Bellwork Get out a blank sheet of paper

Bellwork : Get out a blank sheet of paper for lab. Write down the following stations. You will need about 7 lines between each station. Station 1: Calcium chloride reacts with sodium carbonate Station 2: Methane (CH 4) burns Station 3: H 2 O 2 is in the presence of a catalyst Mn. O 2 Station 4: Hydrochloric acid reacts with solid magnesium Station 5: Magnesuim oxide reacts with water *

For each station: a)Observations b) State the type of Reaction c) Predict the products of the reaction d) Balance the equation Station 1: Calcium chloride reacts with sodium carbonate Station 2: Methane (CH 4) burns Station 3: H 2 O 2 is in the presence of a catalyst Mn. O 2 Station 4: Hydrochloric acid reacts with solid magnesium Station 5: Magnesium oxide reacts with water *
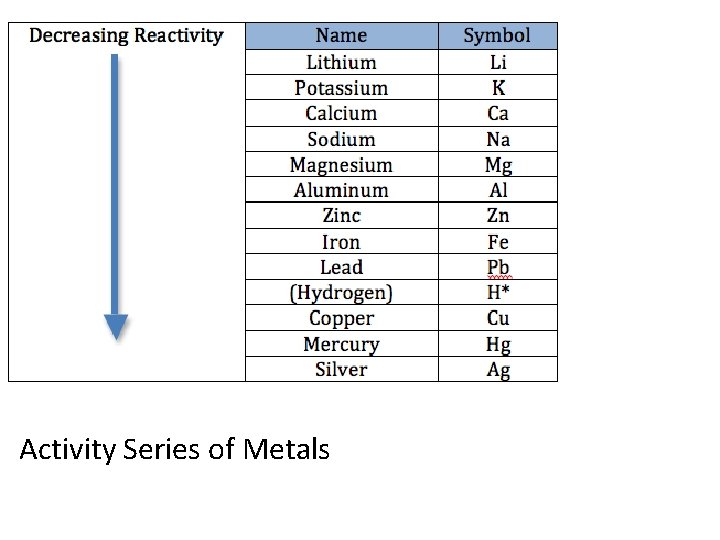
Activity Series of Metals
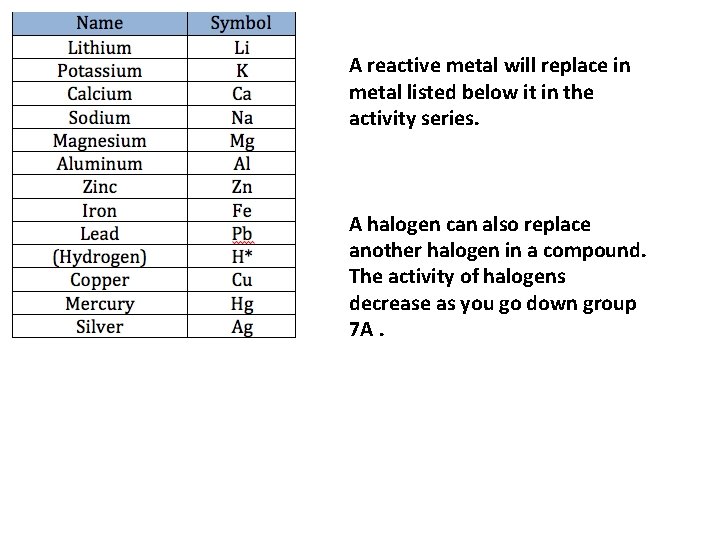
A reactive metal will replace in metal listed below it in the activity series. A halogen can also replace another halogen in a compound. The activity of halogens decrease as you go down group 7 A.

Complete and Balance 1) C 3 H 8 is totally burned. 2) Al(NO 3)3 + Ca 3) Rb. NO 3 + Be. F 2 4) Ca. O + F 2 5) Al. Br 3

Complete and Balance 1) C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O 2) 2 Al(NO 3)3 + 3 Ca(NO 3)2 + 2 Al 3) 2 Rb. NO 3 + Be. F 2 2 Rb. F + Be(NO 3)2 4) 2 Ca. O + 2 F 2 2 Ca. F 2 + O 2 5) 2 Al. Br 3 2 Al + 3 Br 2
- Slides: 6