Benzyne Mechanism Reactant is halobenzene with no electronwithdrawing
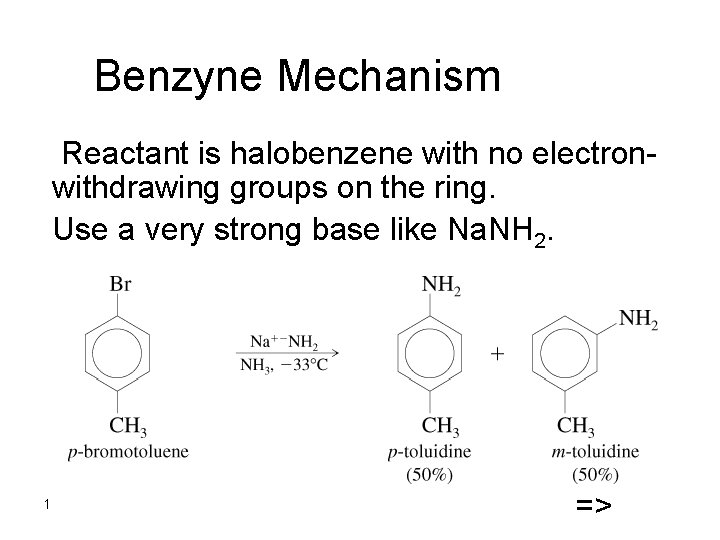
Benzyne Mechanism Reactant is halobenzene with no electronwithdrawing groups on the ring. Use a very strong base like Na. NH 2. 1 =>

Benzyne Intermediate => 2

Chlorination of Benzene Addition to the benzene ring may occur with high heat and pressure (or light). The first Cl 2 addition is difficult, but the next 2 moles add rapidly. The product, benzene hexachloride, is an insecticide. => 3

Catalytic Hydrogenation Elevated heat and pressure is required. Possible catalysts: Pt, Pd, Ni, Ru, Rh. Reduction cannot be stopped at an intermediate stage. => 4

Birch Reduction: Regiospecific • A carbon with an e--withdrawing group is reduced. • A carbon with an e--releasing group is not reduced. => 5

Birch Mechanism => 6

Side-Chain Oxidation Alkylbenzenes are oxidized to benzoic acid by hot KMn. O 4 or Na 2 Cr 2 O 7/H 2 SO 4. => 7

Side-Chain Halogenation Benzylic position is the most reactive. Chlorination is not as selective as bromination, results in mixtures. Br 2 reacts only at the benzylic position. => 8

SN 1 Reactions Benzylic carbocations are resonancestabilized, easily formed. Benzyl halides undergo SN 1 reactions. 9 =>

SN 2 Reactions Benzylic halides are 100 times more reactive than primary halides via SN 2. Transition state is stabilized by ring. => 10

Reactions of Phenols Some reactions like aliphatic alcohols: phenol + carboxylic acid ester phenol + aq. Na. OH phenoxide ion Oxidation to quinones: 1, 4 -diketones. => 11

Quinones Hydroquinone is used as a developer for film. It reacts with light-sensitized Ag. Br grains, converting it to black Ag. Coenzyme Q is an oxidizing agent found in the mitochondria of cells. => 12

Electrophilic Substitution of Phenols and phenoxides are highly reactive. Only a weak catalyst (HF) required for Friedel. Crafts reaction. Tribromination occurs without catalyst. Even reacts with CO 2. => 13
- Slides: 13