Best Implant Surface in Current and Future Implantology

Best Implant Surface in Current and Future Implantology Merit Dental Clinic Yin-Shik Hur, Periodontist

Today’s Topics in Implant Surfaces 1. Protein or peptides coated surface 2. Sol-gel coated surface 3. Alkali-heat treated surface 4. Fluoride Incorporated surface 5. Anodizing surface 6. Conclusion
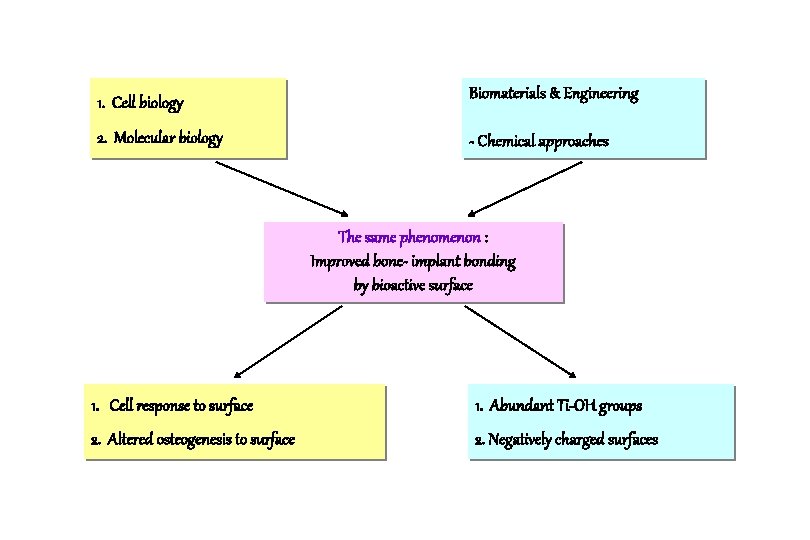
1. Cell biology Biomaterials & Engineering 2. Molecular biology - Chemical approaches The same phenomenon : Improved bone- implant bonding by bioactive surface 1. Cell response to surface 1. Abundant Ti-OH groups 2. Altered osteogenesis to surface 2. Negatively charged surfaces
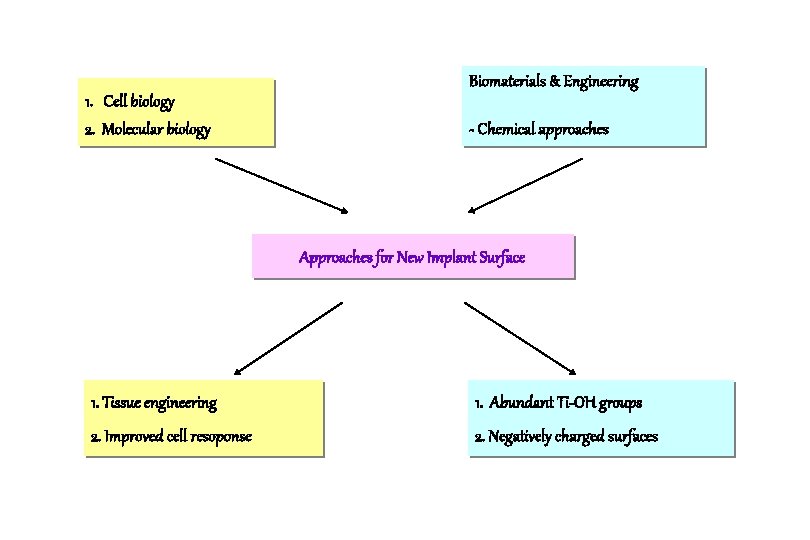
Biomaterials & Engineering 1. Cell biology 2. Molecular biology - Chemical approaches Approaches for New Implant Surface 1. Tissue engineering 1. Abundant Ti-OH groups 2. Improved cell resoponse 2. Negatively charged surfaces

Three Different Approaches for Bioactive Surfaces 1. Cell and molecular biology 2. Biomaterials and engineering 3. Histomorphometry
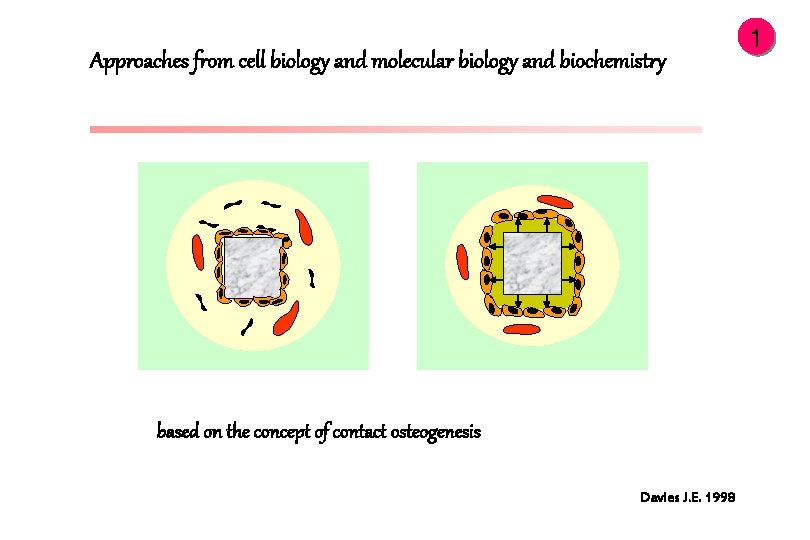
Approaches from cell biology and molecular biology and biochemistry based on the concept of contact osteogenesis Davies J. E. 1998 1

Biochemical modifications using specific peptides 1. Surface-immobilized peptides, proteins, or growth factor. 2. Guiding of cell behavior on an ultrathin layer of bioactive molecules. 3. Improving the biocompatibility of a surface without adversely affecting the bulk properties of the system.
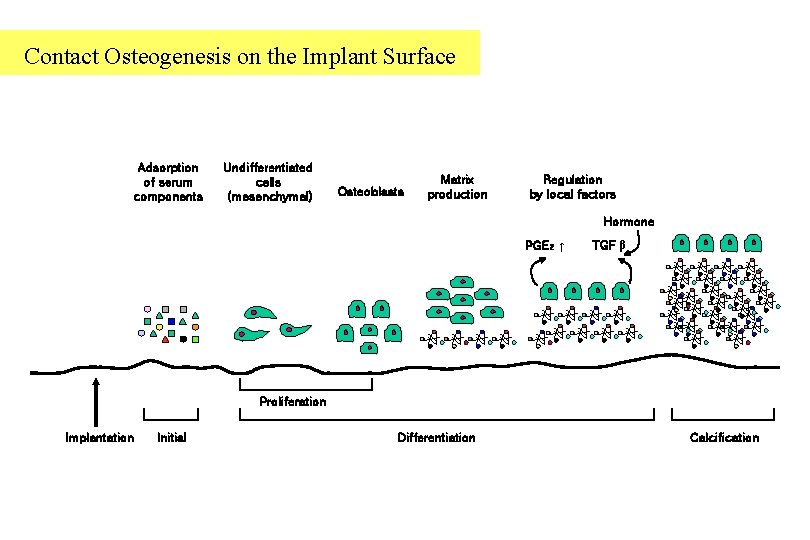
Contact Osteogenesis on the Implant Surface Adsorption of serum components Undifferentiated cells (mesenchymal) Osteoblasts Matrix production Regulation by local factors Hormone PGE 2 ↑ TGF β Proliferation Implantation Initial Differentiation Calcification
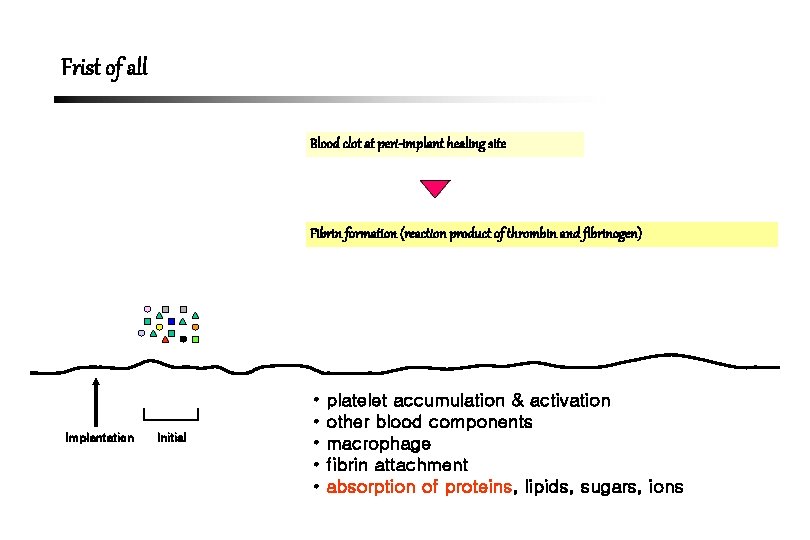
Frist of all Blood clot at peri-implant healing site Fibrin formation (reaction product of thrombin and fibrinogen) Implantation Initial • • • platelet accumulation & activation other blood components macrophage fibrin attachment absorption of proteins, lipids, sugars, ions
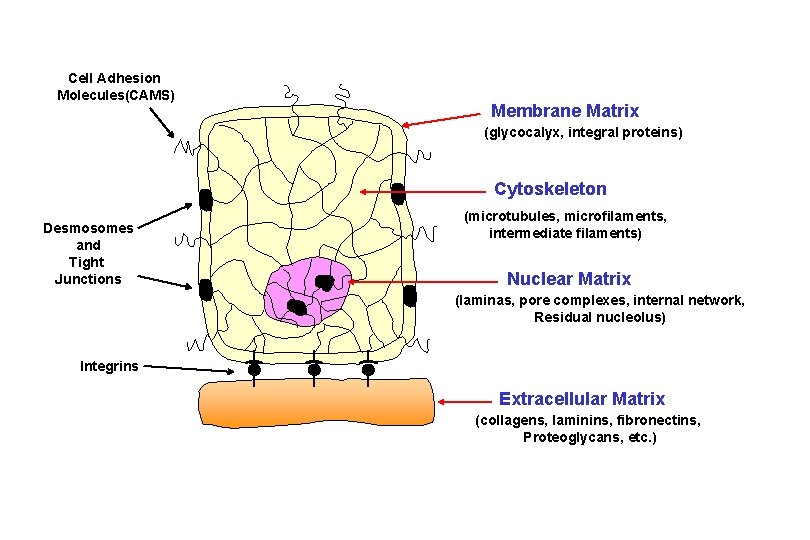
Cell Adhesion Molecules(CAMS) Membrane Matrix (glycocalyx, integral proteins) Cytoskeleton Desmosomes and Tight Junctions (microtubules, microfilaments, intermediate filaments) Nuclear Matrix (laminas, pore complexes, internal network, Residual nucleolus) Integrins Extracellular Matrix (collagens, laminins, fibronectins, Proteoglycans, etc. )
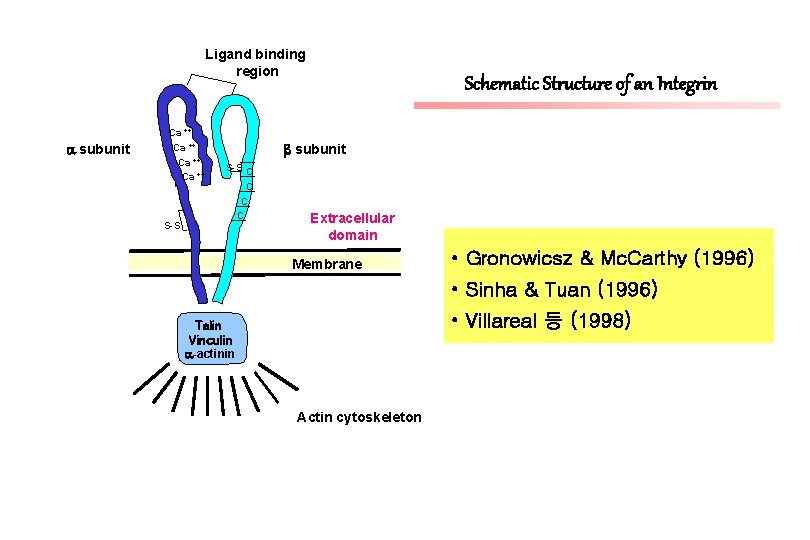
Ligand binding region subunit Ca ++ S-S Schematic Structure of an Integrin subunit S-S C C Extracellular domain Membrane • Gronowicsz & Mc. Carthy (1996) • Sinha & Tuan (1996) • Villareal 등 (1998) Talin Vinculin -actinin Actin cytoskeleton

RGD proteins 1. Arginine-glycine-aspartic acid binding site sequence 2. These protein allow it to mediate mesenchymal cell atachment by integrins. 3. Fibronectin, vitronectin, 4. By pre-conditioning it with specific RGD proteins, the types of adherent compounds and their sequence of adsorption can be modified.

In vivo studies of RGD-coated titanium implant 1. A significant increase in bone thickness for RGD implants at 4 weeks while control samples did not show any significant change. 2. Mechanical pull-out testing conducted at 4 weeks revealed the average interfacial shear strength of peptide modified rods was 38% greater than that for control rods, although it should be noted that this difference was not statistically significant. Ferris et al. , Biomaterials, 1999

In vivo studies of RGD-coated titanium implant 3. These pilot data suggest that an RGD peptide coating may enhance the titanium-rod osseointegration in the rat femur. Ferris et al. , Biomaterials, 1999

Biochemical Methods for specific peptides bonding 1. Physical adsorption process (physi- and chemisorption) 2. Chemical binding

Physical adsorption 1. Occurs immediately on the surfaces by dipping into the biomolecular solutions (amino acids, peptides, proteins and blood components) 2. Adsorption is often reversible and the adsorbed species can easily be either washed away with fresh buffers or replaced by other molecules in solution. 3. Generally simple

Chemical binding 1. Covalent attachment of the target molecule to a solid surface 2. Experimentally more delicate, but the resultant irreversible binding with high levels of surface coverage makes this approach more popular.

Chemical binding 3. The bound molecule is much more stable under physiological conditions and is more resistant to disruption under harsh environmental conditions. 4. In some cases, chemical binding can alter the conformation or orientation of the biomolecule and disturb the bioactive center of the molecule, causing a reduction in activity
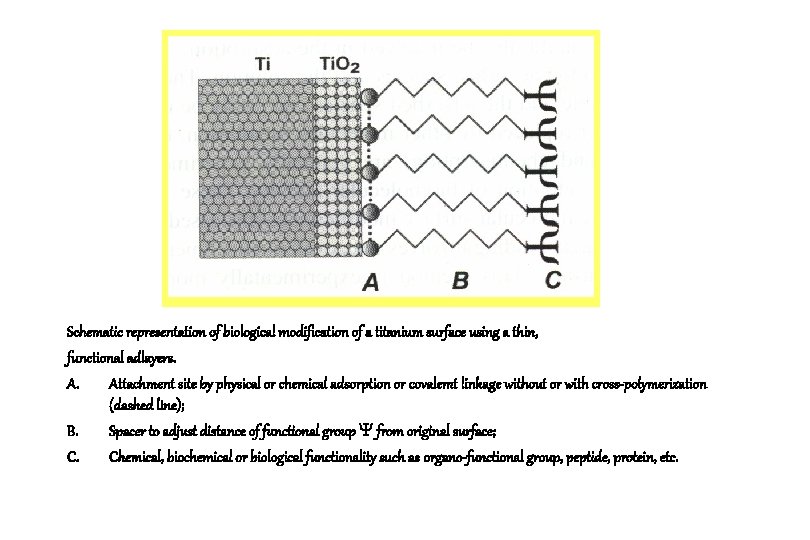
Schematic representation of biological modification of a titanium surface using a thin, functional adlayers. A. Attachment site by physical or chemical adsorption or covalemt linkage without or with cross-polymerization (dashed line); B. Spacer to adjust distance of functional group from original surface; C. Chemical, biochemical or biological functionality such as organo-functional group, peptide, protein, etc.

Reactivity of titanium for biomechanical modification 1. Since Ti. O 2 is a relatively inert surface, only a few organic reagents are able to form strong chemical bonds with its surface. 2. Examples of these reagents include organosilanes, organophosphate, and photosensitive chemicals.
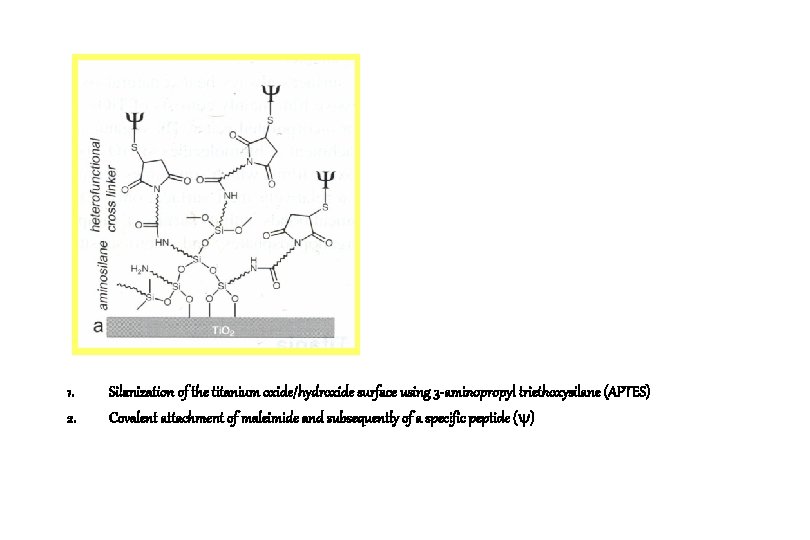
1. 2. Silanization of the titanium oxide/hydroxide surface using 3 -aminopropyl triethoxysilane (APTES) Covalent attachment of maleimide and subsequently of a specific peptide ( )
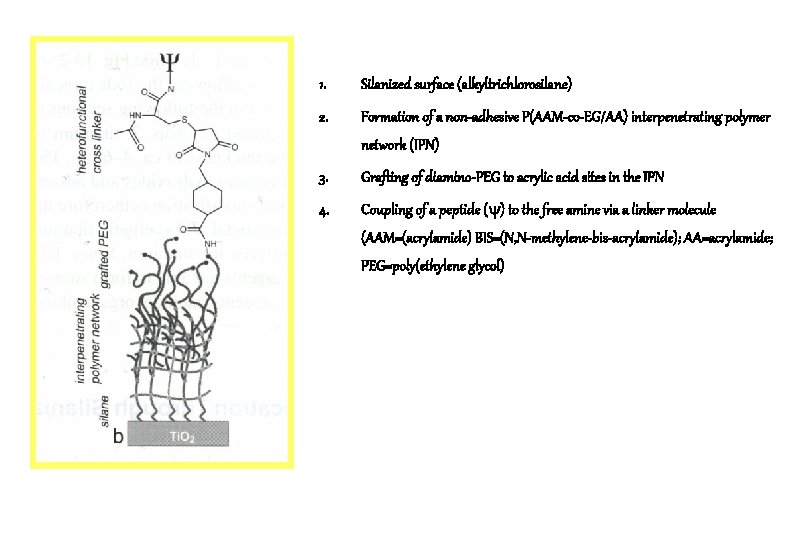
1. Silanized surface (alkyltrichlorosilane) 2. Formation of a non-adhesive P(AAM-co-EG/AA) interpenetrating polymer network (IPN) 3. Grafting of diamino-PEG to acrylic acid sites in the IPN 4. Coupling of a peptide ( ) to the free amine via a linker molecule (AAM=(acrylamide) BIS=(N, N-methylene-bis-acrylamide); AA=acrylamide; PEG=poly(ethylene glycol)
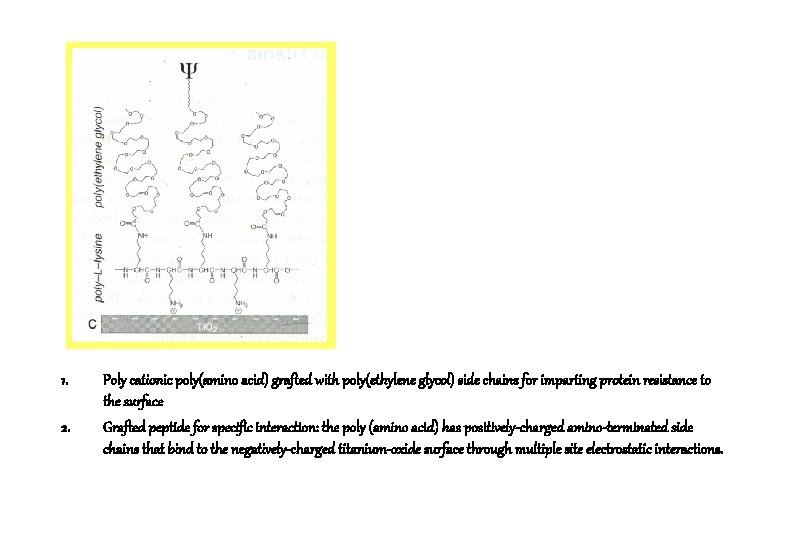
1. 2. Poly cationic poly(amino acid) grafted with poly(ethylene glycol) side chains for imparting protein resistance to the surface Grafted peptide for specific interaction: the poly (amino acid) has positively-charged amino-terminated side chains that bind to the negatively-charged titanium-oxide surface through multiple site electrostatic interactions.
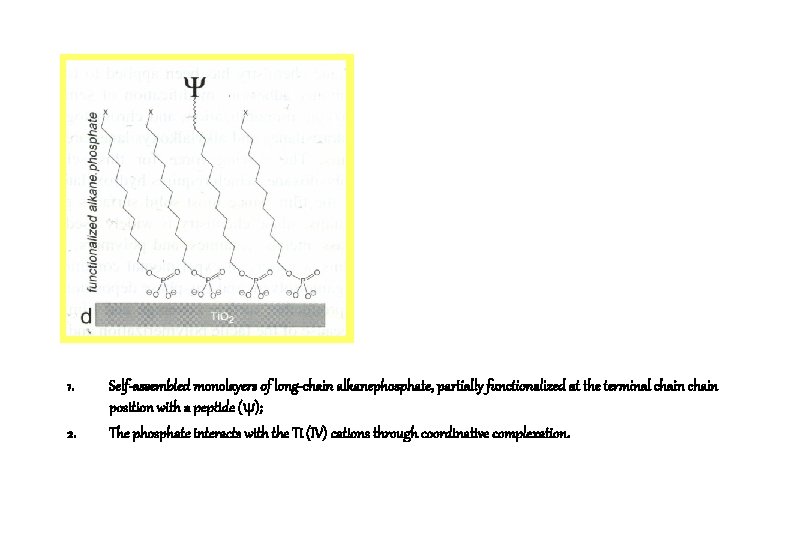
1. 2. Self-assembled monolayers of long-chain alkanephosphate, partially functionalized at the terminal chain position with a peptide ( ); The phosphate interacts with the Ti (IV) cations through coordinative complexation.

Modification Through Silanized Titania 1. Silane chemistry has been applied to titania surfaces for metal-metal and metal-polymer adhesion, modification of semiconductor Ti. O 2 films as electrodes, enzyme immobilization, and chromatography 2. Self-assembled films of alkylchlorosilanes and alkylalkoxysilanes are two of the most extensively studied systems.

Definition of Silane (coupling agent) Compounds of silicon and hydrogen of the formula Sin. H 2 n+2, and other monomeric silicon compounds have the ability to bond inorganic materials such as glass, mineral fillers, metals and metallic oxides to orgaic resins.

Properites of Silane for Silanization Due to the active reactivity of alkylchlorosilanes and alkylalkoxysilanes, a complete synthesis of compounds bearing both bioactive species and silanol groups is nearly impossible. Therefore the stepwise reaction is often used in silane chemistry
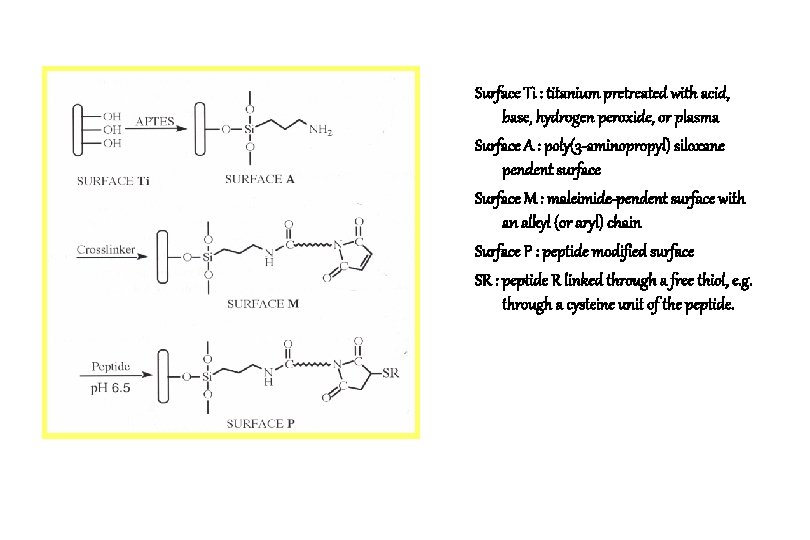
Surface Ti : titanium pretreated with acid, base, hydrogen peroxide, or plasma Surface A : poly(3 -aminopropyl) siloxane pendent surface Surface M : maleimide-pendent surface with an alkyl (or aryl) chain Surface P : peptide modified surface SR : peptide R linked through a free thiol, e. g. through a cysteine unit of the peptide.

Most Common Residues used for Chemical Modification 1. Thiol group of cysteine, 2. Amino groups of lysine and N-terminal residues 3. Carboxyl groups of glutamic acid, aspartic acid and C-terminal residues.

Thiol-Directed Immobilization 1. The thiol moeity, with the thiolate ion as the active species, is the most reactive functional group in a protein. 2. The thiol-reactive functional groups are primarily alkylating reagents, including alkyl halides, benzyl halides, -haloacetyls and maleimides

Definition of Thiol 1. Any of a class of sulfur-containing organic compounds with the formula, RSH, where R is an organic group. 2. Also called ( not in technical usage ) mercaptan.

Merits of Thiol-Directed Immobilization 1. Mild reaction conditions : Three-step reactions can be carried out at room temperature and in common organic solvents such as methanol and ethanol, as well as in aqueous buffer near neutral p. H 2. Higher surface coverage up to 0. 1 nmol/cm 2 compared to other reported method.

Merits of Thiol-Directed Immobilization 3. Specific binding : No polymerization reactions occur in the second step. The maleimide/thiol reaction is very fast, and in most cases, can be completed in just a few minutes.

Merits of Thiol-Directed Immobilization 4. Multiple choice : The technique is not limited to maleimide, and a variety of other crosslinking groups such as iodoacetyl, succinimidyl ester, benzophenone and aryl azide can be grafted to surface in the second step. Consequently, many kinds of biomolecules possessing different reactive residues can be immobilized.

Amino(-NH 2) Groups-Directed Immobilization 1. Because the amino acid is a strong nucleophile that is abundant and omnipresent in peptides and proteins, it is the most important target for chemical modification, particularly in cases where cysteine residues are absent. 2. Due to the relatively high p. Ka of the ammonium ion, most of the reagents that react with the amino group will also react with other functionalities

Amino (-NH 2) Groups-Directed Immobilization 3. Many stable acrylated products are formed only with the amino groups, providing a clear basis of selectivity. 4. Most common reactions of amines are alkylation and acrylation reactions. 5. Reagents such as N-succinimidyl ester, imidoester, glutaraldehyde and isocyanate react with amino groups.

Carboxyl (-COOH) Groups-Directed Immobilization 1. High frequency in natural proteins (Asp, Glu and C-terminus) and are typical reactive sites for attachment to surfaces. 2. The most important reagent for the chemical modification of carboxyl groups is carbodiimide.

Drawbacks of Carboxyl Groups-Directed Immobilization 1. Carboimide reacts not only with carboxylic acids but also with alcohols, amines, water and other nucleophiles. 2. Undergo hydrolysis slowly and may react with other unwanted nucleophilic residues to form different carboxylated derivatives. These reactions decrease the efficiency of coupling and may cause inactivation of the modified protein.
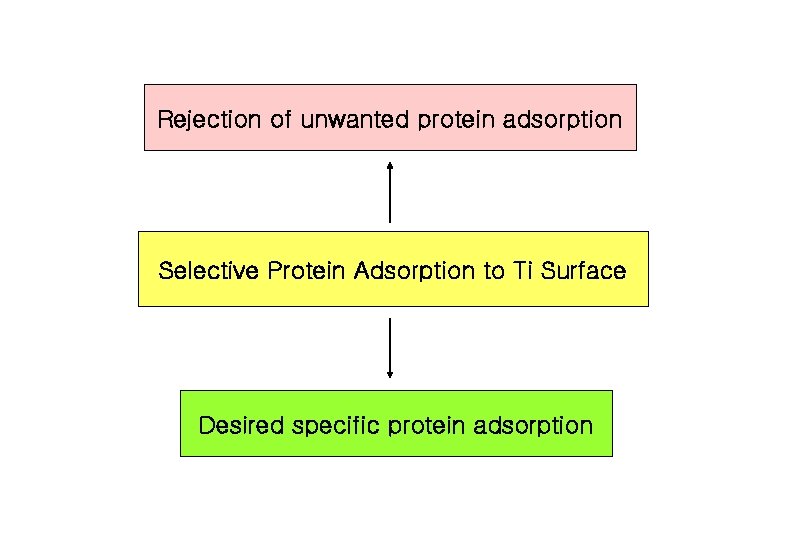
Rejection of unwanted protein adsorption Selective Protein Adsorption to Ti Surface Desired specific protein adsorption
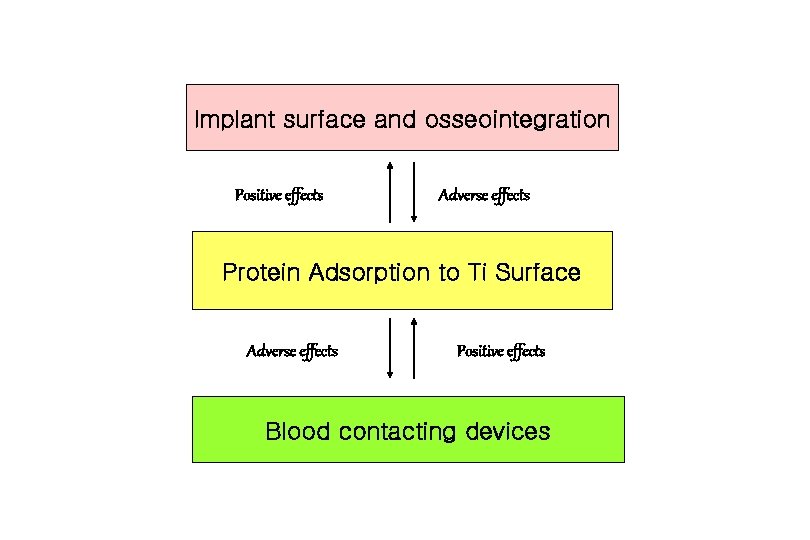
Implant surface and osseointegration Positive effects Adverse effects Protein Adsorption to Ti Surface Adverse effects Positive effects Blood contacting devices

Techniques for Specific Protein Adsorption 1. Blocking the other proteins by pre-adsorption with albumin (inert protein) 2. Surface immobilization by surface-chemical functioanlities

Protein Albumin and Unwanted Adsorption 1. Modification of titania surfaces within lanthanum ions increased the subsequent albumin adsorption roughly five-fold. 2. This enhanced adsorption of albumin was found to inhibit osseointegration-the integration of bone into implant. Ellingsen & Pinholt, J. Biomed. Mater. Res. 1995

Protein Albumin and Wanted Adsorption 1. The simplest approach to controlling protein adsorption at the titania interface is to passivate the surface with a layer of an inert protein such as albumin 2. By the blocking effect of inert albumin adsorption, subsequent proteins cannot attach.

Effects and Limits of Albumin Adsorption 1. The effectiveness of this method is limited by the fact that albumin binding to titania is often partially reversible, and thus subsequent proteins would be able to replace albumin within the protein layer.

Protein concentration and Ca-P depostion 1. Fibronectin concentration of 0. 01 mg/m. L had little effect ; but at 0. 05 mg/m. L concentration, calcium phosphate deposition onto the titanium oxide surface was strongly inhibited. 2. Fibrinogen in blood is found at a concentration level of approximately 0. 2 mg/m. L. 3. Preadsorption of fibronectin on the titanium surface was also found to inhibit the deposition process. do Serro et al. , J. Biomed. Mater. Res. 2000
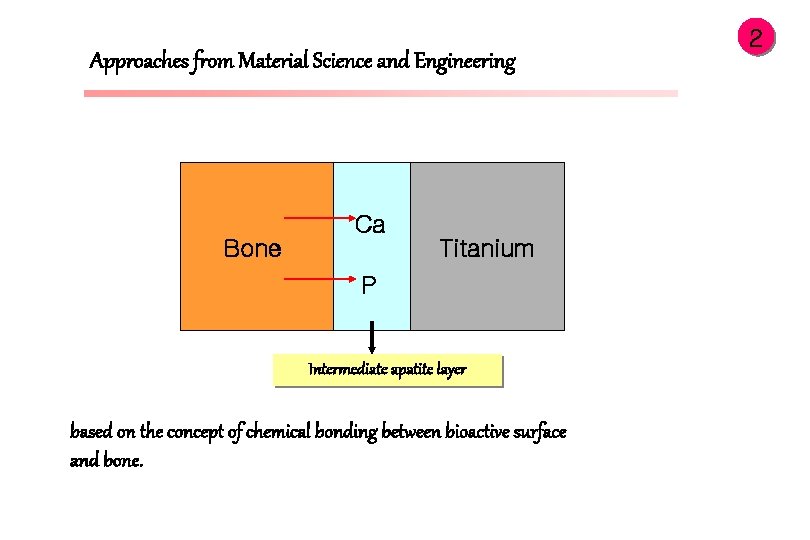
Approaches from Material Science and Engineering Ca Bone Titanium P Intermediate apatite layer based on the concept of chemical bonding between bioactive surface and bone. 2
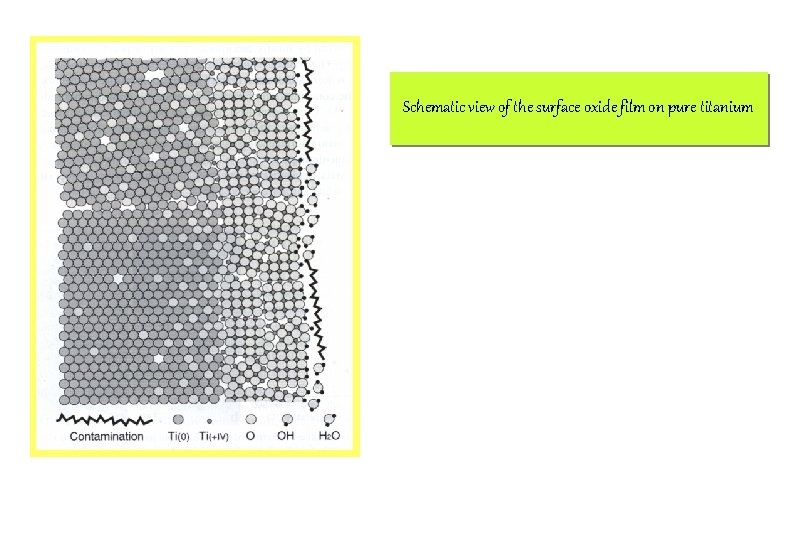
Schematic view of the surface oxide film on pure titanium

Early Interfacial Processes Taking Place at the Interface between the Titanium Oxide and Biological Fluid in Contact with the Implant Merit Dental Clinic Hur, Yin Shik, Periodontist

1 Hydroxylation and hydration of the titanium oxide film in contact with aqueous fluid
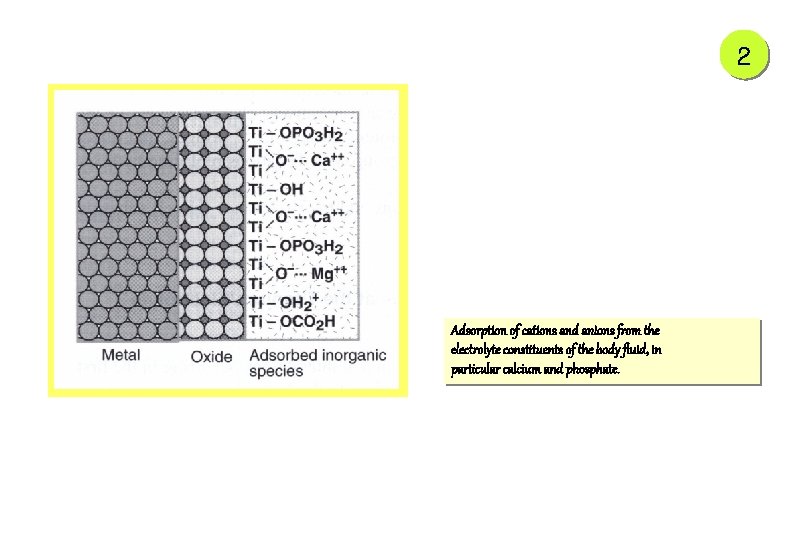
2 Adsorption of cations and anions from the electrolyte constituents of the body fluid, in particular calcium and phosphate.
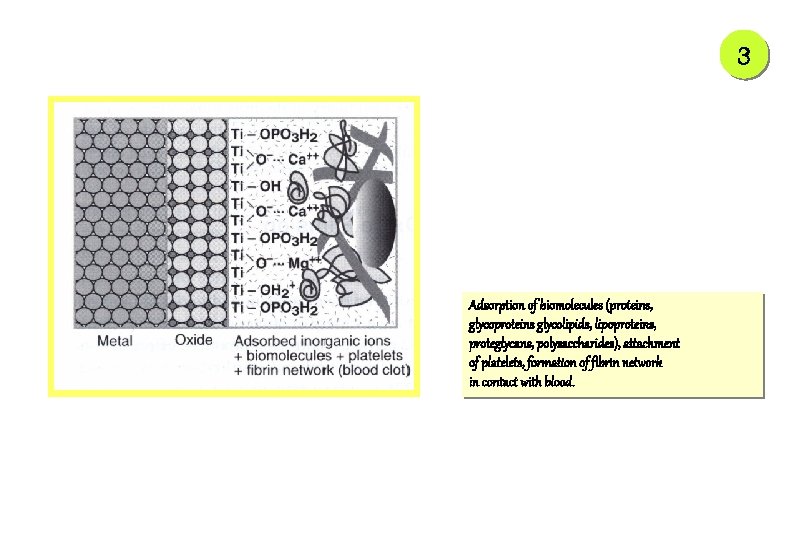
3 Adsorption of biomolecules (proteins, glycoproteins glycolipids, lipoproteins, proteglycans, polysaccharides), attachment of platelets, formation of fibrin network in contact with blood.

4 Further modifications of the interface : growth of the oxide/hydroxide layer (porous, gel-like outer layer) and precipitation of calcium phosphates containing also other species (magnessium, carbonate, fluoride, etc. ) present in the fluid. exchange reactions and reorganization within the proteinaceous film (“biofilm”)
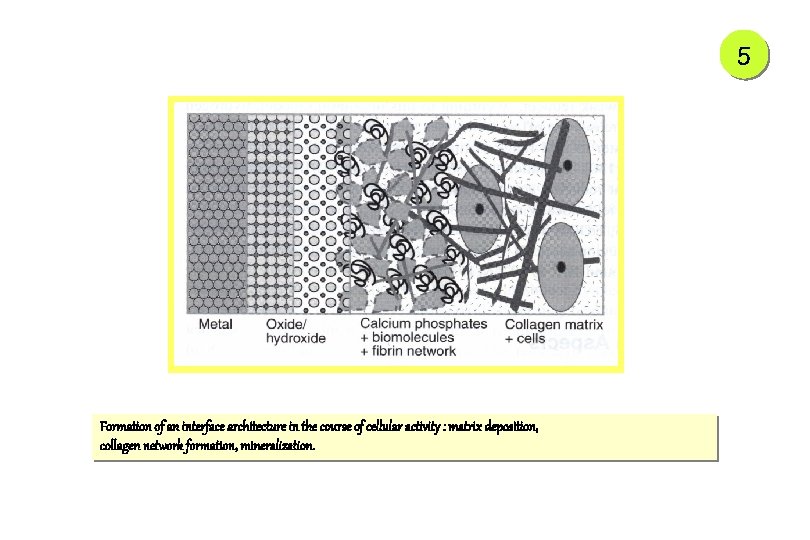
5 Formation of an interface architecture in the course of cellular activity : matrix deposition, collagen network formation, mineralization.

New Calcium Phosphate Coating Method on Titanium Implant Merit Dental Clinic Yin-Shik Hur, Periodontist

Why plasma sprayed hydroxyapatite coating method is declined ?
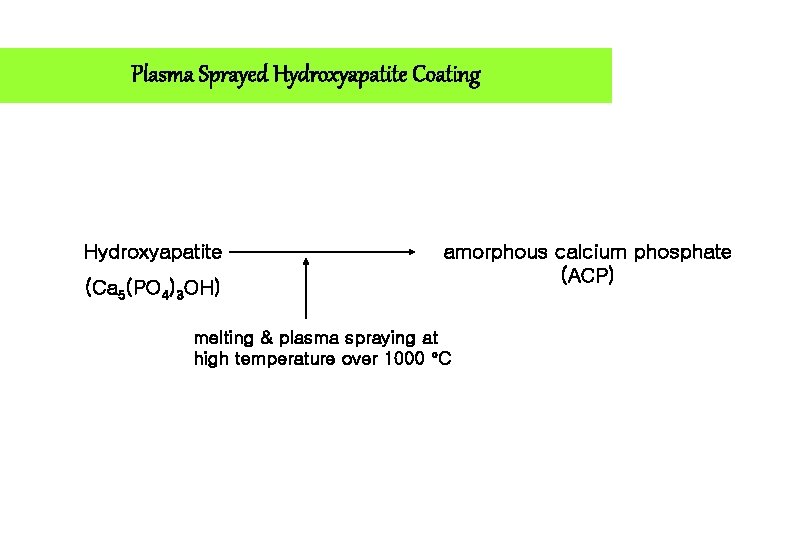
Plasma Sprayed Hydroxyapatite Coating Hydroxyapatite (Ca 5(PO 4)3 OH) amorphous calcium phosphate (ACP) melting & plasma spraying at high temperature over 1000 C
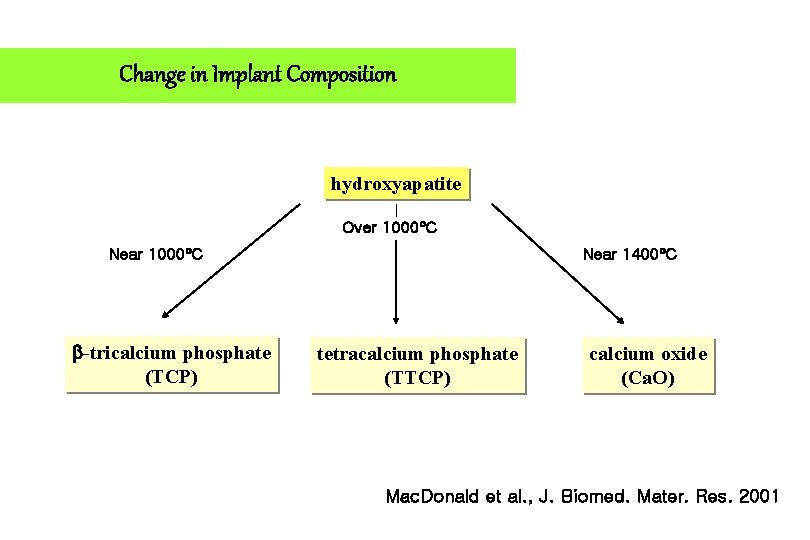
Change in Implant Composition hydroxyapatite Over 1000 C Near 1000 C -tricalcium phosphate (TCP) Near 1400 C tetracalcium phosphate (TTCP) calcium oxide (Ca. O) Mac. Donald et al. , J. Biomed. Mater. Res. 2001

Change in Implant Composition Therefore, HA coating after plasma spraying procedure is not longer HA, but contains non-HA constituents(TTCP, ACP and Ca. O). Mac. Donald et al. , J. Biomed. Mater. Res. 2001

Less Crystallinity and Coating Dissolution Constituents of less crystallinity (ACP, TTCP, TCP and Ca. O) within th e c o a tin g w h ic h d e m o n s tra te a g re a te r p re d is p o s itio n to w a rd d is s o lu tio n w ill p a s s in to th e b io lo g ic a l e n v iro n m e n t to a g re a te r degree than more highly crystalline phases, most notably HA. Mac. Donald et al. , J. Biomed. Mater. Res. 2001

Results of non-HA constituents solubilization 1. Resultant microvoids develop between the HA splats 2. Loss of these soluble Ca. P phases may potentially compromise the integrity of the remaining coating 3. Solubilized Ca. P inorganics potentially reprecipitating, 4. The coating interacting with bone and proteins facilitating the formation and entry of bone into these microvoids. Mac. Donald et al. , J. Biomed. Mater. Res. 2001

Results of non-HA constituents solubilization 5. As a result of coating disintegration and fragmentation within the biological environment, these HA particles may incorporate into the surrounding bone or potentially stimulate an inflammatory reaction with localized bone loss. Mac. Donald et al. , JBMR 2001

Current and future perspectives of Ca-P coating 1. 2. 3. 4. 5. 6. Sol-gel coating Electrophoretic deposition Pulsed laser deposition Alkali and heat treatment Biomimetic deposition Sputter deposition

Sol-gel Technology for Bioactive Surface Merit Dental Clinic Yin-Shik Hur, Periodontist

What it means, Sol-gel ? 1. A sol is a colloidal suspension of solid particles in a liquid; a gel is a substance that contains a continuous solid skeleton enclosing a continuous liquid phase. 2. In a sol-gel process, a ceramic is built starting from its molecular components in solution by a carefully controlled condensation reaction.

What it means, Colloid ? 1. A colloid is a suspension in which the dispersed phase is so small (about 1 -1000 nm) that gravitational forces are negligible and interactions dominated by short range forces such as van der Waals attraction and surface charge.

Molecular Mixture of Precursors in Sol-gel Process 1. A high degree of homogeneity in the film can be expected. 2. This intimate mixture of the coating components allows lower processing temperatures to be applied during sintering. 3. This can be important for coating compositions, which easily decompose or otherwise undergo undesired phase transitions or possess volatile components.

Sol-gel Process 1. Inorganic polymerization procedure based on the hydrolysis and condensation reactions of metal alkoxides. 2. Conventional sol-gel precursor solutions often contain metal alkoxides diluted in ethanol, water and acid. Vititala et al. , Thin Solid Films 2000

Sol-gel Process 3. Titanium alkoxides are known to react readily with water. 4. Solution composition and preparation procedures play a key role in the sol-gel manufacturing of titania thin films. Vititala et al. , Thin Solid Films 2000

Advantages of Sol-gel Process 1. Better control of the chemical composition of the coating 2. Preparation of homogenous films 3. Reduction of densification temperature of the ceramic 4. Control of the film microstructure 5. Considerably less equipment 6. Potentially less expensive than many of the alternative coating techniques

Applications of Sol-gel Technology for Surface Treatment 1. Sol-gel titania coatings 2. Ca-P coatings produced by the sol-gel route 3. Ti. O 2 -Ca. P composite coating

1 Sol-gel Titania Surface Merit Dental Clinic Hur, Yin Shik, Periodontist

Five Steps in Sol-gel Process 1. Hydrolysis and polycondensation 2. Gelation 3. Aging 4. Drying 5. Densification and crystallization

Sol-gel Processed Titania 1. Hydroxy(OH) groups present at the surface of titanium implants played a key role in biocompatibility aspects by favoring osseointegration 2. Sol-gel titania, which is rich in Ti. OH groups, should induce calcium phosphate formation and may therefore be able to contribute to an enhanced “bonding” to bone. Titanium in medicine 2001

Apatite formation by Sol-gel method and bone-apatite The thickness of the layer, presenting a crystallinity very similar to that of bone apatite, may reach up to 10 m after two weeks in SBF. Li et al. J. Am. Ceram. Soc. 1994

Apatite precipitation on sol-gel derived coatings after 2 weeks in SBF Li et al. , J. Am. Ceram. Soc. 1994

Sintering temperature on apatite precipitation 1. An optimum was found for coatings treated at 500 C. 2. Above 500 C it is supposed that the amount of Ti. OH required for calcium phosphate formation decreases as a consequence of dehydration. Peltola et al. J. Biomed. Mater. Res. 1998

2 Ca-P Sol-gel Coated Surface Merit Dental Clinic Hur, Yin Shik, Periodontist

Processing Routes for Sol-gel Ca-P Coatings 1. Phenyldichlorophosphine (C 6 H 5 PCl 2)+ calcium nitrate(Ca(NO 3)2) 2. Partially hydrolyzed phosphoric acid esters + calcium nitrate, calcium glycoxide, calcium acetate 3. Triethyl phosphite, P(OEt)3 + calcium diethoxide, Ca(OEt)2 Titanium in Medicine, 2001

Modification for Hydroxyapatite/Fluoroapatite Mixture 1. One part of calcium nitrate, Ca(NO 3)2 is replaced by calcium tri-fluoroacetate, Ca(CF 3 COO)2. The stoichiometry of the precursor exactly matches the stoichiometry of the pure ceramic. Ca: P ratio is set to 1. 67. Titanium in Medicine, 2001

Sintering temperature and Crystalline phase 1. The crystalline HA phase builds up between 350 C and 400 C. 2. It remains pure even after the ceramic is sintered at a higher temperature, between 900 C and 1000 C. 3. The absence of additional phases such as -tricalcium phosphate, tetracalcium phosphate or calcium oxide in this temperature region is the sign that the stoichiometry of HA is respected. Titanium in Medicine, 2001

3 Ti. O 2 - Ca. P Sol-gel Coated Surface Merit Dental Clinic Hur, Yin Shik, Periodontist

Advantages of Ti. O 2 –Ca. P Coatings by Sol-gel Routes 1. Calcium phosphate coatings are bioactive but present a poor adhesion strength to the substrate. 2. Ti. O 2 coatings strongly adhere to titanium but their bioactivity is limited. Titanium in Medicine, 2001

Advantages of Ti. O 2 –Ca. P Coatings by Sol-gel Routes 3. The idea behind the use of Ti. O 2 – Ca. P is to combine the advantages of both materials : the adhesion strength of the titanium dioxide on the substrate and the bioactivity of the calcium phosphate. Titanium in Medicine, 2001

Advantages of Ti. O 2 –Ca. P Coatings by Sol-gel Routes 4. The use of an amorphous mixture of titanium, calcium and phosphorus allows a better control of the crystalline and chemical composition of the layer. Titanium in Medicine, 2001

Advantages of Ti. O 2 –Ca. P Coatings by Sol-gel Routes 5. By changing the Ti: Ca ratio of the precursor between the deposition of each layer, it is possible to introduce a concentration gradient inside of the coating with the calcium phosphate concentration increasing towards the coating/bone interface. Titanium in Medicine, 2001

4 Organic- Inorganic Sol-gel Coated Surface Merit Dental Clinic Hur, Yin Shik, Periodontist

Synthesis of Hybrid Organic-Inorganic Compounds 1. Organic molecules with a simple or more complex(e. g. protein) structure or even living organisms are mixed with the liquid precursor and finally entrapped in the oxide matrix. 2. The entrapped enzyme or organism can communicate with outer world and act on it.

Synthesis of Hybrid Organic-Inorganic Compounds 3. While classical ceramics fabrication relies on high temperature processing, sol-gel chemistry is performed at room temperature.

Synthesis of Hybrid Organic-Inorganic Compounds 4. By carefully controlling the solvent polarity and p. H during the synthesis, it is possible to retain the structural and chemical properties and activity of such sensitive molecules or living organisms (bacteria or cells) and to host them in an inert, protective oxide matrix.

Various Sol-gel Processes 1. Dip-coating method 2. Spin-coating method 3. Aerosol-gel method

Dip-Coating Methods 1. Based on linear spreading of the sol on the substrate 2. The film thickness is essentially controlled by the solution viscosity and the linear speed of the substrate. Vititala et al. , Thin Solid Films 2000
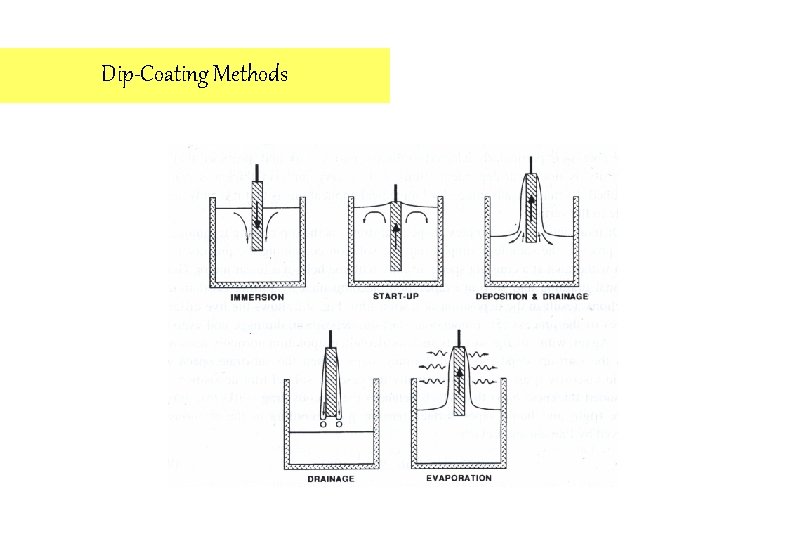
Dip-Coating Methods

Sol-gel Preparation by Dip-coating 1. Viscosity of precursor : 1. 95 10 -2 Pa s, measured at 26ºC. 2. Withdrawal speed : Thin (1 m) coating: 0. 57± 0. 05 mm/s Thick(5 m) coating: 4. 26± 0. 05 mm/s Piveteau et al. , Biomaterials 2000

Dip-Coating Methods 3. For complex shaped substrates 4. Gravitational drainage and solvent evaporation, accompanied by further condensation reactions, result in the deposition of a solid film. Titanium in medicine 2001

Spin-Coating Methods 1. Based on centrifugal spreading of the sol on the substrate 2. The film thickness is essentially controlled by the solution viscosity and the centrifugal speed of the substrate. Vititala et al. , Thin Solid Films 2000

Spin-Coating Methods 3. Its main disadvantage for biomedical applications is that it is only applicable to flat surfaces. Titanium in medicine 2001

Sol-gel Preparation by Spin-coating 1. Single angular velocity : 2800 r. p. m. 2. Viscosity of precursor : measured at 26ºC. Thin (1 m) coating: 3. 52 10 -3 Pa s, Thick(5 m) coating: 1. 95 10 -2 Pa s, Piveteau et al. , Biomaterials 2000
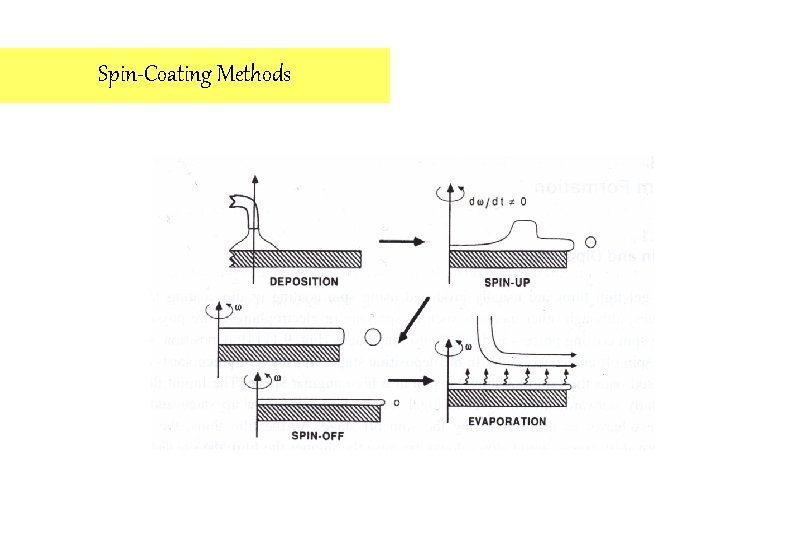
Spin-Coating Methods

Aerosol-gel Techniques 1. Based on the sol-gel polymerization of a liquid film deposited from an ultrasonically generated aerosol. 2. This process displays several advantages compared to conventional sol-gel deposition techniques. Vititala et al. , Thin Solid Films 2000
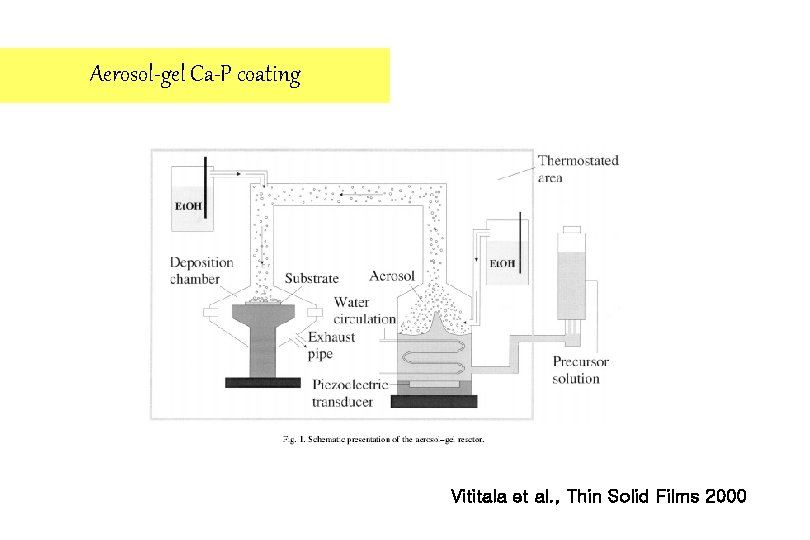
Aerosol-gel Ca-P coating Vititala et al. , Thin Solid Films 2000

Materials of Aerosol-gel Techniques 1. Titanium i-propoxide {Ti(OC 3 H 7)4}(TPT) solutions 2. Dopant precursor : Calcium nitrate {Ca(NO 3)2· 4 H 2 O } and triethylphosphate {PO(OC 2 H 5)3}(TEP) 3. After film deposition, thermally treated in preheated furnace for 10 min at 80 -500ºC 4. Deposition time 10 -50 s Film thickness 20 -100 nm Vititala et al. , Thin Solid Films 2000

Advantages of Aerosol-gel Techniques 1. Due to aerosol it is possible to coat substrates with complex shapes. 2. The layer thickness can be accurately controlled by variation of several experimental parameters, including dilution of the precursor, applied ultrasonic excitation power, deposition duration and carrier gas flow rate. Vititala et al. , Thin Solid Films 2000

Advantages of Aerosol-gel Techniques 3. A suitable adjustment of the substrate temperature enables the manufacturing of films with different morphologies, ranging from smooth optical quality layers to rough or non-continuous coatings. 4. From an industrial point of view, the aerosol-gel process is fully compatible with inline technologies for highly efficient production. Vititala et al. , Thin Solid Films 2000

Importance of Hydroxyl Groups on Surface Technology Merit Dental Clinic Hur, Yin Shik, Periodontist

Apatite formation on hydrated titania 1. Calcium ions are bound to be caught around the titania surface of negative charges, because of Coulomb’s force. 2. A hydroxyl group in the titania may catch the phosphate groups through a hydrogen bond and cause them to concentrate around the surface. Li et al. , J. Am. Ceram. Soc. 1994

Apatite formation on hydrated titania 3. The enrichment of both Ca and P increases the local supersaturation degree with respect to hydroxyapatite. 4. Apatite nuclei are initiated once the supersaturation rises over the critical level necessary for heterogenous nucleation of apatite Li et al. , J. Am. Ceram. Soc. 1994

The role of hydrated silica, titania, and alumina in inducing apatite on implants Panjian Li, Chikara Ohtsuki, Tadashi Kokubo, Kazuki Nakanishi, Naoshiro Soga, and Klaas de Groot J. Biomed. Mater. Res. 1994
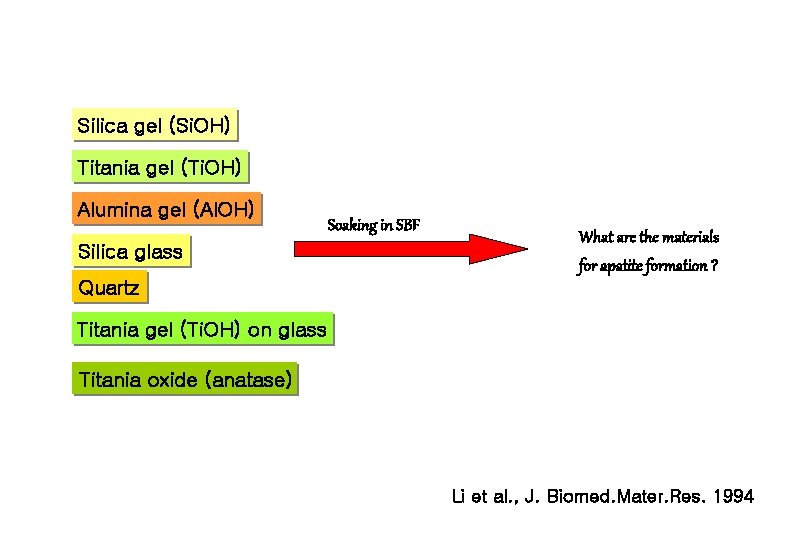
Silica gel (Si. OH) Titania gel (Ti. OH) Alumina gel (Al. OH) Silica glass Quartz Soaking in SBF What are the materials for apatite formation ? Titania gel (Ti. OH) on glass Titania oxide (anatase) Li et al. , J. Biomed. Mater. Res. 1994
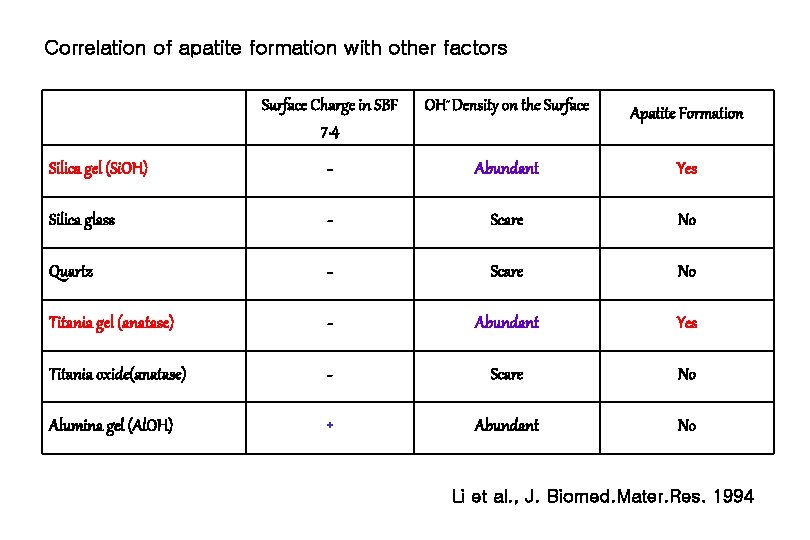
Correlation of apatite formation with other factors Surface Charge in SBF 7. 4 OH- Density on the Surface Apatite Formation Silica gel (Si. OH) - Abundant Yes Silica glass - Scare No Quartz - Scare No Titania gel (anatase) - Abundant Yes Titania oxide(anatase) - Scare No Alumina gel (Al. OH) + Abundant No Li et al. , J. Biomed. Mater. Res. 1994

The Point of Zero Charge (pzc) of an Oxide 1. 2. 3. 4. Defined as the p. H at which the surface charge of oxide is zero. The pzc of pure silica gel : p. H 2 The pzc of titania gel : p. H 6. 8 The pzc of Alumina gel : p. H 7. 8 It can be expected that alumina gel is charged positively at the surface in SBF with p. H 7. 4, while silica gel and titania gel are negatively charged. Li et al. , J. Biomed. Mater. Res. 1994

Successful Apatite Inducers 1. Negatively charged surfaces 2. Abundant OH groups … in physiologically-related fluid. Li et al. , J. Biomed. Mater. Res. 1994

Substrate Conditions for sol-gel coating 1. Mirror-like polished surface (grinding with 220 grains, grinding with 400 grains, brushing and stuff grinding) 2. Mechanically polished surface (polished in bonded abrasive, cylinder-shaped products made of ceramics) 3. Alumina-blasted surface (blasted with alumina particles with sharp edges, size between 0. 06 and 0. 1 mm) Piveteau et al. , Biomaterials 2000

Adhesion, Coating thickness, Roughness 1. For all surface pre-treatment, the tensile strength was reduced by 20 -40% for a thick coating. 2. Alumina-blasted samples showed a 30 -50% higher adhesion strength. Piveteau et al. , Biomaterials 2000

Combination of Alkali Treatment and Heat Treatment for Ca-P Deposition Merit Dental Clinic Yin-Shik Hur, Periodontist

The effect of heat treatment on bone-forming ability of alkali-treated titanium Shigeru Nishiguchi, Takashi Nakamura, Masahiko Kobayashi, Hyun-Min Kim, Fumiaki Miyaji, Todashi Kokubo Biomaterials 1999
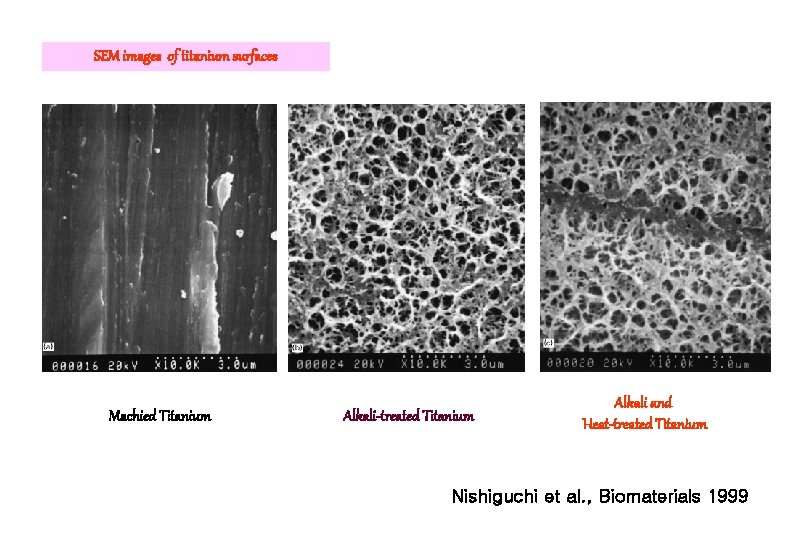
SEM images of titanium surfaces Machied Titanium Alkali-treated Titanium Alkali and Heat-treated Titanium Nishiguchi et al. , Biomaterials 1999

Machied Cp titanium Alkali and Heat-treated Titanium Apatite formed titanium Nashio et al. , J. Biomed. Mater. Res. 2000
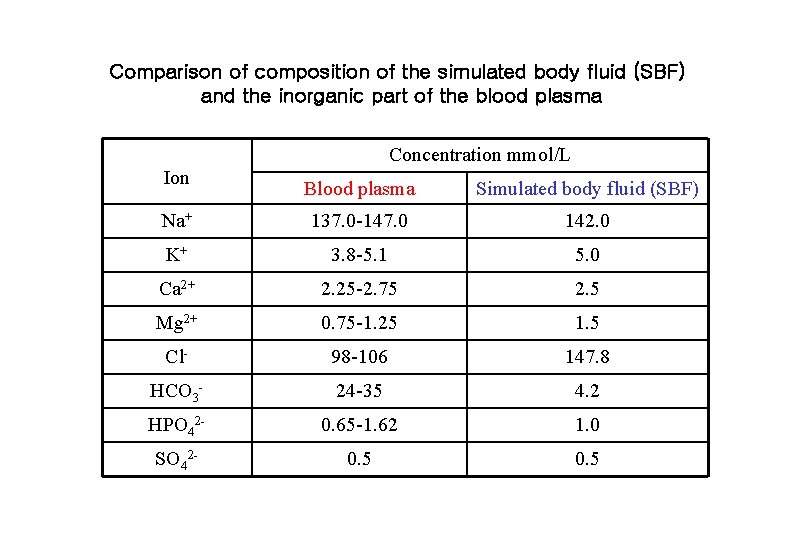
Comparison of composition of the simulated body fluid (SBF) and the inorganic part of the blood plasma Concentration mmol/L Ion Blood plasma Simulated body fluid (SBF) Na+ 137. 0 -147. 0 142. 0 K+ 3. 8 -5. 1 5. 0 Ca 2+ 2. 25 -2. 75 2. 5 Mg 2+ 0. 75 -1. 25 1. 5 Cl- 98 -106 147. 8 HCO 3 - 24 -35 4. 2 HPO 42 - 0. 65 -1. 62 1. 0 SO 42 - 0. 5

Surface structural and morphologic feature of the surface of Ti metal subjected to alkali and heat treatments and the subsequent soaking in SBF for 4 weeks. (SEM image) Yan et al. , J. Biomed. Mater. Res. 1997

Procedure for Alkali and Heat Treatment 1. Pure titanium plates were soaked in 5 mol Na. OH aqueous solution at 60ºC for 24 h. 2. The plates were washed gently with acetone and distilled water, and dried at 40ºC for 24 h in an air atmosphere. 3. The plates were heated to 600 ºC at a rate of 5 ºC min-1 in an electric furnace, kept at a given temperature for 1 h. Nishiguchi et al. , Biomaterials 1999
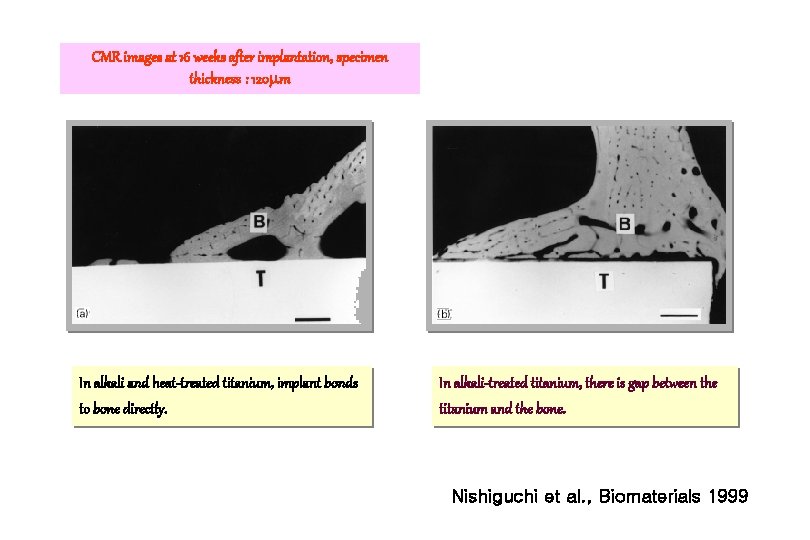
CMR images at 16 weeks after implantation, specimen thickness : 120 m In alkali and heat-treated titanium, implant bonds to bone directly. In alkali-treated titanium, there is gap between the titanium and the bone. Nishiguchi et al. , Biomaterials 1999
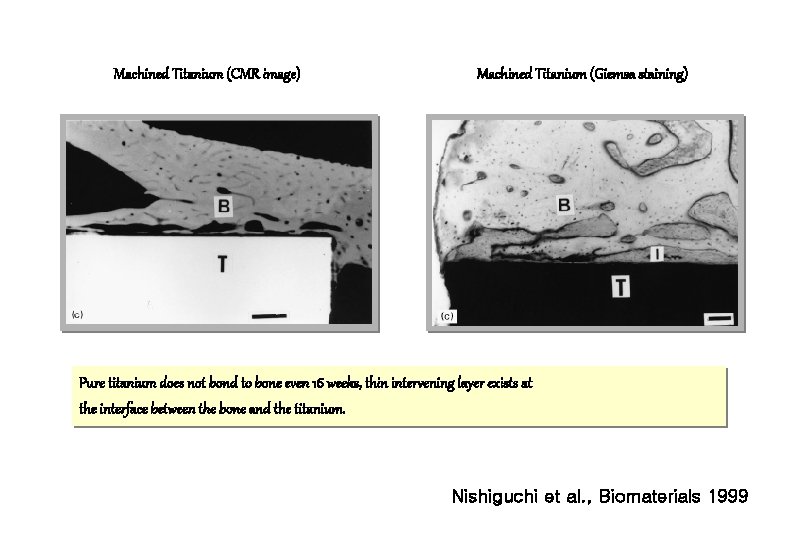
Machined Titanium (CMR image) Machined Titanium (Giemsa staining) Pure titanium does not bond to bone even 16 weeks, thin intervening layer exists at the interface between the bone and the titanium. Nishiguchi et al. , Biomaterials 1999
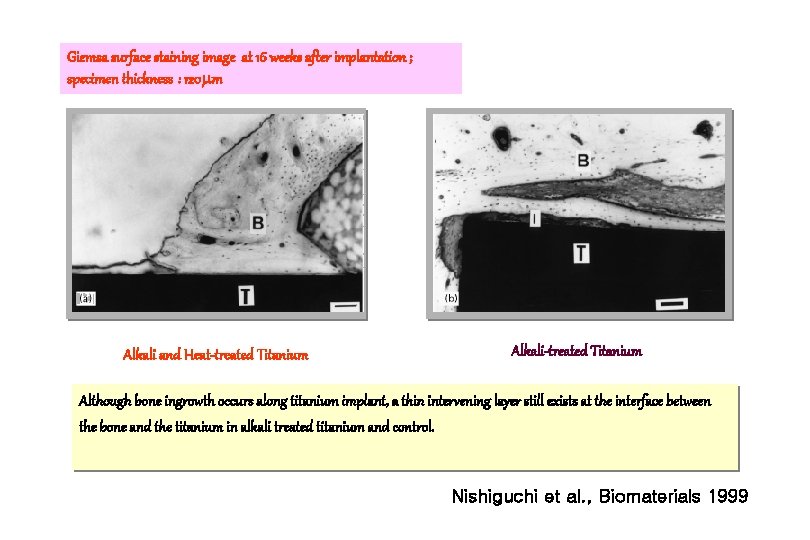
Giemsa surface staining image at 16 weeks after implantation ; specimen thickness : 120 m Alkali and Heat-treated Titanium Alkali-treated Titanium Although bone ingrowth occurs along titanium implant, a thin intervening layer still exists at the interface between the bone and the titanium in alkali treated titanium and control. Nishiguchi et al. , Biomaterials 1999

Alkali Titanate Hydrogel Layer 1. Titanium forms an alkali titanate hydrogel layer on its surface when soaked in an alkali solution. 2. This layer is unstable as to lose its apatite-forming ability during preservation or implantation. 3. Kim et al. (1997) recommended that heat treatment should be added to form a more stable alkali titanate layer. Nishiguchi et al. , Biomaterials 1999
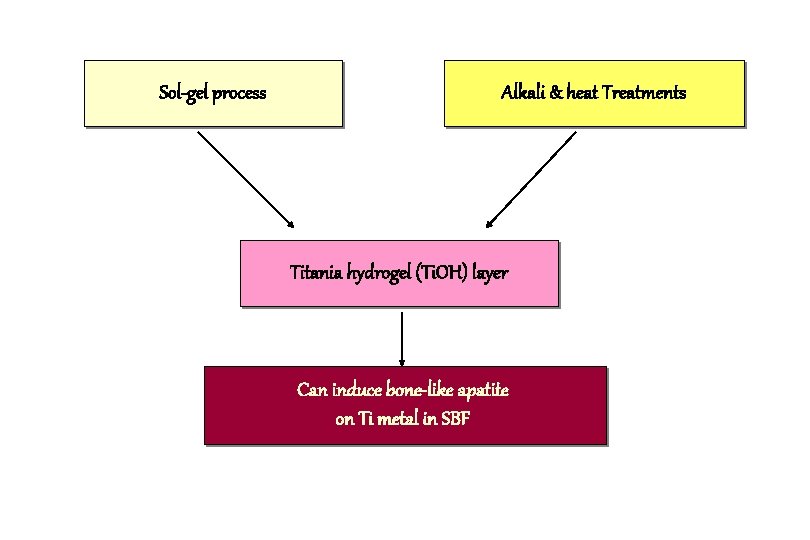
Sol-gel process Alkali & heat Treatments Titania hydrogel (Ti. OH) layer Can induce bone-like apatite on Ti metal in SBF
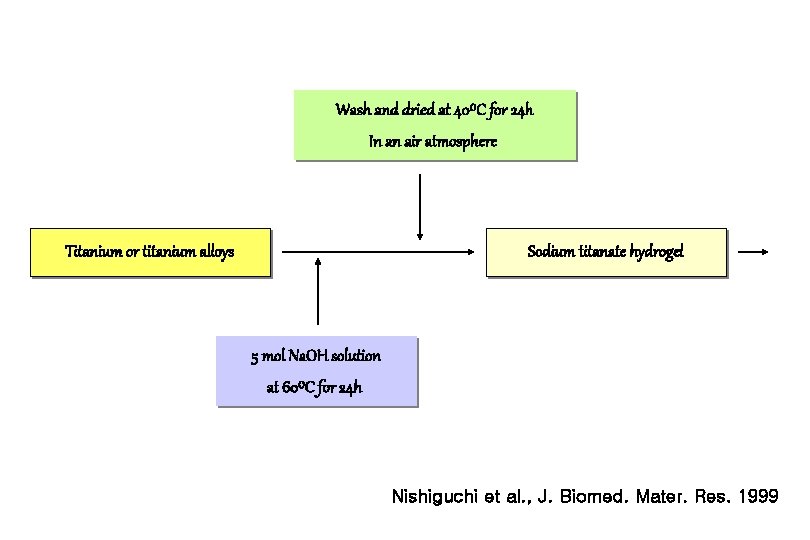
Wash and dried at 40⁰C for 24 h In an air atmosphere Titanium or titanium alloys Sodium titanate hydrogel 5 mol Na. OH solution at 60⁰C for 24 h Nishiguchi et al. , J. Biomed. Mater. Res. 1999
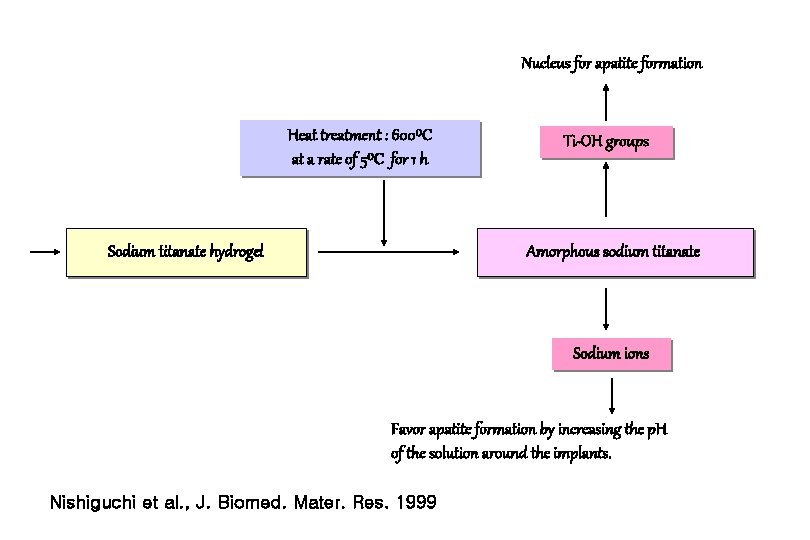
Nucleus for apatite formation Heat treatment : 600⁰C at a rate of 5⁰C for 1 h Sodium titanate hydrogel Ti-OH groups Amorphous sodium titanate Sodium ions Favor apatite formation by increasing the p. H of the solution around the implants. Nishiguchi et al. , J. Biomed. Mater. Res. 1999
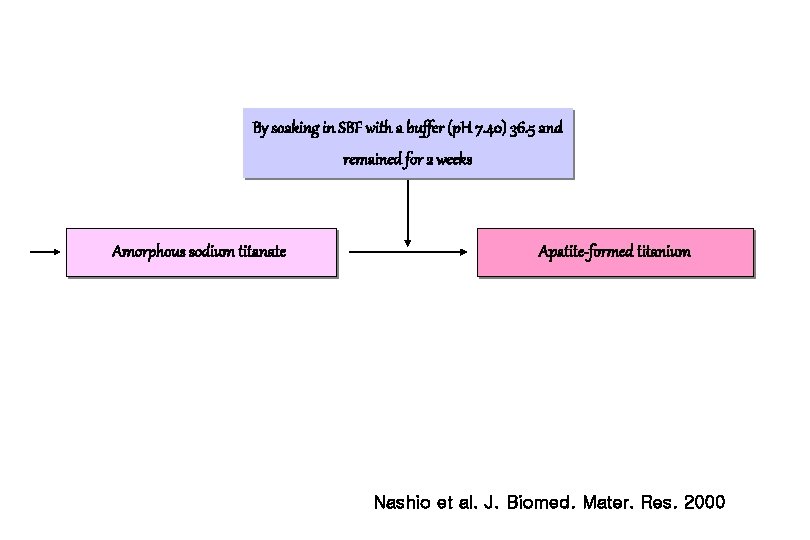
By soaking in SBF with a buffer (p. H 7. 40) 36. 5 and remained for 2 weeks Amorphous sodium titanate Apatite-formed titanium Nashio et al. J. Biomed. Mater. Res. 2000

Conclusion of Nishiguchi et al. (1999) 1. Although alkali-treated titanium has the ability to form apatite in SBF, it dose not serve as a bone-bonding material in practical use. 2. Both alkali and heat treatments are essential for inducing bioactivity in titanium. Nishiguchi et al. , Biomaterials 1999

Conclusion of Nishiguchi et al. (1999) 3. In our study we measured the tensile strength, which is less affected by surface roughness. Also, alkali-treated samples had almost the same surface topography. 4. Thus it is supposed that chemical bonding between titanium and bone via an apatite layer plays a major role in bone-bonding behavoir Nishiguchi et al. , Biomaterials 1999

Conclusion of Nishiguchi et al. (1999) 1. Alkali-and heat treated Ti 6 Al 4 V may be more useful as a bioactive metal than alkaliand heat treated pure titanium, due to its enhanced mechanical properties. 2. T i 15 Mo 5 Zr 3 Al may be more useful rather than Ti 6 Al 4 V because of low elastic modulus and lack of vanadium. Nishiguchi et al. , J. Biomed. Mater. Res. 1999

Advantages of Alkali and Heat Treatment over Plasma-Spray Coating 1. Can be applied uniformly on complicated surface texture. 2. Since reactive layer is as thin as 1 m, can be applied without changing existing implant design. 3. Simple and econnomical compared with the H plasma-spray technique. Nishiguchi et al. , Biomaterials 1999

TPS control AW-glass ceramic TPS AH-TPS Nishiguchi et al. , J. Biomed. Mater. Res. 2001

TPS control and AW-GC coated TPS implant : bone ingrowth into pores was observed, but intervening fibrous tissue usually existed at the interface between the bone and the titanium. Alkali and heat treated TPS implant : direct bonding between the bone and the titanium. Nishiguchi et al. , J. Biomed. Mater. Res. 2001

Combination of Alkali and Heat Treatment on the TPS surface 1. Alkali and heat treatments on porous titanium (TPS) implants may provide earlier stabilization and better clinical results of porous titanium (TPS) implants. 2. Alkali and heat treatments do not reduce the pore space available for bone ingrowth, because they cause little change in the surface morphology of the implant, affecting only the top 1 m of the surface. Nishiguchi et al. , J. Biomed. Mater. Res. 2001

Combination of Alkali and Heat Treatment on the TPS surface 3. Direct bonding between bone and the titanium implant obtained by alkali and heat treatments may prevent the invasion of fibrous tissue, metal particles, subsequent osteolysis. 4. Their effects extend all over the irregular surface of the implant, because the implants are immersed into Na. OH solution. Nishiguchi et al. , J. Biomed. Mater. Res. 2001

Fluoride-Modified Titanium Implant Merit Dental Clinic Yin-Shik Hur, Periodontist

“Rough is not Enough”, Enhanced Bone Integration by Surface Modified Titanium • EAO 2002 in Brussel • Jan Eirik Ellingsen • Oral Research Laboratry and Department of Prosthetic Dentistry and • Oral Function • Dental Facultu • University of Oslo • Norway

Effects of fluoride in bone 1. 2. 3. 4. 5. 6. Interacts with hydroxyapatite to form fluoridated hydroxyapatite and fluoroapatite Stimulates osteoprogenitor cells in vitro Increases trabecular bone density Induces calcification of bone Increases the alkaline phosphatase activity Reduces the adsorption of proteoglycans and glycosaminoglycans to HAp Jan Eirik Ellingsen, EAO, 2002
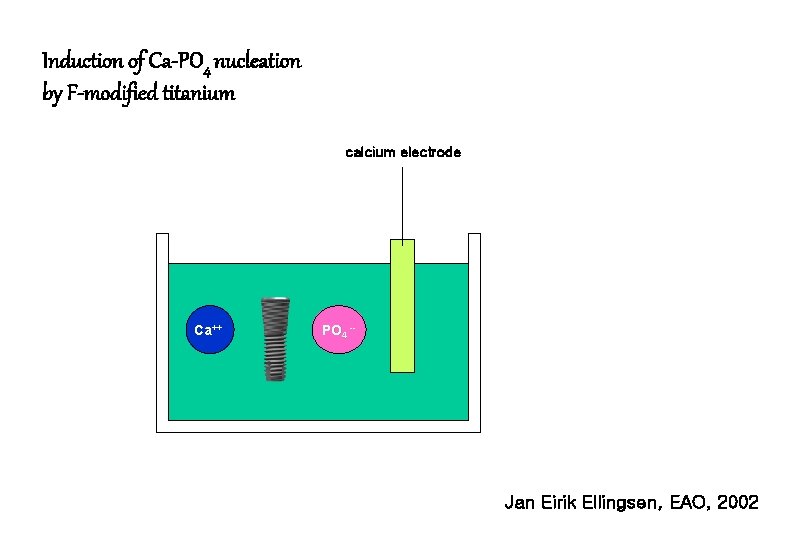
Induction of Ca-PO 4 nucleation by F-modified titanium calcium electrode Ca++ PO 4 -- Jan Eirik Ellingsen, EAO, 2002
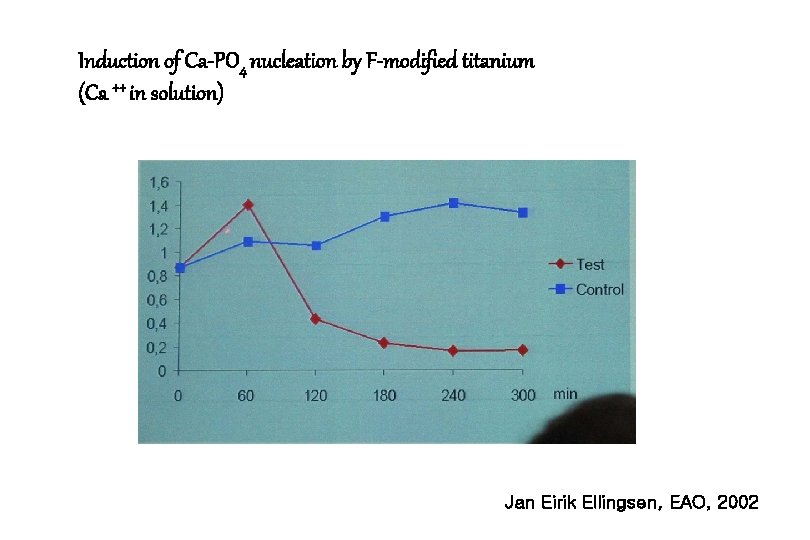
Induction of Ca-PO 4 nucleation by F-modified titanium (Ca ++ in solution) Jan Eirik Ellingsen, EAO, 2002
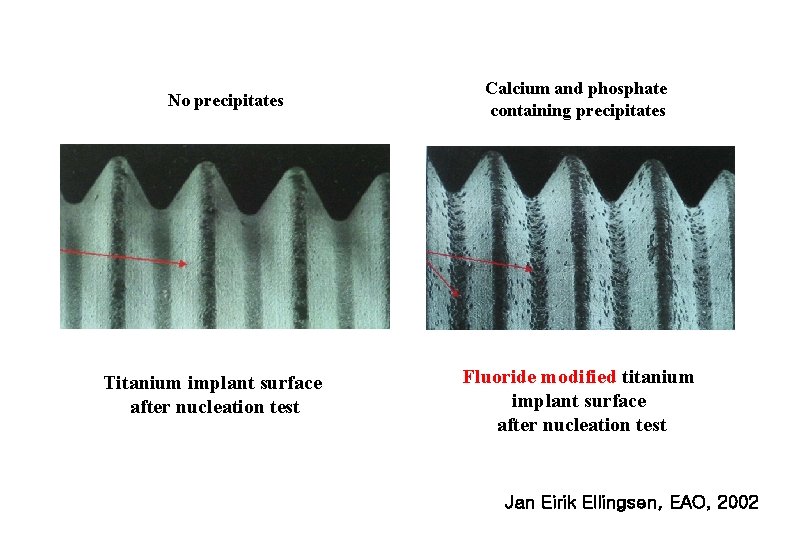
No precipitates Titanium implant surface after nucleation test Calcium and phosphate containing precipitates Fluoride modified titanium implant surface after nucleation test Jan Eirik Ellingsen, EAO, 2002

In vivo study 1. Removal torque test 2. Mean % bone to implant contact (1 month, 3 months) 3. Mean % bone area (1 month, 3 months) 4. Shear strength (3 months) 거의 모든 항목에서 F-modified implant가 기존 blasted implant 보다 월등히 좋은 결과를 나타냄 Jan Eirik Ellingsen, EAO, 2002

Coming Soon ! Fluoridate Implant The Name of “ Osseo. Speed” Astra Tech Implant

Electrochemically Anodized Titanium Surface Merit Dental Clinic Yin-Shik Hur, Periodontist
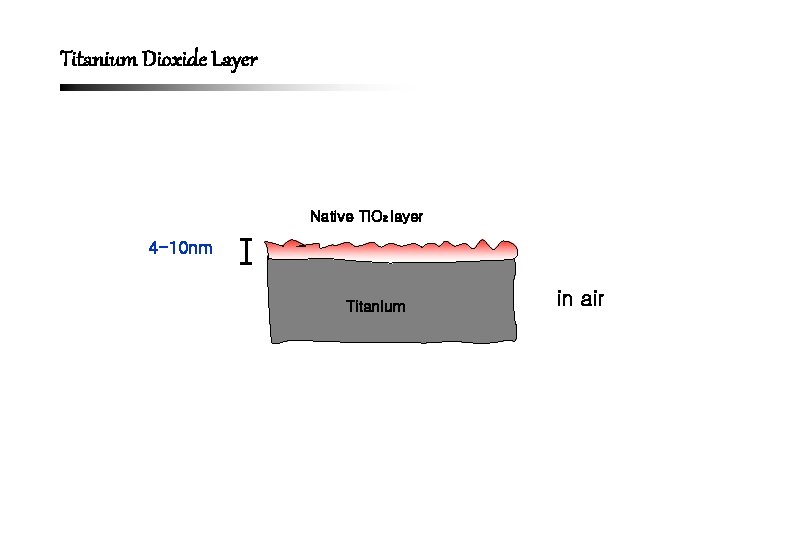
Titanium Dioxide Layer Native Ti. O 2 layer 4 -10 nm Titanium in air
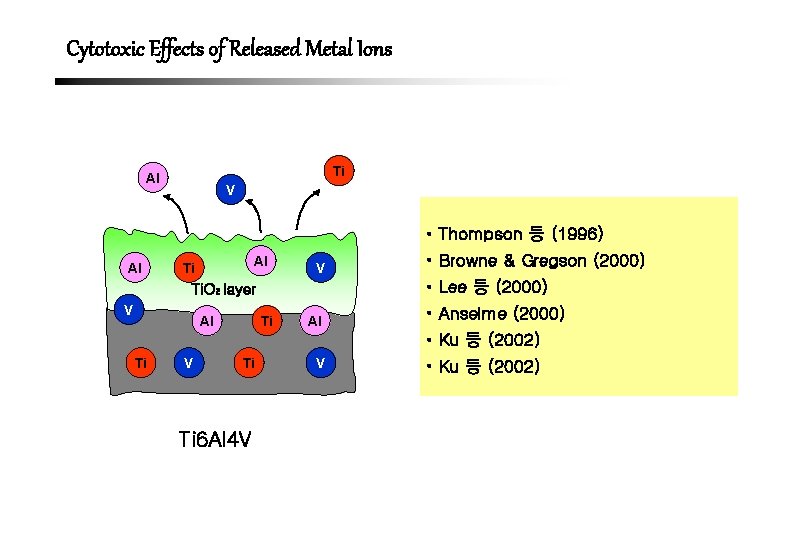
Cytotoxic Effects of Released Metal Ions Ti Al V • Thompson 등 (1996) Al Al Ti V • Lee 등 (2000) Ti. O 2 layer V Al • Browne & Gregson (2000) Ti Al • Anselme (2000) • Ku 등 (2002) Ti V Ti Ti 6 Al 4 V V • Ku 등 (2002)
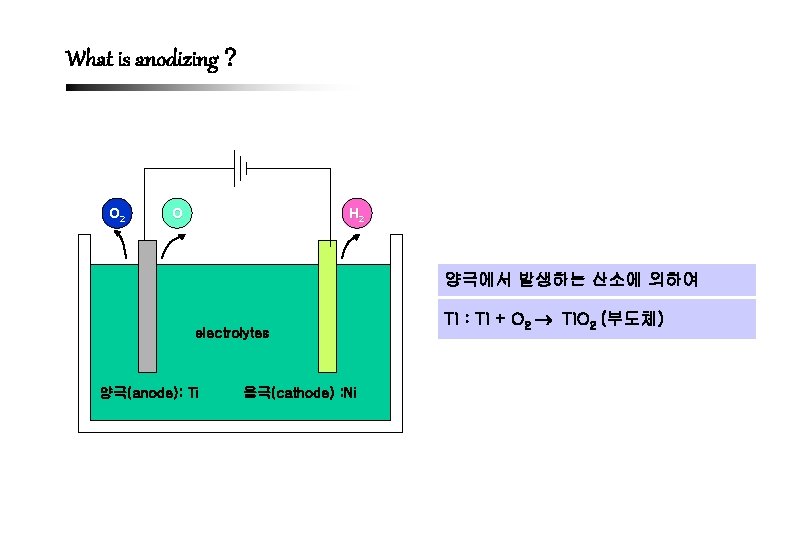
What is anodizing O 2 O H 2 양극에서 발생하는 산소에 의하여 electrolytes 양극(anode): Ti 음극(cathode) : Ni Ti : Ti + O 2 Ti. O 2 (부도체)
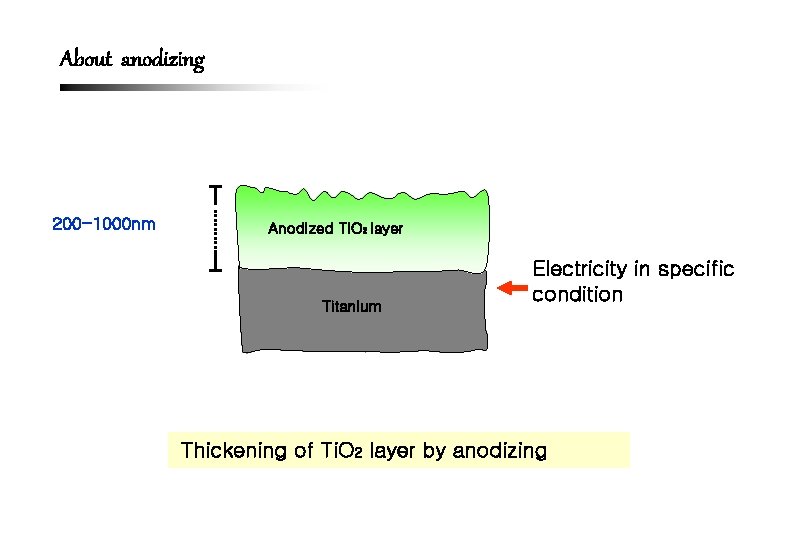
About anodizing 200 -1000 nm Anodized Ti. O 2 layer Titanium Electricity in specific condition Thickening of Ti. O 2 layer by anodizing
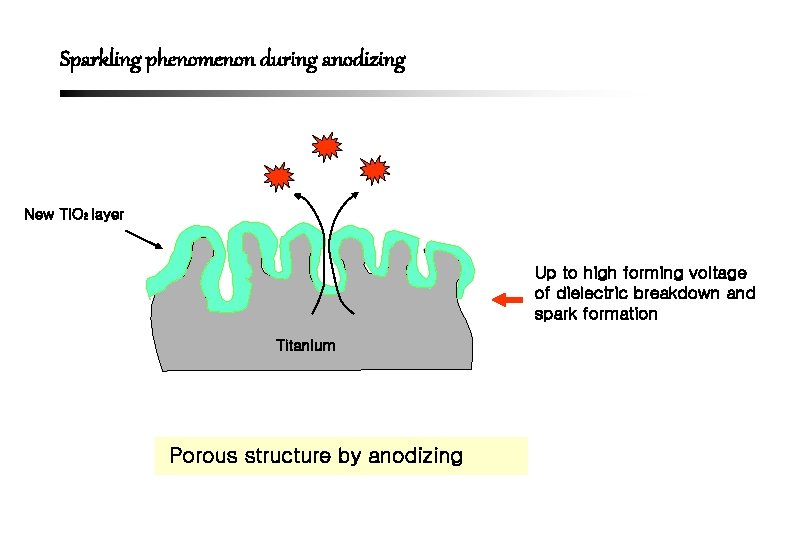
Sparkling phenomenon during anodizing New Ti. O 2 layer Up to high forming voltage of dielectric breakdown and spark formation Titanium Porous structure by anodizing
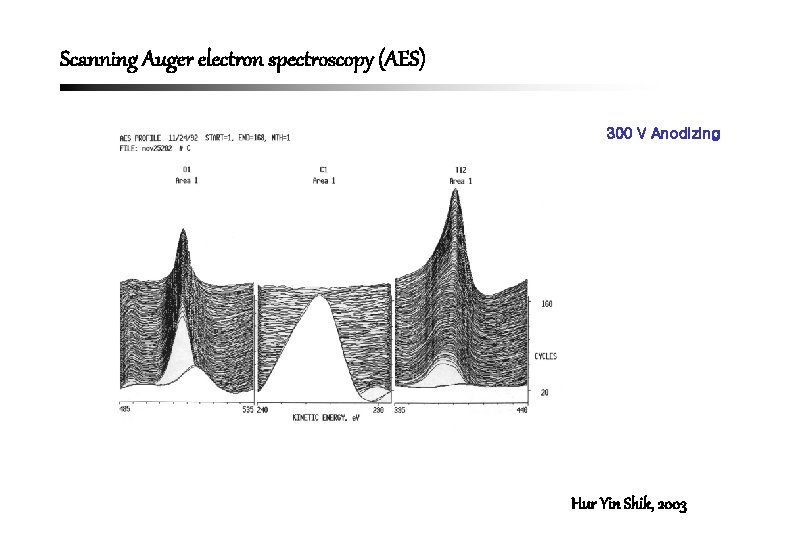
Scanning Auger electron spectroscopy (AES) 300 V Anodizing Hur Yin Shik, 2003
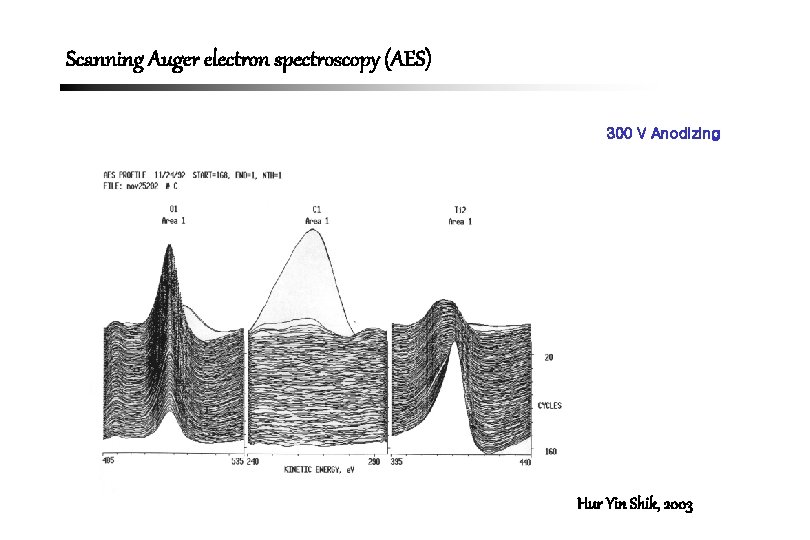
Scanning Auger electron spectroscopy (AES) 300 V Anodizing Hur Yin Shik, 2003
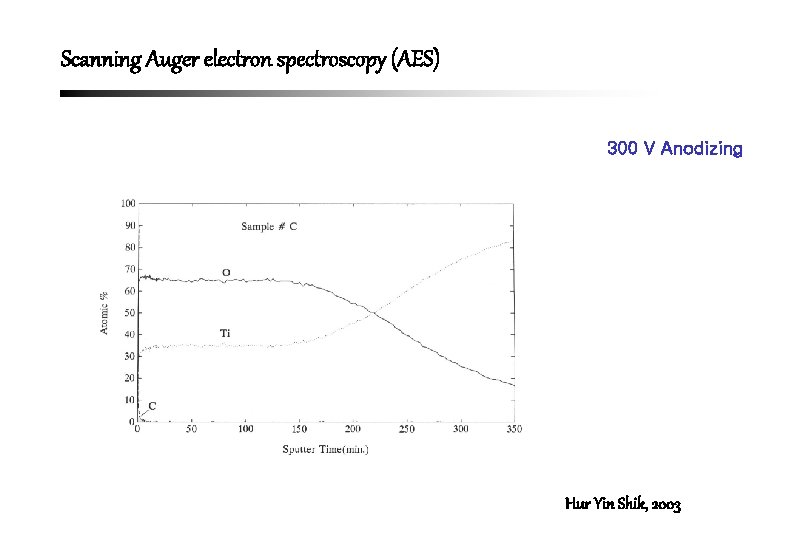
Scanning Auger electron spectroscopy (AES) 300 V Anodizing Hur Yin Shik, 2003

Thickness of Titanium Oxide Layer by AES • Control : 0. 01 -0. 05 m (10 -50 nm) • 200 V : about 2 m (2000 nm) • 250 V : about 3 m (3000 nm) • 300 V : about 4 m (4000 nm) Hur Yin Shik, 2003

Surface Roughness (Ra) by Long Scan Profiler • Control : 0. 8291 m • 200 V : 1. 2560 m • 300 V : 1. 7450 m Hur Yin Shik, 2003
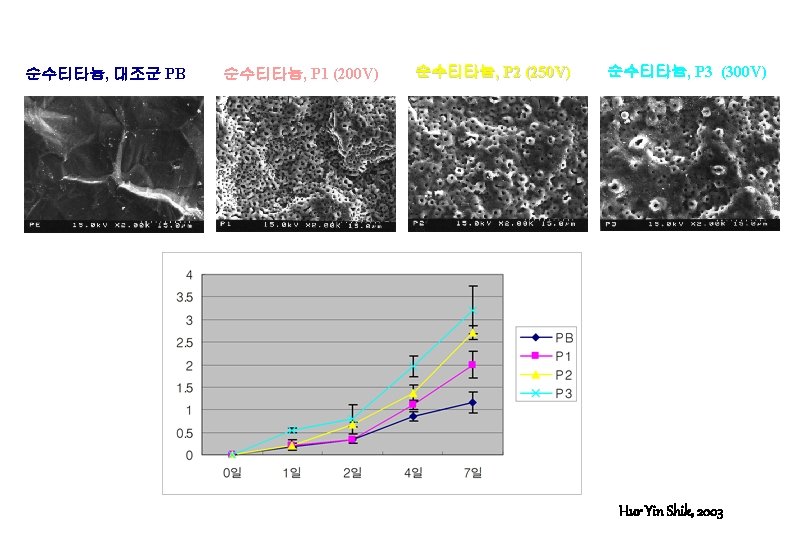
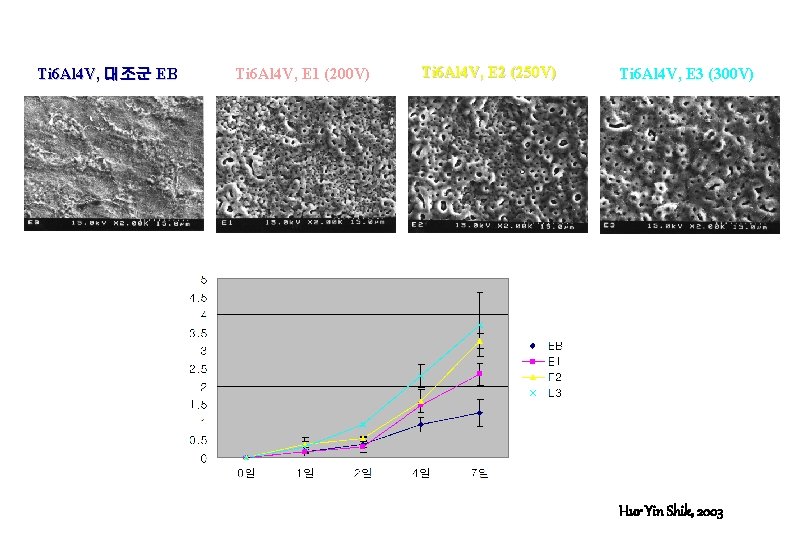
Ti 6 Al 4 V, 대조군 EB Ti 6 Al 4 V, E 1 (200 V) Ti 6 Al 4 V, E 2 (250 V) Ti 6 Al 4 V, E 3 (300 V) Hur Yin Shik, 2003

Advantages of anodizing 1. Cost effective surface treatment 2. Enhanced biocompatibility by thickened and dense oxide structures 3. Various modification by changing experimental condition. 4. Optimal roughness suitable to theory of Wennerberg (Ra 1. 5 um (1996), Ra 1 -1. 5 um(2000))
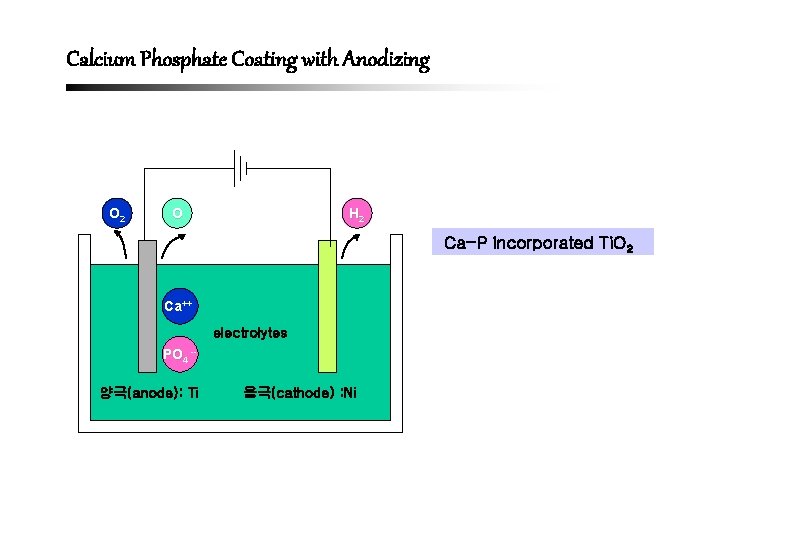
Calcium Phosphate Coating with Anodizing O 2 O H 2 Ca-P incorporated Ti. O 2 Ca++ electrolytes PO 4 -양극(anode): Ti 음극(cathode) : Ni

Ca-P Incorporated Anodizing (HA-Ti. O 2 Coating) 1. Substantial proportions of Ca and P can be incorporated into the Ti. O 2 matrix and believed to be present as ions rather than as defined calcium phosphate phases. 2. No crystalline calcium phosphate phases could be detected by X-ray diffraction in the anodic oxide coatings as produced. 3. Hydrothermal post-treatment has been shown to be effective in converting the (originally non-crystalline) calcium phosphate in oxide layer into crystalline hydroxyapatite.

Merits of Ca-P Incorporated Anodized Surface 1. “Natural”, favorable porosity and roughness of coatings 2. Suitable for coating complex-shaped titanium implants 3. Less abrupt changes in chemistry (and mechanical properties) at the interface between the HA layer and the titanium substrate (implant) in comparison to pure “ add-on” coatings such as plasma sprying.
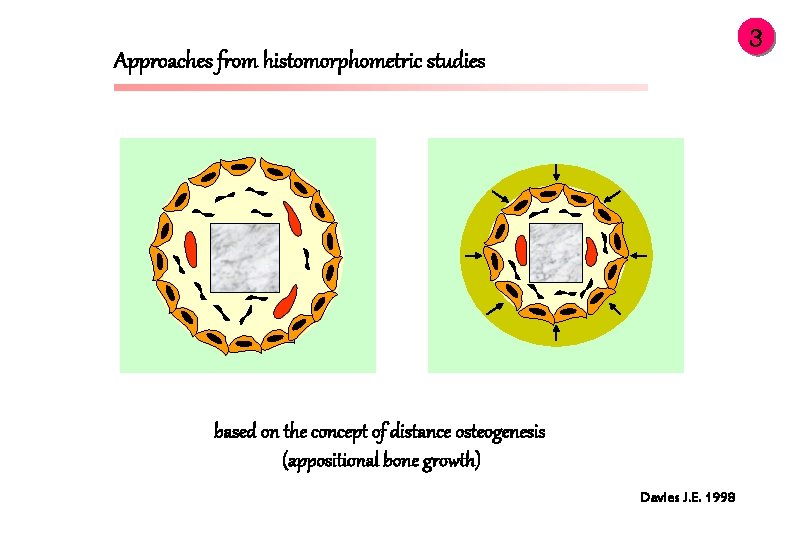
3 Approaches from histomorphometric studies based on the concept of distance osteogenesis (appositional bone growth) Davies J. E. 1998
What is the mechanism of bone formation around the implant ?
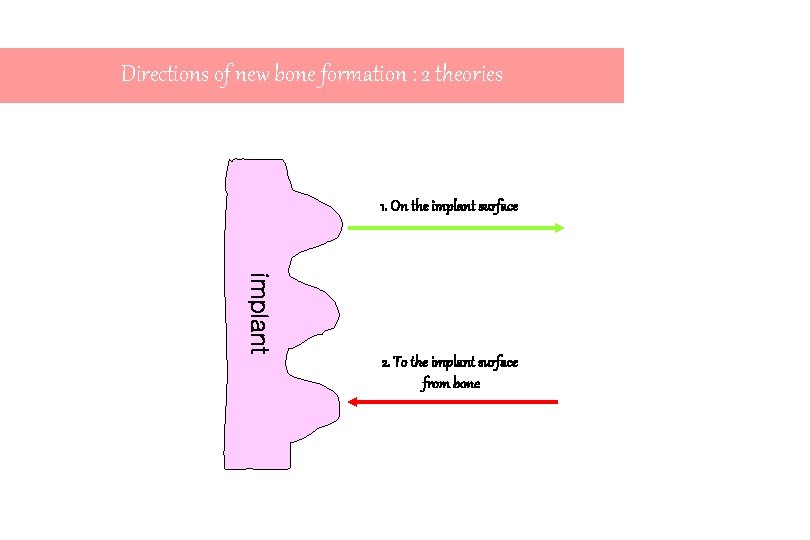
Directions of new bone formation : 2 theories 1. On the implant surface implant 2. To the implant surface from bone
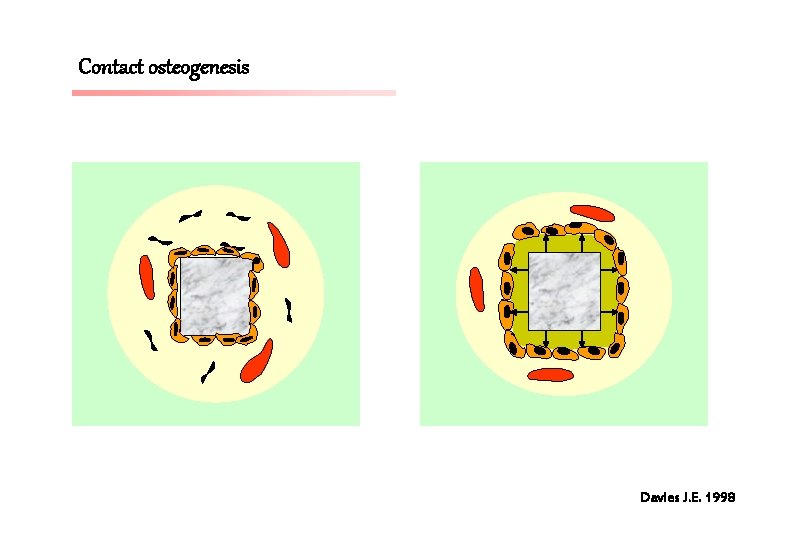
Contact osteogenesis Davies J. E. 1998
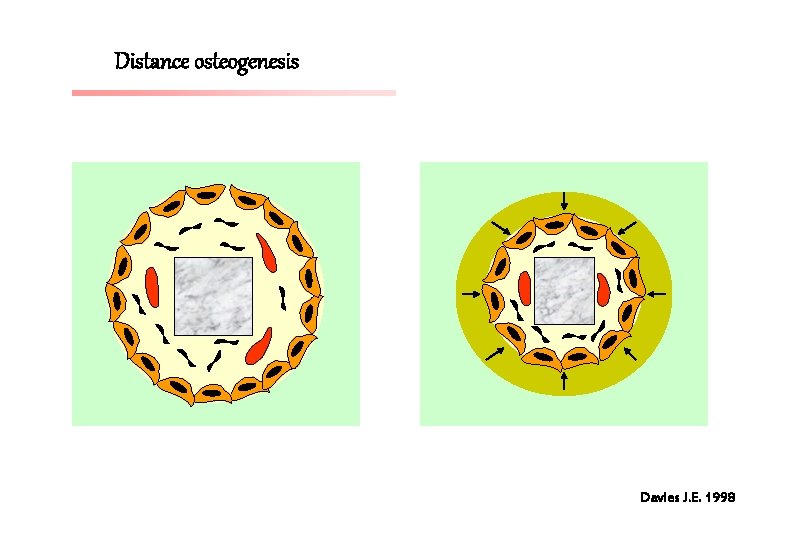
Distance osteogenesis Davies J. E. 1998
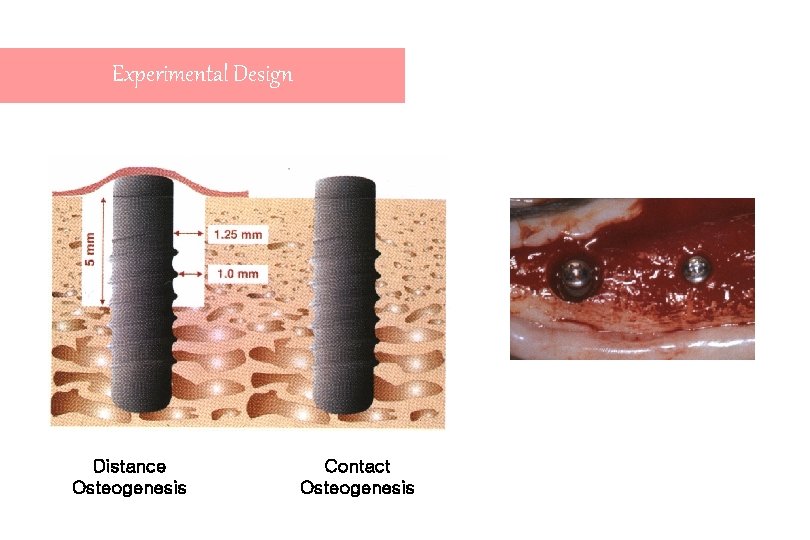
Experimental Design Distance Osteogenesis Contact Osteogenesis

The jumping distance revisited An experimental study in the dog Daniele Botticelli, Tord Berglundh, Daniel Buser, Jan Lindhe Clinical Oral Implants Research, 2003

After 1 month of healing (ground section) Contact Osteogenesis Distance Osteogenesis
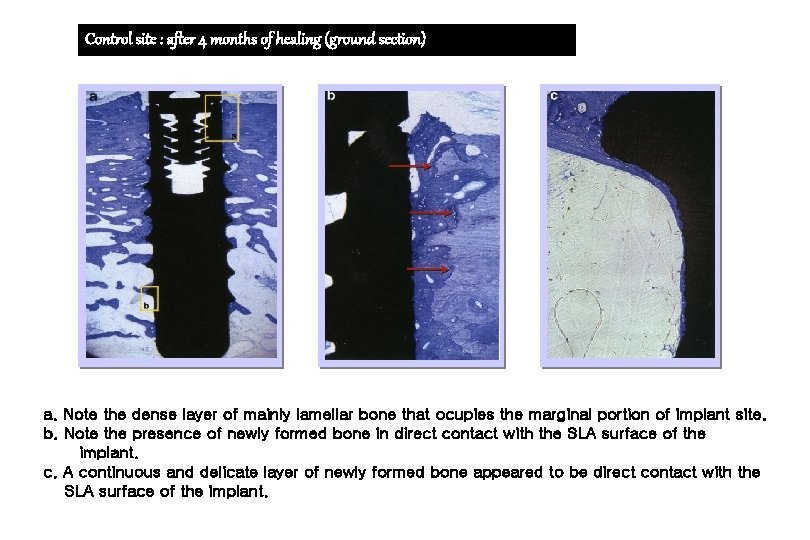
Control site : after 4 months of healing (ground section) a. Note the dense layer of mainly lamellar bone that ocupies the marginal portion of implant site. b. Note the presence of newly formed bone in direct contact with the SLA surface of the implant. c. A continuous and delicate layer of newly formed bone appeared to be direct contact with the SLA surface of the implant.
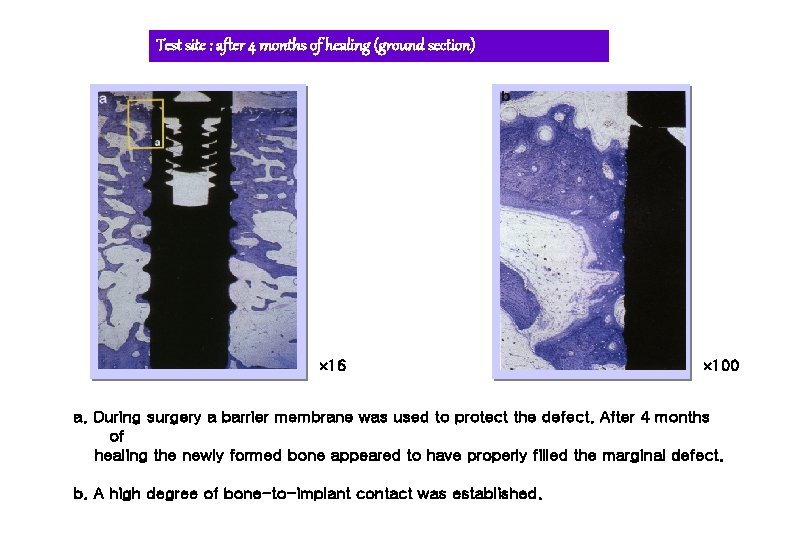
Test site : after 4 months of healing (ground section) 16 100 a. During surgery a barrier membrane was used to protect the defect. After 4 months of healing the newly formed bone appeared to have properly filled the marginal defect. b. A high degree of bone-to-implant contact was established.
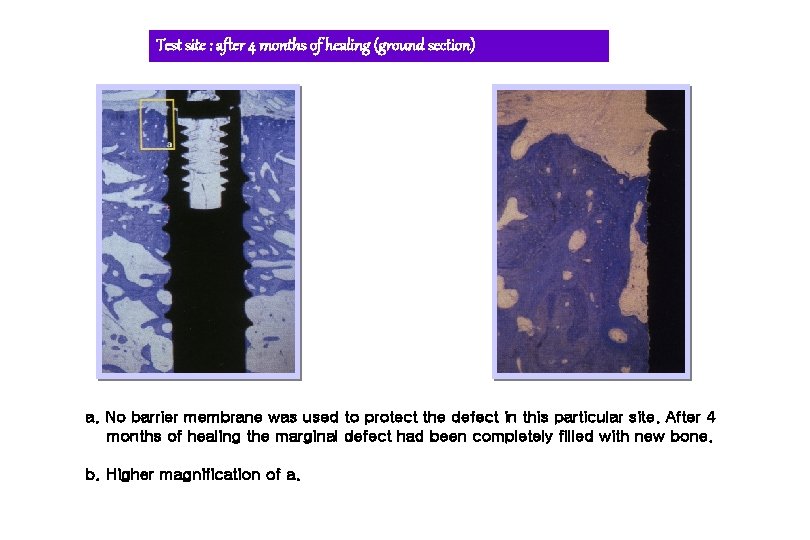
Test site : after 4 months of healing (ground section) a. No barrier membrane was used to protect the defect in this particular site. After 4 months of healing the marginal defect had been completely filled with new bone. b. Higher magnification of a.

microporous sodium titanate surface created by alkali tx. It is evident that the collagen compartment of the bone is separated from the implant surface by a continuous submicron-thick layer comprising individual, fused globules. This is the cement line matrix. The scalloped appearance of both the collagen compartment and cement line has been caused by osteoclastic resorption (Howship’s lacunae). Davies J. E. , Int. J. Prosthodont. 1998
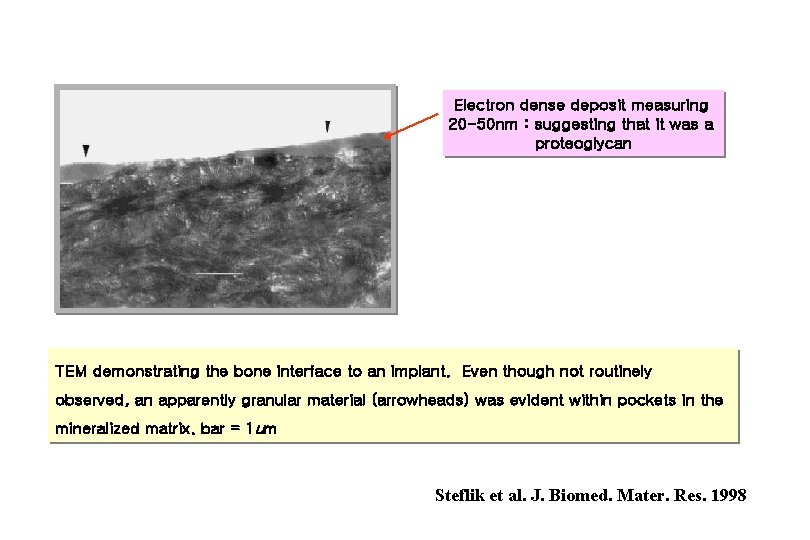
Electron dense deposit measuring 20 -50 nm : suggesting that it was a proteoglycan TEM demonstrating the bone interface to an implant. Even though not routinely observed, an apparently granular material (arrowheads) was evident within pockets in the mineralized matrix. bar = 1 um Steflik et al. J. Biomed. Mater. Res. 1998

14 days after insertion of titanium implant in rabbit tibia Collagen fibrils extend into the amorphous layer (am); co, collagen; OC, portion of osteocyte Sennerby et al. , J. Mater. Sci. : Mater. Med. 1995

Interesting Contradictions !!! 1. Bone response was enhanced by surface modifications through the different concept and methods. 2. How can these modifications affect bone response ?
Distance Osteogenesis ? Chemical Bonding ? Reailty of “Enhanced Bone Response” Contact Osteogenesis ?

Conclusion Implant Surface = Biological Stability of Implant Hur, Yin Shik, F&I Meeting 2003

Conclusion Following situations need longer biological stability period (Unfavorable healing conditions for implant) 1. Gaps between the bone and the implant (about 1 mm) 2. Primary instability of the implant at the time of placement The properties of implant surface are very important !

Conclusion 1. Making the hard and soft tissue environments for the intimate bone to implant contact 2. Using the implant system devised for enhanced osteogenesis Hur, Yin Shik, F&I Meeting 2003

Conclusion Implant system devised for enhanced osteogenesis 1. Macroscopic implant design for mechanical stability 2. Implant surface modification for biological stability Hur, Yin Shik, F&I Meeting 2003

Conclusion For enhanced hard and soft tissue environment Blessed are those who don’t hurry, for they will get a good results ! Hur, Yin Shik, F&I Meeting 2003

Closing Comment Bone physiology treats doctors without discrimination. Hur, Yin Shik, F&I Meeting 2003

We can see, but it’s not all ! Reality is a reality in my knowledge and experience till now. Hur, Yin Shik, F&I Meeting 2003

Implant Surface, Clinical Application of Our Knowledge Merit Dental Clinic Yin-Shik Hur, Periodontist
- Slides: 185