Benzoic Acid Plant Please click on our logo

Benzoic Acid Plant Please click on our logo or any link in this presentation to be redirected to our website, email or other documentation. Thank You! 17 A Marlen Drive w Hamilton, NJ 08691 w USA Tel: (609) 586 -8004 w Fax: (609) 586 -0002 www. ippe. com
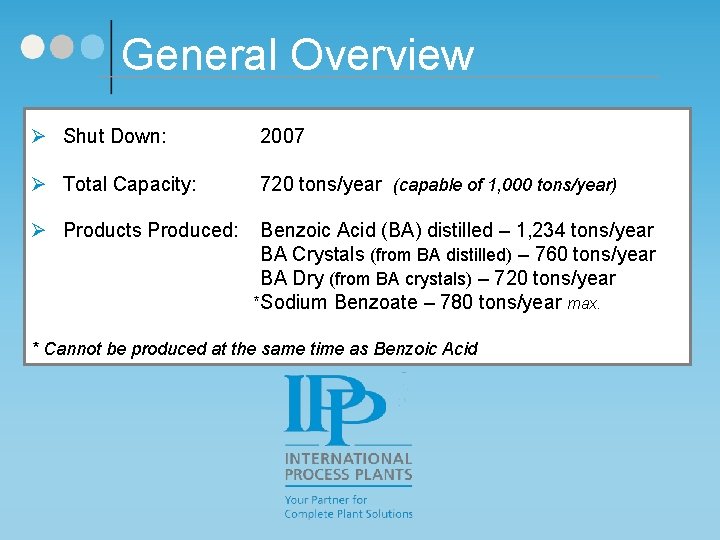
General Overview Ø Shut Down: 2007 Ø Total Capacity: 720 tons/year (capable of 1, 000 tons/year) Ø Products Produced: Benzoic Acid (BA) distilled – 1, 234 tons/year BA Crystals (from BA distilled) – 760 tons/year BA Dry (from BA crystals) – 720 tons/year *Sodium Benzoate – 780 tons/year max. * Cannot be produced at the same time as Benzoic Acid
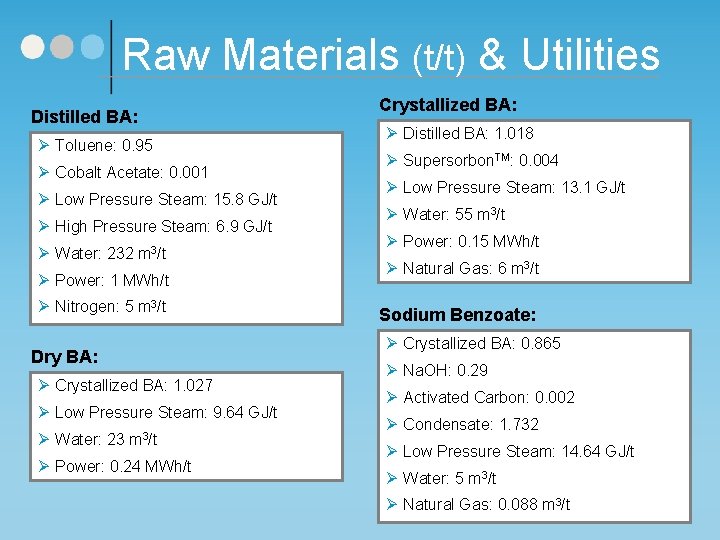
Raw Materials (t/t) & Utilities Distilled BA: Ø Toluene: 0. 95 Ø Cobalt Acetate: 0. 001 Ø Low Pressure Steam: 15. 8 GJ/t Ø High Pressure Steam: 6. 9 GJ/t Ø Water: 232 m 3/t Ø Power: 1 MWh/t Ø Nitrogen: 5 m 3/t Dry BA: Ø Crystallized BA: 1. 027 Ø Low Pressure Steam: 9. 64 GJ/t Ø Water: 23 m 3/t Ø Power: 0. 24 MWh/t Crystallized BA: Ø Distilled BA: 1. 018 Ø Supersorbon. TM: 0. 004 Ø Low Pressure Steam: 13. 1 GJ/t Ø Water: 55 m 3/t Ø Power: 0. 15 MWh/t Ø Natural Gas: 6 m 3/t Sodium Benzoate: Ø Crystallized BA: 0. 865 Ø Na. OH: 0. 29 Ø Activated Carbon: 0. 002 Ø Condensate: 1. 732 Ø Low Pressure Steam: 14. 64 GJ/t Ø Water: 5 m 3/t Ø Natural Gas: 0. 088 m 3/t

Process Description Summary of Operations I. Sodium Benzoate (BS) BS produced by the neutralization of benzoic acid and sodium hydroxide. A water solution of Na. OH is added with constant mixing to the water suspension of Benzoic Acid (BA). The solution is then bleached with Carborafíne, filtered at vacuum with a nutsche filter and after the final control of density and acidity, dried with a drum dryer. Dried BS is scraped to the Turk feeder and delivered to the elevator. The BS falls from the elevator to the storage bin and is packaged.

Process Description Summary of Operations II. Benzoic Acid Crystals The Benzoic Acid (BA) Crystals are produced by cleaning the Distilled BA which contains 0. 5 -1. 5% additive compounds. The principles of production are to remove all the additives by extraction, stripping by water steam, adsorption at activated carbon, washing and drying of the crystals. First the Distilled BA is mixed with the parent lye and then separated into a water solution of BA and recurrent BA. After extraction, most of the BA is in the saturated water solution which is then stripped by the water steam. In this process the volatile compounds are separated. After condensation and separation of water, the compounds are burned. The solution of BA, is then purified on the activated carbon where the compounds with higher boiling point are held. The activated carbon is regenerated by boiling water steam. After adsorption, the BA Crystals are crystallized, centrifuged and dried or processed to the production of the dried BA dried and Sodium Benzoate.

Process Description Summary of Operations III. Benzoic Acid Distilled In high pressure and high temperature reactors, the Distilled BA is produced by oxidation of toluene and catalyzed by cobalt acetate. It is an exothermic reaction where reaction heat is - 439. 103 J/mol. It is a radical mechanism reaction. Besides the main products of BA and water, there are many by-products like benzaldehyde, benzyl alcohol, diphenyl, o-, m-, and p-methyldiphenyl, aliphatic acids – formic acid, acetic acid and esters. Oxidation is made in 3 high pressure continuous reactors under the conditions of overpressure 1. 1 -1. 3 MPa and temperature of 160°-190°C. Toluene, recurrent toluene, (1) catalyst and air are brought into the first reactor; recurrent toluene and air into the second; and air into the third. Heat from the reaction is conducted away mostly by circulation of the reaction compound through the exchanger which is cooled by boiling water and vaporization of toluene in the reactors. For start-up, there is a heating of high pressure steam in the reactors.

Process Description Summary of Operations III. Benzoic Acid Distilled continued Reaction gas is degassed from each reactor to the coolers where the toluene and reaction water condensate. The condensate is separated from the reaction water which is drained out of the reactor; toluene is returned back to the first and second oxidation reactors. After passing through all three reactors, the oxidation compound is separated by distillation. At first the toluene and part of the benzaldehyde are distillated at atmospheric pressure in the evaporator. Distillation of toluene and benzaldehyde is finished in the first rectification column. The second reactor, separation of other parts like compound of diphenyl derivates and low boiling esters occurs. Distillates from the first and second column of the evaporator 35 are returned to the oxidation. In the third rectification column BA is distillated from high boiling parts which are separated in the evaporator. The distillate which is returned to the previous rectification and distillate residue which is processed to the efficient waste.

Process Flow

Process Flow

Process Flow

Process Flow

Process Flow

Photos

Photos

Photos

Photos

Photos

Contact Information MICHAEL JOACHIM DIRECTOR, PLANTS DEPT. Tele: 609 -838 -5930 (direct) Mobile: 609 -516 -9107 Michael. J@ippe. com SANJEEV REGE VP GLOBAL PLANT SALES Tele: 609 -838 -5938 (direct) Mobile: 609 -510 -2616 Sanjeev. R@ippe. com
- Slides: 18