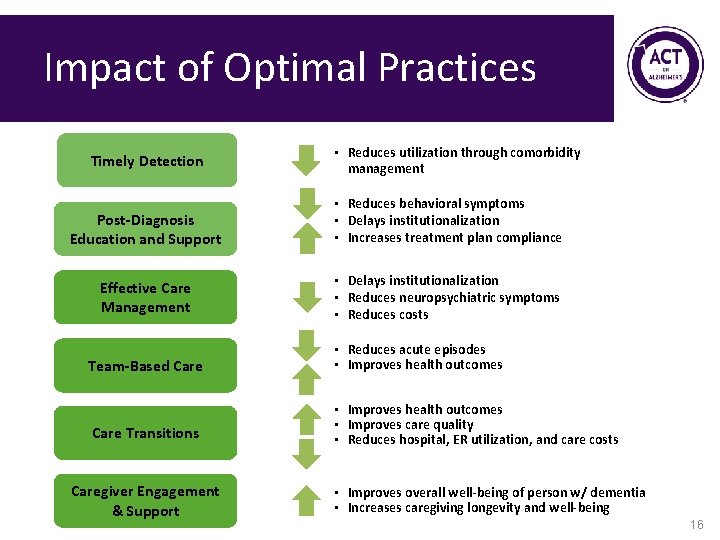
Impact of Optimal Practices Timely Detection • Reduces utilization through comorbidity management Post-Diagnosis Education and Support • Reduces behavioral symptoms • Delays institutionalization • Increases treatment plan compliance Effective Care Management • Delays institutionalization • Reduces neuropsychiatric symptoms • Reduces costs Team-Based Care • Reduces acute episodes • Improves health outcomes Care Transitions • Improves health outcomes • Improves care quality • Reduces hospital, ER utilization, and care costs Caregiver Engagement & Support • Improves overall well-being of person w/ dementia • Increases caregiving longevity and well-being 16

Changing National & Local Landscape • National Alzheimer’s Project Act (NAPA) – Awareness, readiness, dissemination, coordination • Annual Wellness Visit – For first time, “detection of cognitive impairment” is core feature of the exam • MN healthcare systems implementing tools – Health. Partners – Park Nicollet – Essentia – Allina 26

Rethinking Everyday Practice • Brain historically ignored, not a focus of routine exam – Is this logical? Consider base rates of dementia • Dementia is simply “brain failure” – Heart failure – Kidney failure – Liver failure • Brain as 6 th Vital Sign 27

Introduction to ACT on Alzheimer’s

Focus on Quality Health Care www. ACTon. ALZ. org 29

ACT Tool Kit • Evidence and consensusbased, best practice standards for Alzheimer’s care • Tools and resources for: – Primary care providers – Care coordinators – Community agencies – Patients and families 30

ACT Tools 31

ACT Tools 32

www. actonalz. org/provider-resources

Clinical Practice Tips 34

Case Study: Sam • 76 y/o retired teacher (master’s degree) • Daughter c/o short-term memory is poor, patient acknowledges problem but does not feel it is significant – Repeats himself, multiple phone calls b/c can’t find belongings • Other family members have noticed changes • Began 2 years ago, getting worse • Hx of hypertension and DM, both fairly well controlled • Wife died unexpectedly last year, lives alone • Conversational presentation fairly intact • Oriented x 3 but vague awareness of current events 35

Case Study: Colleen 66 y/o retired accountant for family business Presents to primary care with memory complaints Daughter agrees that short-term memory is poor Began 2 years ago, seems to be worsening Hx of Low blood sugar, heart attack x 1, repeat ER visits and hospitalizations for atrial flutter • Frequent medication changes, managing independently • Lives with husband who is still running the family business • • •

Signs and Symptoms of AD • • • • Memory loss Confusion Disorientation to time or place Getting lost in familiar locations Impairment in speech/language Trouble with time/sequence relationships Diminished insight Poor judgment/problem solving Changes in sleep and appetite Mood/personality/behavior changes Wandering Deterioration of self care, hygiene Difficulty performing familiar tasks, functional decline Alzheimer’s Association, 2009 37

Practice Tips • Unfortunately, most of us do not recognize signs and symptoms until they are quite pronounced – Attribution error: “What do you expect? She is 80 years old. ” • Clinical interview – Let patient answer questions without help – Remember: Social skills remain intact until late stage dementia – Easy to be fooled by a sense of humor, reliance on old memories, or quiet/affable demeanor

Practice Tips • Red flags – – – – Repetition (not normal in 7 -10 min conversation) Tangential, circumstantial responses Losing track of conversation Frequently deferring answers to family member Over reliance on old information/memories Inattentive to appearance Unexplained weight loss or “failure to thrive”

Practice Tips • Family observations: – ANY instances whatsoever of getting lost while driving, trouble following a recipe, asking same questions repeatedly, mistakes paying bills – Take these concerns seriously: by the time family report problems, symptoms have typically been present for quite a while and are getting worse • Raise your expectation of older adults: – If this patient was alone on a domestic flight across the country and the trip required a layover with a gate change, would he/she be able to manage that kind of mental task on his/her own? • If answer is “not likely” for a patient of any age: RED FLAG

Practice Tips • Intact older adult should be able to: – Describe at least 2 current events in adequate detail (who, what, when, why, how) – Describe events of national significance • September 11, 2001 – Name or describe the current President and an immediate predecessor – Describe their own recent medical history and report the conditions for which they take medication
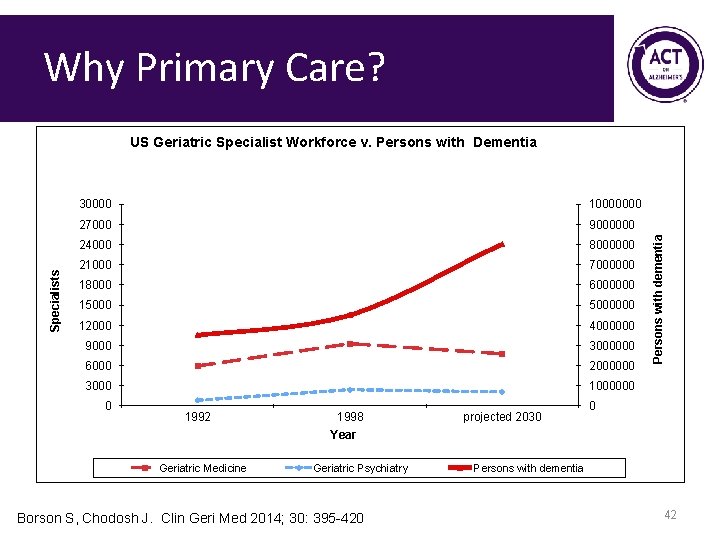
Why Primary Care? 30000 10000000 27000 9000000 24000 8000000 21000 7000000 18000 6000000 15000000 12000 4000000 9000 3000000 6000 2000000 3000 1000000 0 1992 1998 projected 2030 Persons with dementia Specialists US Geriatric Specialist Workforce v. Persons with Dementia 0 Year Geriatric Medicine Geriatric Psychiatry Borson S, Chodosh J. Clin Geri Med 2014; 30: 395 -420 Persons with dementia 42

Detecting Cognitive Impairment 43

Show of Hands • How many routinely use an objective cognitive assessment instrument as part of a memory loss work-up? – What happens after a patient ‘fails’? • How many are doing Medicare Annual Wellness Exams for patients? 44

Workflow • Step 1: Trigger – Annual exam (e. g. , Medicare AWV) – Signs and symptoms – Patient / family report • Step 2: Cognitive Assessment • Step 3: Work-up • Step 4: Referral 45

Is Screening Good Medicine? 2014 US Preventative Services Task Force (USPSTF) • Purpose: Systematically review the diagnostic accuracy of brief cognitive screening instruments and the benefits/harms of medication and non-medication interventions for early cognitive impairment. • Limitation: Limited studies in persons with dementia other than AD and sparse reporting of important health outcomes. • Conclusion: Brief instruments to screen for cognitive impairment can adequately detect dementia, but there is no empirical evidence that screening improves decision making. Long et al. , 2014 46

Show of Hands • How long, on average, is the time lapse between the first pathophysiological changes in the brain and presentation of clinical symptoms? Answer: a) 3 -5 years b) 5 -10 years c) 10 -20 years d) 20 -30 years 47

Alzheimer’s is Insidious Accumulation of neuropathology in the brain 10 -20 years before symptoms appear

Fundamentals of Medicine • Research on risk factors helps us identify population at highest risk of disease • Goal is to identify disease states before damage to organ system is too great • Objective measures are used to monitor patients’ health and detect disease whenever possible – Brain should be no different – Early symptoms of dementia are detectable using simple, objective screening instruments – Subjective impressions FAIL to detect cognitive impairment in early stages 49

Provider Perspective “Avoiding detection of a serious and life changing medical condition just because there is no cure or ‘ideal’ medication therapy seems, at worst, incredibly unethical, and, at best, just bad medicine. ” George Schoephoerster, MD Family Practice Physician 50

Clinical Provider Practice Tool • Easy button workflow for: 1. Identification 2. Dementia work-up 3. Treatment / care www. actonalz. org/provi der-practice-tools 51

Identifying Cognitive Impairment • Initial considerations – Timing • Routine, annual check-ups or only when patients become obviously symptomatic? – Practice recommendation: Annual, objective cognitive assessment at 65+ – Objective cognitive assessment meant to uncover insidious disease – Doesn’t add much if you can already detect impairment in basic conversation – Research • Which tools are best? • Balance b/w time and sensitivity/specificity

Identifying Cognitive Impairment – Clinic flow • Who will administer? – Rooming nurses, social workers, allied health professionals, MDs • What happens when patients fail? 53

Objective Measures • Wide range of options – – – Mini-Cog™ (MC) Mini-Mental State Exam© (MMSE) St. Louis University Mental Status Exam™ (SLUMS) Montreal Cognitive Assessment™ (Mo. CA) Rowland Universal Dementia Assessment (RUDAS) • All but MMSE free, in public domain, and online Borson et al. , 2000; Folstein et al. , 1975; Nasreddine 2005; Tariq et al. , 2006

Administration Best Practices • Try not to: – Use the words “test” or “memory” • Instead: “We’re going to do something next that requires some concentration” – Allow patient to give up prematurely or skip questions – Deviate from standardized instructions – Offer multiple choice answers – Be soft on scoring – Score ranges already padded for normal errors – Deduct points where necessary – be strict
Cognitive Impairment Identification Flow Chart 56

Mini-Cog™ Contents • Verbal Recall (3 points) • Clock Draw (2 points) Advantages • Quick (2 -3 min) • Easy • High yield (executive fx, memory, visuospatial) Borson et al. , 2000 Subject asked to recall 3 words Leader, Season, Table +3 Subject asked to draw clock, set hands to 10 past 11 +2
www. actonalz. org/sites/default/ files/documents/Mini-Cog_. pdf 58

Mini-Cog Pass • > 4 Fail • 3 or less NOTE: A cut point of <3 on the Mini-Cog has been validated for dementia screening, but many individuals with clinically meaningful cognitive impairment will score higher. When greater sensitivity is desired, a cut point of <4 is recommended as it may indicate a need for further evaluation of cognitive status. Borson et al. , 2000; Borson, Scanlan, Chen et al. , 2003; Borson, Scanlan, Watanabe et al. , 2006; Lessig, Scanlan et al. , 2008; Mc. Carten, Anderson et al. , 2011; Mc. Carten, Anderson et al. , 2012; Tsoi, Chan et al. , 2015
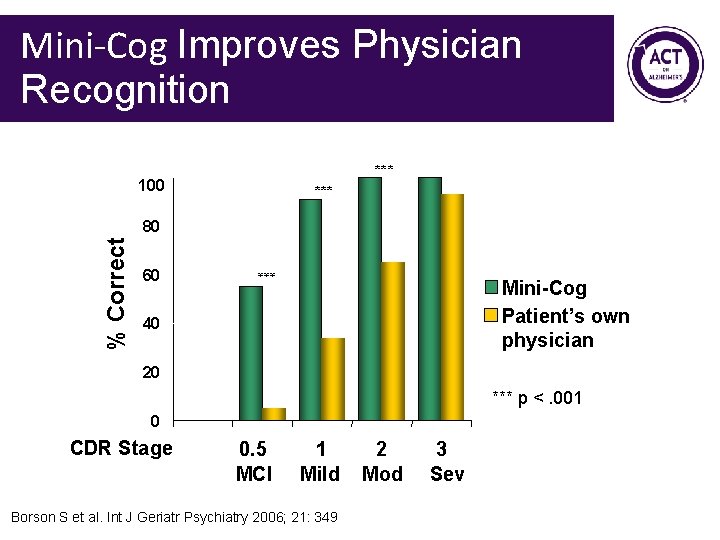
Mini-Cog Improves Physician Recognition *** 100 *** 60 *** % Correct 80 Mini-Cog Patient’s own physician 40 20 *** p <. 001 0 CDR Stage 0. 5 MCI 1 Mild Borson S et al. Int J Geriatr Psychiatry 2006; 21: 349 2 Mod 3 Sev

Mini-Cog Research • Performance unaffected by education or language • Borson Int J Geriatr Psychiatry 2000 • Sensitivity and specificity similar to MMSE (76% vs. 79%; 89% vs. 88%) • Borson JAGS 2003 • Does not disrupt workflow & increases rate of diagnosis in primary care • Borson JGIM 2007 • Failure associated with inability to fill pillbox • Anderson et al The Consultant Pharmacist 2008

Mini-Cog: Sam www. actonalz. org/video-tutorials 62

Mini-Cog Scoring: Sam

Mini-Cog Scoring: Sam

Mini-Cog: Colleen www. actonalz. org/video-tutorials 65

Colleen’s Clock

Colleen’s Score

Mini-Cog Exercise Form groups of 2 • Administer Mini. Cog to each other • Score sample clocks 68
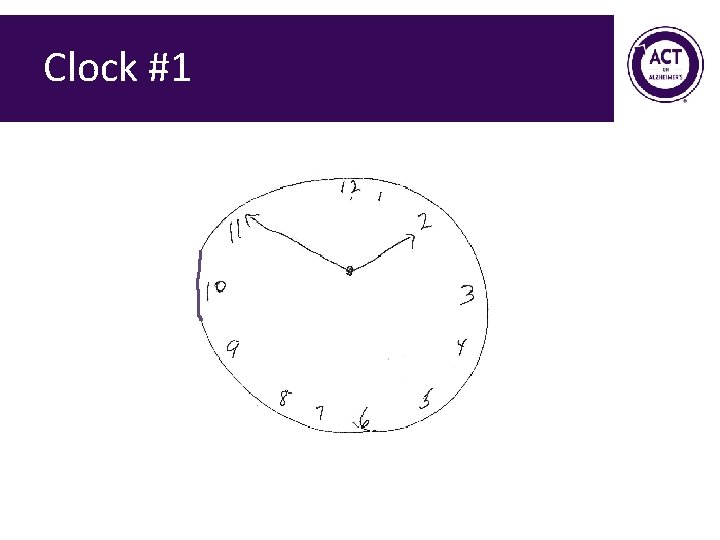
Clock #1
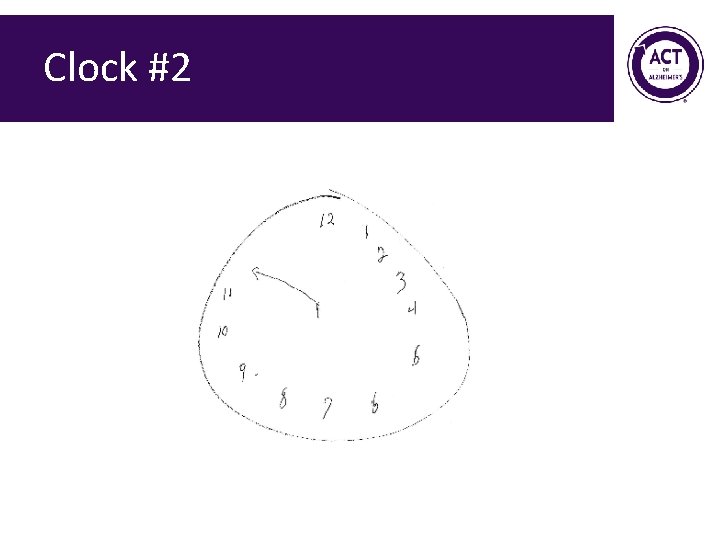
Clock #2
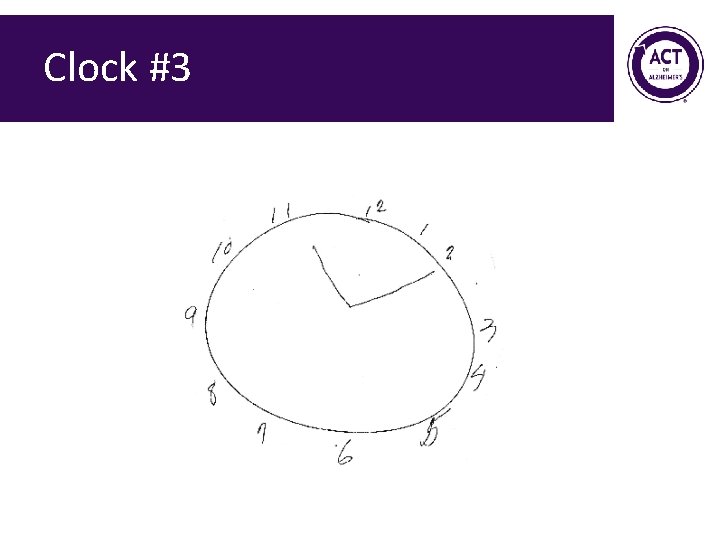
Clock #3
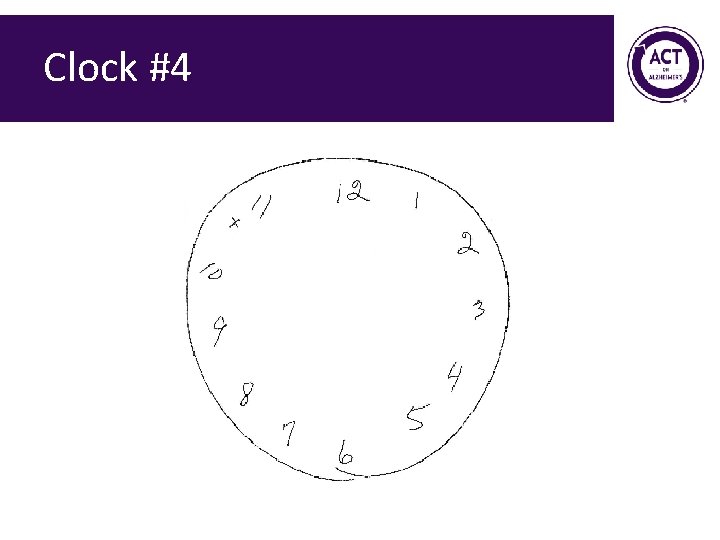
Clock #4
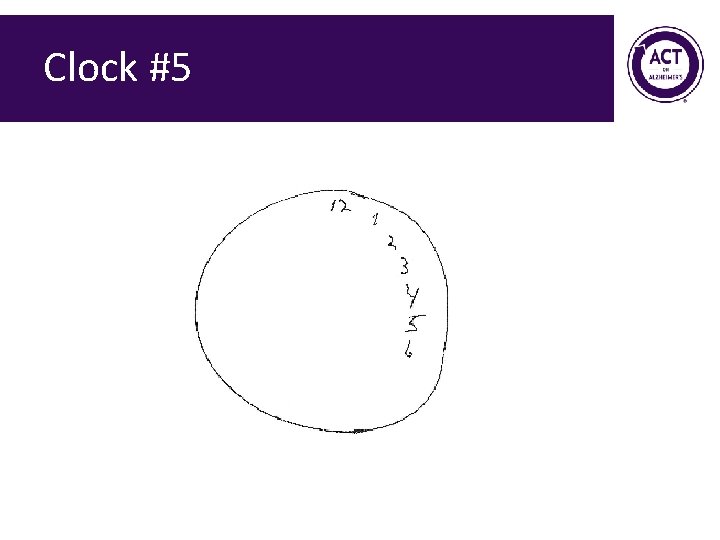
Clock #5
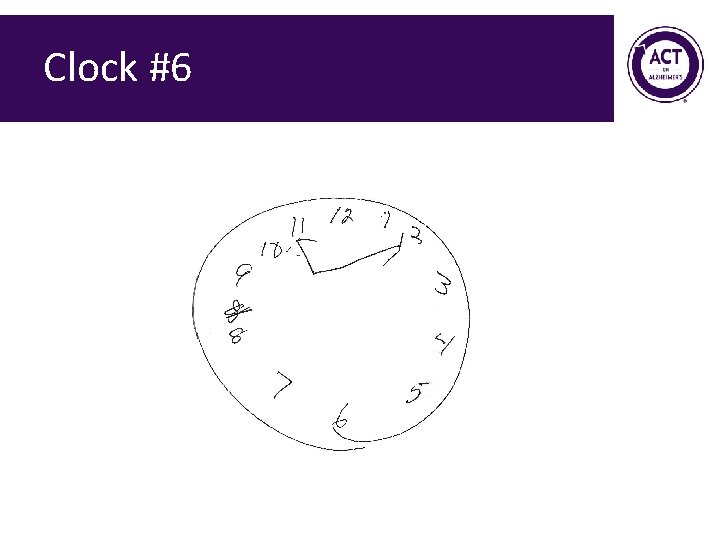
Clock #6

Clock #7

Clock #8

SLUMS Tariq et al. , 2006

SLUMS High School Diploma Less than 12 yrs education Pass > 27 > 25 Fail 26 or less 24 or less Tariq SH, Tumosa N, Chibnall et al. Comparison of the Saint Louis University mental status examination and the mini-mental state examination for detecting dementia and mild neurocognitive disorder--a pilot study. Am J Geriatr Psychiatry. 2006 Nov; 14(11): 900 -10. 78

SLUMS: Colleen www. actonalz. org/video-tutorials 79

SLUMS Scoring: Colleen • Interactive scoring exercise 80

SLUMS Scoring: Colleen 81

SLUMS Scoring: Colleen 82

SLUMS Scoring: Colleen 83
Mo. CA Nasreddine et al. , 2005

Mo. CA Pass • > 26 Fail • 25 or less Nasreddine 2005 85

Mo. CA: Sam www. actonalz. org/video-tutorials 86

Mo. CA Scoring: Sam • Interactive scoring exercise 87
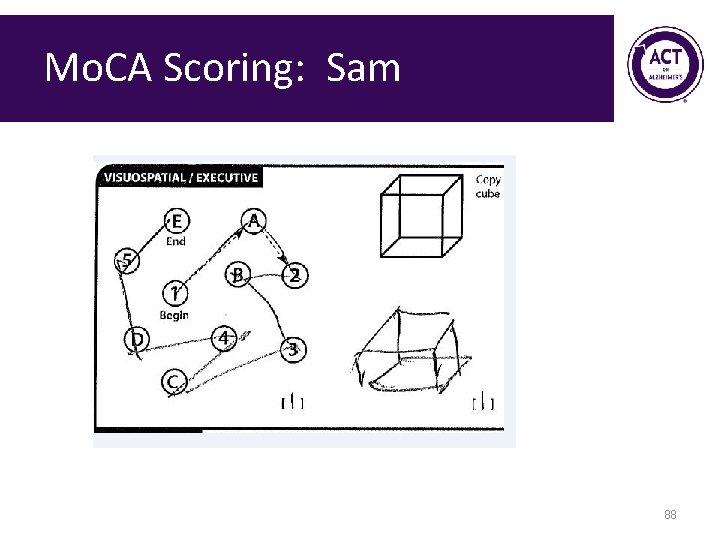
Mo. CA Scoring: Sam 88

Mo. CA Scoring: Sam 89
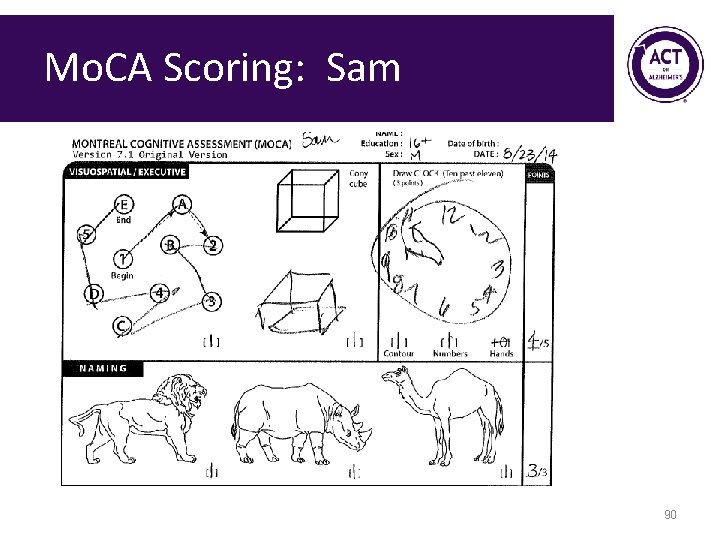
Mo. CA Scoring: Sam 90
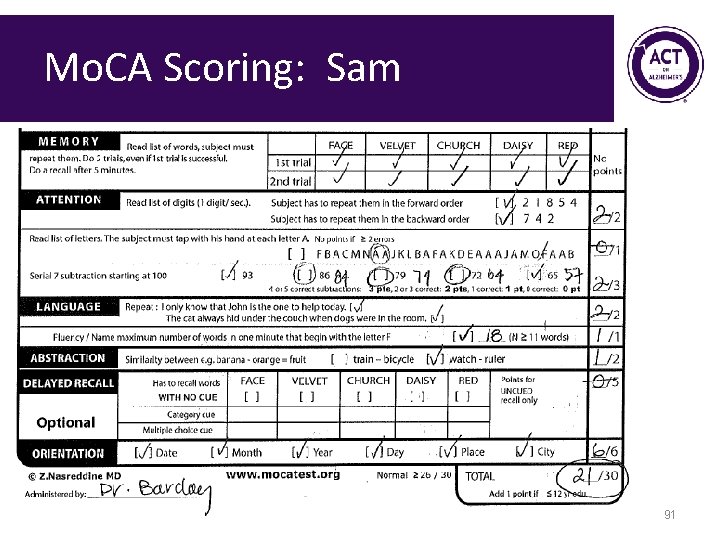
Mo. CA Scoring: Sam 91

Objective Measure Selection Montreal Cognitive Assessment (Mo. CA) • Sensitivity: • Specificity: 90% for MCI, 100% for dementia 87% St. Louis University Mental Status (SLUMS) • Sensitivity: • Specificity: 92% for MCI, 100% for dementia 81% Mini-Mental Status Exam (MMSE) • Sensitivity: • Specificity: 18% for MCI, 78% for dementia 100% Larner 2012; Nasreddine et all, 2005; Tariq et al. , 2006; Ismail et al. , 2010

Alternative Tools • Virtually all tools based upon a euro-centric cultural and educational model • Consider: country and language of origin, type/quality/length of education, disabilities (visual, auditory, motor) • Alternative tools my be less biased • More information – Screening Diverse Populations on the ACT website 93

RUDAS • Developed intentionally for patients with: • low levels of education • limited language fluency • diverse cultural histories • Measures wide variety of cognitive abilities • Score range 0 -30 • Track change over time • Strong psychometric properties • 10 -15 minutes to administer 94

95

96

Family Questionnaire www. actonalz. org/pdf/Family. Questionnaire. pdf
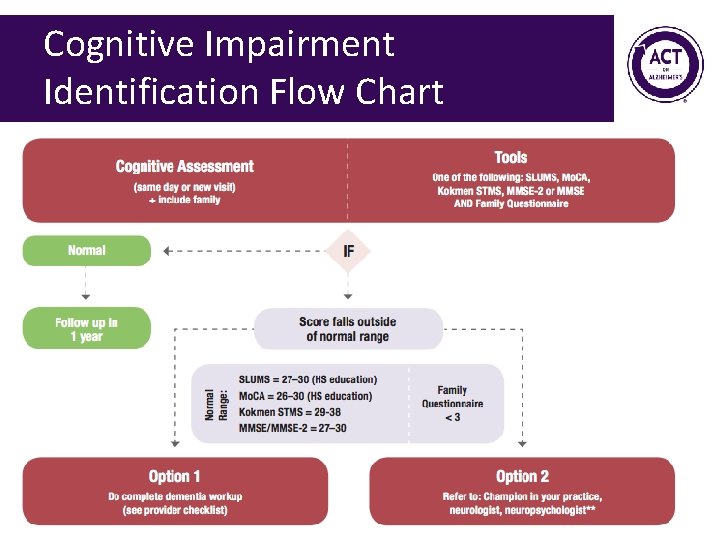
Cognitive Impairment Identification Flow Chart 98

Communicating Cognitive Assessment Results 99

Scripting: Framing the Purpose • Regardless of a passing or failing score, explain the patient’s test result by first reminding them of the assessment purpose “The purpose of this task was to check on the health of the brain and determine if there is any need for further evaluation of your thinking or memory. ” 100

Scripting: Passing Score • “You obtained a normal score on this measure, which is good news. No additional action is needed. ” • “However, if you have concerns about your thinking or memory, talk to your provider. ” 101

Scripting: Failing Score • Nurse/Allied health professional: – “Your doctor will review the results with you today during your visit. ” 102

Scripting: Failing Score • Physician/Provider: – “Your score on the measure was a little low today. Have you been having any trouble with your memory lately? ” – “I would like you to schedule a follow-up appointment with me (or neurology, the memory clinic, psychiatry, etc. ) on your way out so we can take a closer look at your memory. This is an important part of your care and an important part of staying healthy. ” – Enter order follow-up/referral 103

Scripting: Failing Score • “Today we did a brief exam to check on the health of the brain. Your score on this measure was a little low. This is like getting blood work back that is outside the normal range. We should follow-up on this. I am going to send you to a specialist so we can determine what this means and what we should do about it. ” 104

Dementia Work-up and Diagnosis 105

106

107

Dementia Work-Up • H&P • Objective cognitive measurement • Diagnostics – Labs – Imaging ? – More specific testing (e. g. , neuropsychometric)? • Diagnosis • Family meeting

Dementia Diagnoses FTD Alzheimer’s disease: 60 -80 % • Includes mixed AD + VD Lewy Body Dementia Vascular Dementia Lewy Body Dementia: 10 -25 % – Parkinson spectrum Alzheimer’ s Disease Vascular Dementia: 6 -10 % – Stroke related Frontotemporal Dementia: 2 -5 % – Personality or language disturbance

Show of Hands • How many of you deliberately use the words Alzheimer’s disease with patients when making a diagnosis vs. terms such as “memory loss” or “dementia”? 110

Delivering the Diagnosis • General guidelines: – Include a family member in the visit if at all possible – Talk directly to the person with dementia – Speak at a slower, relaxed pace using plain words • Try not to fill the time with words – less is more – Explain why tests were ordered and what results mean – Provide a specific diagnosis – Ask more than once whether the patient / family has any questions – Acknowledge how overwhelming the information feels; provide empathy, support, reassurance 111

Delivering the Diagnosis • Focus on wellness, healthy living, and optimizing function – Sleep – Exercise – Social and mental stimulation – Nutrition and hydration – Stress reduction – Increase structure at home Zaleta AK and Carpenter BD. Patient-Centered Communication During the Disclosure of a Dementia Diagnosis. AM J ALZHEIMERS DIS OTHER DEMEN 2010, 25: 513 112

Delivering the Diagnosis • Connect patient/family to community resources – Care for both patient and caregiver – Examples: Senior linkage line, Alzheimer’s Association • Discuss follow-up – Want to see patient and family member at regular intervals (e. g. , q 6 months) for proactive care – Discuss involvement of care coordinator • Provide written summary of visit 113

Delivering the Diagnosis • Address immediate problems: – Management of medications, finances, meals – Driving – Home safety – Caregiver burnout – Social isolation – Inactivity/lack of exercise • Encourage family involvement/assignments – Family need to accompany patient to doctor appts. 114

Common Questions • • • How is Alzheimer’s different from dementia? Is there any treatment? What can we do? Does [latest news report] work? How fast is this going to progress? How often do we see you? What’s next? 115

Follow-up Messages • Recommend future actions – Create a ‘Plan B’ • What if primary caregiver is suddenly unavailable? – Develop medication management plan, consider medication review (Pharm. D Consult) – Patient/Family Disease Education / Planning – Fall Risk Assessment – Investigate home care, AL, LTC, other living options – Develop hospitalization plan – Complete Advance Directives – Consider future medical care—how aggressive? Zaleta & Carpenter 2010 116

Delivering the Diagnosis: Sam http: //www. actonalz. org/video-tutorials 117

Delivering the Diagnosis: Sam • Discussion – Observations? Reactions? – What was done well? – What could have been done differently, better? – What elements would you incorporate into your practice? – If Sam was American Indian what, if anything, would you do differently? 118

Dementia Care and Treatment 119
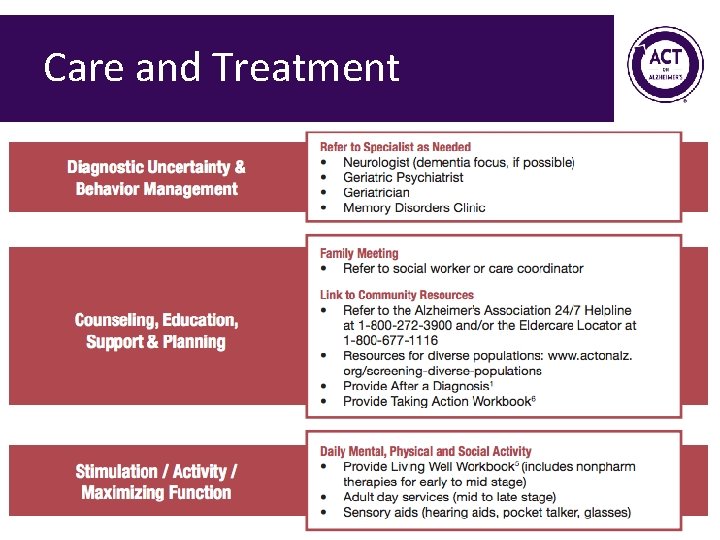
Care and Treatment 120
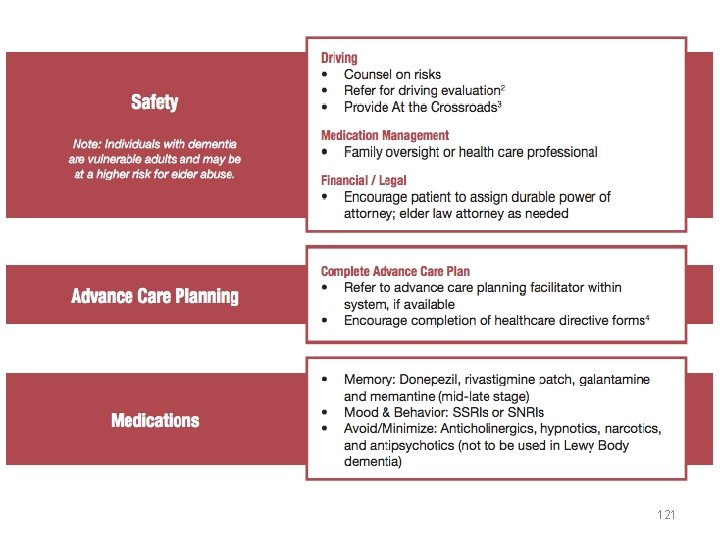
Care and Treatment 121

Treatment: Medications • Cholinesterase inhibitors – Donepezil, Rivastigmine, Galantamine, Cognex – Possible side effects: nausea, vomiting, syncope, dizziness, anorexia • NMDA receptor antagonist – Memantine – Possible side effects: tiredness, body aches, dizziness, constipation, headache 122

Care and Treatment • The care for patients with Alzheimer’s has very little to do with pharmacology and more to do with psychosocial interventions • Involve care coordinator • Connect patient and family to experts in the community – Example: Alzheimer’s Association – Refer every time, at any stage of disease, and for every kind of dementia – Stress this is part of their treatment plan and you expect to hear about their progress at next visit 123

After A Diagnosis - Partnering with doctors - Understanding the disease - Planning ahead - How to ask for help - Using community resources - Role of care coordinator

ACT EMR Tools • Use EMR to automate and standardize: – Screening – Work-up – After visit summary with dementia education – Orders and referrals – Community supports www. actonalz. org/provider-practice-tools 125

Screening 126

Labs and Orders 127
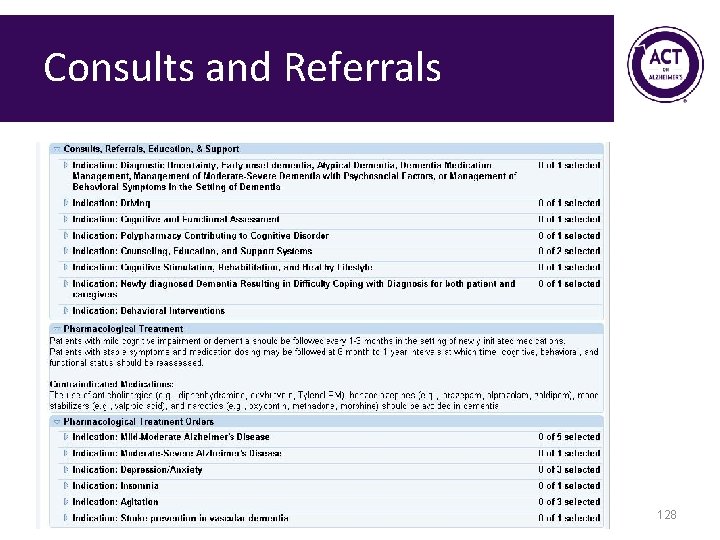
Consults and Referrals 128

Consults and Referrals 129

Pharmacological Treatment 130

Managing Mid to Late Stage Dementia 131

Managing Dementia Across the Continuum www. actonalz. org/provider-practice-tools 132

Mood and Behavioral Symptoms • Neuropsychiatric symptoms common: – 60% of community dwelling patients with dementia – > 80% of nursing home residents with dementia • Nearly all patients with dementia will experience mood or behavioral symptoms during the course of their illness Ferri et al. , 2005; Jeste et al. , 2008 133

Adverse Outcomes • • • Decreased quality of life Increased hospital length of stay Increased system-wide costs Increased caregiver distress, depression, burnout Independently associated with NH placement ? Increased mortality Jeste et al. , 2008; Finkel et al. , 1996 134

135

ACT to the Rescue! 136

Systematic Approach to Management • • Step 1: Step 2: Step 3: Step 4: Define behavior Categorize target symptom Identify reversible causes Use non-drug interventions first to treat target symptoms 137

Step 1: Define Behavior • Examples – Attention seeking behaviors • Verbal outbursts – Aggression during cares – Hitting, pushing, kicking – Sexual disinhibition – Restless motor activity, pacing, rocking – Calling out 138

Step 2: Categorize Target Symptom • Psychosis – Delusions – Hallucinations • Mood symptoms – Anxiety – Dysphoria – Irritability – Lability • Aggression • Spontaneous disinhibition 139

Step 3: Identify Reversible Causes • • Delirium Untreated medical illness (e. g. , UTI) Medication side effects, polypharmacy Environmental triggers Undiagnosed psychiatric illness Inexperienced caregivers Unrealistic expectations 140

Step 3: Identify Reversible Causes • Common root causes: – Anxiety, fear or uncertainty – Touch or invasion of personal space – Loss of control, lack of choice – Lack of attention to personal needs or wishes – Frustration, grief due to loss of function or ability – Pain or fear of pain

Step 3: Identify Reversible Causes • Unmet needs – Boredom – Meaning, purpose – Over/under stimulation – Safety – Environmental stressors • Caregiver reactions – Limited knowledge about disease process or behaviors 142

Step 4: Non-pharmacologic Interventions • Think like a behavioral analyst – Detective work, ask: • • • Who (is involved/present) What (exact description, be specific) When (time dependent? only in morning? triggers? ) Where (location specific? ) Why (what happens right before, right afterwards? what do family think is cause? ) – ABC approach (antecedent, behavior, consequence) 143

Step 4: Non-pharmacologic Interventions • Teach family caregivers to: – Validate Join Distract – Understand that behavior = communication – Ask themselves: • Is this really a problem, and for whom? • What is the feeling or underlying message this behavior is trying to communicate? • How can I address the underlying need? • How long will this solution last? 144

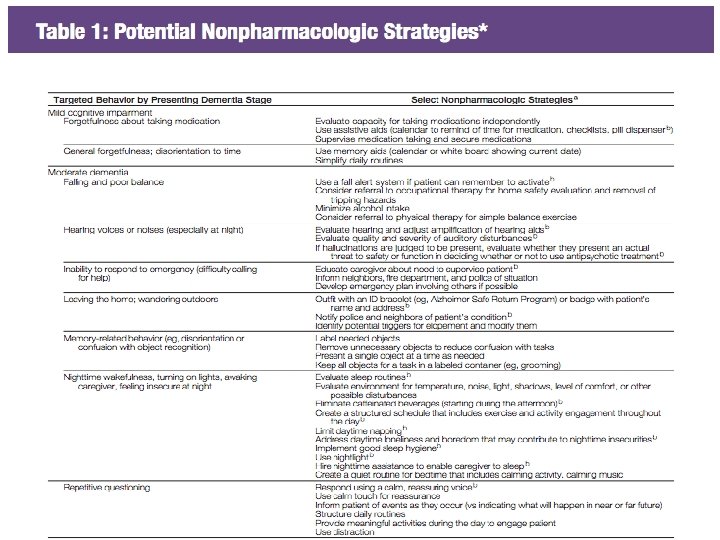
Step 4: Non-pharmacologic Interventions • Activity planning – Tap into preserved capabilities and previous interests – Involve repetitive motion • Communication – Slow down, offer simple choices – Help individual find words for self expression • Simplify Environment – Remove clutter, minimize stimuli during activity – Establish routines • Caregiver support – Self care, minimize confrontation/arguing with loved one – Identify support network Gitlin, et al. , 2012 145
146

Pharmacological Treatment • • Antipsychotics Antidepressants Mood stabilizers Cognitive enhancers 147

Antipsychotic Medications in Dementia • 1952: First generation antipsychotic: haloperidol – Extrapyramidal symptoms – Tardive dyskinesia • 1989: Second generation antipsychotic: clozapine – Agranulocytosis • 1990’s: More second generation antipsychotics – Risperidone, olanzapine, quetiapine, ziprasidone, aripiprazole – Less motor side effects, better tolerated – Utilization of these agents broadens • THEN in 2005 … Jeste et al. , 2008 148

2005 FDA Box Warning Elderly patients with dementiarelated psychosis treated with atypical antipsychotic drugs are at an increased risk of death compared to placebo. 149

Bottom Line with Atypical Antipsychotics • Modest efficacy in the treatment of psychotic and neuropsychiatric symptoms • Increased risk of negative outcomes: DEATH, STROKE, HIP FRACTURE, FALLS • Share the decision with healthcare proxies • Monitor: – Falls, orthostatic BP, EPS, tardive dyskinesia, glucose – Regularly attempt to wean/discontinue 150

Optimizing Medication Therapy Professional Resources • AGS Beers Criteria (2012) • START (Screening Tool to Alert Doctors to the Right Treatment) • STOPP (Screening Tool of Older Persons’ Potentially inappropriate Prescriptions) 151

Advanced Care Planning • Discussion of goals of care, values • Identification AND engagement of HCPOA – Honoring Choices – PREPARE • Introduce concept of palliative care, educate about hospice • Document in EMR, healthcare directive • Provider Orders for Life Sustaining Treatment (POLST) 152

Assessing Caregiver/Family Needs • Be alert for signs of: – Burnout, depression, neglected self-care, elder abuse • Promote: – Respite services – Support groups – Activities to optimize health and well-being • Refer to one-stop-shop for support: – Alzheimer’s Association – Senior Linkage Line 153

Patient Engagement: Research Participation • Alzheimer’s Association Trial Match – Free, easy-to-use clinical studies matching service that connects individuals with Alzheimer's, caregivers, healthy volunteers and physicians with current studies. – http: //www. alz. org/research/clinical_trials/find _clinical_trials_trialmatch. asp • National Institute of Health (NIH) – http: //clinicaltrials. gov 154

HIPAA: Q & A • HIPAA (Health Insurance Portability and Accountability Act) • Federal law that protects medical information • Allows only certain people to see information – Doctors, nurses, therapists and other health care professionals on the patient’s medical team – Family caregivers and others directly involved with a patient’s care (unless the patient says he/she does not want this information shared with others) www. nextstepsincare. org, United Hospital Fund, 2002 155

HIPAA: Sharing Patient Information • If the patient is present and has the capacity to make health care decisions: – Health care providers may discuss the patient’s health information with a family member, friend, or other person if the patient agrees or, when given the opportunity, does not object. • If patient is not present or is incapacitated: – Health care providers may share the patient’s information with family, friends or others as long as the provider determines (based on professional judgment) that it is in the best interest of the patient. www. nextstepsincare. org, United Hospital Fund, 2002 156

Top Resources for Patients and Families 157

#1 Promoting Wellness & Function http: //www. alz. org/documents/mndak/alz_living_well_workbook_2011 v 2_web. pdf 158

#2 Addressing Behavioral Challenges 159

#3 Caregiver Support Alzheimer’s Association 24/7 Helpline 800. 272. 3900 | www. alz. org One stop shop for: – Care Consultation (social work intervention) – Support Groups (Memory Club) – 24/7 Helpline 160

#4 Medication Review Pharm. D Consult • Medication review, simplification • Reminder strategies • Family support, supervision 161

ACKNOWLEDGEMENTS This presentation was created by ACT on Alzheimer’s, an award-winning, nationally recognized, volunteer-driven collaborative seeking to create supportive environments for everyone touched by Alzheimer’s disease and to prepare Minnesota for its impacts. ACT on Alzheimer’s® Executive Co-Leads: Olivia Mastry, JD, MPH, Michelle Barclay, MA & Emily Farah-Miller, MS Lead Presentation Authors: Terry Barclay, Ph. D and Michelle Barclay, MA Special thanks to the ACT on Alzheimer’s Detection and Quality Health Care Leadership Group and Medical Speaker’s Bureau Members Visit www. actonalz. org/provider-practice-tools for more information and to access supportive tools and resources.

ACKNOWLEDGEMENTS This project is/was supported by funds from the Bureau of Health Professions (BHPr), Health Resources and Services Administration (HRSA), Department of Health and Human Services (DHHS) under Grant Number UB 4 HP 19196 to the Minnesota Area Geriatric Education Center (MAGEC) for $2, 192 (7/1/2010— 6/30/2015). This information or content and conclusions are those of the author and should not be construed as the official position or policy of, nor should any endorsements be inferred by the BHPr, HRSA, DHHS or the U. S. Government. Minnesota Area Geriatric Education Center (MAGEC) Grant #UB 4 HP 19196 Director: Robert L. Kane, MD Associate Director: Patricia A. Schommer, MA

References & Resources • • • • Alzheimer’s Association (2014). Alzheimer’s Disease Facts and Figures, Alzheimer’s & Dementia, Volume 10, Issue 2. Anderson K, Jue S & Madaras-Kelly K (2008). Identifying Patients at Risk for Medication Mismanagement: Using Cognitive Screens to Predict a Patient's Accuracy in Filling a Pillbox. The Consultant Pharmacist, 6(14), 459 -72. Balasa M, et al. Neurology. 2011; 76: 1720 -1725. Barry PJ, Gallagher P, Ryan C, & O‘mahony D. (2007). START (screening tool to alert doctors to the right treatment)--an evidence-based screening tool to detect prescribing omissions in elderly patients. Age and Ageing, 36(6): 632 -8. Blendon RJ, Benson JM, Wikler, EM, Weldon, KJ, Georges, J, Baumgart, M, Kallmyer B. (2012). The impact of experience with a family member with Alzheimer’s disease on views about the disease across five countries. International Journal of Alzheimer’s Disease, 1 -9. Boise L, et al. Am J Alzheimer's Dis. 1999: 20 -26. Boise L, Neal MB, & Kaye J (2004). Dementia assessment in primary care: Results from a study in three managed care systems. Journals of Gerontology: Series A; Vol 59(6), M 621 -26. Borson S, Scanlan J, Brush M, Vitaliano P, Dokmak A. (2000). The mini-cog: a cognitive “vital signs” measure for dementia screening in multi-lingual elderly. Int J Geriatr Psychiatry, 15(11): 1021 -1027. Borson S, Scanlan JM, Chen P, Ganguli M. (2003). The Mini-Cog as a screen for dementia: validation in a population-based sample. J Am Geriatr Soc; 51(10): 1451 -1454. Borson S, Scanlan J, Hummel J, Gibbs K, Lessig M, & Zuhr E (2007). Implementing Routine Cognitive Screening of Older Adults in Primary Care: Process and Impact on Physician Behavior. J Gen Intern Med; 22(6): 811– 817. Borson, S. , Scanlan, JM, Watanabe, J et al. (2006). Improving identification of cognitive impairment in primary care. Int J Geriatr Psychiatry; 21: 349 -355. Boustani M, Peterson B, Hanson L, et al. (2003). Systematic evidence review. Agency for Healthcare Research and Quality; Rockville, MD: Screening for dementia. Boustani M, Callahan CM, Unverzagt FW, Austrom MG, Perkins AJ, Fultz BA, Hui SL, Hendrie HC (2005). Implementing a screening and diagnosis program for dementia in primary care. J Gen Intern Med. Jul; 20(7): 572 -7. 164

References & Resources • • • • Ferri CP, Prince M, Brayne C, et al. (2005). Alzheimer’s Disease International Global prevalence of dementia: A Delphi consensus study. Lancet, 366: 2112– 2117. Finkel, SI (Ed. ) (1996). Behavioral and Psychological Signs of Dementia: Implications for Research and Treatment. International Psychogeriatrics, 8(3). Folstein MF, Folstein SE, & Mc. Hugh PR (1975). "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res, Nov 12(3): 189 -98. Gallagher P & O’Mahony D (2008). STOPP (Screening Tool of Older Persons’ potentially inappropriate Prescriptions): Application to acutely ill elderly patients and comparison with Beers’ criteria. Age and Ageing, 37(6): 673 -9. Gitlin LN, Kales HC, Lyketsos CG, & Plank Althouse E (2012). Managing Behavioral Symptoms in Dementia Using Nonpharmacologic Approaches: An Overview. JAMA, 308(19): 2020 -29. Holroyd S, Turnbull Q, & Wolf AM (2002). What are patients and their families told about the diagnosis of dementia? Results of a family survey. Int J Geriatr Psychiatry, Mar; 17(3): 218 -21. Holzer C, Warshaw G. Arch Fam Med. 2000; 9: 1066 -1070. Ismail Z, Rajji TK, & Shulman KI (2010). Brief cognitive screening instruments: An update. Int J Geriatr Psychiatry, 25: 111– 20. Jeste DV, Blazer D, Casey D et al. (2008). ACNP White Paper: Update on Use of Antipsychotic Drugs in Elderly Persons with Dementia. Neuropsychopharmacology, 33(5): 957 -70. Larner AJ (2012). Screening utility of the Montreal Cognitive Assessment (Mo. CA): In place of – or as well as – the MMSE? Intern Psychogeriatrics, 24, 391– 396. Lessig, M, Scanlan, J et al. (2008). Time that tells: Critical clock-drawing errors for dementia screening. Int Psychogeriatr; 20(3): 459 -470. Lin JS, O’Connor E, Rossom RC, Perdue LA, Burda BU, Thompson M, & Eckstrom E (2014). Screening for Cognitive Impairment in Older Adults: An Evidence Update for the U. S. Preventive Services Task Force. Agency for Healthcare Research and Quality, Evidence Syntheses, 107. Long KH, Moriarty JP, Mittelman MS, & Foldes SS (2014). Estimating The Potential Cost Savings From The New York 165 University Caregiver Intervention In Minnesota. Health Affairs, 33(4), 596 -604.

References & Resources • • • • Mc. Carten JR, Anderson, P et al. (2011). Screening for cognitive impairment in an elderly veteran population: Acceptability and results using different versions of the Mini. Cog. J Am Geriatr Soc; 59: 213 -309. Mc. Carten JR, Anderson P Kuskowski MA et al. (2012). Finding dementia in primary care: The results of a clinical demonstration project. J Am Geritr Soc; 60(2): 210 -217. Mittelman MS, Haley WE, Clay OJ, & Roth DL (2006). Improving caregiver well-being delays nursing home placement of patients with Alzheimer disease. Neurology, November 14(67 no. 9), 1592 -1599. Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, & Chertkow H. (2005). The Montreal Cognitive Assessment, Mo. CA: A Brief Screening Tool For Mild Cognitive Impairment. J Amer Ger Soc, 53(4), 695 -99. National Chronic Care Consortium and the Alzheimer’s Association. 1998. Family Questionnaire. Revised 2003. Patel, A. , Parikh, R. et al. (2015). Mini-cog performance: A novel marker of post discharge risk among patients hospitalized for heart failure. Heart Failure; 8(1): 8 -16. Robinson, T. , Wu, D. , Pointer, L. , Dunn, C. & Moss, M. (2012). Preoperative cognitive dysfunction is related to adverse postoperative outcomes in the elderly. Journal American College of Surgeons, 215: 12 -18. Scanlan, J & Borson, S (2001). The Mini-Cog: Receiver operating characteristics with the expert and naïve raters. Int J Geriatr Psychiatry; 16: 216 -212. Schubert, C. C. , Boustani, M. , Callahan, C. M. , Perkins, A. J. , Carney, C. P. , Fox, C. , Unverzaget, F. & Hendrie, H. C. (2006). Comorbidity profile of dementia patients in primary care: are they sicker? Journal of the American Geriatric Society, 54: 104 -109. Silverstein NM & Maslow K (Eds. ) (2006). Improving Hospital Care for Persons with Dementia. New York: Springer Publishing CO. Tariq SH, Tumosa N, Chibnall JT, Perry MH, & Morley E. (2006). Comparison of the Saint Louis University mental status examination and the mini-mental state examination for detecting dementia and mild neurocognitive disorder: A pilot study. Am J Geriatr Psychiatry, Nov; 14(11): 900 -10. Tsoi, K, Chan, J et al. (2015). Cognitive tests to detect dementia: A systematic review and meta-analysis. JAMA Intern Med; E 1 -E 9. Turnbull Q, Wolf AM, & Holroyd S (2003). Attitudes of elderly subjects toward “truth telling” for the diagnosis of Alzheimer’s disease. J Geriatr Psychiatry Neurol, Jun; 16(2): 90 -3. Zaleta AK & Carpenter BD (2010). Patient-Centered Communication During the Disclosure of a Dementia Diagnosis. Am J Alzheimers Dis Other Demen, 25, 513. 166

References & Resources • • 2012 Updated AGS Beers Criteria: http: //www. americangeriatrics. org/files/documents/beers/2012 Beers. Criteria_JAGS. pdf After a Diagnosis (ACT): http: //www. actonalz. org/sites/default/files/documents/ACT-After. Diagnosis. pdf Alzheimer’s Association • Basics of Alzheimer’s Disease: https: //www. alz. org/national/documents/brochure_basicsofalz_low. pdf • Caregiver Notebook - http: //www. alz. org/care/alzheimers-dementia-caregiver-notebook. asp • Driving Center: www. alz. org/care/alzheimers-dementia-and-driving. asp • Facts & Figures video: http: //youtu. be/waeuks 1 -3 Z 4 • Facts & Figures Report: http: //www. alz. org/documents_custom/2016 -facts-and-figures. pdf • Family Questionnaire: http: //www. alz. org/mnnd/documents/Family_Questionnaire. pdf • Know the 10 Signs. http: //www. alz. org/national/documents/checklist_10 signs. pdf • Living with Alzheimer’s – Mid Stage: https: //www. alz. org/documents_custom/middle-stage-caregiver-tips. pdf • Living with Alzheimer’s – Late Stage: https: //www. alz. org/documents_custom/late-stage-caregiver-tips. pdf • Living Well workbook: http: //www. alz. org/documents/mndak/alz_living_well_workbook_2011 v 2_web. pdf • Taking Action Workbook: http: //www. alz. org/mnnd/documents/2010_taking_action_e-book(1). pdf • Trial Match: http: //www. alz. org/research/clinical_trials/find_clinical_trials_trialmatch. asp • HOPE for Alzheimer’s ACT Fact Sheet: http: //act. alz. org/site/Doc. Server/hope_for_alz_fact_sheet. pdf? doc. ID=3021 167

References & Resources • • • • AD 8 Dementia Screening Interview: http: //knightadrc. wustl. edu/About_Us/PDFs/AD 8 form 2005. pdf At the Crossroads: http: //www. thehartford. com/sites/thehartford/files/at-the-crossroads-2012. pdf Caring for a Person with Alzheimer’s Disease: http: //www. nia. nih. gov/sites/default/files/caring_for_a_person_with_alzheimers_disease_0. pdf Coach Broyles Playbook on Alzheimer’s: http: //www. caregiversunited. com Coping with Behavior Change in Dementia: A Caregier’s Guide: http: //www. amazon. com/Coping-Behavior. Change-Dementia-Caregivers/dp/0692385444 Honoring Choices Minnesota: http: //www. honoringchoices. org Hospitalization Happens: http: //www. nia. nih. gov/sites/default/files/hospitalization_happens_0. pdf Medicare Annual Wellness Visit: http: //www. cms. gov/Outreach-and-Education/Medicare-Learning-Network. MLN/MLNMatters. Articles/downloads/MM 7079. pdf Mini. Cog™ http: //www. alz. org/documents_custom/minicog. pdf MN Health Care Home Care Coordination Tool Kit: http: //www. health. state. mn. us/healthreform/homes/collaborative/lcdocs/cliniccarecoordtoolkit. pdf Montreal Cognitive Assessment (Mo. CA)http: //www. mocatest. org National Alzheimer’s Project Act: http: //aspe. hhs. gov/daltcp/napa/Natl. Plan. pdf Next Step in Care: http: //www. nextstepincare. org Physician Orders for Life Sustaining Treatment (POLST): http: //www. polst. org 168

References & Resources • • • St. Louis University Mental Status (SLUMS) examination http: //medschool. slu. edu/agingsuccessfully/pdfsurveys/slumsexam_05. pdf The Alzheimer’s Action Plan: http: //www. amazon. com/The-Alzheimers-Action-Plan-Know/dp/0312538715 Zarit Caregiver Burden Interview: http: //www. uconnaging. uchc. edu/patientcare/memory/pdfs/zarit_burden_interview. pdf 169
- Slides: 169