Benzene and Aromatic Compounds Chem160 9162020 D r

Benzene and Aromatic Compounds Chem-160 9/16/2020 D r. Seemal Jelani 1
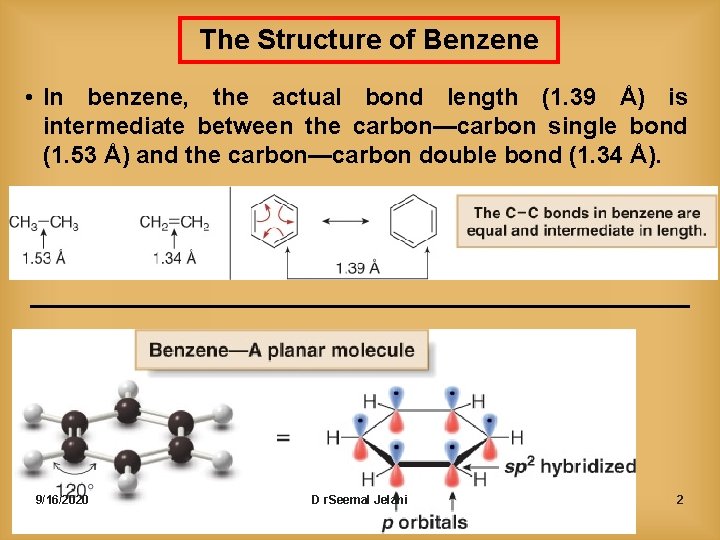
The Structure of Benzene • In benzene, the actual bond length (1. 39 Å) is intermediate between the carbon—carbon single bond (1. 53 Å) and the carbon—carbon double bond (1. 34 Å). 9/16/2020 D r. Seemal Jelani 2

Interesting Aromatic Compounds • Benzene and toluene, the simplest aromatic hydrocarbons obtained from petroleum refining, are useful starting materials for synthetic polymers. They are also two of the components of the BTX mixture added to gasoline to boost octane ratings. • Compounds containing two or more benzene rings that share carbon—carbon bonds are called polycyclic aromatic hydrocarbons (PAHs). Naphthalene, the simplest PAH, is the active ingredient in mothballs. 9/16/2020 D r. Seemal Jelani 3

9/16/2020 Reagent Cyclohexene KMn. O 4 oxidation no reaction Br 2/CCl 4 addition no reaction HI addition no reaction H 2/Ni reduction no reaction D r. Seemal Jelani Benzene 4

Benzene + 3 H 2, Ni, room temp. NR Benzene + 3 H 2, Ni, 200 o. C, 1500 psi cyclohexane Although highly unsaturated, benzene does not react like alkenes, dienes, cycloalkenes, or alkynes (addition reactions) rather it undergoes substitution reactions instead. 9/16/2020 D r. Seemal Jelani 5

Reactions of benzene: 1. Nitration C 6 H 6 + HNO 3, H 2 SO 4 C 6 H 5 NO 2 + H 2 O 2. Sulfonation C 6 H 6 + H 2 SO 4, SO 3 C 6 H 5 SO 3 H + H 2 O 3. Halogenation C 6 H 6 + X 2, Fe C 6 H 5 X + HX 4. Freidel-Crafts alkylation C 6 H 6 + RX, Al. Cl 3 C 6 H 5 R + HX substitutions 9/16/2020 D r. Seemal Jelani 6

e) From X-ray, all of the C—C bonds in benzene are the same length and intermediate in length between single and double bonds. C—C single bonds 1. 50 Å C = C double bonds 1. 34 Å The bonds in benzene are all equal and 1. 39 Å but 1, 3, 5 -cyclohexatriene has three double bonds and three single bonds! 9/16/2020 D r. Seemal Jelani 7

Key Ideas on Benzene Unusually stable heat of hydrogenation 150 k. J/mol lower than a cyclic triene Planar hexagon: bond angles are 120° carbon–carbon bond lengths 139 pm Undergoes substitution not addition Resonance hybrid One more important factor is the number of electrons in the cyclic orbital 9/16/2020 D r. Seemal Jelani 8

Aromaticity E Huckel (1931) Aromaticity is a property of certain molecules Chemistry would be similar to benzene Meet the following criteria Planar Mono cyclic system Conjugated pi system Contains 4 n + 2 electrons Can apply rules to variety of compounds and determine aromatic nature. 9/16/2020 D r. Seemal Jelani 9
9/16/2020 D r. Seemal Jelani 10
9/16/2020 D r. Seemal Jelani 11

Aromaticity and the 4 n + 2 Rule Huckel’s rule, based on calculations – a planar cyclic molecule with alternating double and single bonds has aromatic stability if it has 4 n+ 2 electrons (n is 0, 1, 2, 3, 4) For n=1: 4 n+2 = 6 benzene is stable and the electrons are delocalized 9/16/2020 D r. Seemal Jelani 12

For example, the benzene molecule, which has 3 π bonds or 6 π electrons, is aromatic. Number of π electrons = 4 n + 2 6 = 4 n + 2 n = 1 9/16/2020 D r. Seemal Jelani 13

For n = 0, we get (4 × 0 + 2) = 2 For n = 1, we get (4 × 1 + 2) = 6 For n = 2, we get (4 × 2 + 2) = 10 For n = 3, we get (4 × 3 + 2) = 14 For n = 4, we get (4 × 4 + 2) = 18 9/16/2020 D r. Seemal Jelani 14

Molecules that have the 3 characteristics listed above (cyclic, conjugated, flat) and have this number of π electrons [4 n +2] will be aromatic. The letter “n” is not a characteristic of the molecule! 9/16/2020 D r. Seemal Jelani 15
- Slides: 15