BernRotterdam Cohort Study Newer generation everolimuseluting stents eliminate

Bern-Rotterdam Cohort Study Newer generation everolimus-eluting stents eliminate the risk of very late stent thrombosis compared with early generation sirolimus-eluting and paclitaxel-eluting stents Lorenz Räber, Michael Magro, Giulio G. Stefanini, Bindu. Kalesan, Ron T. van Domburg, Yoshinobu. Onuma, Peter Wenaweser, Joost Daemen, Bernhard Meier, Peter Jüni, Patrick W. Serruys, Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern University Hospital, Switzerland Thoraxcenter, Erasmus Medical Center, Rotterdam, The Netherlands

Disclosures No disclosures Funding Intramuralgrants of CTU Bern and a grant of the Swiss National Science Foundation

BR Cohort Study - Background I • Stentthrombosis (ST) is a rare butpotentiallydevastatingcomplicationfollowing coronarystentimplantation and isassociatedwithdeathormyocardialinfarction in up to 90% of cases. • Whereasearly and late ST occurwithsimilarfrequencyamongpatientstreate dwithearlygenerationdrug-elutingstents (DES) and bare metal stents (BMS), verylate ST ismorecommonwithearlygeneration DES with an annualrisk of up to 0. 6% duringlongtermfollow-up.

BR Cohort. Study - Background II • The newergenerationeverolimus-elutingstent (EES) is a thinstrut, cobaltchromiumstent and releaseseverolimus, a sirolimusanaloguefrom a fluoropolymer. • Whetherthenewergeneration EES reducestherisk of verylate ST as compared to earlygeneration DES has not beeninvestigated in an adequatelypoweredstudywithsufficientlongtermfollow-up.

BR Cohort. Study - Objective To comparethesafety of theunrestricteduse of EES (XIENCE/PROMUSTM) comparedwith early generation sirolimus-eluting (CYPHERTM) (SES) and paclitaxel-elutingstents (TAXUS Express. TM) (PES) forcoronaryrevascularization in a large, consecutivelyenrolledpatient populationduringlong-termfollow-up.

BR Cohort Study - Patient Population Inclusion. Criteria • All consecutivepatientstreatedwith EES, SES, and PES at Bern University Hospital and the Thoraxcenter, Erasmus University Hospital in thesetting of stable angina, silentischemia, and acutecoronary syndromes (UA, NSTEMI, STEMI) • Diameter stenosis>50% • Number of lesions: no limitation • Number of vessels: no limitation • Lesionlength: no limitation Exclusion. Criteria • Implantation of morethanonestent type

BR Cohort. Study - Endpoints Primary Endpoint • ARC definite ST through 4 years Secondary Endpoints • • • ARC verylate definite ST ARC definite orprobable ST ARC verylate definite orprobable ST Cardiac death Myocardialinfarction (MI) Cardiac deathor MI

BR Cohort Study - Statistical Analysis • Propensity scores for receiving EES were estimated using a probit model including age, gender and pre-treatment variables associated with stent selection at p<0. 10 and used to derive inverse probability of treatment weights (ITPW). Comparisons between stents were performed using a Cox proportional hazards model, crude and adjusted by weighting using ITPW. • years. Clinical events are expressed as counts and cumulative incidence rates per 100 patient

BR Cohort Study - Clinical Trial Organization Event Adjudication. Committee • Salvatore Brugaletta, MD, Barcelona, Spain • Josep Gomez Lara, MD, Barcelona, Spain • Gerrit Hellige, MD, Aarau, Switzerland • Niklas Millauer, MD, Bern, Switzerland Central Data Monitoring • Clinical Trials Unit Bern, Switzerland Statistical Analysis • Peter Jüni, Bindu Kalesan Institute of Social and Preventive Medicine, University of Bern, Switzerland

BR Cohort Study - Patient Flow 12339 Patients. Undergoing PCI EES 4212 consecutivepatients 11/2006 – 3/2009 SES 3819 consecutivepatients 3/2002 -1/2006 PES 4308 consecutivepatients 3/2002 -1/2006 Fup rate* 97. 4% Meanfupduration 2. 5 years (1. 8 -3. 1) Fup rate 97. 5% Meanfupduration 4. 0 years (3. 1 -4. 0) Fup rate 95. 9% Meanfupduration 3. 0 years (2. 1 -3. 6) *F/U rate atthe time of latestfollow-up

BR Cohort Study - Antithrombotic. Drug Regimen Preorduringprocedure Acetylsalicylicacid: > 100 mg Clopidogrel: 300 -600 mg loading dose Unfractionatedheparin - Bolus of at least 5000 IU i. v. or 70 IU/kg Glycoprotein. IIb/IIIaantagonists - Operator discretion Post procedure Acetylsalicylicacid: 100 mg/d indefinitely Clopidogrel 75 mg/d for 3 -12 months
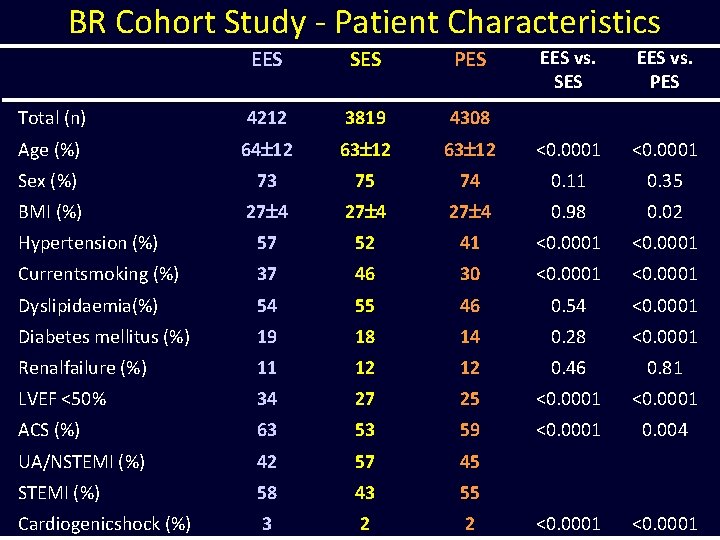
BR Cohort Study - Patient Characteristics EES SES PES Total (n) 4212 3819 4308 Age (%) 64 12 63 12 Sex (%) 73 BMI (%) EES vs. SES EES vs. PES 63 12 <0. 0001 75 74 0. 11 0. 35 27 4 0. 98 0. 02 Hypertension (%) 57 52 41 <0. 0001 Currentsmoking (%) 37 46 30 <0. 0001 Dyslipidaemia(%) 54 55 46 0. 54 <0. 0001 Diabetes mellitus (%) 19 18 14 0. 28 <0. 0001 Renalfailure (%) 11 12 12 0. 46 0. 81 LVEF <50% 34 27 25 <0. 0001 ACS (%) 63 53 59 <0. 0001 0. 004 UA/NSTEMI (%) 42 57 45 STEMI (%) 58 43 55 Cardiogenicshock (%) 3 2 2 <0. 0001
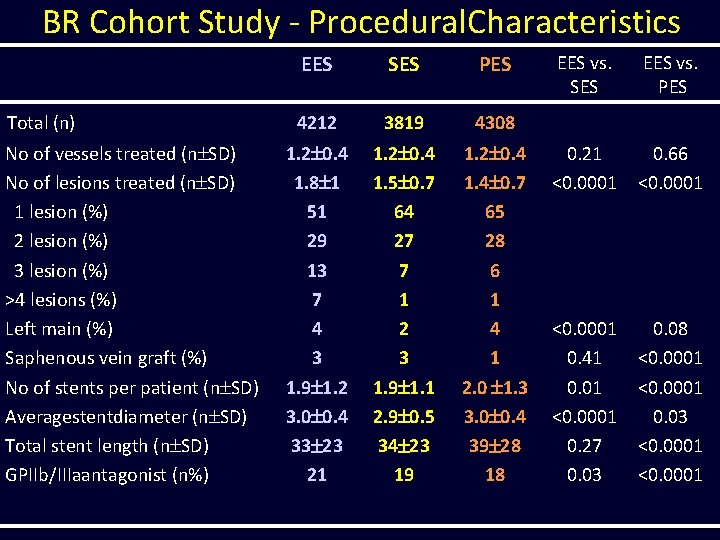
BR Cohort Study - Procedural. Characteristics Total (n) No of vessels treated (n SD) No of lesions treated (n SD) 1 lesion (%) 2 lesion (%) 3 lesion (%) >4 lesions (%) Left main (%) Saphenous vein graft (%) No of stents per patient (n SD) Averagestentdiameter (n SD) Total stent length (n SD) GPIIb/IIIaantagonist (n%) EES SES PES 4212 3819 4308 1. 2 0. 4 1. 8 1 51 29 13 7 4 3 1. 9 1. 2 3. 0 0. 4 33 23 21 1. 2 0. 4 1. 5 0. 7 64 27 7 1 2 3 1. 9 1. 1 2. 9 0. 5 34 23 19 1. 2 0. 4 1. 4 0. 7 65 28 6 1 4 1 2. 0 1. 3 3. 0 0. 4 39 28 18 EES vs. SES EES vs. PES 0. 21 <0. 0001 0. 66 <0. 0001 0. 41 0. 01 <0. 0001 0. 27 0. 03 0. 08 <0. 0001 0. 03 <0. 0001
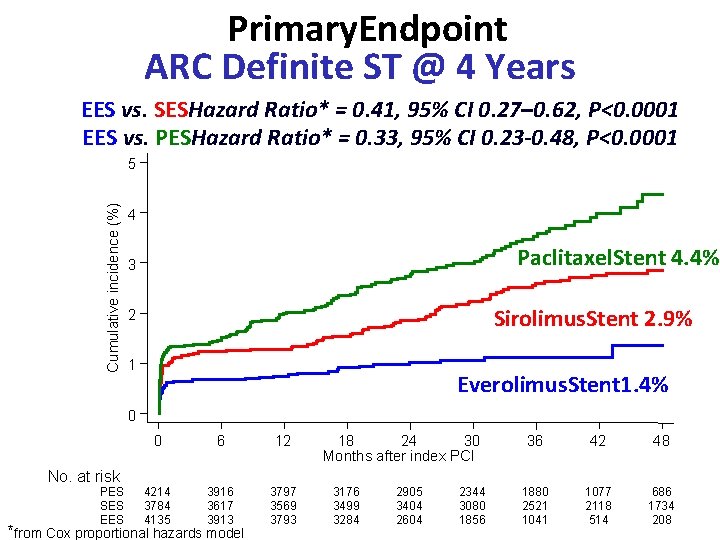
Primary. Endpoint ARC Definite ST @ 4 Years EES vs. SESHazard Ratio* = 0. 41, 95% CI 0. 27– 0. 62, P<0. 0001 EES vs. PESHazard Ratio* = 0. 33, 95% CI 0. 23 -0. 48, P<0. 0001 Cumulative incidence (%) 5 4 Paclitaxel. Stent 4. 4% 3 Sirolimus. Stent 2. 9% 2 1 Everolimus. Stent 1. 4% 0 0 6 12 4214 3784 4135 3916 3617 3913 3797 3569 3793 18 24 30 Months after index PCI 36 42 48 1880 2521 1041 1077 2118 514 686 1734 208 No. at risk PES SES EES *from Cox proportional hazards model 3176 3499 3284 2905 3404 2604 2344 3080 1856
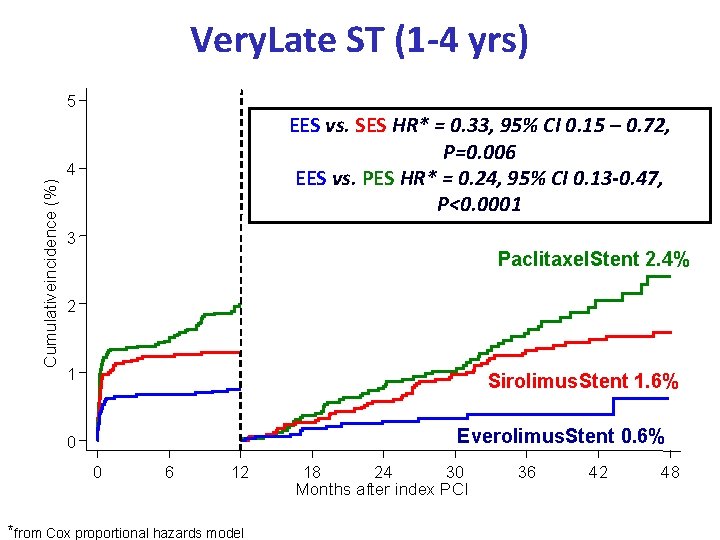
Very. Late ST (1 -4 yrs) 5 EES vs. SES HR* = 0. 33, 95% CI 0. 15 – 0. 72, P=0. 006 EES vs. PES HR* = 0. 24, 95% CI 0. 13 -0. 47, P<0. 0001 Cumulativeincidence (%) 4 3 Paclitaxel. Stent 2. 4% 2 1 Sirolimus. Stent 1. 6% Everolimus. Stent 0. 6% 0 0 6 12 *from Cox proportional hazards model 18 24 30 Months after index PCI 36 42 48
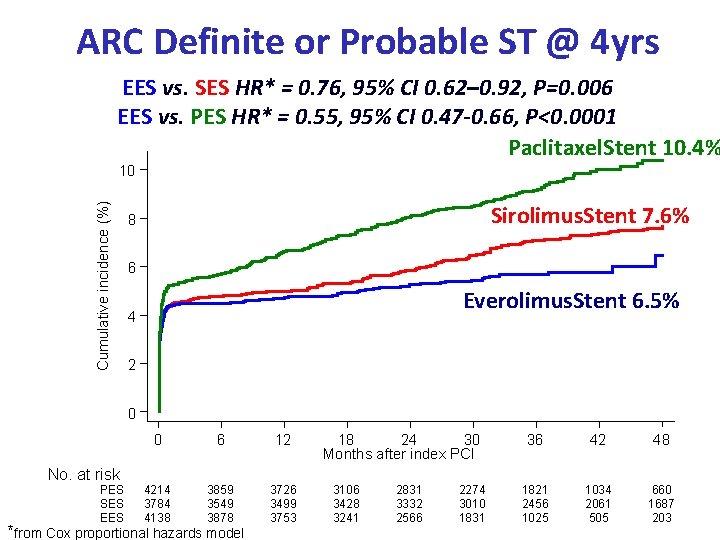
ARC Definite or Probable ST @ 4 yrs EES vs. SES HR* = 0. 76, 95% CI 0. 62– 0. 92, P=0. 006 EES vs. PES HR* = 0. 55, 95% CI 0. 47 -0. 66, P<0. 0001 Paclitaxel. Stent 10. 4% Cumulative incidence (%) 10 Sirolimus. Stent 7. 6% 8 6 Everolimus. Stent 6. 5% 4 2 0 0 6 12 4214 3784 4138 3859 3549 3878 3726 3499 3753 18 24 30 Months after index PCI 36 42 48 1821 2456 1025 1034 2061 505 660 1687 203 No. at risk PES SES EES *from Cox proportional hazards model 3106 3428 3241 2831 3332 2566 2274 3010 1831
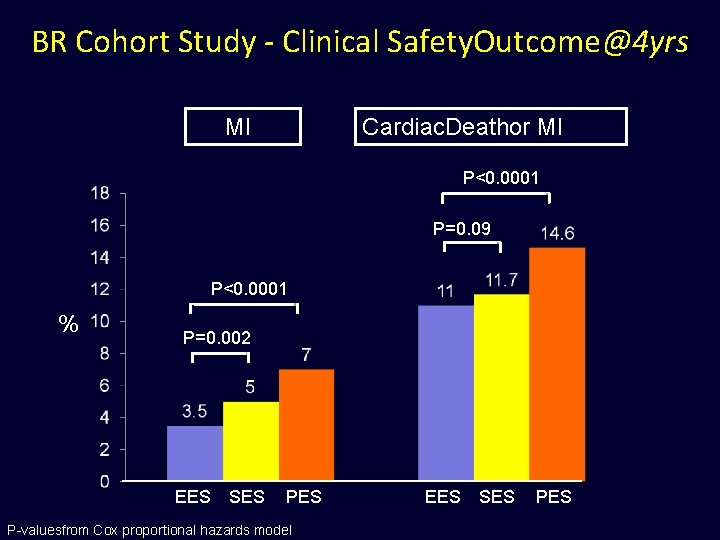
BR Cohort Study - Clinical Safety. Outcome@4 yrs MI Cardiac. Deathor MI P<0. 0001 P=0. 09 P<0. 0001 % P=0. 002 EES SES P-valuesfrom Cox proportional hazards model EES SES PES
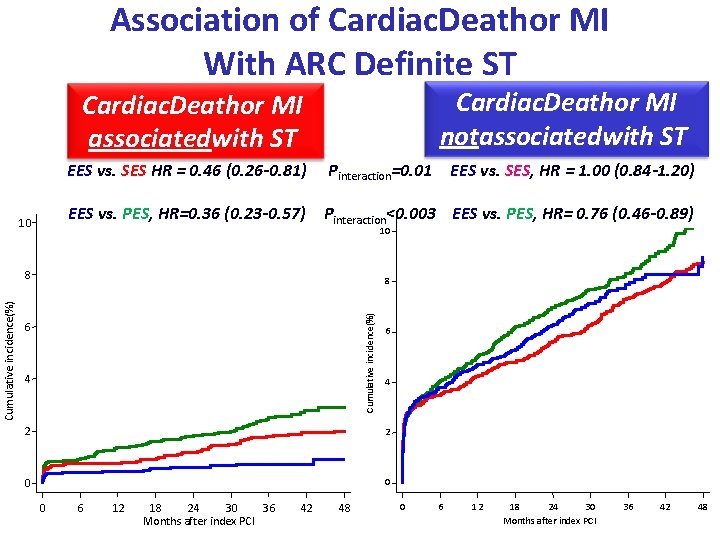
Association of Cardiac. Deathor MI With ARC Definite ST Cardiac. Deathor MI notassociatedwith ST Cardiac. Deathor MI associatedwith ST 10 Pinteraction=0. 01 EES vs. PES, HR=0. 36 (0. 23 -0. 57) Pinteraction<0. 003 EES vs. PES, HR= 0. 76 (0. 46 -0. 89) 10 8 8 Cumulative incidence(%) EES vs. SES, HR = 1. 00 (0. 84 -1. 20) EES vs. SES HR = 0. 46 (0. 26 -0. 81) 6 4 2 2 0 0 0 6 12 18 24 30 36 Months after index PCI 42 48 0 6 12 18 24 30 Months after index PCI 36 42 48
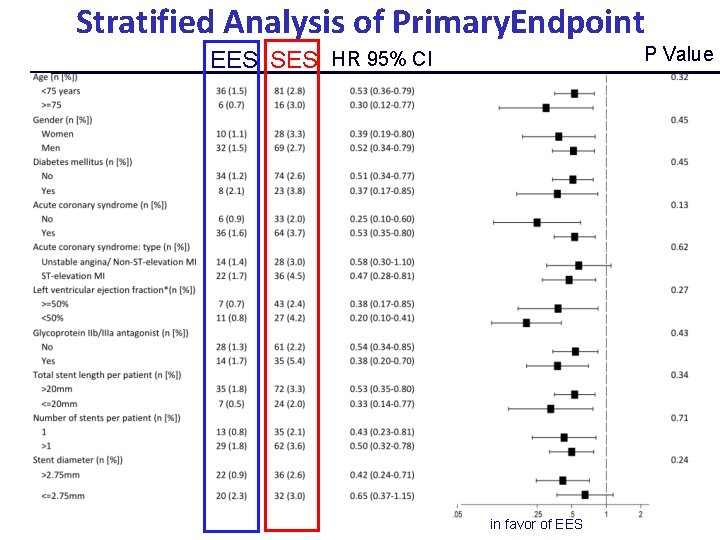
Stratified Analysis of Primary. Endpoint P Value EES SES HR 95% CI in favor of EES
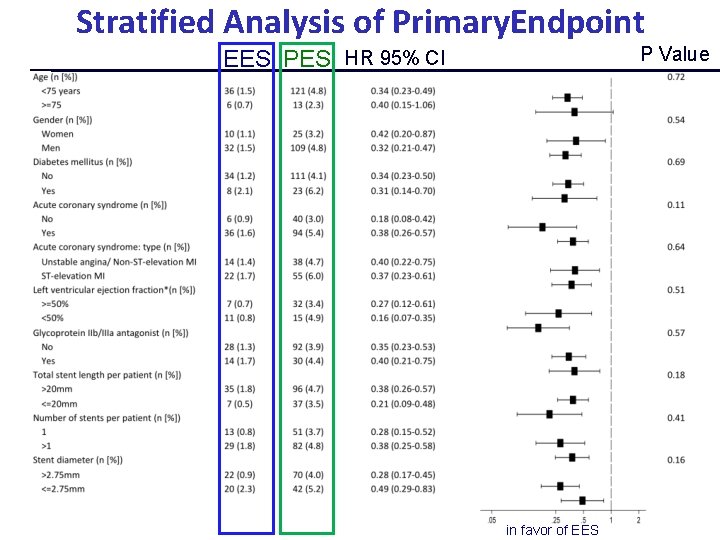
Stratified Analysis of Primary. Endpoint P Value EES PES HR 95% CI in favor of EES

BR Cohort. Study - Limitations • Non-randomizedobservationalstudy - analyseswereadjustedfordifferencesusinginverse probability of treatmentweighting (ITPW) - differences in favour of EES were large and consistentamongsubgroups • Patientswereenrolledsequentially and advances in implantationtechnique and prolongation of DAPT to oneyearmayhavefavored EES - Study focused on verylate. ST - Sequentialenrollmentreducestherisk of selectionbias - EES patientswere at higherrisk

BR Cohort. Study - Conclusions • In thisobservational, prospectivecohortstudy, theunrestricteduse of a EES was associatedwith a lowerrisk of overall ARC definite and ARC definite orprobable ST up to fouryears of follow-up. • The benefit in favor of a EES was most pronounced during the very late period with a 67% and 76% reduced risk of definite ST compared with SES and PES, resulting in a near elimination of very late ST. • The reduced risk of VLST with the unrestricted use of EES overcomes the principal limitation of early generation DES and constitutes an important advance in DES safety.
- Slides: 22