BENIGN DISEASES OF THE UTERUS AND CERVIX Rukset

BENIGN DISEASES OF THE UTERUS AND CERVIX Rukset Attar, MD, Ph. D Department of Obstetrics and Gynecology

Benign Disorders of The Uterine Servix n Congenital Anomalies of the Cervix n n n Cervical agenesis İncomplete Mullerian fusion Failure of resorption Cervical Anomalies due to DES Cervical Injuries n n n Laserations Perforations Ulcerations Cervical stenosis Annular detachment

Benign Disorders of The Uterine Servix n Cervical Infections n n Acute cervicitis Chronic cervicitis Granulomatous Infections of the cerviks n Tbc Rare Infections n Lymphogranuloma venereum n Cervical actinomycosis n Schistosomias of the cervix n Echinococcal cysts

Benign Disorders of The Uterine Servix n Cystic Abnormalities n n Nabothian cyts Mesonephric cysts Cervical Stenosis Benign Neoplasms n n microglandular hyperplasia of the endocervical mucosa cervical polyps papillomas leiomyomas

Congenital Anomalies of Cervix n n Cervix develops from paramesonephric ducts in the sixth week of gestation After fusion of the two Mullerian ducts in the midline there is resoption of the septum In the absence of paramesonephric ducts there is agenesis of cervix and uterus Cervical agenesis n n n Failure of Mullerian duct canalisation Abnormal epithelial proliferation after canalisation Usu with the absence of uterine corpus and upper vagina

Congenital Anomalies of Cervix n Incomplete Mullerian Fusion n Uterine didelphys ( 2 separate uterine horns) Bicornuate uterus ( due to partial incomplete fusion) Renal abnormalities are seen in 20 -30% of women with Mullerian defects

Cervical Abnormalities Due To DES n n n DES is a nonsteroid estrogen Cervical abnormalities Infertility At risk for miscarriage, ectopic pregnancy, premature delivery Cervical incompetency
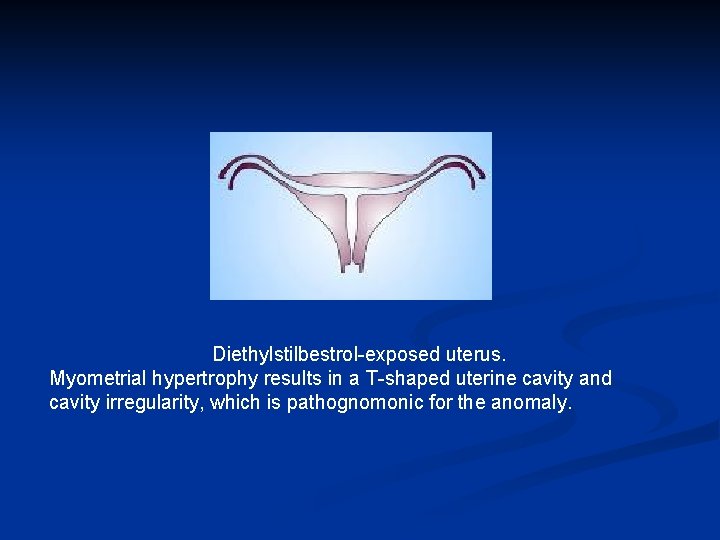
Diethylstilbestrol-exposed uterus. Myometrial hypertrophy results in a T-shaped uterine cavity and cavity irregularity, which is pathognomonic for the anomaly.

Cervical Injuries n Lacerations n n n Vaginal Delivery (most common at the 3 and 6 o'clock positions ) D&C n particularly on the postmenopausal patient n Preop use of laminaria or misoprostol may reduce it Resecting LOOPin hysteroscopic surgery , rollerball (during hysterescopy) Excessive traction of the ant lip of cervix Perforations n n Self induced abortion Cervical dilatation Insertion of radioactive sources Conization
Cervical Injuries n Ulcerations n n n Due to vaginal pessary Due to cervical stem pessary May be due to uterine prolapse when the cervix protrudes through the vaginal introitus n Annular Detachment n Rare complication due to compression necrosis of the cervix during labor n Occurs when the ext os fails to dilate and the blood supply is compromised by pressure of the fetal head

Cervical Injuries n Cervical stenosis n n usually occurs at the level of the internal os In the premenopausal woman, it may be responsible for obstruction of menstrual flow, leading to amenorrhea and pelvic pain, and infertility A postmenopausal woman with cervical stenosis may have pyometra, requiring evacuation of uterine contents and biopsy to rule out endometrial carcinoma. Causes; n n n Cervical injury, surgical procedures such as cone biopsy, loop excision, or ablative techniques for treatment of dysplasia, and inflammatory Excision by loop diathermy tends to remove less cervical stroma and therefore is less likely to cause cervical stenosis than a cold knife cone biopsy. Radiation therapy, neoplasia, and atrophic changes are more common causes in the postmenopausal woman.

Cervical Infections n n Acute Cervicitis Chronic Cervicitis n Chlamydia trachomatis n N. gonorrhea n HSV n HPV n Trichomonas vaginalis n Candidial inf n Bacterial vaginosis

Cervical Infections n Chlamydia trachomatis n n n may infect the fetus during its passage through the birth canal, or it may ascend via the endometrial cavity to the fallopian tubes causing salpingitis, pelvic and perihepatic peritonitis. It has been implicated as the agent responsible for the Fitz. Hugh and Curtis syndrome (violin-string adhesions between the liver and the parietal peritoneum). C trachomatis and N gonorrhoeae often are coinfecting agents in the etiology of acute and chronic cervicitis and salpingitis. may be transmitted to the eyes, where it causes trachoma and inclusion conjunctivitis.

Cervical Infections n N. gonorrhea n n n common cause of cervicitis also infecting the columnar epithelium of the endocervix. The mature squamous epithelium of the adult cervix and vagina is resistant to the invading organism. As in the case of Chlamydia infections, the cervix acts as a nidus for ascending infection of the endometrium and the fallopian tubes, with upward invasion often occurring after a menstrual period and loss of the protective mucus plug.

Cervical Infections n HSV n produces cervical lesions similar to those found on the vulva. n The lesion is vesicular at first and then becomes ulcerative. n Primary infections may be extensive and severe, producing constitutional symptoms of low-grade fever, myalgia, and malaise lasting approximately 2 weeks. n Recurrences of lesser severity and duration are common. n HSV-2 in more than 90%, HSV-1 n After the initial infection has healed, the virus continues to reside in the epithelial cells of the cervix, and viral shedding occurs in asymptomatic patients. n Infection of infants during their passage through the birth canal has in women with active infection at term. n Women with antibodies to HSV-2 have a higher incidence of intraepithelial neoplasia as well as invasive malignancy (a direct etiologic link has not been established).

Cervical Infections n HPV n They are flatter and moister than the typical genital warts (condylomata acuminata) seen on the vulva and perianal skin. n In fact, they often are invisible to the naked eye, becoming visible only after application of a dilute solution of acetic acid (acetowhite epithelium) or by colposcopic examination (white epithelium, mosaicism, and coarse punctation). n More than 65 types of HPV have been identified. n Benign lesions of the cervix are associated with types 6, 11, 42, 43, 44, 53, 54, and 55, whereas types 16, 18, 31, 33, 35, 39, 45, and 56 are more often found in association with cervical intraepithelial neoplasia and invasive cancers. n Approximately one-third of women with HPV infection have coexistent cervicitis caused by other organisms. n The presence of cervicitis does not significantly affect the clinical course of HPV lesions.

Cervical Infections n n HPV infection is characterized by squamous epithelial cell enlargement, multinucleation, and the perinuclear "halo" effect of koilocytosis. The so-called "balloon cell" is almost pathognomonic of this condition. Cellular changes of mild dysplasia (low-grade squamous intraepithelial lesion [SIL]), moderate or severe dysplasia (carcinoma in situ [CIS], high-grade SIL), and even invasive cancer may be associated findings. Greatly enlarged, multinucleated cells with ground-glass cytoplasm and nuclei containing characteristic inclusion bodies are indicative of HSV infection

Cervical Infections n Complications n Cervical hemorrhage n Salpingitis n Leukorrhea n Cervical stenosis n Infertility

Granulomatous Infections Of The Cervix n Tuberculosis n n n n Always sec to disease elsewhere usu pulmonary Abdominal pain, irregular bleeding Diagnosis made by biopsy n Histologically, the disease is characterized by tubercles undergoing central caseation n such lesions may be caused by other entities such as amoebiasis, schistosomiasis, brucellosis, tularemia, sarcoidosis, and foreign body reaction Tbc bacillus must be demonstrated by acid-fast stain or culture Medical therapy or surgery (ATH+BSO after a trial of CT) Tertiary syphilis Granuloma inguinale

Rare Infectious Diseases Of The Cervix n n Lymphogranuloma venereum n A chlamydial inf and chancroid caused by Haemophilus Ducreii Cervical actinomyces n Instruments n RIA Schistosomiasis of the cervix n Involvement of pelvic and uterine veins by S. Haematobium Echinococcal cysts

Cystic Abnormalities Of The Cerviks n n Nabothian Cysts Mesonephric Cysts

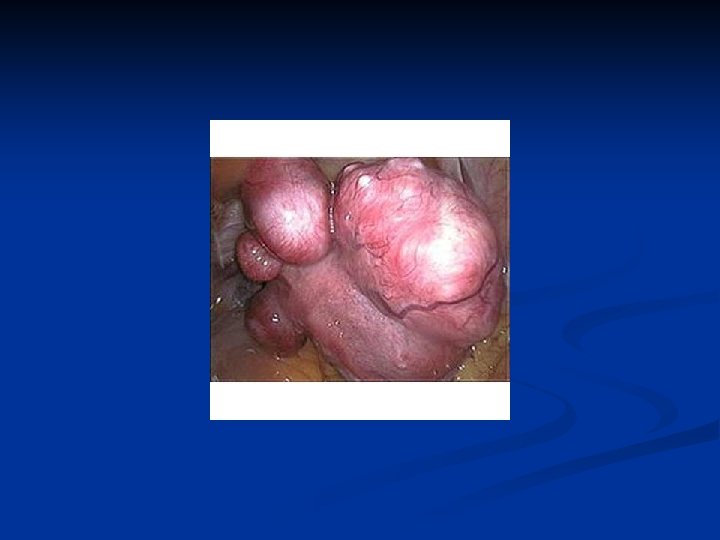
Benign Neoplasms of The Cervix n n Microglandular Hyperplasia of the Endocervical Mucosa n Due to OC, pregnancy, inflammation Cervical polyps n Arise as a result of focal hyperplasia of the endocervix n Due to chronic inflammation or abnormal responsiveness to hormonal stimulation or a localized vascular congestion of cervical blood vessels n Offen found in association with endometrial hyperplasia (hyperestrogenism may play a significant etiologic role) n malignant change is less than 1%, squamous cell carcinoma is the most common type; adenocarcinomas have been reported. n Endometrial cancer may involve the polyp secondarily. n Sarcoma rarely develops within a polyp.

Benign Neoplasms of The Cervix n Papillomas of the cerviks n n n Asympomatic There are 2 types n Solitary ( cause unknown) n Condyloma accuminata n HPV n STD Leiomyomas of the cerviks

Benign Disorders of The Uterine Corpus n n n n Congenital Anomalies of the uterine corpus( Mullerian fusion anomalies) Uterine Corpus Anomalies due to DES Leiomyoma Uteri Adenomyosis Endometrial Polyps Endometritis Asherman Syndrome

Congenital Anomalies of Uterine Corpus n Incomplete Mullerian Fusion n n Uterine didelphys ( 2 separate uterine horns) Bicornuate uterus ( due to partial incomplete fusion) Unicornuate uterus Arcuate uterus Renal abnormalities are seen in 20 -30% of women with Mullerian defects

Normal uterus

Arcuate uterus

Bicornuate uterus

Didelphys uterus

Unicornuate uterus

Congenital Anomalies of Uterine Corpus n Failure of Resorption n n Septate uterus If obstetric complications occur surgery has to be done
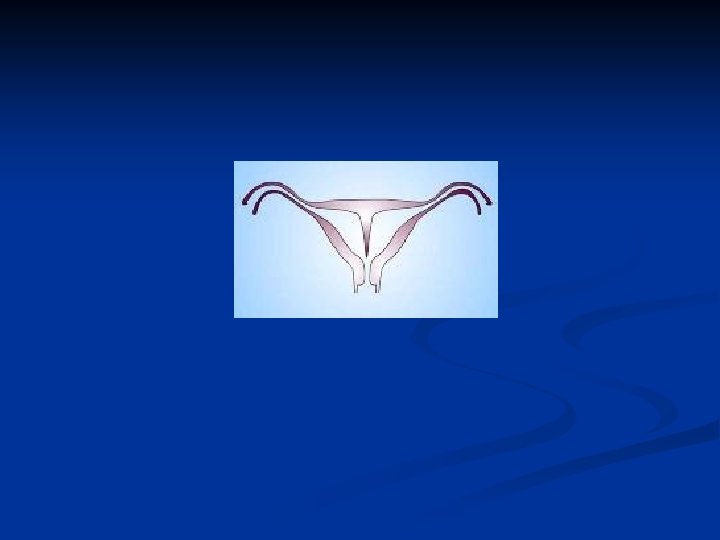

Uterine Corpus Abnormalities Due To DES n n n DES is a nonsteroid estrogen Cervical abnormalities Infertility At risk for miscarriage, ectopic pregnancy, premature delivery Cervical incompetency
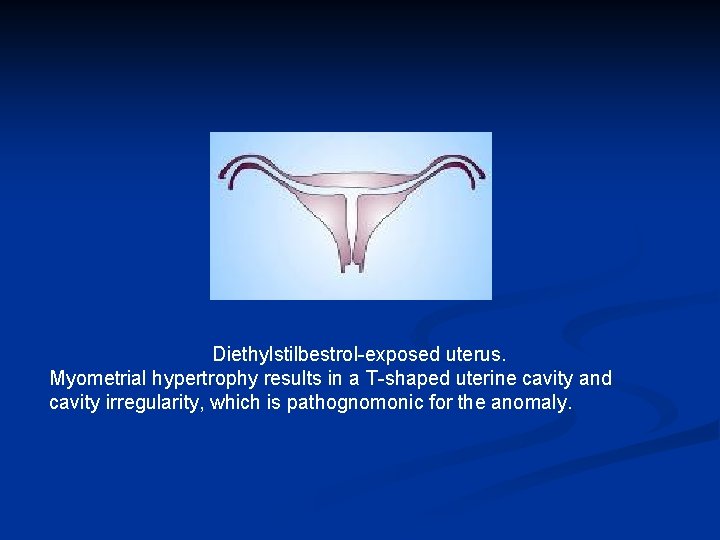
Diethylstilbestrol-exposed uterus. Myometrial hypertrophy results in a T-shaped uterine cavity and cavity irregularity, which is pathognomonic for the anomaly.

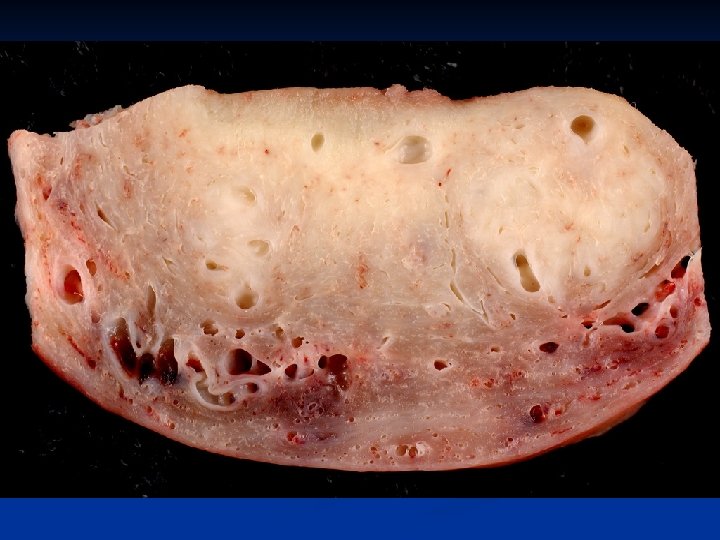
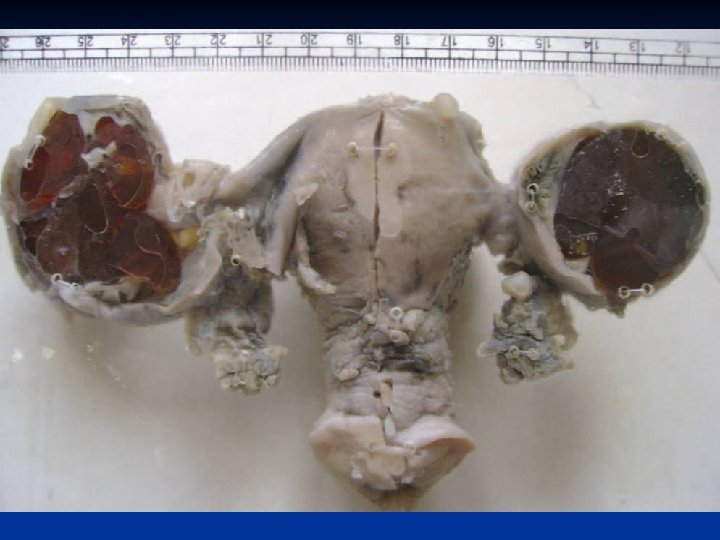
Leiomyoma Uteri n n n Benign neoplasm arising from smooth muscle cells in the uterine wall Pseudocapsule 20 -25% More commonly multiple Symptoms n n n Usually asymptomatic Metrorrhagia Menorrhagia Pain İnfertility
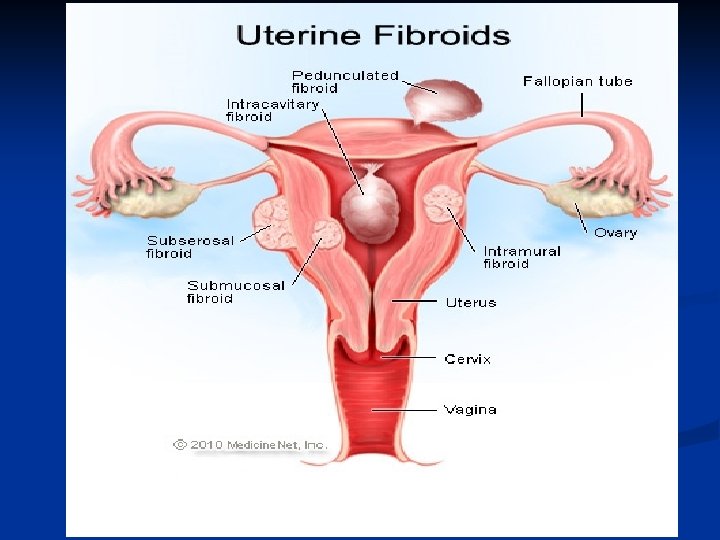
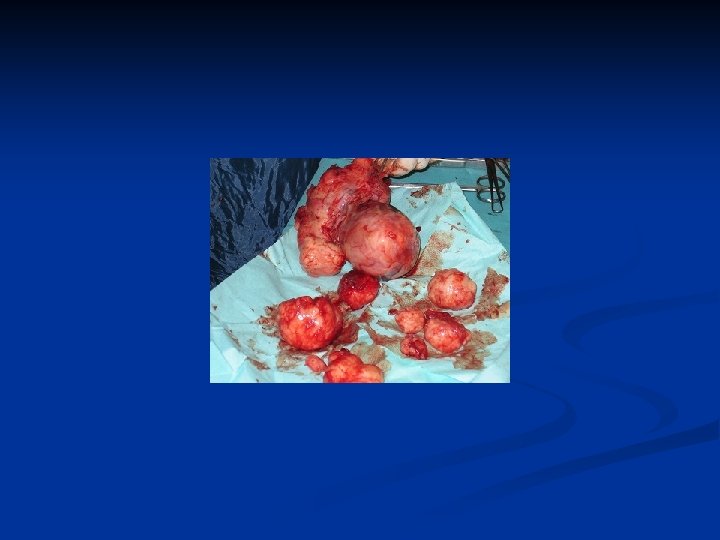

Leiomyoma Uteri n Etiology n n n Not known Unicellular in origin Estrogens are important in growth of myomas Progestins increases mitotic activity HPL may cause growth Classification n Submucous Intramural or interstitial Subserous

Leiomyoma Uteri n Changes n n n Hyalinisation Liquefaction Calcification Hemorrhage Inflammation Degeneration ( atrophic, hyaline, cystic, calcific, septic, carneous, fatty )

Leiomyoma Uteri n Clinical findings n n n Abnormal uterine bleeding 30% Pain Pressure effects n n Infertility n n n Intestinal obstruction Compress to ureters, bladder or rectum Pelvic venous compression Relationship is unknown 27 -40% of women with myomas are infertile Spontation abortion n 2 times more

Leiomyoma Uteri n Complications n Myomas and Pregnancy n n n n Rapid growth Degeneration Pain Fetal malpresentation Obstruction of the birth canal Uterine inertia Nonpregnant Women n n Heavy bleeding Urinary or bowel complication

Leiomyoma Uteri n Treatment n depends on the n n n n patients age parity symptoms general health pregnancy status reproductive plans Expectant Medical therapy n n n Gn. RHa OC Levonorgestrel releasing intrauterine device

Leiomyoma Uteri n Surgery n n n n Myomectomi Hysterectomi Uterine fibroid embolisation Endometrial ablation Myolsis Laparoscopic uterine artery occlusion MR guided focused USG surgery

Adenomyosis n n Is defined by the presence of endometrial glands and stroma within the myometrium Can be diffuse or localised ( adenomyosis) Incidence is 20% Pathogenesis is unkown n Might be sec to postpartum endometritis because of the endom line break down An arrest of Mullerian cells in the myometrium and later de nova endometrial gland development Animal models suggest PRL and FSH stimulates growth
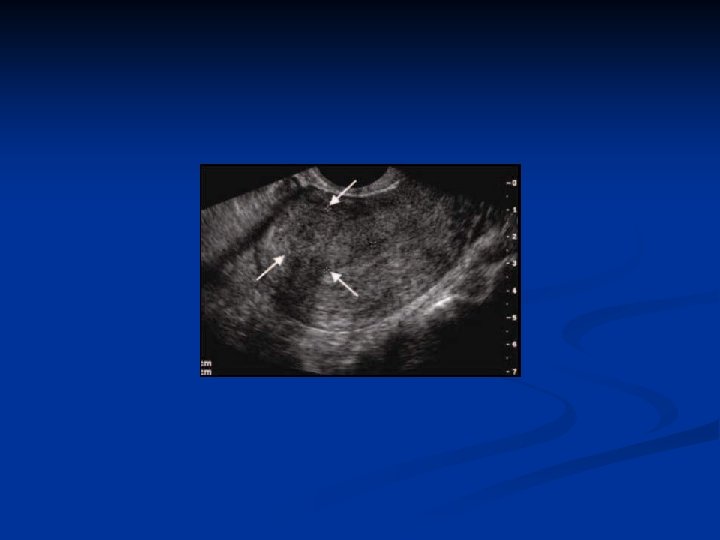

Adenomyosis n Symptoms and signs n n n Menorrhagia Dysmenorrhea Treatment n n Hysterectomi Gn. RHa

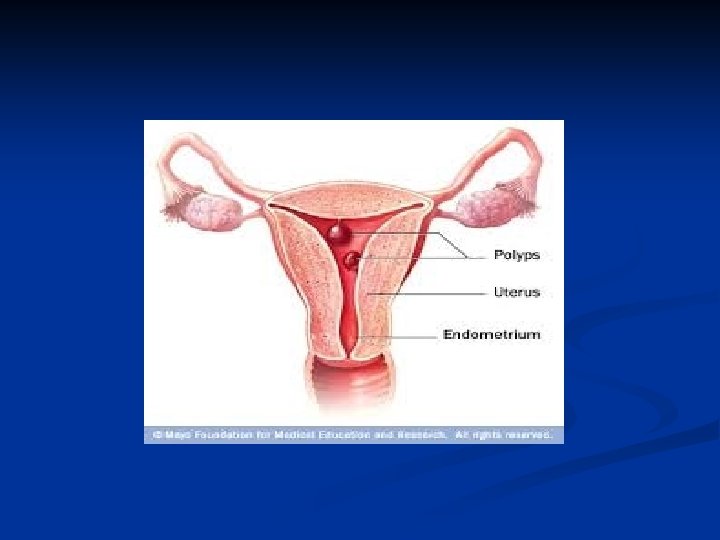
Endometrial Polyps n n Is hyperplastic growth of the endometrium Incidence increases directly with age, peaks in the fifth decade and declines with menopause 10 -24% Risk factors n n n HT Obesity Tamoxifen therapy Are considered to be estrogen sensitive Rarely undergo malignant changes

Endometrial Polyps n Clinical Findings n n n Metrorrhagia Prolapsus Staining Spotting Treatment n n n Surgical excision Gn. RHa Hysterectomi
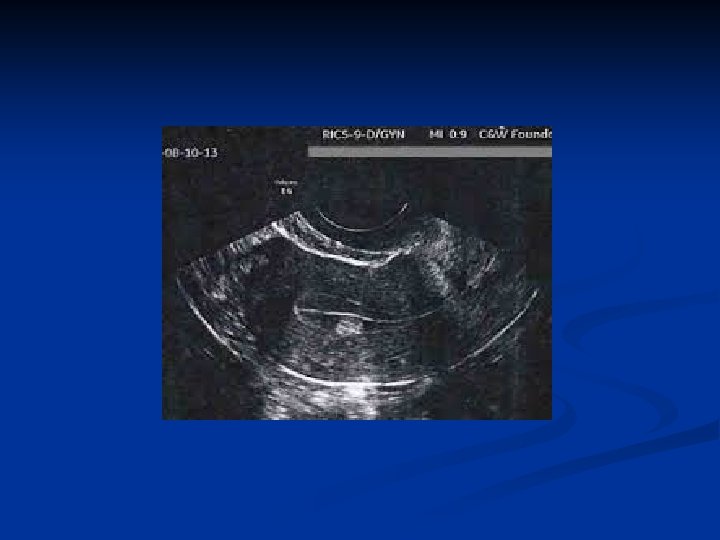
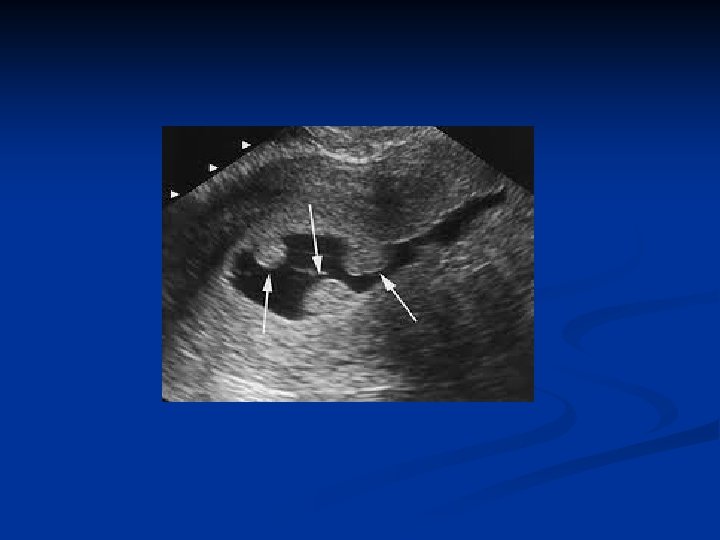
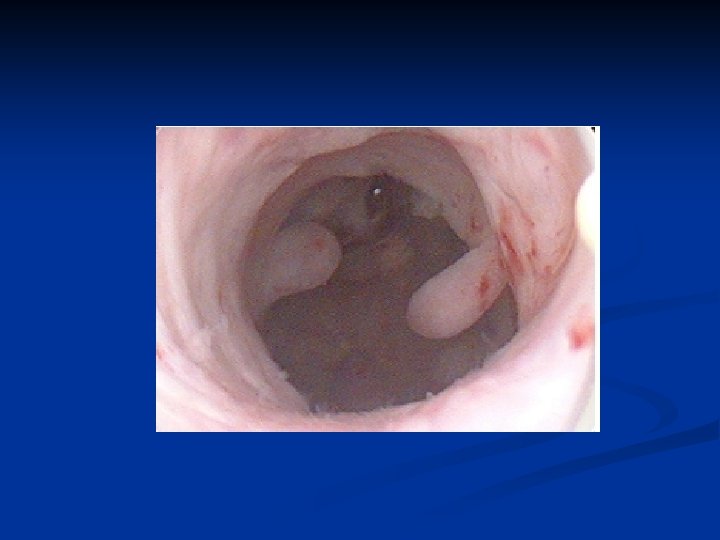
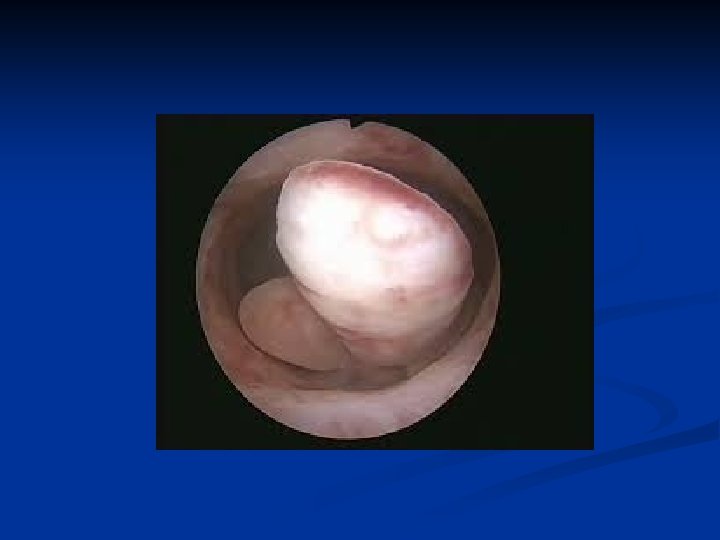

Endometritis n Symptoms n n Uterine tenderness Fever Foul smelling discharge May be chronic n n Usu postmenopause May be due to foreign body

Asherman Syndrome n n Due to recurrent D&C, abortus Sec amenorrhea, infertility, hypomenorrhea

BENIGN DISEASES OF THE OVARIES Rukset Attar, MD, Ph. D Department of Obstetrics and Gynecology

Benign Disorders of the Ovaries n n n Are common in reproductive ages Operation only when rupture and hemorrhagia occurs Physiologic Enlargement n n n Functional cysts Hyperthecosis PCO Luteoma of pregnancy Ovarian Neoplasms n n n Epithelial tumors Sex cord stromal tumors Germ cell tumors

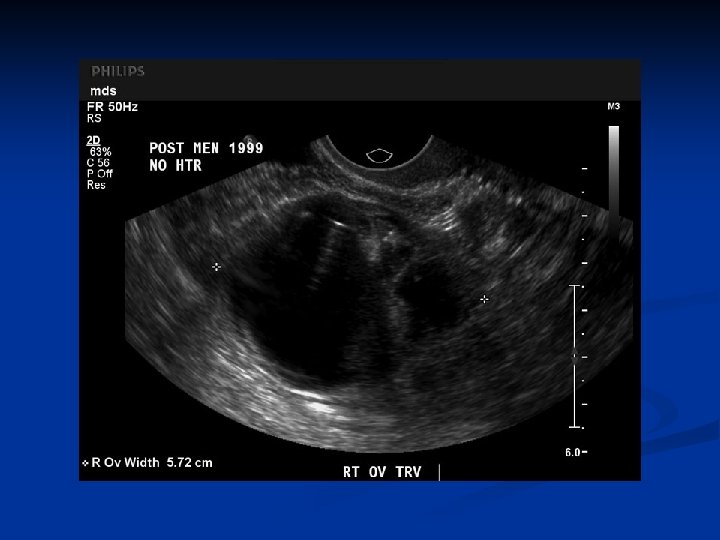
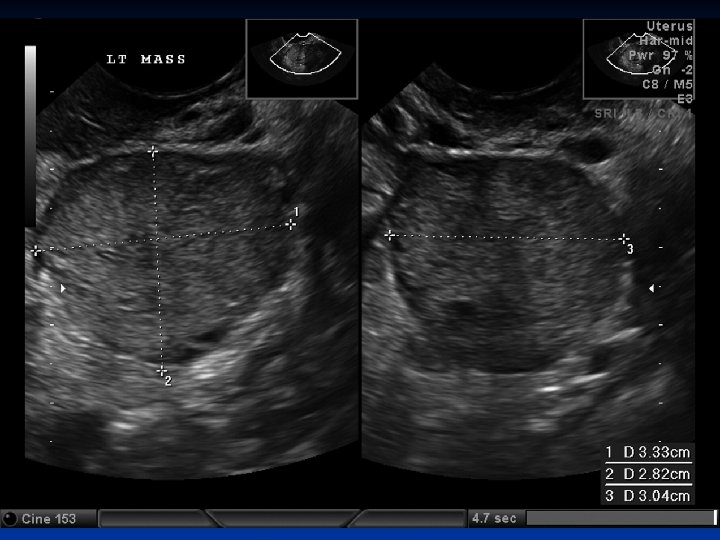
Physiologic Enlargement n Functional cysts n Follicular cysts n 3 -8 cm n Result from a failure of ovulation most likely sec to disturbances in the release of the pituitary gonadotropins n Typically asymp n May cause pain, dysparonia, AUB, bleeding and tortion n Expectant management or OC n Disappear within 60 days

Physiologic Enlargement n Endometriomas n 6 -8 n cm Corpus Luteum Cysts n 3 -11 cm n May cause pain, tenderness, amenorrhea, delayed menstruation n Regress after 1 -2 months n Symptomatic therapy unless acute complication occurs

Physiologic Enlargement n Theca Lutein Csyts n Caused by elevated levels of HCG n Hydatyform mole n Choriocarcinoma n h. CG or CC therapy n Rarely seen in pregnancy n Usually bilateral n Disappear spontaneously after termination n Surgery when complications occur

Physiologic Enlargement n Hypertecosis n n No gross enlargement of the ovary Lesions are demonstrated only by histologically In premenopausal women is associated with virilisation, and clinical findings similar to PCO n n n Characterized by persistent anovulation Enlarged ovaries Sec amenorrhea Oligomenorrhea Infertility

Physiologic Enlargement n n n Prevalance is 5 -10% 50% of patients are hirsute, 30 -75% are obese Lipid anomalies, insulin resistance, increased LH/FSH ratio

Physiologic Enlargement n Luteoma of Pregnancy n n n Often multifocal and bilateral Up to 20 cm Usually 5 -10 cm

Ovarian Neoplasms n n n Epithelial tumors Sex cord stromal tumors Germ cell tumors

Ovarian Neoplasms-Epithelial Tumors n n n 60 -80% Usually asymptomatic Usually cystic 20 -60 years of ages Types: n Serous n Mucinous n Clear-cell n Transitional cell tumors (Brenner) n Endometriod lesions

Ovarian Neoplasms-Epithelial Tumors Serous Cystadenoma n n n n In all ages 25 -30% of epithelial tumors 5 -15 cm, ≈ 10 cm, sometimes bigger usu. smaller than mucinous tm 12 -20% bilateral Age 20 s-50 s Usu unilocular İnner lining may be flat or partially covered by papillary projections; simple cuboidal epithelial cells resembling the ovarian surface epithelium – tall columnar cells resembling the fallopian tube, ciliated and secretory cells may be present

Ovarian Neoplasms-Epithelial Tumors n n n Mitoses are rare, nuclear atypia is absent Psammoma bodies (concentric calcifications) are present in 15% Multiple large calcified deposits may be visible on radiologic examination The preop workup depends on age and degree of susp for malignancy Therapy: USO / cystectomi / BSO /TAH + BSO ) They have a distinc pattern of genetic alterations

Ovarian Neoplasms-Epithelial Tumors n Mucinous Cystadenoma n n n 10 -20% Age 30 s-50 s 2 -3% bilat, the largest tumors in the body up to 50 cm 15 reported tumors weighting more than 70 kgs Usually multilocular, epithelium consists of single layer tall columnar cells that resemble a picket fence in the endocervical type or may contain goblet, argentaffin, and paneth cells in the Gİ type Arises from the surface epithelium of the ovary resembling müllerian type epithelium of the endocervix, intestinal epithelium or both of these types
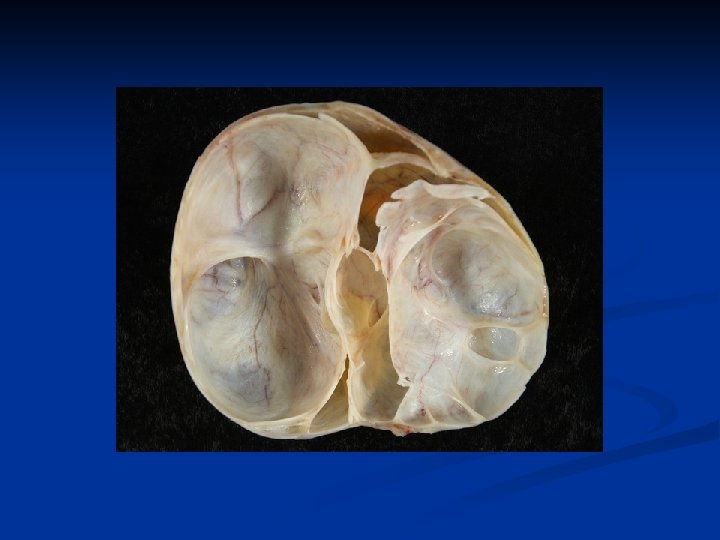

Ovarian Neoplasms-Epithelial Tumors n Mucosel of the appendix may accompany therefore apendectomy is recommended n Pseudomixoma peritoneum is a rare complication n Are asymptomatic n Presents with abdominal mass or nonspesific abdominal discomfort n In postmenopausal patients luteinisation of the stroma results in hormone secretion and endom hyperplasia n Therapy: surgery (cystectomy/ TAH + BSO) + apendectomy

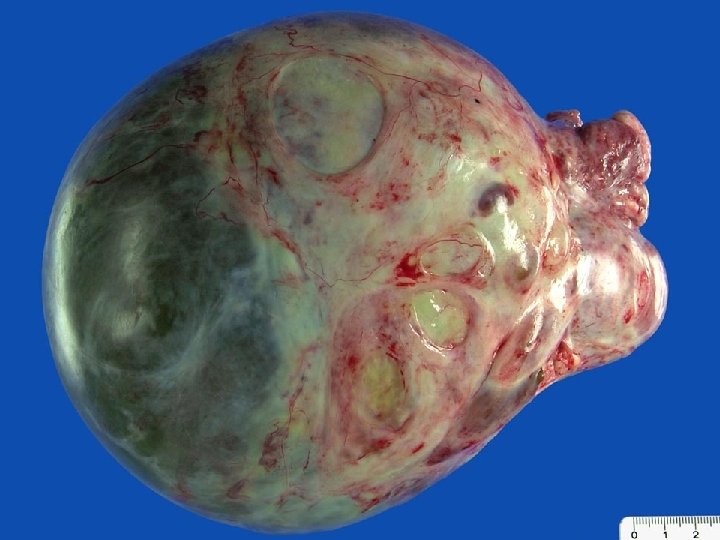
Ovarian Neoplasms-Epithelial Tumors n Cystadenofibroma n n n a variant of serous cystadenoma, containing both cystic and solid components Usu unilat Papillary-solid

Ovarian Neoplasms-Epithelial Tumors n Transitional Cell ( Brenner ) Tumors n 1 -2% n 30 s-70 s ages, mean age 50 years n Derived from surface epithelium that undergoes a metaplastic transformation to cells resemling urothelium n May occurs with mucinous cystadenoma n Most benign, but malign transformation may occur n Asymp-palpable mass, occ with bleeding due to hormonal activity n 95% unilateral, 98% are benign
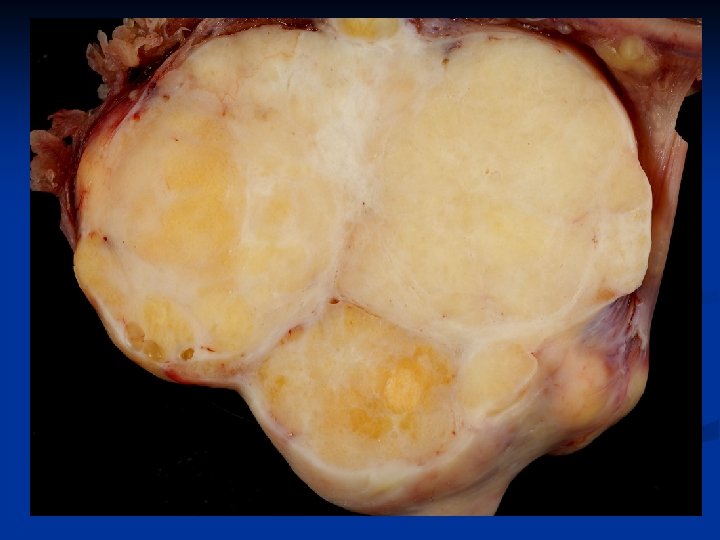

Ovarian Neoplasms-Epithelial Tumors n frequently are so small that they are incidental operative findings, but may be 5 -8 cm, max 30 cm n On section they are firm and pale yellow or white n The epithelium is composed of nests of cells with ovoid nuclei having a prominent longitudinal groove ("coffee-bean nuclei"). n Occasionally there is a mucinous metaplasia of the cells in the center of one or more of these nests, which may account for the 10% incidence of mucinous cystadenomas found associated with Brenner tumors.

Ovarian Neoplasms-Epithelial Tumors n Endometrioid Lesions n Are characterised by proliferation of benign nonspesific stroma in which endometrial-type glands may be found n Types n n Endometrioid adenofibroma Proliferative endometrioid adenofibroma

Ovarian Neoplasms-Epithelial Tumors n Clear Cell Tumors n Are rare n a solid proliferation of nonspecific stroma contains small cytologically bland glands formed by columnar cells with clear cytoplasm. n Clinically they appear like any other benign ovarian mass and are diagnosed only on histologic examination. n The prognosis is excellent

Ovarian Neoplasms-Germ Cell Tumors n n n %20 of all ovarian tumors Usually in children and young women Results from primitive germ cells (derived from embryonic yolk sac) %50 -60 of all tumors seen under age 20, %90 of prepubertal tumors 1/3 of germ cell tumors of children are malign while %4 malign in all ages cystic teratoma is %95 of all benign germ cell tumors of the ovary and %20 of all ovarian tumors

Ovarian Neoplasms-Germ Cell Tumors n Patients present with n n n Abdominal pain Pelvic or abdominal mass Ovarian rupture Hemorrhage Ovarian tortion Tumor markers: n n n AFP (endodermal sinus tm, embryonal carinoma, immature cystic teratoma, polyembryoma) β-h. CG (dysgerminoma, embryonal carinoma, polyembryoma, chorio. CA) LDH (dysgerminoma) CA 125 (endodermal sinus tm, immature cystic teratoma, dysgerminoma) CEA (endodermal sinus tm, immature cystic teratoma)

Ovarian Neoplasms-Germ Cell Tumors- WHO Classification n Germ Cell Tumors n n n n Dysgerminoma Endodermal sinus tumor ( yolk sac tumor) Teratomas Embryonal carcinoma Choriocarsinoma Polyembryoma Combinations of germ cell tumors

Ovarian Neoplasms-Germ Cell Tumors- WHO Classification n Mixt germ cell and sex cord stromal tumors Gonadoblastom n Others n n Germ cell tumors of disgenetic gonads Pure gonadal dysgenesis n Mixt gonadal dysgenesis n Turner Syndrome n Testicular feminisation n

Ovarian Neoplasms-Germ Cell Tumors n Teratomas n Mature teratomas n Solid mature teratomas n Cystic mature teratomas ( dermoid cysts ) n Cystic with malign transformation Immature teratomas n Monodermal n n Struma ovarii n Carsinoid n Others

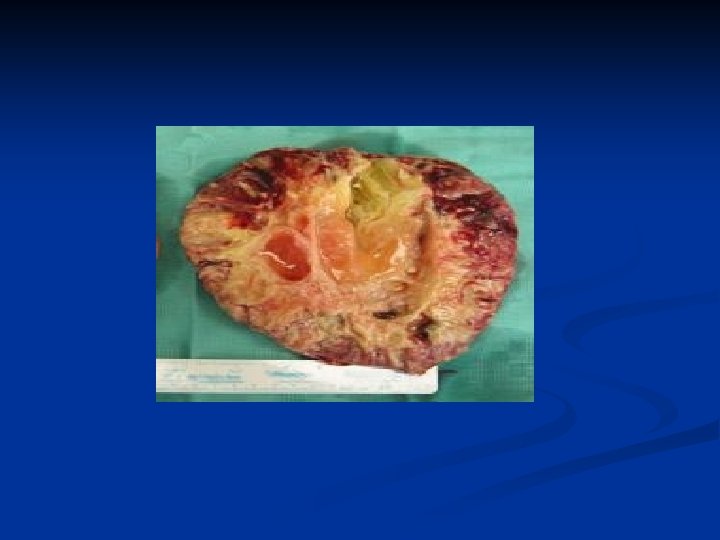
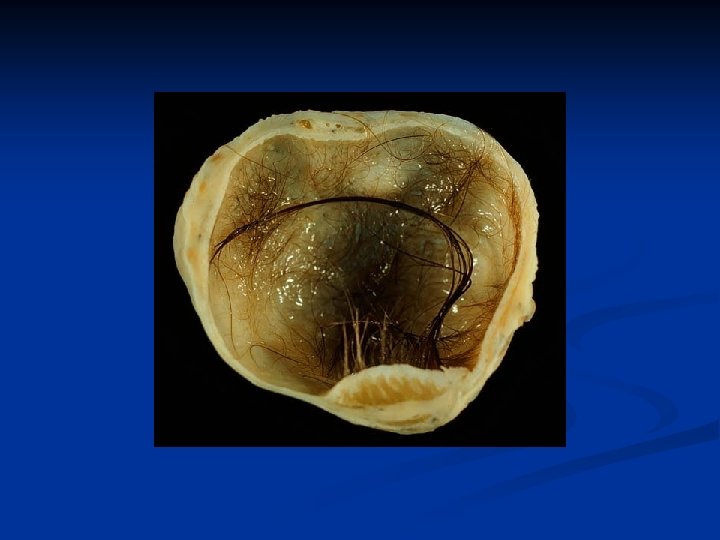
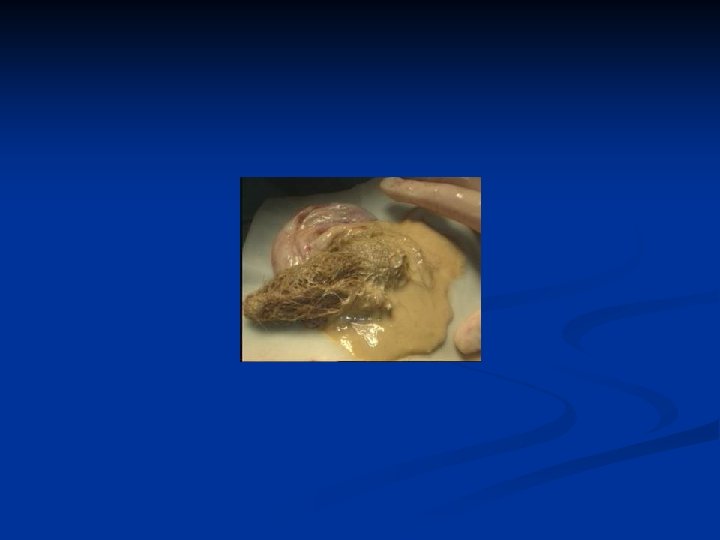
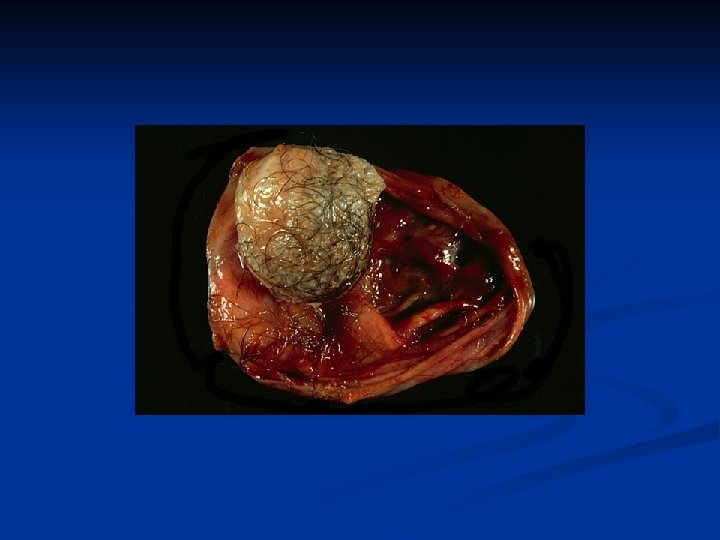
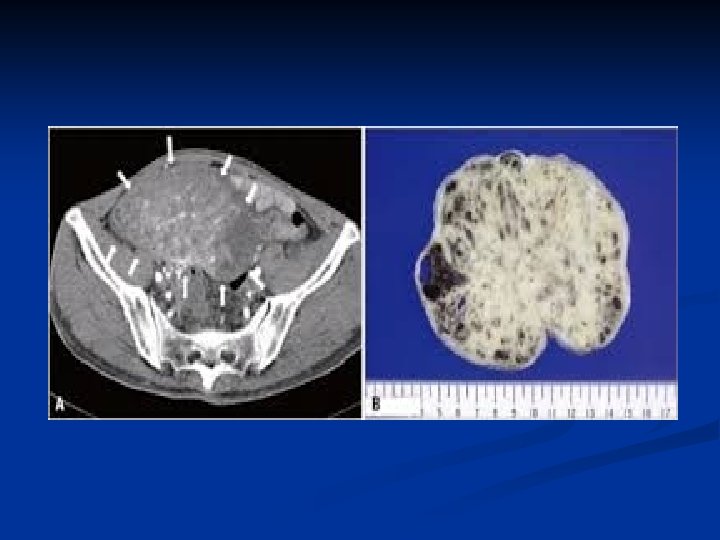
Ovarian Neoplasms-Germ Cell Tumors n Mature Teratomas n Solid n n mature teratomas Usu unilat rare n Mature n n cystic teratomas (dermoid cysts) %95 of all germ cell tumors and %20 of all ovarian tumors Derived from any of the 3 germ cell layers, well differentiated Usually asymptomatic Mostly seen in 20 -40 years of age

Ovarian Neoplasms-Germ Cell Tumors n n n %10 -15 bilateral Rarely bigger than 15 cm Usually unilocular Filled with yellowish coloured fluid with hair, tooth and bones in it There is an irregular solid nodule (Rokitansky nodule) on the inner side of the cyst which consists bones, cartilages and/or fat Complications n n Tortion ( the most)- usually in children and women Rupture ( very rare) Infection Malign transformation ( %1 -2) ( mostly squam. cell carsinoma)

Ovarian Neoplasms-Germ Cell Tumors n n n Diagnosis: TV-USG, MRI Therapy: ovarian cystectomi + examination of the contralateral ovary Immature Teratomas n n n Most common malign germ cell tumors Different from mature cystic teratomas can be seen at all ages, but usu. at reproduc. ages esp first three decades Usu grows quickly outside the capsule and causes adhesions to nearby structures and peritonel cavity implants Usu unilat, AFP may rise 9 -28 cm

Ovarian Neoplasms-Germ Cell Tumors n n n Mature cystic teratoma may co-exist in the other ovary Most common tissue is neuronal tissue Hystological grade is based on the amount of mature and immature neuroepithelial tissue, mitotic activity and differentiation n n n Grade 0: only mature tissue Grade I: little immature tissue and mitotic activity Grade II: moderate immature tissue and mitotic activity Grade III: very high immature tissue and mitotic activity Prognosis depends on the grade Ia/Grade I: explorative laparotomy + USO + staging Advanced disease : surgery + adjuvant CT

Ovarian Neoplasms-Germ Cell Tumors n Monodermal Teratomas n Struma n n n ovarii The most %1 of cystic teratomas Contains thyroid tissue (%5 of the patients present with thyrotoxicosis) Usually seen in 50 -60 years of age malign transformation is rare ( mostly follicular type, %30 metastatic disease) Therapy as in thyroid CA

Ovarian Neoplasms-Germ Cell Tumors n Carsinoid n n n Rare Usually unilateral Usually with GIS or respiratory epithelium and thyroid tissue 2 types insular and trabecular Must be consider as metastasis until proven primer ovarian Secretes bradykinin, serotonin and other peptide hormones directly into the systemic circulation ( bypassing inactivation by the first pass effect in the liver they are hormonally active)

Ovarian Neoplasms-Germ Cell Tumors n n ≈ %50 > 4 cm ≈ %50 presents with Carsinoid Syndrome n n n Episodic flushing Diarrhea Bronchospasm

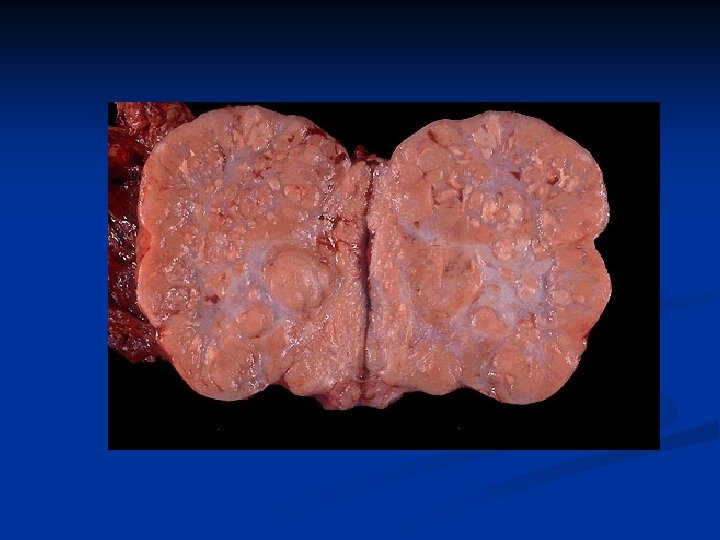
Ovarian Neoplasms-Germ Cell Tumors n Dysgerminoma n n n n is the female counterpart of the seminoma in the male. primarily in young females and accounts for app. 30– 40% of germ cell tumors. unilateral in 85– 90% of cases. It is a solid neoplasm which may contain areas of softening as a result of degeneration. Histologically, it mimics the pattern seen in the primitive gonad, with nests of germ cells that appear as large, rounded cells with central nuclei that contain one or two prominent nucleoli surrounded by undifferentiated stroma Lymphocytes may invade the stroma and occasionally giant cells are identified. A lymphocytic infiltrate is considered a favorable prognostic indicator.

Ovarian Neoplasms-Germ Cell Tumors n n n Presents with precocious puberty and primary amenorrhea %10 -15 bilat LDH is always high (therapy and follow-up) Β-h. CG is high in <%10 (therapy and follow-up) %33 of malign germ cell tumors Surgery, CT ( VAC, VBP, BEP), RT

Ovarian Neoplasms-Germ Cell Tumors n Endodermal sinus tumor (yolk sac tumor) n n n is the third most common germ cell neoplasm. If bilateral metastatic disease must be ruled out These lesions are friable, focally necrotic, and hemorrhagic. Patients commonly present with an acute abdomen. is composed of primitive epithelial cells that form architectural patterns that recapitulate the primitive gut and the primitive liver The pathognomonic finding is the Schiller-Duval body, which is a single papilla lined by tumor cells with a central blood vessel.


Ovarian Neoplasms-Germ Cell Tumors n n AFP is useful in following the response to therapy. Patterns: n n n Endodermal (Schiller Duval Body) Alveolar Alveolaglandular Polyvesiculler vitellin pattern Solid Conservative surgery + staging laparotomy followed by CT (VAC, VBP, BEP)

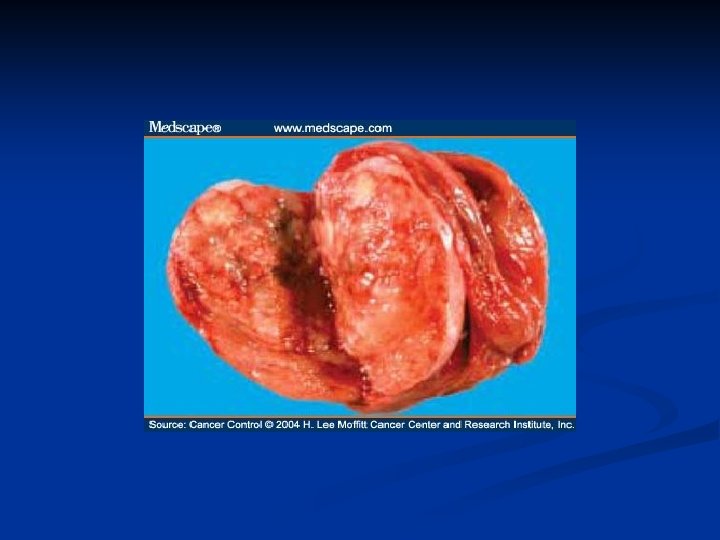
Ovarian Neoplasms-Germ Cell Tumors n Embryonal carcinoma n n n a very rare germ cell tumor a mean age of diagnosis at 15 years. The neoplasm has a highly aggressive growth pattern with early extensive spread. Usu a part of mixt germ cell tms consists of solid sheets of large polygonal cells with pale, eosinophilic cytoplasms that appear to merge together as a syncytium because the cell membranes are poorly defined. n precocious puberty, abnormal bleeding n Serum human chorionic gonadotropin (h. CG) and serum AFP levels are usually elevated. In addition, estrogens can be produced by these tumors and may serve as a serum marker. n

Ovarian Neoplasms-Germ Cell Tumors n Polyembryoma n n n n Characterised with multiple embryoid body Usu a part of mixt germ cell tms Usu at 15 years Usu unilat 10 cm-fullfills the abdominal cavity Embryonic bodies containing embryonic disc, amniotic cavity, yolk sac, extraembryonic mesenchime AFP, h. CG, sometimes h. PL

Ovarian Neoplasms-Germ Cell Tumors n Chorio. CA n n n Pure or mixt form Rare Pure: usu prepubertal Isosex precocious puberty, uterine bleeding, or amenorrhea. Microscopically, this neoplasm is composed of cytotrophoblasts, intermediate trophoblasts, and syncytiotrophoblasts. Unlike gestational choriocarcinoma, primary ovarian choriocarcinoma is associated with somewhat lower elevations of h. CG.

Ovarian Neoplasms-Germ Cell Tumors n Gonadoblastom n n n n Always in congenitally abnormal gonads with sex development disorder Most of the time they have pure or mixt gonadal dysgenesis or male pseudohermaphrodism Usu 46 XY, 45 X/46 XY mosaicism: rarely 46 XX and 45 X Female phenotype / male phenotype 4/1 Usu with dysgerminoma, sometimes with yolk sac tm, embryonal CA and chorio. CA are more common in the right ovary than in the left, usually occur during the second decade of life are found in patients with abnormal gonadal development in the presence of a Y chromosome.

Ovarian Neoplasms-Germ Cell Tumors n n n Primary amenorrhea, virilisation, abnormal genitalia Abnormal sexual development + pelvic mass= always ask for karyotype After removal of the tm menapausal symptoms such as flushing %38 bilat Bilat gonadectomy is the treatment Prognosis is good if with dysgerminoma, bad with others

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n n Sex cord or genital tubercule %5 of all ovarian tumors, ( 3 rd most) Usually hormonally active % 90 of functional ovarian tumors Hormonal activity of the tumor differs ( estrogenic, androgenic, gestational) Usually benign, rarely malignent transformation %50 of sex cord tumors is fibromas ( no hormonal activity) Symptoms: Endocrin disorders ( except fibromas)

Ovarian Neoplasms-Sex Cord Stromal Tumors Classification n Granulosa-Stromal Cell Tumors n Granulosa Cell Tumor n n n Adult Type Juvenile Type Thecoma-Fibroma Tumors n n n Thecoma Fibroma Celluler Fibroma Fibrosarcom Sclerosan stromal tumors
Ovarian Neoplasms-Sex Cord Stromal Tumors Classification n Sertoli-Leydig Cell Tumors n n n n Sertoli Cell Tumors Leydig Cell Tumors Sertoli- Leydig Cell Tumors Sex Cord Tumor with Annuler Tubulus Unclassified Gynandroblastoma Steroid-cell Tumors

Ovarian Neoplasms-Sex Cord Stromal Tumors n Thecoma n n n n 2% nonpalpable-40 cm Always unilateral, solid, lobulated and yellow, almost never malign Most common in postmenopause, rarely before 35 years Produce estrogen Abnormal bleeding or postmenopausal bleeding Occasionally with adeno. CA of endom, endom hyperplasia as a result of unopposed estrogen productıon by tumor A luteinized thecoma sometimes occurs usu in younger women

Ovarian Neoplasms-Sex Cord Stromal Tumors n Fibroma n n n n n The most frequent gonadol stromal tumor %4 of all ovarian tumors, benign At all ages esp 40 -60 years Usually 3 -15 cm, sometimes more than 20 cm Unilateral but multinodular Mostly prior to menopause, later than 40 years of age Produce no hormones May be a part of Meigs Syndrome ( pelvic mass + ascites + hydrothorax ) May be a part of basal cell carcinoma ( mesenteric cysts + dura calcification + keratocysts of the jaw )

Ovarian Neoplasms-Sex Cord Stromal Tumors n Fibroma n n Hyalinization and intercellular edema are characteristics Patients with Meigs syndrome ascites + hydrothorax resolves after resection of the tumor

Ovarian Neoplasms-Sex Cord Stromal Tumors n Granulosa Cell Tumor n n n Usually unilateral Primarily estrogenic activity Adult type n n n usually in peri and postmenapausal women %95 of granulosa cell tumors Juvenile type n n First 3 decades of life especially before puberty Causes precox puberty

Ovarian Neoplasms-Sex Cord Stromal Tumors n Granulosa Cell Tumors n Juvenile Granulosa Cell Tumors n n n %44 occurs in the first decade, %97 in the first three decades Isosex pseudopuberty precox Ollier disease (enchondromatosis) Maffucci syndrome (enchondromatosis+hemangiomas) Abnormal karyotypes with ambigious genitalia Symptoms n n Abdominal pain İcreasing abdominal girth Hemotoperitonium Occasionally it may be associated with pregnancy

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n Microsc features distinguishing juvenile from adult form are; hyperchromatism of the tumor cells, which generally lack grooves and the frequent luteinisation of theca and granulosa cells The follicules are immature Call-Exner bodies, patogonomic of adult granulosa cell tumor are rarely present Treatment USO+complete staging Survival rate for stage 1 disease >%90, yet it behaves agressively in advanced disease with a survival rate of <%50 Isolated reports of succesful treatment wıth CT, but standart CT regiments has not emerged due to the rarety of these tumors

Ovarian Neoplasms-Sex Cord Stromal Tumors n Adult Granulosa Cell Tumor n n Accounts for %1 -2 of all ovarian tumors and %95 of granulosa cell tumors Average 10 -12 cm, a pelvic mass is not always detected 50 -55 years Most of the tumors secrete esrogen manifested as n Menstrual irregularities n Menorrhagia n Amenorrhea n Metrorrhagia n Potmenapausal bleeding due to endometrial stimulation, n endometrial CA ( X 2 premenapausal women) n endometrial hyperplasia ( X 2 premenapausal women)

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n Solid, gray-white or yellow with cystic areas and hemorrhage Micr they are composed of granulosa cells, theca cells or fibroblasts or both Hystologic patterns n Microfollicular pattern with its distinctive Call-Exner bodies n Macrofollicular n Insular n Trabecular n Solid-tubular n Hollow tubular n Watered silk ( less differentitaed) n Diffuse pattern( less differentitaed)

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n A granulosa cell tumor rarely undergoes malign transformation İnhibin may be a usefull immunohistochemical marker and can be used to monitor clinical course Although most granulosa cell tumors have a very low potential for malignant behavior with 90% of tm being stage Ia they have a propensity for late recurrence up to 10 -20 years or more after initial diagnosis Full surgical staging+TAH+BSO in postmenapause. Conservative surgery for future fertility if confined to one ovary Stage I disease no difference in 5 year survival rate between conservative surgery and standard surgical staging ( 98% vs 97% )

Ovarian Neoplasms-Sex Cord Stromal Tumors n n Patients age and stage appear to be the most important prognostic factors Other prognostic factors; n n n n n Capsular rupture Tumor size Nuclear atypia Mitotic activity Histologic pattern CT efficiency? As they are rare Platinium based therapy including VBP and BEP regimens has been reported to be the most successful CT for a 5 year survival rate 50% for advanced disease Taxane-based regiments effective BEP regiment-high toxicity

Ovarian Neoplasms-Sex Cord Stromal Tumors n RT varying success n Recurrent disease surgery followed by CT may offer the best model

Ovarian Neoplasms-Sex Cord Stromal Tumors n Sertoli-Leydig Cell Tumors n n n n Named as androblastoma or arrhenoblastoma in the past, also named as Sertoli-stromal tumors Usually in reproductive ages (20 -30 years) Usually unilateral Hormone production is not always associated with these tumors Usually has androgenic activity (some has no hormonal activity or some has estrogenic activity) May result in masculine or feminine phenotypes if E or P is produced Stage and tumor differentiation seem to be important prognostic factor

Ovarian Neoplasms-Sex Cord Stromal Tumors n Leydig (Hilus) Cell Tumor n n n n n Are a subtype of Leydig cell tm originating from ovarian hilum Other type is nonhilar-type Leydig cell tumor-very rare, similar clinical and pathological features 58 years Contains Reinke crystals ( eosinophilic crystalloids) Always unilateral 0. 7 -15 cm, usually small Usually in peri and postmenopausal women Symptoms are seen after ≈ 7 years from the beginning of the tumor Abnormal menstruation, hirsutism, virilization, estrogenic manifestation

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n Almost benign Solid, soft, red-yellow Tm enlargement is usu min and tm is often undetectable Sometimes accidentally found while examinig for another pathology USO / ovarian cystectomy

Ovarian Neoplasms-Sex Cord Stromal Tumors n Sertoli Cell Tumor n n n n Mean age 27 -33, ranging from 7 to 79 years Usu present with pelvic/abdominal mass Causes precox puberty, DUB, postmenopausal bleeding, endometrial hyperplasia (estrogenic effect 2/3) Androgenic and progestogenic effects may also occur Usually unilocular, solid, yellow-brown, lobulated, ≈ 9 cm, Usu at stage I, usu benign or at early stage-cured with surgery (USO) Association with Peutz-Jeghers syndrome

Ovarian Neoplasms-Sex Cord Stromal Tumors n Sertoli-Leydig Cell Tumor n < 0. 5% n 25 years n Fewer than 10% >50 years; Fewer than 5% prepubertal n Often assoc with androgen production, however virilization develops in only 50% of patients- may be due to lack of ho production or insufficient androgen produc. n Typically patients complain of n oligomenorrhea followed by amenorrhea n Breast atrophy n Acne n Hirsutism n Temporal balding

Ovarian Neoplasms-Sex Cord Stromal Tumors Deepening of the voice ( may not resove after surgery) n Enlargement of clitoris ( may not resove after surgery) n Abdominal sweelling n Pain n Menorrhage ( due to E produc by Sertoli cell component or per androgen conversion) n Menometrorrhagia ( due to E produc by Sertoli cell component or per androgen conversion) Must be distinguished from virilisating tumors such as adrenal tumors n n

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n n Urinary 17 -KS level is normal or only slightly elevated ( it is high in adr tms) AFP may be increased and can be a used as a tm marker 12 -15 cm Tan-yellow, cystic Hemorrhage and necrosis in poorly differentiated tms Micr sertoli + leydig + undiff gonadal stromal cells with or without heterologus components in varying proportions and degrees of differentiation Intermediate or poorly diff tms are charactarized by more immature components of rhe Sertoli and Leydig cells

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n Cartilage, mucinous epithelium, skeltal mussle, and other heterologous elements are found in 20% and 25% of these tms most of which are intermediate differantiation When heterologous elements are present in poorly differentiated neoplasm the tms are clinically malignant Treatment depends on n age n tm stage, n degree of differ and n presence of heterologous elements

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n n Stage Ia + poorly differ / heterologous elements: adjuvant therapy is recommended Patients with heterologous elements or metastasis: adjuvant therapy is recommended Stage Ia + well differ: USO + staging Advanced disease or postmenapause: TAH+BSO+staging Due to the rarety of cases no standard adjuvant therapy has been accepted Treatment of advanced sex cord stromal tm, unlike germ cell tms has not met with success Platinium based therapies have the best resullts (overall survival rate 50%) – VAC, VBP; BEP As CT RT has been used successfully in limited cases

Ovarian Neoplasms-Sex Cord Stromal Tumors n Steroid Cell Tumors not Otherwise Spesified (NOS) n n n n termed lipid cell or lipoid tumors in the past Are composed entirely of cells resembling typical steroid secreting cells ( eg lutein cells, Leydig cells, adrenal cortical cells) except that specific features such as location of origin in the hilus or crystalloids of Reinke are not identified 0. 1% of all ovarian tms 43 -60 age androgenic changes occuring in 75 -90% of patients may be of many years duration E ve P changes are occationally noted Cushing syndrome may also be present accompanied by elevated cortisol levels

Ovarian Neoplasms-Sex Cord Stromal Tumors n n n E manifestation may be a result of E production, the aromatization of androgen to E 2 in adipose tissue may be more plausable Diagnosis is often dependent on clinical manifestationof virilization or the rare occasion of isosex pseudoprecocity Tm removal results in rapid resolution of most of the hormonal effects except deepening of the voice and enlargement of clitoris Yellow-orange-tan 5 -8 cm Soft, lobulated, hemorrhage, necrosis and cystic degeneration are occ observed

Ovarian Neoplasms-Sex Cord Stromal Tumors n n Microsc resemble Leydig or hilar cells, sometimes adrenocortical cells- has led to speculate that they may arise from the adrenocortical rests Rarely malignant; 10 -15% of them recur or metastasize Young stage Ia: USO Advanced disease / beyond reproductive age: TAH + BSO + +staging + removal of extraovarian disease

Ovarian Neoplasms-Sex Cord Stromal Tumors n Typical presentation Hirsutism n Virilization (%80) n Menstruation n Rarely attain palpable size n Acne Testesteron levels are high whereas androstenodione and n n DHEA levels are low

Ovarian Neoplasms-Tumors of Low Malignant Potential ( Atypical Proliferating Tumors) n n n n A group of tumors showing greater epithelial proliferation that seen benign serous cystadenoma although they are by definition noninvasive 15% of all epithelial tumors Mean age 40 years Parity, OC, breast feeding, multiple births are protective İnfertility, use of infertility drugs may increase the risk Asymptom, but usu with pelvic mass, increasing abdominal girth, or abnormal bleeding USG, CT

Ovarian Neoplasms-Tumors of Low Malignant Potential ( Atypical Proliferating Tumors) n n n n CA 125 is not always elevated Usu serous No markers The most common types are serous or mucinous The absence of stromal invasion is an absolute criterion for making the diagnosis Mean diameter is 12 cm, 33 -75% bilateral Usu cystic with mural clusters of papillary projections Mucinous are larger than serous ( 17 -20 cm), rarely bilat, multiloculated

Ovarian Neoplasms-Tumors of Low Malignant Potential ( Atypical Proliferating Tumors) n n Survival rate better than EOT with a 5 year survival rate of 95% for stage I disease 0. 8% developed invasive carcinoma Patient outcome depends on the presence or absence of micropapillary features within the ovarian tumor and invasive versus noninvasive implants 5 and 10 year survival rates with micropapillary ca are 81% and 71%; serous borderline tms without invasive implants > 98% and with invasive implants 60% and 70%

Ovarian Neoplasms-Tumors of Low Malignant Potential ( Atypical Proliferating Tumors) n The risk of developing invasive disease; n n n Stage of disease Extraovarian implant status Stromal invasion in the primary tumor Micropapillary architecture Micropapillary serous CA n n is characterised by thin, elongated micropapillae with min or no fibrovascular support arising directly from thick, more centrally located papillary structures Mitotic activity may be seen in some of the cases 1 -3 figures/10 high power field
- Slides: 151