Bernie Betlach CLS MTASCP Laboratory Consultant MediCal Xerox

Bernie Betlach CLS, MT(ASCP) Laboratory Consultant / Medi-Cal Xerox State Healthcare, LLC 1 November 2014 1

Paper Claim Submission §Clear and Scannable §Send Only Necessary Documentation §Do Not Use Highlight Markers §Do Not Use Pencils §Do Not Submit Handwritten Forms 2 2

Multiple Procedure Billing §Use a Single Claim Line to bill the same Lab Procedure, more than once, on the same day § Indicate Quantity in the Units Column §Exceptions are Surgical Pathology CPT-4 codes 88300 through 88309 when billed for separate sites must be billed on separate claim lines with a maximum of one unit for each line. See: path bil cms 2, path surg 2 3 3

Claim Form Units Field § On Appeals submit the original claim with the total number of units § Corrected if necessary §Medi-Cal will only pay what is billed See: appeal form 1 4 4

Laboratory Services Reservation System (LSRS) Allows Laboratories to Reserve or Determine if a Procedure is Within the Medi-Cal Frequency Limits for that Monthly Frequency Limits for all Lab Services are: ▪ Per Recipient ▪ Per Service 5

LSRS (cont’d) 3 Ways to Make a Reservation: §Web Based, on the Internet §Calling the Help Desk §Allowing the System to Make a Reservation if Available by Submitting a Claim 6 a

LSRS (cont’d) Common Mistake When Making a Web Based Reservation: Use only the Patient’s 9 character Identification Number: Use this: 87709090 D Do Not Use: 87709090 D 71249 Exception: The Longer Identification Numbers as with Presumptive Eligibility 7

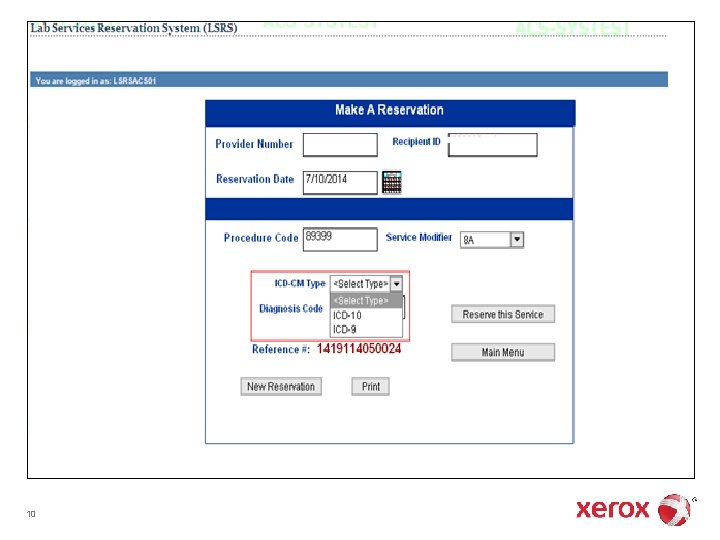
ICD-10 § Primary baseline crosswalk is CMS General Equivalency Maps (GEMS) § Mandatory implementation date October 1, 2015 8
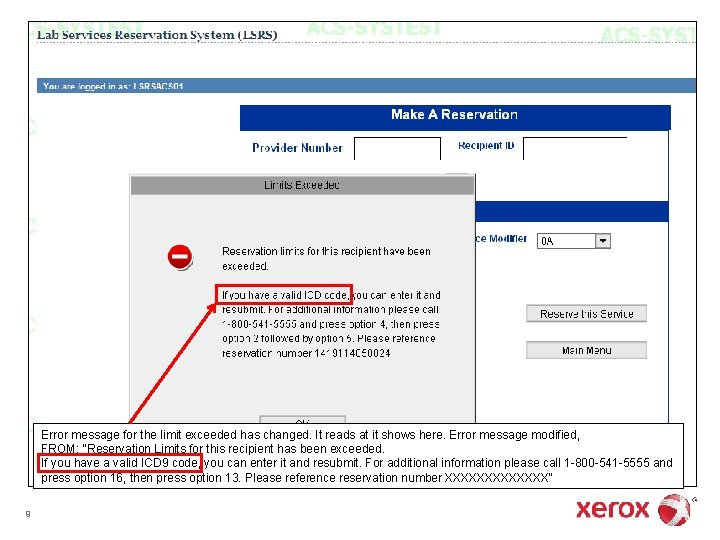
Error message for the limit exceeded has changed. It reads at it shows here. Error message modified, FROM: "Reservation Limits for this recipient has been exceeded. If you have a valid ICD 9 code, you can enter it and resubmit. For additional information please call 1 -800 -541 -5555 and press option 16, then press option 13. Please reference reservation number XXXXXXX" 9
10
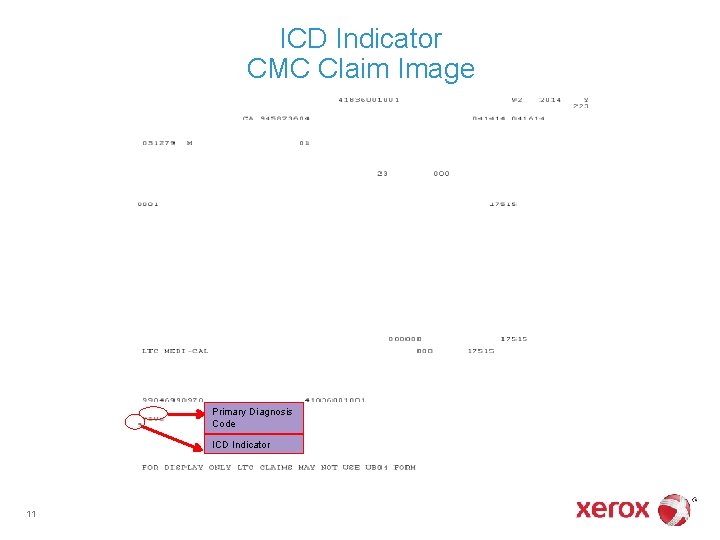
ICD Indicator CMC Claim Image Primary Diagnosis Code ICD Indicator 11

Paper Claim Image – UB-04 Outpatient claim (With Overlay) Primary Diagnosis Code ICD Indicator 12

Paper Claim Image – CMS 1500 claim Primary Diagnosis Code 13 ICD Indicator

Electronic Treatment Authorization Request (e. TAR) forms. Effective September 22, 2014, an ICD code and an ICD indicator are required on electronic Treatment Authorization Request (e. TAR) forms.

Quantitative Drug Tests Requiring Justification Documentation must be Submitted with a Claim Justifying the use of a Quantitative Determination of Drug level rather than Qualitative Determination (screening). CPT-4 code Description 82101 82145 82205 82649 83840 83925 83992 Alkaloids, urine, quantitative Amphetamine or methamphetamine Barbiturates, not elsewhere specified Dihydromorphinone Methadone Opiate(s), drug and metabolites, each procedure Phencyclidine (PCP) See: path drug 1, 2 15

Maximum Reimbursement Laboratory Services are paid based on the least amount of the following: § § The amount billed The charge to the general public Medicare’s maximum allowance Medi-Cal’s maximum allowance See: Cal. Code Regs. , tit. 22, § 51529, subd. (a)(2)(B) 16

Rate Update for Laboratory Services Effective on or after January 1, 2013: Reimbursement rates for certain laboratory services that were higher than 80 percent of the 2013 Medicare rate will change. Claims processed for dates of service on or after January 1, 2013, will be subject to the updated rates. See: California Code of Regulations (CCR), Title 22, Section 51137. 2 17

Benefit Update Effective November 1, 2013 CPT-4 codes 88740 (hemoglobin, quantitative, transcutaneous, per day, carboxyhemoglobin) and 88741 (quantitative, transcutaneous, per day methemoglobin) Both are non-benefits for Medi-Cal. See: path bil 5 18

Every Women Counts New Benefits Cytopathology CPT Codes Effective retroactively for dates of service on or after July 1, 2013, the following CPT-4 and HCPCS codes have been added as benefits of Every Woman Counts (EWC): 88143 88172 88342 See: ev woman (16– 31) 19

Presumptive Eligibility Program New Benefit Effective for dates of service on or after January 1, 2014 CPT-4 code 87077 aerobic bacterial identification, definitive identification is reimbursable to Presumptive Eligibility (PE) recipients. See: presum (19, 20) 20

Presumptive Eligibility Program New Benefit Cystic Fibrosis Effective for dates of service on or after August 1, 2014 CPT-4 code 81220 (CFTR [cystic fibrosis transmembrane conductance regulator][eg, cystic fibrosis] gene analysis; common variants [eg, ACGM/ACOG guidelines]) is reimbursable to Presumptive Eligibility (PE) recipients with aid code 7 G. See: gene coun (4); pre sum (19) 21

Fetal Screening Procedures Reimbursable Under PE Program Effective for dates of service on or after May 1, 2014 CPT-4 code 81508 (fetal congenital abnormalities, biochemical assays of two proteins [PAPP-A and h. CG (any form)], utilizing maternal serum, algorithm reported as risk score) and CPT-4 code 81511 (fetal congenital abnormalities, biochemical assays of four analytes [AFP, u. E 3, h. CG (any form), DIA], utilizing maternal serum, algorithm reported as risk score) are reimbursable under the Presumptive Eligibility (PE) program. Reimbursable only once for women in the first and/or second trimester of pregnancy. See: presum (20) 22

Noninvasive prenatal testing for fetal aneuploidy is billed with A TAR for the test requires documentation of the following criteria: Patient with singleton gestation only. The patient has an increased risk of aneuploidy due to one or more of the following: • Maternal age 35 years or older at delivery • Fetal ultrasonographic findings indicating an increased risk of aneuploidy • History of a prior pregnancy with a trisomy • Positive test result for aneuploidy, including first trimester, sequential, or integrated screen, or a quadruple screen • Parental balanced Robertsonian translocation with increased risk of fetal trisomy 13 or trisomy 21 • See: path molec 28 23

BRCA Testing Procedure Code Rate Update Effective retroactively for dates of service on or after January 1, 2014 The rate for CPT-4 code 81211 is being increased. An Erroneous Payment Correction will be issued, allowing automatic resubmission of claims with dates of service on or after January 1, 2014. When procedure code 81211 is billed with modifier QP, the payment will be the difference between the rate for code 81211 and the rate for either CPT-4 code 81215 or CPT-4 code 81217, depending on the previously performed BRCA test. 24

Immunoassay for Tumor Antigens: Diagnostic Restrictions Updated Effective for dates of service on or after April 1, 2014 CPT-4 code 86304 (immunoassay for tumor antigen, quantitative; CA 125) is reimbursable only when billed in conjunction with one of the following ICD-9 -CM diagnosis codes: 158. 8 158. 9 180. 0 182. 0 183. 2 183. 8 184. 8 198. 6 198. 82 236. 0 236. 1 236. 2 236. 3 See: path chem (7) 25 338. 3 789. 39 795. 82 795. 89 V 10. 41 V 10. 42 V 10. 43 V 10. 44

Molecular Pathology and Diagnostics Only the Procedures listed in the Provider Manual are Medi-Cal Covered Services. Reimbursement Requirements for Each Procedure include some or all of the following: § Treatment Authorization Request (TAR) and claim documentation requirements. § Allowable diagnosis (ICD-9 -CM) codes. § Once-in-a-lifetime and other frequency limitations for reimbursement. See: path molec 26

TAR/SAR/CCS/GHPP Override for Genetic Codes For CPT-4 codes 81200 -81408: • Over-rides include non-benefits listed under Tier 1 Genetics. • Authorization must include one of the following: • CCS/GHPP Stamp • Hardcopy Authorization • TAR, TAR ending in 8 or 4 • SAR Prefix 91, 97 or 99 27

New Benifit Hepatitis C, IL 28 B testing Effective for dates of service on or after October 1, 2014 Bill with CPT-4 code 81400 (Mo. Path Level 1). Document “IL 28 B” on the claim form or on an attachment. The following conditions which must be documented: • The patient has genotype 1 hepatitis C virus infection, and, • Treatment will be contingent on the test results. See: path molec (15) 28

Family PACT and Medi-Cal Family Planning Code Conversions Effective for dates of service on or after December 30, 2013 §DHCS has discontinued the use of the Family PACT Program and Medi-Cal Family Planning local diagnosis codes (the S-Codes). §The local codes will have been replaced with HCPCS national HIPAA compliant codes. Ref: 1996 Public Law 104 -191, 45 CFR 162. 1000 See: lab 2 – 34 (FPACT Manual) 29

HIPAA Code Conversion for Local Modifier ZS DHCS will discontinue use of local modifier ZS. ZS will be replaced with a Blank in the modifier position. Anticipated Implementation is May 2015 30

Bernie Betlach CLS, MT(ASCP) Medical Lab Consultant Xerox State Healthcare, LLC 820 Stillwater Road West Sacramento, CA 95605 bernard. betlach@xerox. com 31 31
- Slides: 31