Best of HCV From AASLD 2013 Paul Kwo

Best of HCV From AASLD 2013 Paul Kwo, MD Indianapolis, Indiana, USA This activity has been supported by an independent medical education grant from Bristol Myers Squibb. 2013 IC-HEP Educational supporters include Bristol Myers Squibb and Janssen Therapeutics EMEA. Supporters do not influence IC-HEP faculty selection or educational content. 1

Abstract #LB-1 Phase 2 b study of the interferon-free and ribavirin-free combination of daclatasvir, asunaprevir, and BMS-791325 for 12 weeks in treatment-naïve patients with chronic HCV genotype 1 infection Gregory T. Everson 1, Karen D. Sims 2, Paul J. Thuluvath 3, Eric Lawitz 4, Tarek Hassanein 5, Maribel Rodriguez-Torres 6, Trevor Hawkins 7, Howard Schwartz 8, Vinod K. Rustgi 9, Federico Hinestrosa 10, James M. Levin 11, Zobair M. Younossi 12, Lynn R. Webster 13, Timothy Eley 2, Shu-Pang Huang 14, Fiona Mc. Phee 15, Dennis M. Grasela 2, David F. Gardiner 2 1. University of Colorado Denver, Aurora, CO, United States. 2. Bristol-Myers Squibb, Hopewell, NJ, United States. 3. Mercy Medical Center, Baltimore, MD, United States. 4. The Texas Liver Institute, University of Texas Health Science Center, San Antonio, TX, United States. 5. Southern California Liver Centers, Coronado, CA, United States. 6. Fundación de Investigación, San Juan, Puerto Rico, United States. 7. Southwest CARE Center, Santa Fe, NM, United States. 8. Miami Research Associates, South Miami, FL, United States. 9. Metropolitan Research, Arlington, VA, United States. 10. Orlando Immunology Center, Orlando, FL, United States. 11. Dean Foundation for Health, Research and Education, Inc, Madison, WI, United States. 12. Inova Fairfax Hospital, Center for Liver Diseases, Falls Church, VA, United States. 13. CRI Lifetree, Salt Lake City, UT, United States. 14. Bristol-Myers Squibb, Princeton, NJ, United States. 15. Bristol-Myers Squibb, Wallingford, CT, United States. 2

Direct-Acting Antiviral Agents • Daclatasvir (DCV) – NS 5 A replication complex inhibitor with potent, pan-genotypic activity in vitro – Studied in over 5500 patients • Asunaprevir (ASV) – NS 3 protease inhibitor active against genotypes (GT) 1, 4, 5, and 6 in vitro – Studied in over 2000 patients • BMS-791325 – Non-nucleoside, NS 5 B polymerase inhibitor active against GT 1, 3, 4, 5, and 6 in vitro – Studied in over 500 patients Everson GT, et al. Abstract #LB-1, AASLD 2013 3
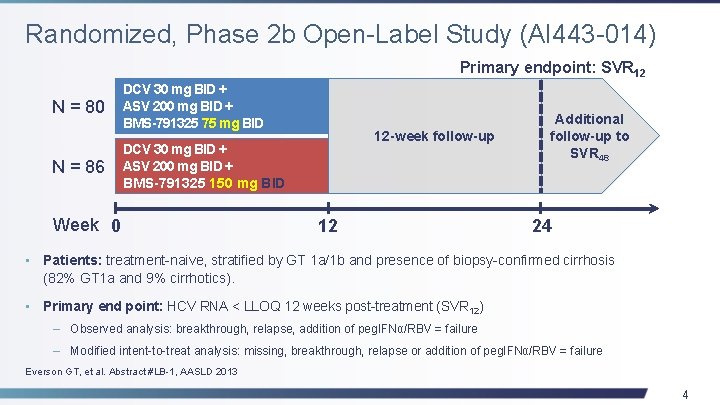
Randomized, Phase 2 b Open-Label Study (AI 443 -014) Primary endpoint: SVR 12 N = 80 DCV 30 mg BID + ASV 200 mg BID + BMS-791325 75 mg BID N = 86 DCV 30 mg BID + ASV 200 mg BID + BMS-791325 150 mg BID Week 0 12 -week follow-up 12 Additional follow-up to SVR 48 24 • Patients: treatment-naive, stratified by GT 1 a/1 b and presence of biopsy-confirmed cirrhosis (82% GT 1 a and 9% cirrhotics). • Primary end point: HCV RNA < LLOQ 12 weeks post-treatment (SVR 12) – Observed analysis: breakthrough, relapse, addition of peg. IFNα/RBV = failure – Modified intent-to-treat analysis: missing, breakthrough, relapse or addition of peg. IFNα/RBV = failure Everson GT, et al. Abstract #LB-1, AASLD 2013 4
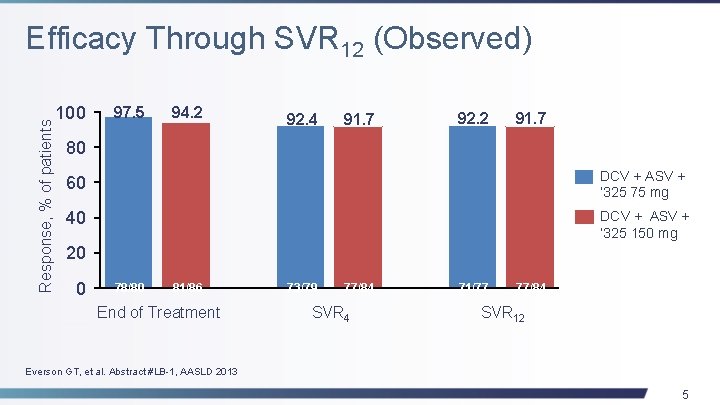
Response, % of patients Efficacy Through SVR 12 (Observed) 100 97. 5 94. 2 92. 4 91. 7 92. 2 91. 7 80 60 DCV + ASV + ‘ 325 75 mg 40 DCV + ASV + ‘ 325 150 mg 20 0 78/80 81/86 End of Treatment 73/79 77/84 SVR 4 71/77 77/84 SVR 12 Everson GT, et al. Abstract #LB-1, AASLD 2013 5
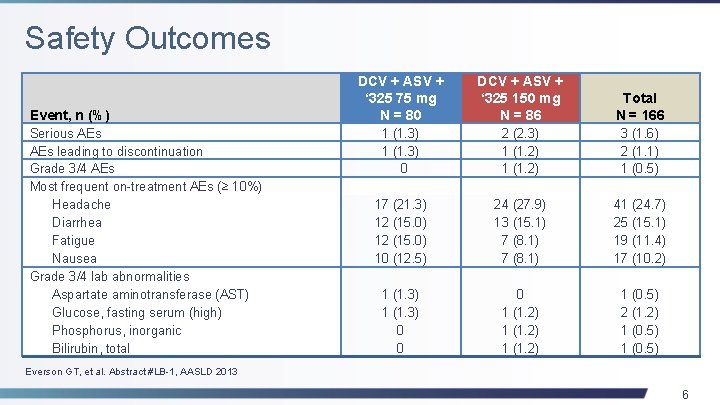
Safety Outcomes Event, n (%) Serious AEs leading to discontinuation Grade 3/4 AEs Most frequent on-treatment AEs (≥ 10%) Headache Diarrhea Fatigue Nausea Grade 3/4 lab abnormalities Aspartate aminotransferase (AST) Glucose, fasting serum (high) Phosphorus, inorganic Bilirubin, total DCV + ASV + ‘ 325 75 mg N = 80 DCV + ASV + ‘ 325 150 mg N = 86 Total N = 166 1 (1. 3) 0 2 (2. 3) 1 (1. 2) 3 (1. 6) 2 (1. 1) 1 (0. 5) 17 (21. 3) 12 (15. 0) 10 (12. 5) 24 (27. 9) 13 (15. 1) 7 (8. 1) 41 (24. 7) 25 (15. 1) 19 (11. 4) 17 (10. 2) 1 (1. 3) 0 0 0 1 (1. 2) 1 (0. 5) 2 (1. 2) 1 (0. 5) Everson GT, et al. Abstract #LB-1, AASLD 2013 6

Abstract #211 All-oral Combination of Daclatasvir Plus Asunaprevir in Interferon Ineligible Naive/Intolerant and Nonresponder Japanese Patients Chronically Infected with HCV Genotype 1 b: Results from a Phase 3 Trial Kazuaki Chayama 1, Yoshiyuki Suzuki 2, Kenji Ikeda 2, Joji Toyota 3, Yoshiyasu Karino 3, Yoshiiku Kawakami 1, Akio Ido 4, Kazuhide Yamamoto 5, Koichi Takaguchi 6, Namiki Izumi 7, Kazuhiko Koike 8, Tetsuo Takehara 9, Norifumi Kawada 10, Michio Sata 11, Hidetaka Miyagoshi 12, Timothy Eley 13, Fiona Mc. Phee 13, Wenhua Hu 13, Hiroki Ishikawa 12, Eric A. Hughes 13, Hiromitsu Kumada 2 1. Hiroshima University, Hiroshima, Japan. 8. University of Tokyo, Japan. 2. Toranomon Hospital, Tokyo, Japan. 9. Osaka University, Osaka, Japan. 3. Sapporo-Kousei General Hospital, Sapporo, Japan. 10. Osaka City University, Osaka, Japan. 4. Kagoshima University, Kagoshima, Japan. 11. Kurume University, Fukuoka, Japan. 5. Okayama University, Okayama, Japan. 12. Bristol-Myers KK, Tokyo, Japan. 6. Kagawa Prefectural Hospital, Kagawa, Japan. 13. Bristol-Myers Squibb, Princeton, NJ, United States. 7. Musashino Red Cross Hospital, Tokyo, Japan. 7
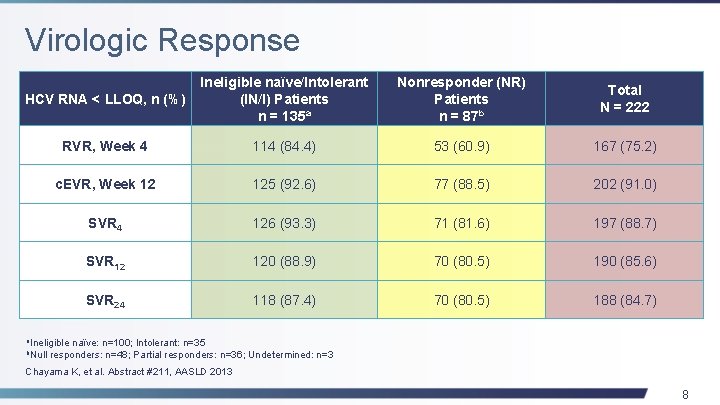
Virologic Response HCV RNA < LLOQ, n (%) Ineligible naïve/Intolerant (IN/I) Patients n = 135 a Nonresponder (NR) Patients n = 87 b Total N = 222 RVR, Week 4 114 (84. 4) 53 (60. 9) 167 (75. 2) c. EVR, Week 12 125 (92. 6) 77 (88. 5) 202 (91. 0) SVR 4 126 (93. 3) 71 (81. 6) 197 (88. 7) SVR 12 120 (88. 9) 70 (80. 5) 190 (85. 6) SVR 24 118 (87. 4) 70 (80. 5) 188 (84. 7) a. Ineligible b. Null naïve: n=100; Intolerant: n=35 responders: n=48; Partial responders: n=36; Undetermined: n=3 Chayama K, et al. Abstract #211, AASLD 2013 8

Abstract #LB-3 SVR results of a once-daily regimen of simeprevir (TMC 435) plus sofosbuvir (GS-7977) with or without ribavirin in cirrhotic and non-cirrhotic HCV genotype 1 treatment-naïve and prior null responder patients: The COSMOS study Ira M. Jacobson 1, Reem H. Ghalib 2, Maribel Rodriguez-Torres 3, Zobair M. Younossi 4, Ana Corregidor 5, Mark S. Sulkowski 6, Edwin De. Jesus 7, Brian Pearlman 8, Mordechai Rabinovitz 9, Norman Gitlin 10, Joseph K. Lim 11, Paul J. Pockros 12, Bart Fevery 13, Tom Lambrecht 14, Sivi Ouwerkerk-Mahadevan 13, Katleen Callewaert 13, William T. Symonds 15, Gaston Picchio 16, Karen Lindsay 16, Maria Beumont-Mauviel 13, Eric Lawitz 17 1. Weill Cornell Medical College, New York, NY, United States. 2. Medicine and Gastroenterology and Hepatology, The Liver Institute, Dallas, TX, United States. 3. Fundación de Investigación, San Juan, Puerto Rico, United States. 4. Department of Medicine, Inova Fairfax Hospital, Falls Church, VA, United States. 5. Borland-Groover Clinic, 4800 Belfort Rd, Jacksonville, FL, United States. 6. Johns Hopkins University School of Medicine, Baltimore, MD, United States. 7. Orlando Immunology Center, Orlando, FL, United States. 8. Atlanta Medical Center, Atlanta, GA, United States. 9. University of Pittsburgh Medical Center, Pittsburgh, PA, United States. 10. Atlanta Gastroenterology Association, Atlanta, GA, United States. 11. Yale School of Medicine, New Haven, CT, United States. 12. Scripps Clinic, La Jolla, CA, United States. 13. Janssen Research & Development, Beerse, Belgium. 14. Novellas Healthcare, Zellik, Belgium. 15. Gilead Sciences Inc, Foster City, CA, United States. 16. Janssen Research & Development LLC, Titusville, NJ, United States. 17. The Texas Liver Institute, University of Texas Health Science Center, San Antonio, TX, United States. 9

Background • Simeprevir (TMC 435) is an investigational, one pill, once- daily, potent oral HCV NS 3/4 A protease inhibitor recently approved in Japan and currently under regulatory review in North America and Europe • Sofosbuvir (GS-7977) is an HCV nucleotide NS 5 B polymerase inhibitor also currently under regulatory review • COSMOS is a Phase IIa, randomized, open-label study investigating simeprevir + sofosbuvir +/- ribavirin • Interim analysis Jacobson IM, et al. Abstract #LB-3, AASLD 2013 10
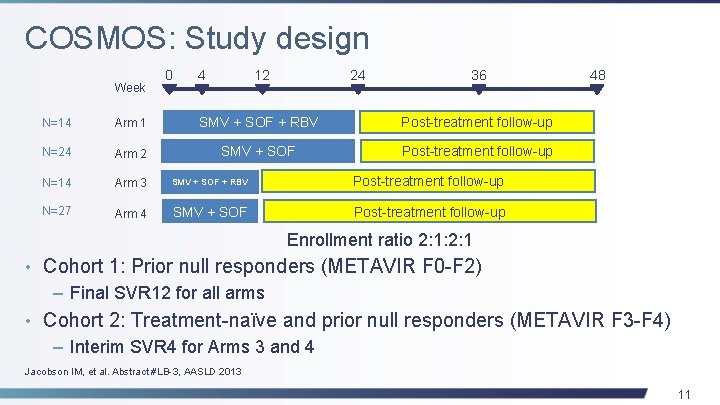
COSMOS: Study design Week 0 4 12 24 36 N=14 Arm 1 SMV + SOF + RBV Post-treatment follow-up N=24 Arm 2 SMV + SOF Post-treatment follow-up N=14 Arm 3 SMV + SOF + RBV Post-treatment follow-up N=27 Arm 4 SMV + SOF Post-treatment follow-up 48 Enrollment ratio 2: 1: 2: 1 • Cohort 1: Prior null responders (METAVIR F 0 -F 2) – Final SVR 12 for all arms • Cohort 2: Treatment-naïve and prior null responders (METAVIR F 3 -F 4) – Interim SVR 4 for Arms 3 and 4 Jacobson IM, et al. Abstract #LB-3, AASLD 2013 11
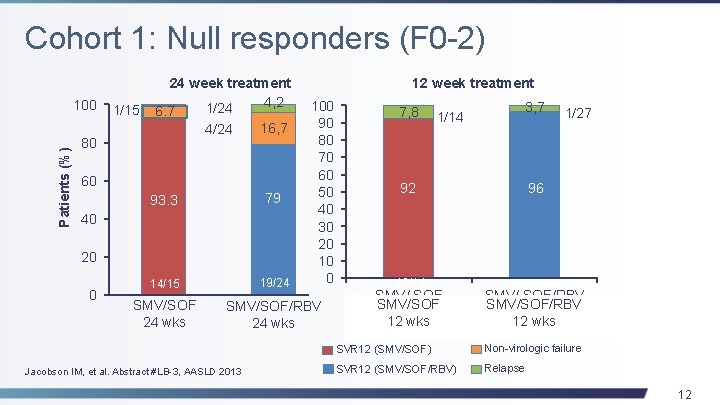
Cohort 1: Null responders (F 0 -2) Patients (%) 100 80 1/15 24 week treatment 4, 2 1/24 6. 7 16, 7 4/24 60 100 90 80 70 60 50 40 30 20 10 0 93. 3 79 14/15 19/24 SMV/SOF 24 wks SMV/SOF/RBV 24 wks 40 20 0 12 week treatment Jacobson IM, et al. Abstract #LB-3, AASLD 2013 7, 8 1/14 3, 7 92 96 13/14 26/27 SMV/ SOF SMV/SOF 12 Wks 12 wks 1/27 SMV/ SOF/RBV SMV/SOF/RBV 12 Wks 12 wks SVR 12 (SMV/SOF) Non-virologic failure SVR 12 (SMV/SOF/RBV) Relapse 12
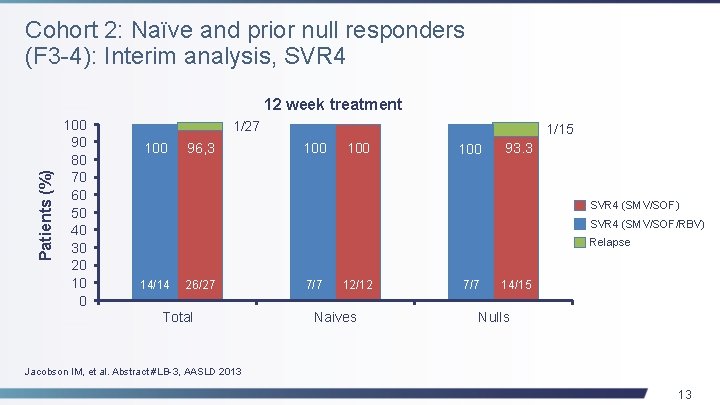
Cohort 2: Naïve and prior null responders (F 3 -4): Interim analysis, SVR 4 Patients (%) 12 week treatment 100 90 80 70 60 50 40 30 20 10 0 1/27 100 96, 3 1/15 100 100 93. 3 SVR 4 (SMV/SOF) SVR 4 (SMV/SOF/RBV) Relapse 14/14 26/27 Total 7/7 12/12 Naives 7/7 14/15 Nulls Jacobson IM, et al. Abstract #LB-3, AASLD 2013 13
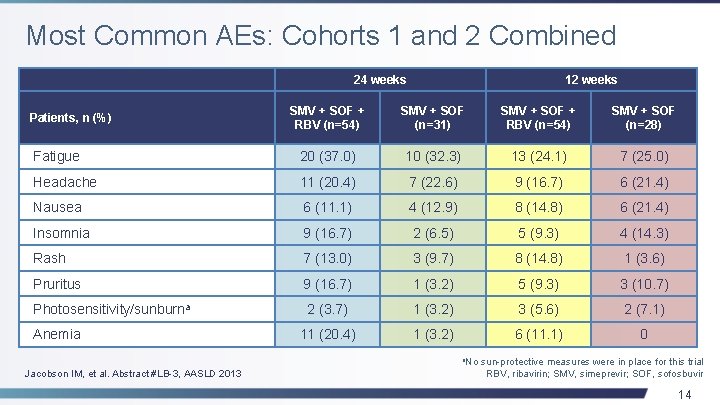
Most Common AEs: Cohorts 1 and 2 Combined 24 weeks 12 weeks SMV + SOF + RBV (n=54) SMV + SOF (n=31) SMV + SOF + RBV (n=54) SMV + SOF (n=28) Fatigue 20 (37. 0) 10 (32. 3) 13 (24. 1) 7 (25. 0) Headache 11 (20. 4) 7 (22. 6) 9 (16. 7) 6 (21. 4) Nausea 6 (11. 1) 4 (12. 9) 8 (14. 8) 6 (21. 4) Insomnia 9 (16. 7) 2 (6. 5) 5 (9. 3) 4 (14. 3) Rash 7 (13. 0) 3 (9. 7) 8 (14. 8) 1 (3. 6) Pruritus 9 (16. 7) 1 (3. 2) 5 (9. 3) 3 (10. 7) Photosensitivity/sunburna 2 (3. 7) 1 (3. 2) 3 (5. 6) 2 (7. 1) 11 (20. 4) 1 (3. 2) 6 (11. 1) 0 Patients, n (%) Anemia a. No Jacobson IM, et al. Abstract #LB-3, AASLD 2013 sun-protective measures were in place for this trial RBV, ribavirin; SMV, simeprevir; SOF, sofosbuvir 14

Conclusion • Treatment with SMV + SOF ± RBV results in: – High SVR 12 rates in HCV GT 1 null responder patients – High SVR 4 rates in naïve and null-responder patients with METAVIR F 3 -F 4 • Addition of RBV to SMV + SOF may not be needed to achieve high rates of SVR in this patient population • 12 weeks of treatment may confer similar SVR rates compared with 24 weeks of treatment • SMV + SOF ± RBV was generally well tolerated Jacobson IM, et al. Abstract #LB-3, AASLD 2013 15

Abstract #73 Once Daily Sofosbuvir/Ledipasvir Fixed Dose Combination with or without Ribavirin: the ELECTRON trial Edward J. Gane 1, Catherine A. Stedman 2, Robert H. Hyland 3, Xiao Ding 3, Evguenia S. Svarovskaia 3, Phil S. Pang 3, William T. Symonds 3 1. Auckland Clinical Studies, Auckland, New Zealand. 2. Christchurch Clinical Studies Trust, Christchurch, New Zealand. 3. Gilead Science, Inc, Foster City, CA, United States. 16

Direct Acting Antiviral Agents Sofosbuvir/Ledipasvir FDC • Once daily, oral fixed-dose (400/90 mg) combination tablet • No food effect • >2000 patients treated SOF Nucleotide Polymerase inhibitor LDV NS 5 A inhibitor GS-9669 • HCV NS 5 B non-nucleoside inhibitor, binding at thumb site II of the polymerase • Potent antiviral activity with QD dosing • Nanomolar potency against GT 1 a and 1 b Gane EJ, et al. Abstract #73, AASLD 2013 17

Study Design F 4 GT 1 Experienced F 3/F 4 GT 1 Naïve Randomized Wk 0 F 0/F 1/F 2 Wk 6 Wk 12 SOF/LDV FDC (n=10) SOF/LDV FDC + RBV (n=25) SVR 12 SOF/LDV FDC + GS-9669 (n=25) SOF/LDV FDC + RBV (n=25) • Primary endpoint: SVR 12 (HCV RNA <LLOQ) • Patients enrolled in ELECTRON or ELECTRON 2 (GT 1, F 3/F 4) • All groups were open label Gane EJ, et al. Abstract #73, AASLD 2013 18
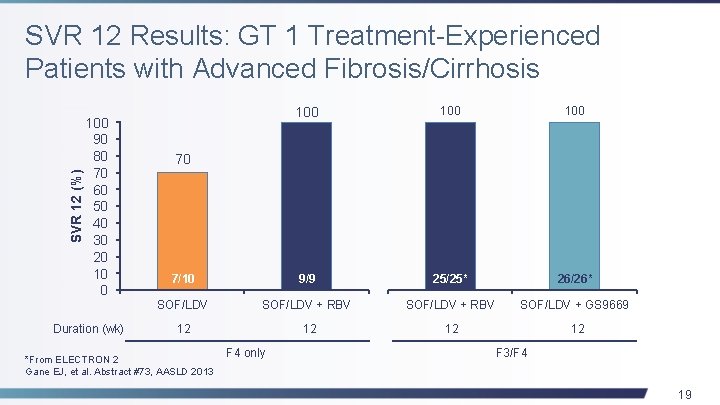
SVR 12 (%) SVR 12 Results: GT 1 Treatment-Experienced Patients with Advanced Fibrosis/Cirrhosis 100 90 80 70 60 50 40 30 20 10 0 Duration (wk) 100 100 7/10 9/9 25/25* 26/26* SOF/LDV + RBV SOF/LDV + GS 9669 12 12 70 *From ELECTRON 2 Gane EJ, et al. Abstract #73, AASLD 2013 F 4 only F 3/F 4 19
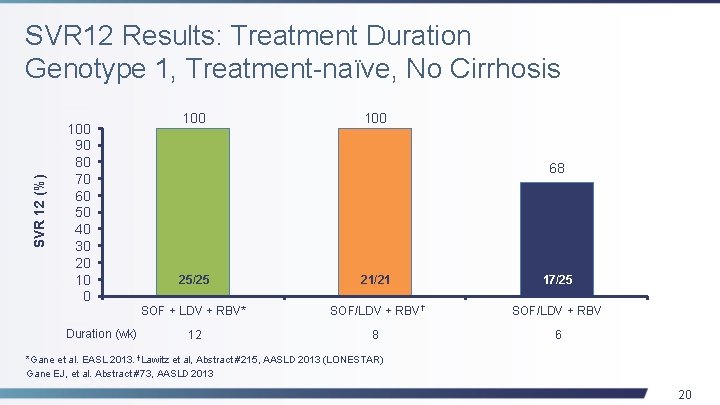
SVR 12 (%) SVR 12 Results: Treatment Duration Genotype 1, Treatment-naïve, No Cirrhosis 100 90 80 70 60 50 40 30 20 10 0 Duration (wk) 100 68 25/25 21/21 17/25 SOF + LDV + RBV* SOF/LDV + RBV † SOF/LDV + RBV 12 8 6 *Gane et al. EASL 2013. †Lawitz et al, Abstract #215, AASLD 2013 (LONESTAR) Gane EJ, et al. Abstract #73, AASLD 2013 20

Conclusions • In treatment-experienced patients with advanced fibrosis/cirrhosis, either RBV or GS-9669 may enhance the efficacy of SOF/LDV given for 12 weeks • The optimal duration of SOF/LDV in treatment-naïve GT 1 patients, even with the addition of RBV, is more than 6 weeks • Regimens of SOF/LDV alone, or with RBV or GS- 9669, were safe and well tolerated Gane EJ, et al. Abstract #73, AASLD 2013 21

Abstract #75 Interferon- and Ribavirin-free Regimen of ABT-450/r + ABT 267 in HCV Genotype 1 b-infected Treatment-naïve Patients and Prior Null Responders Eric Lawitz 1, Christophe Hezode 2, Peter Varunok 3, Paul J. Thuluvath 4, Tolga Baykal 5, Mudra Kapoor 5, Sandra S. Lovell 5, Tianli Wang 5, Tami Pilot-Matias 5, Regis A. Vilchez 5, Barry Bernstein 5 1. The Texas Liver Institute, University of Texas Health Science Center, San Antonio, TX, United States. 2. Assistance Publique Hopitaux de Paris, France. 3. Premier Medical Group of the Hudson Valley, PC, Poughkeepsie, NY, United States. 4. The Institute for Digestive Health and Liver Disease at Mercy, Baltimore, MD, United States. 5. Abb. Vie Inc. , North Chicago, IL, United States. 22
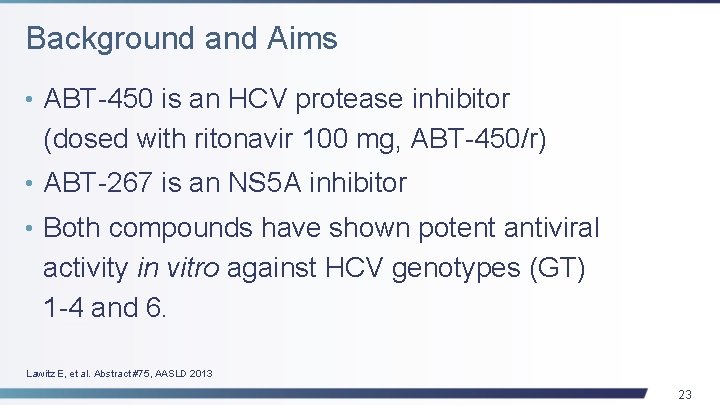
Background and Aims • ABT-450 is an HCV protease inhibitor (dosed with ritonavir 100 mg, ABT-450/r) • ABT-267 is an NS 5 A inhibitor • Both compounds have shown potent antiviral activity in vitro against HCV genotypes (GT) 1 -4 and 6. Lawitz E, et al. Abstract #75, AASLD 2013 23
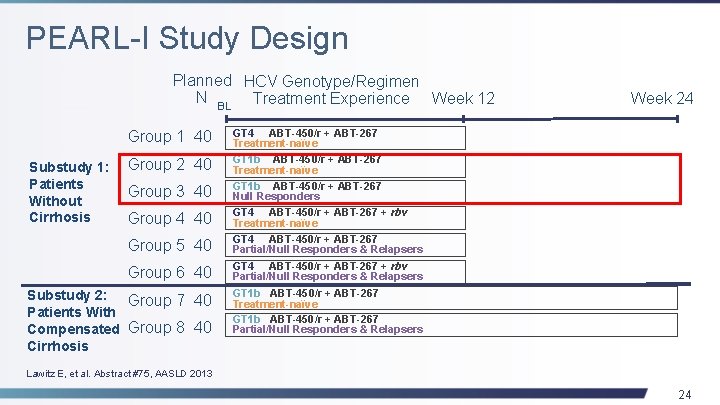
PEARL-I Study Design Planned HCV Genotype/Regimen N BL Treatment Experience Week 12 Group 1 40 Substudy 1: Patients Without Cirrhosis Group 2 40 Group 3 40 Group 4 40 Group 5 40 Group 6 40 Substudy 2: Group 7 40 Patients With Compensated Group 8 40 Cirrhosis Week 24 GT 4 ABT-450/r + ABT-267 Treatment-naïve GT 1 b ABT-450/r + ABT-267 Null Responders GT 4 ABT-450/r + ABT-267 + rbv Treatment-naïve GT 4 ABT-450/r + ABT-267 Partial/Null Responders & Relapsers GT 4 ABT-450/r + ABT-267 + rbv Partial/Null Responders & Relapsers GT 1 b ABT-450/r + ABT-267 Treatment-naïve GT 1 b ABT-450/r + ABT-267 Partial/Null Responders & Relapsers Lawitz E, et al. Abstract #75, AASLD 2013 24
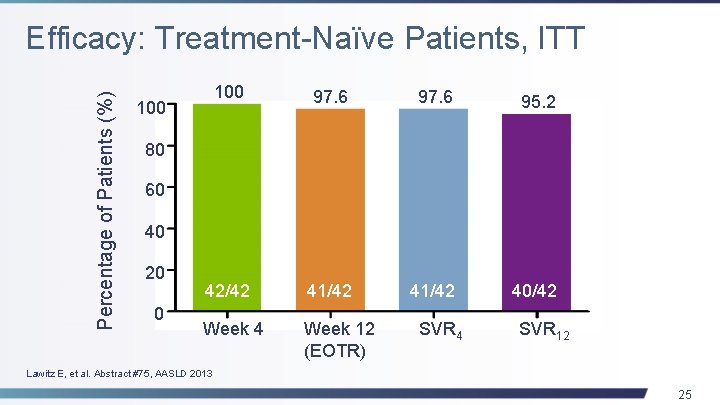
Percentage of Patients (%) Efficacy: Treatment-Naïve Patients, ITT 100 97. 6 95. 2 42/42 41/42 40/42 Week 4 Week 12 (EOTR) 100 80 60 40 20 0 SVR 4 SVR 12 Lawitz E, et al. Abstract #75, AASLD 2013 25
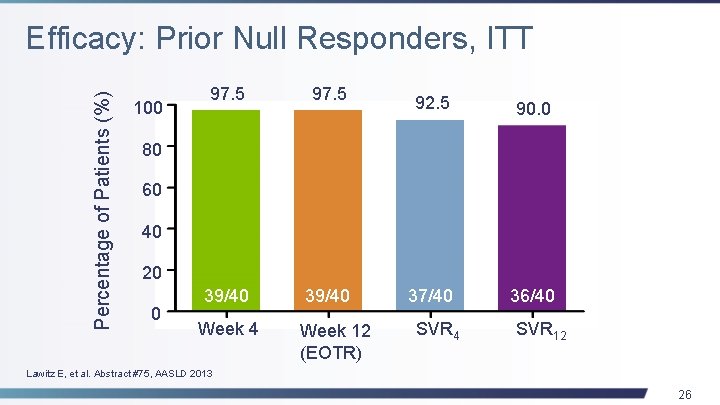
Percentage of Patients (%) Efficacy: Prior Null Responders, ITT 100 97. 5 39/40 92. 5 90. 0 37/40 36/40 80 60 40 20 0 Week 4 Week 12 (EOTR) SVR 4 SVR 12 Lawitz E, et al. Abstract #75, AASLD 2013 26
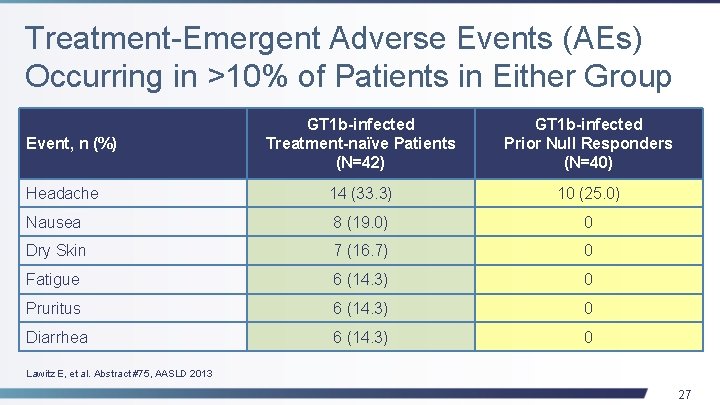
Treatment-Emergent Adverse Events (AEs) Occurring in >10% of Patients in Either Group GT 1 b-infected Treatment-naïve Patients (N=42) GT 1 b-infected Prior Null Responders (N=40) Headache 14 (33. 3) 10 (25. 0) Nausea 8 (19. 0) 0 Dry Skin 7 (16. 7) 0 Fatigue 6 (14. 3) 0 Pruritus 6 (14. 3) 0 Diarrhea 6 (14. 3) 0 Event, n (%) Lawitz E, et al. Abstract #75, AASLD 2013 27

Abstract #215 Once Daily Sofosbuvir/Ledipasvir Fixed Dose Combination with or without Ribavirin Resulted in ≥ 95% Sustained Virologic Response In Patients with HCV Genotype 1, Including Patients with Cirrhosis: the LONESTAR trial Eric Lawitz 1, Fred Poordad 1, Robert H. Hyland 2, Xiao Ding 2, Christy Hebner 2, Phil S. Pang 2, William T. Symonds 2, John G. Mc. Hutchison 2, Fernando E. Membreno 1 1. The Texas Liver Institute, University of Texas Health Science Center, San Antonio, TX, United States. 2. Gilead Science, Inc, Foster City, CA, United States. 28
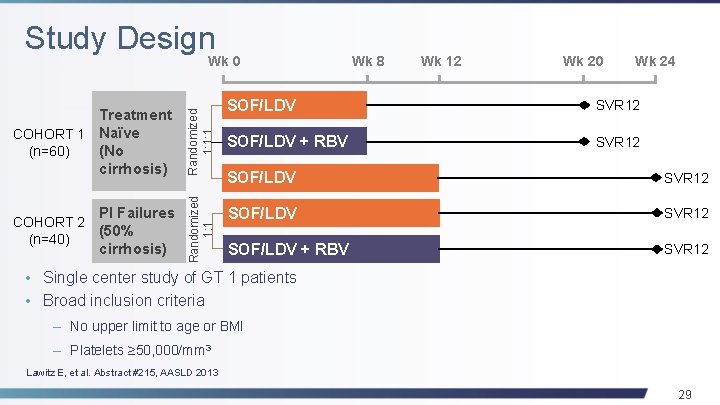
Study Design Treatment COHORT 1 Naïve (n=60) (No cirrhosis) Randomized 1: 1: 1 PI Failures (50% cirrhosis) Randomized 1: 1 Wk 0 COHORT 2 (n=40) Wk 8 Wk 12 Wk 20 Wk 24 SOF/LDV SVR 12 SOF/LDV + RBV SVR 12 • Single center study of GT 1 patients • Broad inclusion criteria – No upper limit to age or BMI – Platelets ≥ 50, 000/mm 3 Lawitz E, et al. Abstract #215, AASLD 2013 29
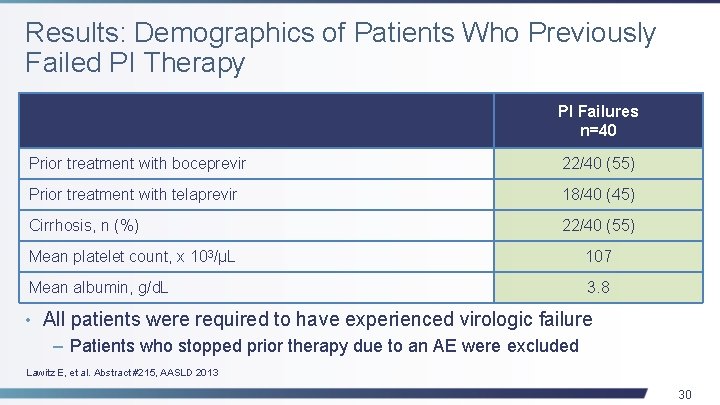
Results: Demographics of Patients Who Previously Failed PI Therapy PI Failures n=40 Prior treatment with boceprevir 22/40 (55) Prior treatment with telaprevir 18/40 (45) Cirrhosis, n (%) 22/40 (55) Mean platelet count, x 103/µL 107 Mean albumin, g/d. L 3. 8 • All patients were required to have experienced virologic failure – Patients who stopped prior therapy due to an AE were excluded Lawitz E, et al. Abstract #215, AASLD 2013 30
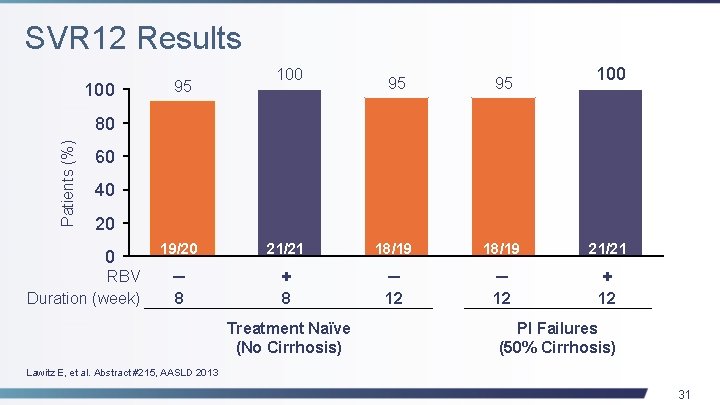
SVR 12 Results 100 95 95 100 Patients (%) 80 60 40 20 0 RBV Duration (week) 19/20 21/21 18/19 21/21 ─ 8 + ─ 12 + 8 Treatment Naïve (No Cirrhosis) 12 PI Failures (50% Cirrhosis) Lawitz E, et al. Abstract #215, AASLD 2013 31
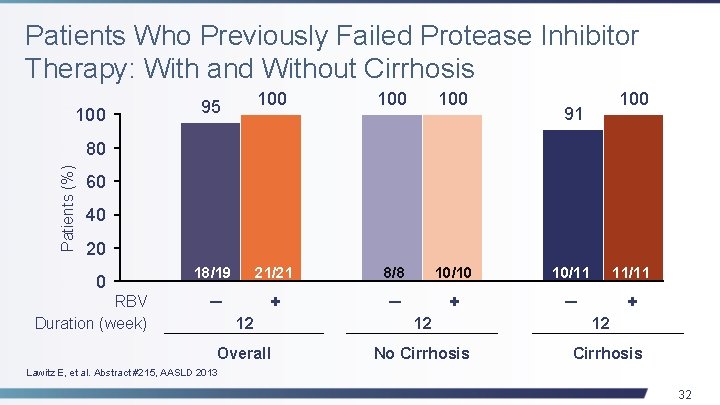
Patients Who Previously Failed Protease Inhibitor Therapy: With and Without Cirrhosis 100 100 18/19 21/21 8/8 10/10 10/11 11/11 ─ + ─ + 95 100 91 Patients (%) 80 60 40 20 0 RBV Duration (week) 12 12 Overall No Cirrhosis 12 Cirrhosis Lawitz E, et al. Abstract #215, AASLD 2013 32
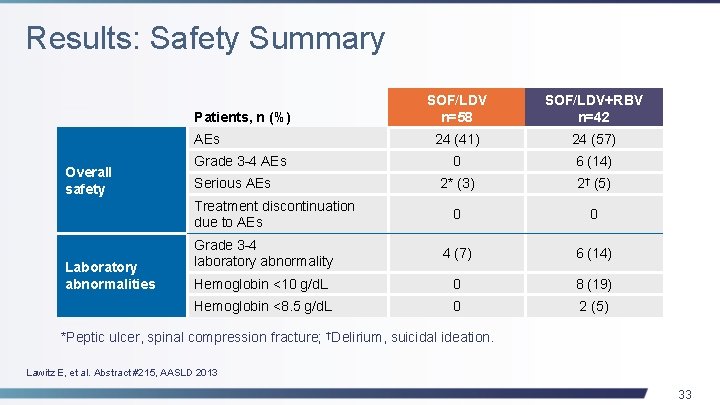
Results: Safety Summary SOF/LDV n=58 SOF/LDV+RBV n=42 24 (41) 24 (57) 0 6 (14) 2* (3) 2† (5) 0 0 Grade 3 -4 laboratory abnormality 4 (7) 6 (14) Hemoglobin <10 g/d. L 0 8 (19) Hemoglobin <8. 5 g/d. L 0 2 (5) Patients, n (%) AEs Overall safety Grade 3 -4 AEs Serious AEs Treatment discontinuation due to AEs Laboratory abnormalities *Peptic ulcer, spinal compression fracture; †Delirium, suicidal ideation. Lawitz E, et al. Abstract #215, AASLD 2013 33

Abstract #LB-4 Sofosbuvir in Combination With Peg. IFN and Ribavirin for 12 Weeks Provides High SVR Rates in HCV-Infected Genotype 2 or 3 Treatment Experienced Patients with and without Compensated Cirrhosis: Results from the LONESTAR-2 Study Eric Lawitz 1, 2, Fred Poordad 1, 2, Diana M. Brainard 3, Robert H. Hyland 3, Di An 3, William T. Symonds 3, John G. Mc. Hutchison 3, Fernando E. Membreno 1, 2 1. Texas Liver Institute, San Antonio, TX, United States. 2. University of Texas Health Science Center, San Antonio, TX, United States. 3. Gilead Science, Inc, Foster City, CA, United States. 34

Study Design Wk 0 GT 2/3 (N=47) • Wk 12 SOF + PEG/RBV Wk 24 Wk 36 SVR 12 Study population – HCV GT 2 or 3 – Failed treatment with pegylated interferon and ribavirin – Approximately 50% with compensated cirrhosis – HIV and HBV coinfected patients excluded Lawitz E, et al. Abstract #LB-4, AASLD 2013 35
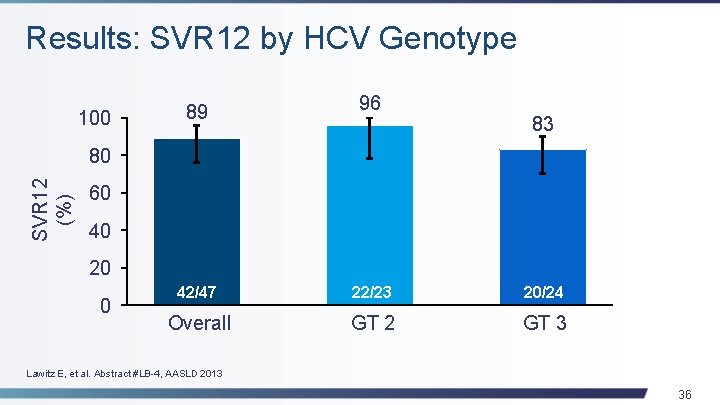
Results: SVR 12 by HCV Genotype 100 89 96 83 SVR 12 (%) 80 60 40 20 0 42/47 22/23 20/24 Overall GT 2 GT 3 Lawitz E, et al. Abstract #LB-4, AASLD 2013 36
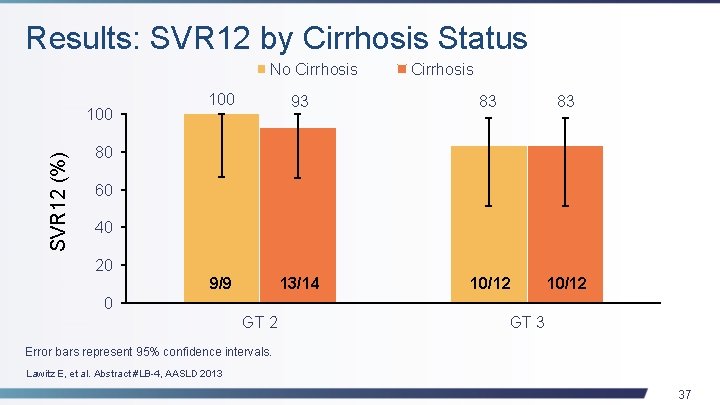
Results: SVR 12 by Cirrhosis Status No Cirrhosis SVR 12 (%) 100 Cirrhosis 100 93 83 83 9/9 13/14 10/12 80 60 40 20 0 GT 2 GT 3 Error bars represent 95% confidence intervals. Lawitz E, et al. Abstract #LB-4, AASLD 2013 37
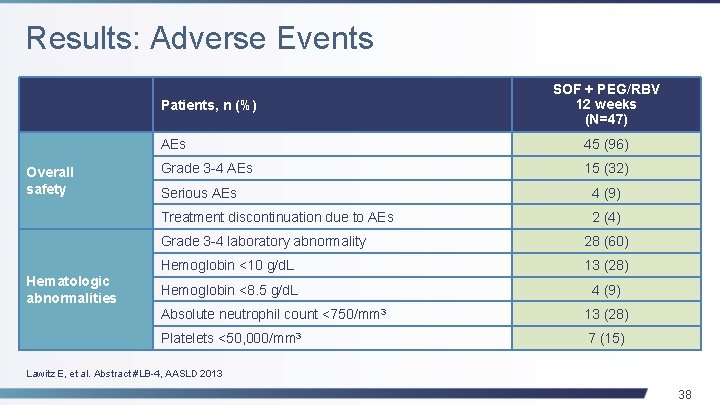
Results: Adverse Events Patients, n (%) Overall safety Hematologic abnormalities SOF + PEG/RBV 12 weeks (N=47) AEs 45 (96) Grade 3 -4 AEs 15 (32) Serious AEs 4 (9) Treatment discontinuation due to AEs 2 (4) Grade 3 -4 laboratory abnormality 28 (60) Hemoglobin <10 g/d. L 13 (28) Hemoglobin <8. 5 g/d. L 4 (9) Absolute neutrophil count <750/mm 3 13 (28) Platelets <50, 000/mm 3 7 (15) Lawitz E, et al. Abstract #LB-4, AASLD 2013 38

Conclusions • SOF + PEG/RBV for 12 weeks demonstrated high efficacy in treatment-experienced GT 2/3 patients who have historically low response rates and limited treatment options – SVR rates were similar in patients with and without cirrhosis • SOF + PEG/RBV was generally safe and well tolerated – Safety profile consistent with PEG/RBV treatment – Low discontinuation rates Lawitz E, et al. Abstract #LB-4, AASLD 2013 39 39

Abstract #1085 Sofosbuvir + Ribavirin for 12 or 24 Weeks for Patients with HCV Genotype 2 or 3: the VALENCE trial Stefan Zeuzem 1, Geoffrey M. Dusheiko 2, Riina Salupere 3, Alessandra Mangia 4, Robert Flisiak 5, Robert H. Hyland 6, Ari Illeperuma 6, Evguenia S. Svarovskaia 6, Diana M. Brainard 6, William T. Symonds 6, John G. Mc. Hutchison 6, Ola Weiland 7, Hendrik W. Reesink 8, Peter Ferenci 9, Christophe Hezode 10, Rafael Esteban 11 1. Johann Wolfgang Goethe University, Frankfurt, Germany. 2. Royal Free and University College School of Medicine, Royal Free Hospital, London, United Kingdom. 3. Tartu University Hospital, Tartu, Estonia. 4. "Casa Sollievo della Sofferenza" Hospital, San Giovanni Rotondo, Italy. 5. Medical University of Bialystok, Poland. 6. Gilead Sciences, Inc. , Foster City, CA, United States. 7. Karolinska Institutet, Karolinska University Hospital Huddinge, Stockholm, Sweden. 8. Academic Medical Center, Amsterdam, Netherlands. 9. Medical University of Vienna, Austria. 10. Hôpital Henri Mondor, Créteil, France. 11. Hospital Universitario Val d’Hebron, Barcelona, Spain. 40
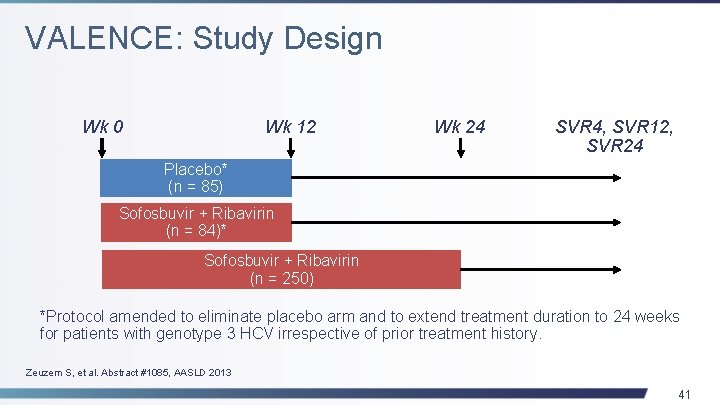
VALENCE: Study Design Wk 0 Wk 12 Wk 24 SVR 4, SVR 12, SVR 24 Placebo* (n = 85) Sofosbuvir + Ribavirin (n = 84)* Sofosbuvir + Ribavirin (n = 250) *Protocol amended to eliminate placebo arm and to extend treatment duration to 24 weeks for patients with genotype 3 HCV irrespective of prior treatment history. Zeuzem S, et al. Abstract #1085, AASLD 2013 41
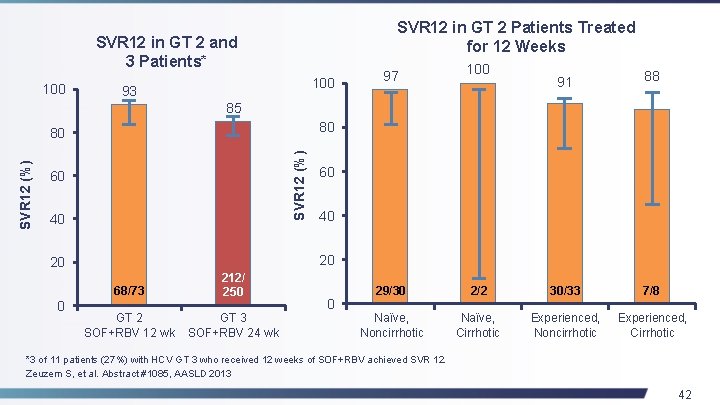
SVR 12 in GT 2 Patients Treated for 12 Weeks SVR 12 in GT 2 and 3 Patients* 100 93 97 100 91 88 85 80 SVR 12 (%) 80 60 40 40 20 20 0 60 68/73 212/ 250 GT 2 SOF+RBV 12 wk GT 3 SOF+RBV 24 wk 0 29/30 2/2 30/33 7/8 Naïve, Noncirrhotic Naïve, Cirrhotic Experienced, Noncirrhotic Experienced, Cirrhotic *3 of 11 patients (27%) with HCV GT 3 who received 12 weeks of SOF+RBV achieved SVR 12. Zeuzem S, et al. Abstract #1085, AASLD 2013 42
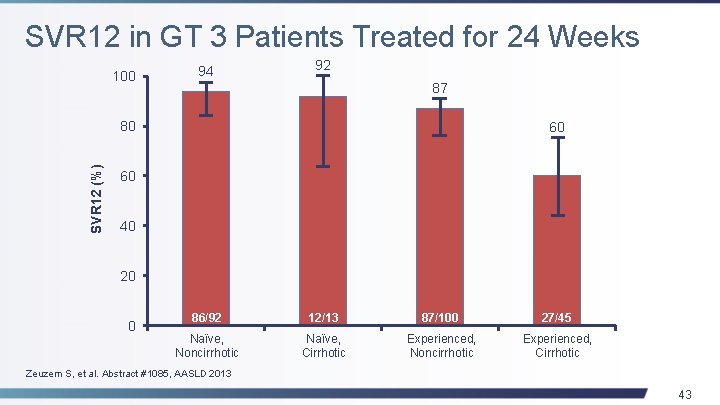
SVR 12 in GT 3 Patients Treated for 24 Weeks 100 94 92 87 SVR 12 (%) 80 60 60 40 20 0 86/92 12/13 87/100 27/45 Naïve, Noncirrhotic Naïve, Cirrhotic Experienced, Noncirrhotic Experienced, Cirrhotic Zeuzem S, et al. Abstract #1085, AASLD 2013 43

Abstract #LB-2 Sofosbuvir and Ribavirin for the Treatment of Established Recurrent Hepatitis C Infection After Liver Transplantation: Preliminary Results of a Prospective, Multicenter Study Michael R. Charlton 1, Edward J. Gane 2, Michael P. Manns 3, Robert S. Brown 4, Michael P. Curry 5, Paul Y. Kwo 6, Robert J. Fontana 7, Richard Gilroy 8, Lewis W. Teperman 9, Andrew J. Muir 10, John G. Mc. Hutchison 11, William T. Symonds 11, Jill M. Denning 11, Lindsay Mc. Nair 11, Sarah Arterburn 11, Norah Terrault 12, Didier Samuel 13, Xavier Forns 14 1. Mayo Clinic, Rochester, MN, United States. 2. Auckland City Hospital, Auckland, New Zealand. 3. Hannover Medical School, Hannover, Germany. 4. Columbia University, New York, NY, United States. 5. Beth Israel Deaconess Medical Center, Boston, MA, United States. 6. Indiana School of Medicine, Indianapolis, IN, United States. 7. University of Michigan, Ann Arbor, MI, United States. 8. Kansas University Medical Center, Lawrence, KS, United States. 9. NYU Medical Center, New York , NY, United States. 10. Duke University Medical Center, Durham, NC, United States. 11. Gilead Sciences, Foster City, CA, United States. 12. University of California, San Francisco, CA, United States. 13. Université Paris-Sud, Villejuif, France. 14. The Liver Unit, Barcelona, Spain. 44

Background • Reinfection of the transplanted liver is universal in patients who are serum HCV RNA-positive at the time of transplantation • Recurrence of HCV is the most common cause of mortality and graft loss following transplantation – 10– 50% of patients with recurrent infection progress to cirrhosis within 5 years 1 • Once cirrhosis is established, the probability of liver graft failure is 42% within 12 months 2 • Current therapies for HCV treatment used after transplantation have poor tolerance, poor efficacy, severe adverse reactions, and significant interactions with immunosuppression medications 1. Berenguer M, et al. Clin Liver Dis 2007; 11: 355– 76; 2. Berenguer M, et al. Hepatology 2002; 36: 202 -10. Charlton MR, et al. Abstract #LB-2, AASLD 2013 45
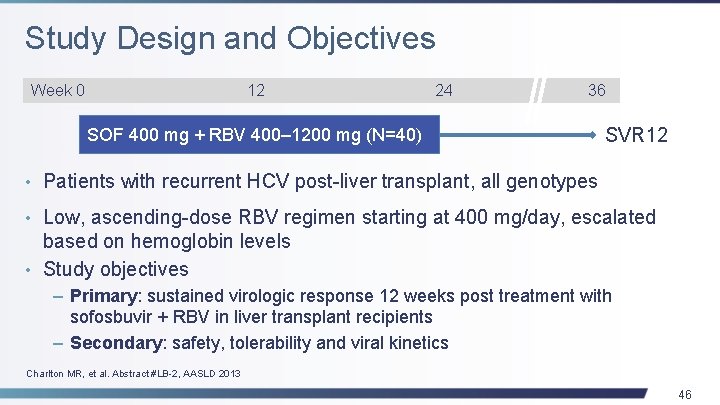
Study Design and Objectives Week 0 12 24 36 SOF 400 mg + RBV 400‒ 1200 mg (N=40) SVR 12 • Patients with recurrent HCV post-liver transplant, all genotypes • Low, ascending-dose RBV regimen starting at 400 mg/day, escalated based on hemoglobin levels • Study objectives – Primary: sustained virologic response 12 weeks post treatment with sofosbuvir + RBV in liver transplant recipients – Secondary: safety, tolerability and viral kinetics Charlton MR, et al. Abstract #LB-2, AASLD 2013 46

Key Inclusion/Exclusion Criteria • Inclusion criteria – Liver transplant ≥ 6 and ≤ 150 months prior to enrollment – Treatment-naïve or experienced – CPT ≤ 7 and MELD ≤ 17 – Primary or secondary, liver alone or liver-kidney transplant – Absence of organ rejection • Exclusion criteria – Current signs of decompensation – Use of corticosteriods at any dose >5 mg of prednisone/day Charlton MR, et al. Abstract #LB-2, AASLD 2013 47
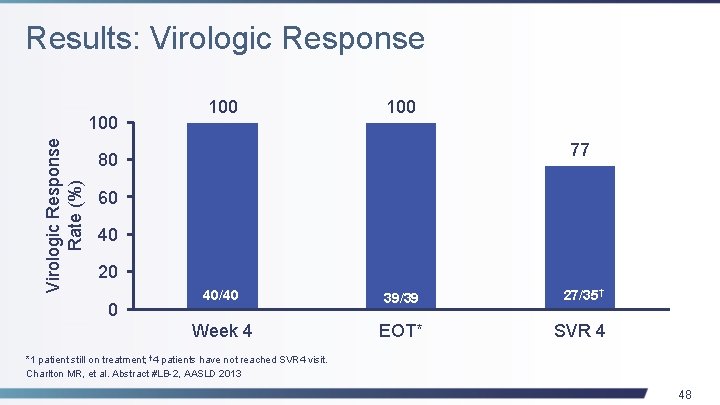
Results: Virologic Response Rate (%) 100 100 77 80 60 40 20 0 40/40 39/39 27/35† Week 4 EOT* SVR 4 *1 patient still on treatment; † 4 patients have not reached SVR 4 visit. Charlton MR, et al. Abstract #LB-2, AASLD 2013 48
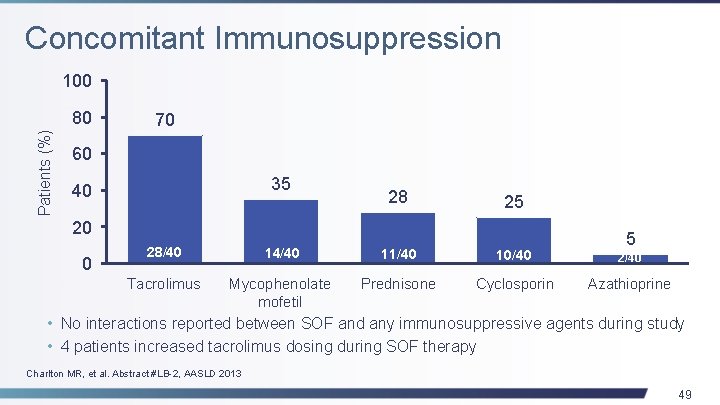
Concomitant Immunosuppression 100 Patients (%) 80 70 60 35 40 28 25 20 0 5 28/40 14/40 11/40 10/40 2/40 Tacrolimus Mycophenolate mofetil Prednisone Cyclosporin Azathioprine • No interactions reported between SOF and any immunosuppressive agents during study • 4 patients increased tacrolimus dosing during SOF therapy Charlton MR, et al. Abstract #LB-2, AASLD 2013 49
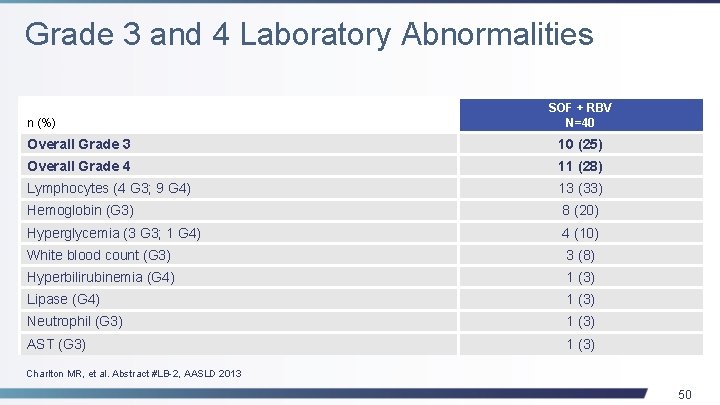
Grade 3 and 4 Laboratory Abnormalities n (%) SOF + RBV N=40 Overall Grade 3 10 (25) Overall Grade 4 11 (28) Lymphocytes (4 G 3; 9 G 4) 13 (33) Hemoglobin (G 3) 8 (20) Hyperglycemia (3 G 3; 1 G 4) 4 (10) White blood count (G 3) 3 (8) Hyperbilirubinemia (G 4) 1 (3) Lipase (G 4) 1 (3) Neutrophil (G 3) 1 (3) AST (G 3) 1 (3) Charlton MR, et al. Abstract #LB-2, AASLD 2013 50

Abstract #213 Pretransplant Sofosbuvir and Ribavirin to Prevent Recurrence of HCV Infection after Liver Transplantation Michael P. Curry 1, Xavier Forns 2, Raymond T. Chung 3, Norah Terrault 4, Robert S. Brown 5, Jonathan M. Fenkel 6, Fredric D. Gordon 7, Jacqueline G. O'Leary 8, Alexander Kuo 9, Thomas D. Schiano 10, Gregory T. Everson 11, Eugene R. Schiff 12, Alex Befeler 13, John G. Mc. Hutchison 14, William T. Symonds 14, Jill M. Denning 14, Lindsay Mc. Nair 14, Sarah Arterburn 14, Dilip Moonka 15, Edward J. Gane 16, Nezam H. Afdhal 1 1. Beth Israel Deaconess Medical Center, Boston, MA, United States. 2. The Liver Unit, Barcelona, Spain. 3. Massachusetts General Hospital, Boston, MA, United States. 4. University of California San Francisco, CA, United States. 5. Columbia University, New York, NY, United States. 6. Thomas Jefferson University Hospital, Philadelphia, PA, United States. 7. Lahey Clinic, Burlington, MA, United States. 8. Baylor University Medical Center, Dallas, TX, United States. 9. University of California San Diego, La Jolla, CA, United States. 10. Mount Sinai School of Medicine, New York, NY, United States. 11. University of Colorado, Denver, CO, United States. 12. University of Miami, FL, United States. 13. St. Louis University, St. Louis, MO, United States. 14. Gilead Sciences, Foster City, CA, United States. 15. Henry Ford Health System, Detroit, MI, United States. 16. Auckland City Hospital, Auckland, New Zealand. 51

Background and Aims: • Recurrent HCV infection of the allograft is universal in patients with detectable HCV RNA at the time of liver transplantation (LT) and may result in accelerated progression to cirrhosis and graft loss. • Interferon-based antiviral treatment before LT can prevent HCV recurrence, but this treatment is poorly tolerated and effective in only a minority of patients. Curry MP, et al. Abstract #213, AASLD 2013 52

Methods: • In this phase 2 open-label study, patients with chronic HCV infection of any genotype (GT) listed for LT for hepatocellular carcinoma (HCC) received up to 48 weeks of SOF 400 mg/day and RBV 1000 -1200 mg/day before LT. • All patients had HCC within Milan criteria and well compensated cirrhosis (Child-Pugh-Turcotte score of ≤ 7). • The primary endpoint was virologic response (HCV RNA <25 IU/m. L) 12 weeks after LT in patients who had HCV RNA <25 IU/m. L at their last measurement prior to LT (SVR 12). • Post-LT immunosuppressive regimen was tacrolimus plus prednisone with or without mycophenolate mofetil. Curry MP, et al. Abstract #213, AASLD 2013 53

Results: • 36 patients included in efficacy analysis • Received a mean of 17. 1 (range 3. 3 to 33. 7) weeks of treatment prior to LT • At the time of writing, 26 patients have reached at least 12 weeks post- transplant, of whom 18 (69%, 90% CI 51% to 84%) achieved SVR 12. • The most frequently reported adverse events were fatigue, anemia, and rash. • Two patients discontinued treatment due to AEs of acute renal failure and pneumonitis, neither was attributed to study drug. • One SAE, anemia, was considered related to study drug. Curry MP, et al. Abstract #213, AASLD 2013 54
- Slides: 54