BELLWORK GET READY FOR BELLWORK 1 12 17

BELLWORK GET READY FOR BELLWORK! 1 -12 -17

1. When elements form mixtures, the elements A. keep their original properties. B. react to form a new substance with new properties. C. combine in a specific mass ratio. D. always change their physical state.
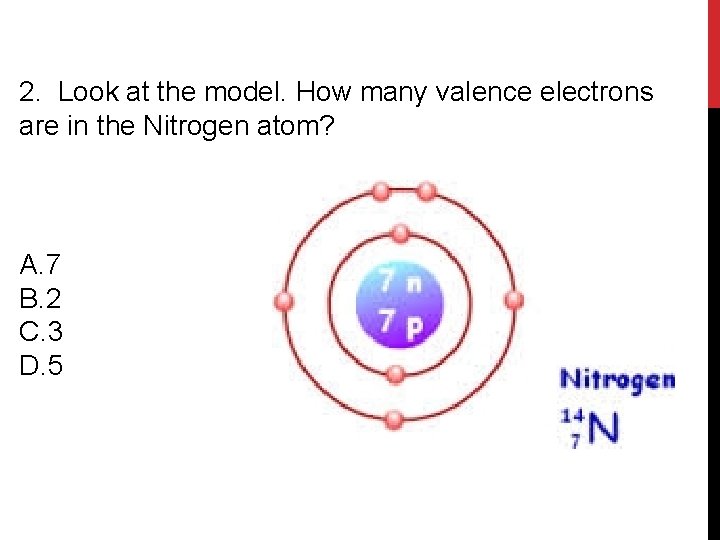
2. Look at the model. How many valence electrons are in the Nitrogen atom? A. 7 B. 2 C. 3 D. 5

3. What is the maximum number of electrons that can be in an atom’s 3 rd energy shell? a. 2 b. 18 c. 24 d. 8

4. What do salt, sugar, baking soda , and water all have in common? A. B. C. D. They are all compounds. They are all solids. They are all mixtures. They are all elements

5. A substance has a mass of 3 grams (g) and a volume of 1 cubic Centimeters (cm 3). What is the density of the substance? A. 3 g/cm 3 B. 30 g/cm 3 C. 60 g/cm 3 D. 675 g/cm 3 Density = Mass Volume D=M V

6. All matter is made of atoms, and matter is all around you. Matter comes in 4 states: (1) Solid, (2) Liquid, (3) Gas, and (4) Plasma. If an item is not one of these four states, then it is not matter, and it is therefore not made up of atoms. Select the item below that IS NOT made of atoms. a. Plant b. Animal c. Time d. Clock

7. The atomic number is the number of ______ found inside the nucleus or the number of ______ found surrounding the nucleus of an atom. A) Protons, Electrons B) Electrons, Neutrons C) Quarks, Protons D) Neutrons, Hydrogen

VIDEO Forming Ionic Bonds

1. When elements form mixtures, the elements A. keep their original properties. B. react to form a new substance with new properties. C. combine in a specific mass ratio. D. always change their physical state.
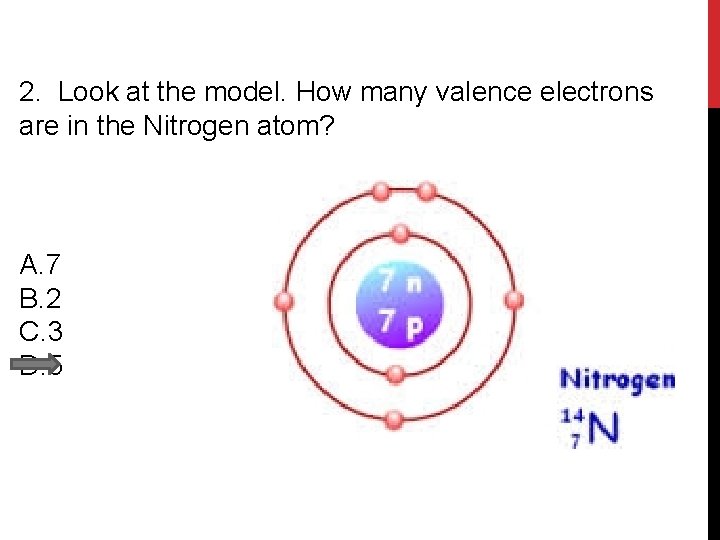
2. Look at the model. How many valence electrons are in the Nitrogen atom? A. 7 B. 2 C. 3 D. 5

3. What is the maximum number of electrons that can be in an atom’s 3 rd energy shell? a. 2 b. 18 c. 24 d. 8

4. What do salt, sugar, baking soda , and water all have in common? A. B. C. D. They are all compounds. They are all solids. They are all mixtures. They are all elements

5. A substance has a mass of 3 grams (g) and a volume of 1 cubic Centimeters (cm 3). What is the density of the substance? A. 3 g/cm 3 B. 30 g/cm 3 C. 60 g/cm 3 D. 675 g/cm 3 Density = Mass Volume D=M V

6. All matter is made of atoms, and matter is all around you. Matter comes in 4 states: (1) Solid, (2) Liquid, (3) Gas, and (4) Plasma. If an item is not one of these four states, then it is not matter, and it is therefore not made up of atoms. Select the item below that IS NOT made of atoms. a. Plant b. Animal c. Time d. Clock

7. The atomic number is the number of ______ found inside the nucleus or the number of ______ found surrounding the nucleus of an atom. A) Protons, Electrons B) Electrons, Neutrons C) Quarks, Protons D) Neutrons, Hydrogen
I CAN… Use the periodic table to determine the properties of an element. TOC #5 Ionic Bonding notes # 6 Ionic Bond Worksheet

Compounds that contain both __metals and __nonmetals__ are usually ionic. Ionic bonds will form when electrons are transferred from one atom to another atom. An atom is neutral because the number of electrons in an atom equals the number of protons. So, the charges cancel each other out. During the ionic bonding, one or more valence electrons are transferred from one atom to another. When an atom gains or loses electrons, it becomes a charged particle called an ion. Ionic bond = Ion formed

Ionic bonds form between a metal and a nonmetal. Atoms of most metals have few valence electrons and tend to lose these valence electrons and form positive ions. The valence level of nonmetal atoms is almost full. So, nonmetal atoms tend to gain electrons and become negative ions. When ions bond, they form a repeating threedimensional pattern called a crystal lattice, such as the one shown below.
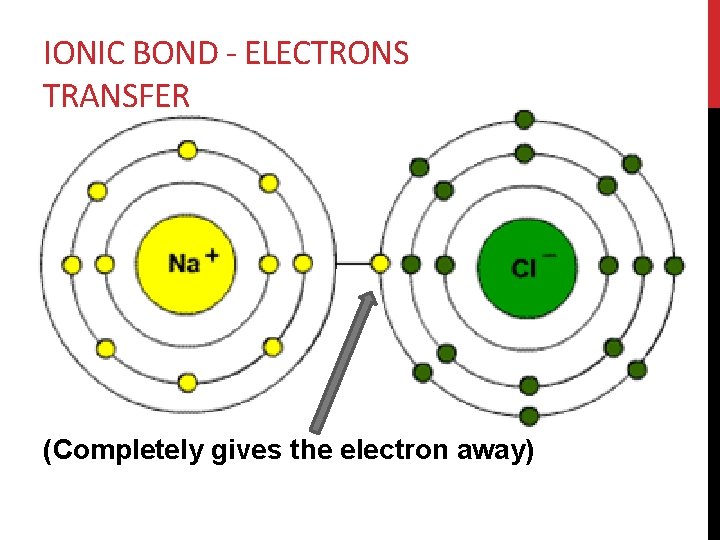
IONIC BOND - ELECTRONS TRANSFER (Completely gives the electron away)

OCTET RULE (ADD TO BOTTOM OF YOUR NOTES) Octet Rule: every atom wants to have eight valence electrons in its outermost electron shell. Atoms will gain or lose electrons to have a full outer shell (valence shell) Happy with 8 valence electrons in the outer shell!

REVIEW: FORMING POSITIVE IONS The atoms that lose electrons form ions that have fewer electrons than protons. Because the positive charges outnumber the negative charges, these ions have a positive charge.
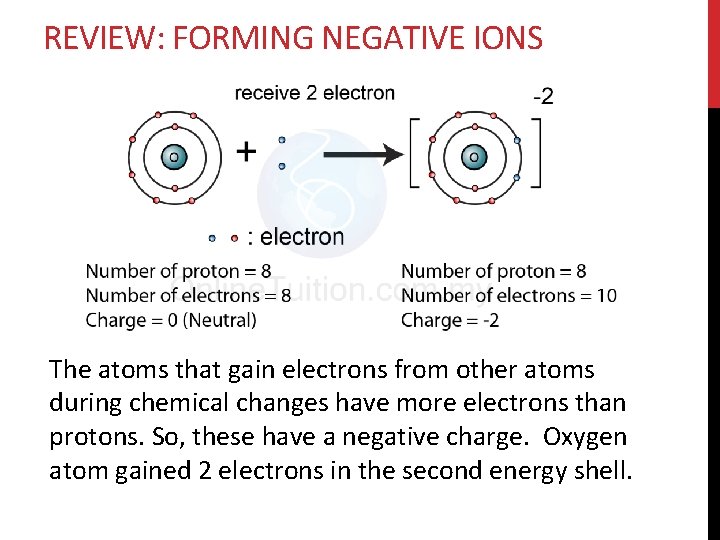
REVIEW: FORMING NEGATIVE IONS The atoms that gain electrons from other atoms during chemical changes have more electrons than protons. So, these have a negative charge. Oxygen atom gained 2 electrons in the second energy shell.

HOW DOES THE BOND BETWEEN SODIUM & CHLORINE FORM? Electron Dot Diagrams only display the valence electrons. Cl Sodium has one valence electron. It’s looking to lose it Na Chlorine has seven valence electrons. It’s looking to gain one
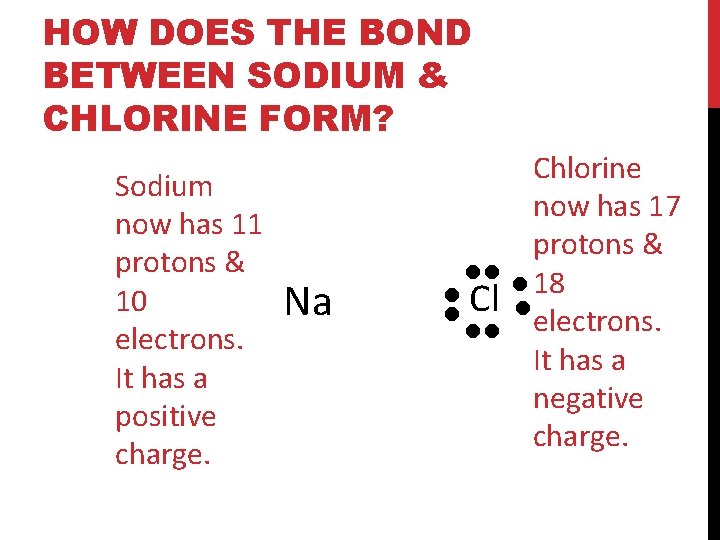
HOW DOES THE BOND BETWEEN SODIUM & CHLORINE FORM? Na Cl Sodium now has 11 protons & 10 electrons. It has a positive charge. Chlorine now has 17 protons & 18 electrons. It has a negative charge.

PROPERTIES OF IONIC COMPOUNDS (ADD TO THE BOTTOM OF YOUR NOTES) Compounds that contain both metals and nonmetals are usually ionic. e. g. Mg. O High melting point • Don’t easily change to liquids (or gases) Solids → Poor conductors Liquids → Good Conductors Shatters when breaks
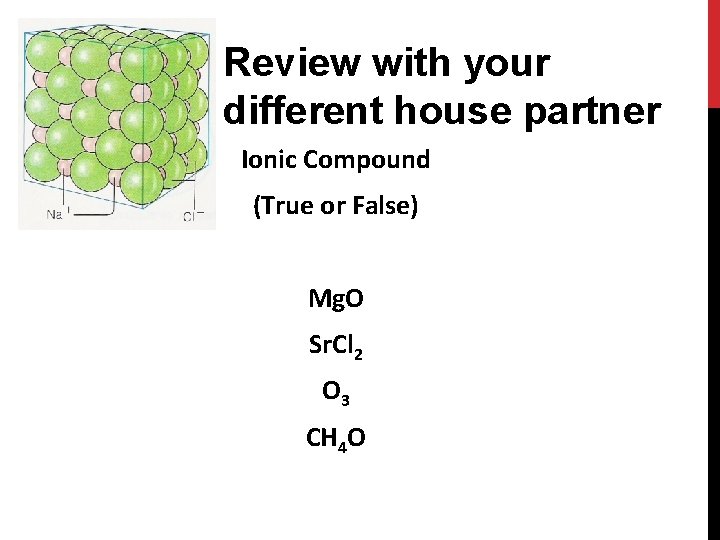
Review with your different house partner Ionic Compound (True or False) Mg. O Sr. Cl 2 O 3 CH 4 O
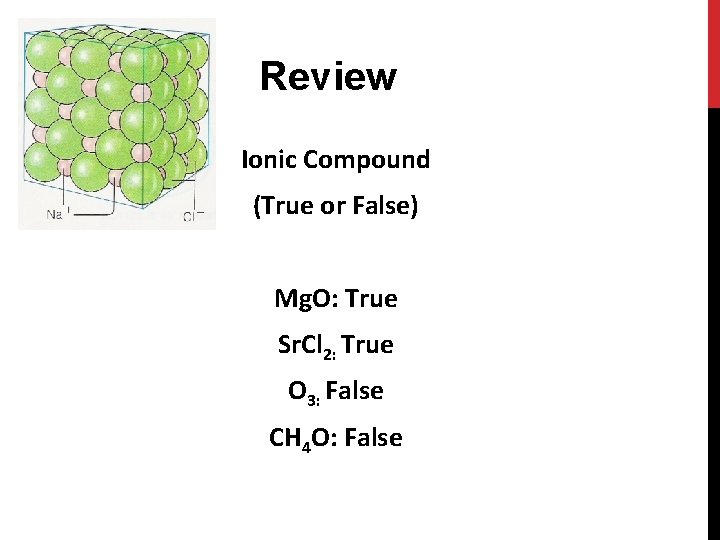
Review Ionic Compound (True or False) Mg. O: True Sr. Cl 2: True O 3: False CH 4 O: False
Group Partner: What is an ionic bond? Atoms will transfer one or more ________ to another to form the bond. Each atom is left with a ________ outer shell. An ionic bond forms between a ______ ion with a positive charge and a ________ ion with a negative charge.

Group Partner: What is an ionic bond? ELECTRONS Atoms will transfer one or more ________ to another to form the bond. COMPLETE Each atom is left with a ________ outer shell. METAL An ionic bond forms between a ______ ion with a NONMETAL positive charge and a ________ ion with a negative charge.
IONIC BONDING WORKSHEET Different # Siblings Partners: Fill in the chart below
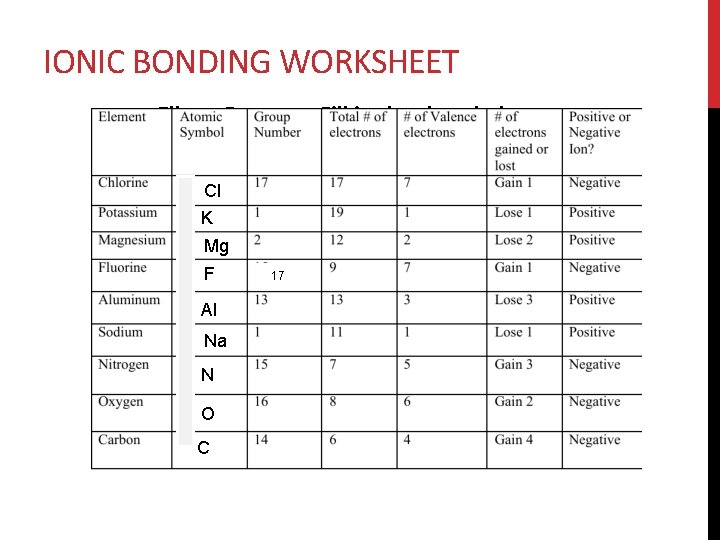
IONIC BONDING WORKSHEET Elbow Partners: Fill in the chart below Cl K Mg F Al Na N O C 17
- Slides: 32