Bellwork Fri Sept 7 On your bellwork sheet

Bellwork Fri. Sept. 7 On your bellwork sheet write “Bill Nye” and on a separate sheet of paper copy down the following: • All matter is made of ____ which are so small we can't even see them with a microscope. • Tiny _______ orbit the nucleus. • Atoms are like letters and when combined they make _______which are like words. • Different materials have unique atoms. It is the number of _______ in the nucleus that makes each materials atom unique. • _______is a fundamental building block of all life on our planet. It is so important it has its own entire branch of chemistry - organic chemistry.

Bellwork, Mon. Sept 10 In a scientific experiment, which variable is the one that is manipulated (changed) by the experimenter?

Bellwork, Tues. Sept 11 Name the 3 atomic particles & list their respective charges. **Don’t forget to pick up a sheet off my lab bench. Have out your periodic table**
Bellwork, Wed. Sept 12 How many protons, neutrons & electrons are in a neutral atom of Calcium? **Don’t forget to have out the story on the back of your periodic table and your atom drawings from yesterday**

Bellwork, Thurs. Sept 13 Name 5 elements that you already know. **Don’t forget to pick up a sheet off my lab bench. Have out your periodic table**

Bellwork, Fri. Sept. 14 Which element is LEAST likely to combine with another element to form a molecule? - Chlorine (Cl), a halogen Iron (Fe), a metal Neon (Ne), a noble gas Silicon (Si), a metalloid

Bellwork, Mon. Sept. 17 The element silicon is best used for which purpose? - as a container to keep coffee hot - as a semiconductor in a computer chip - as a material to make airplane frames - as a malleable material for coins and jewelry **I am stamping Atomic Math work from Friday**

Bell Work, Tues. Sept. 18 How can a scientist, using the periodic table, find an element with properties similar to another element? - by comparing density - by finding it’s periodic group - by comparing malleability - by comparing atomic weight

Atom ATOMS, are the basic particle from which all matter is made. They are so small they cannot be seen even with a microscope.

Elements ELEMENTS are one or more identical ATOMS and each element has its own unique atom that looks different than all the other elements.

Examples An ELEMENT is a pure substance that cannot be broken down into any other substance by chemical or physical means. Such as: • aluminum • zinc • oxygen
Subatomic Particles Each element’s atom is unique due to the number of each subatomic particle that it contains. Nucleus contains: • Protons – positive + charge • Neutrons – neutral / no charge Orbiting the nucleus are: • Electrons – negative - charge
The Periodic Table The PERIODIC TABLE contains information about all the different ELEMENTS.
Natural vs. Synthetic The majority of the known elements are naturally occurring, however all elements above 92, are SYNTHETIC meaning they were created in a lab.
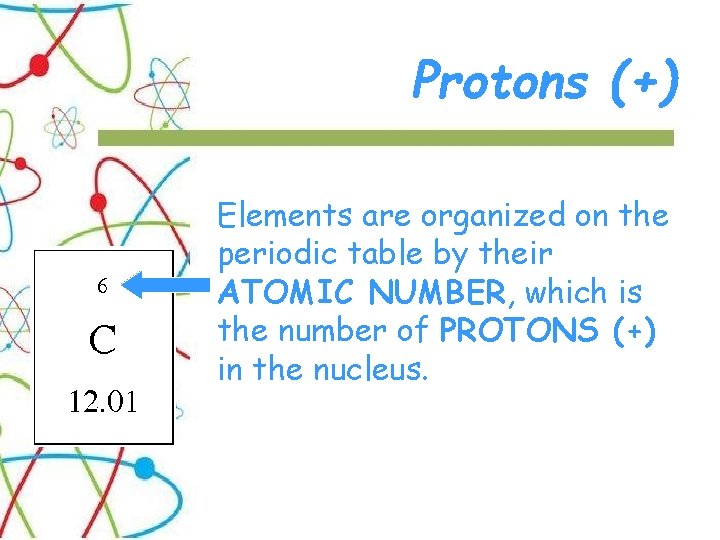
Protons (+) Elements are organized on the periodic table by their ATOMIC NUMBER, which is the number of PROTONS (+) in the nucleus.

Electrons (-) One of an atom’s main goals in life is to remain neutral. Since protons have a positive charge, it would need to have the same number of negative charges (electrons) to remain neutral. The ATOMIC NUMBER, therefore tells us the number of electrons in a neutral atom as well. 5 Positive + -5 Negative = 0 Neutral

Isotopes Atoms of the same element can have different numbers of NEUTRONS; the different possible versions of which are called ISOTOPES.
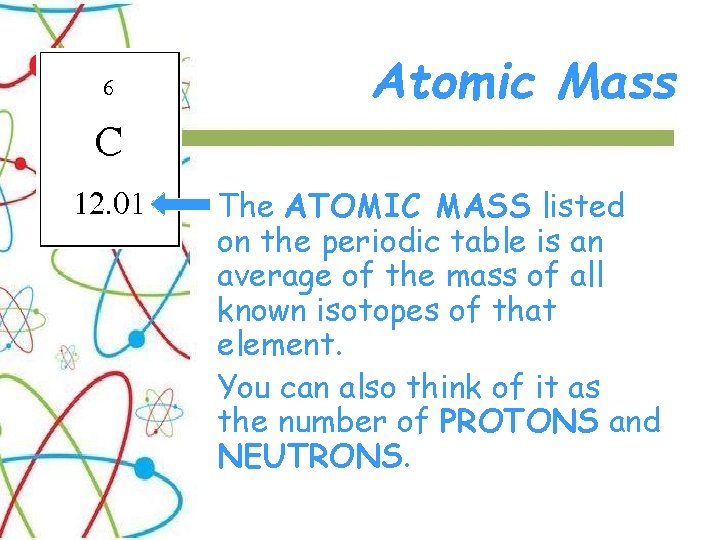
Atomic Mass The ATOMIC MASS listed on the periodic table is an average of the mass of all known isotopes of that element. You can also think of it as the number of PROTONS and NEUTRONS.
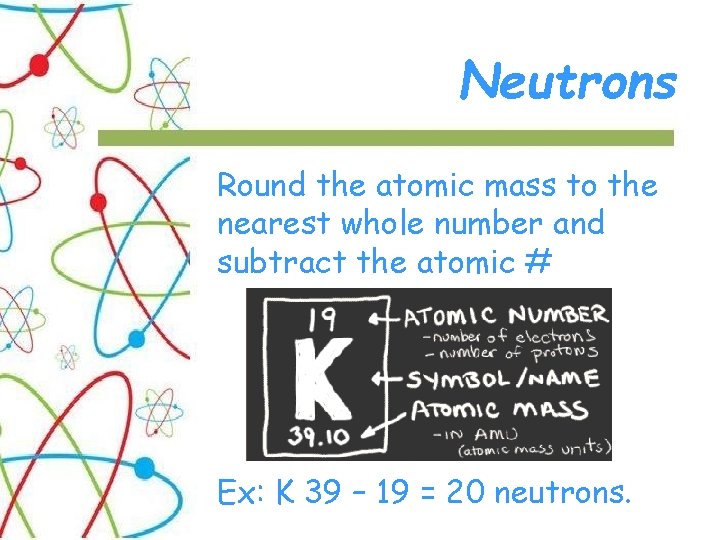
Neutrons Round the atomic mass to the nearest whole number and subtract the atomic # Ex: K 39 – 19 = 20 neutrons.

Symbols Hg – Mercury Au – Gold Pb – Lead Sn – Tin Ag – Silver Cu – Copper Fe – Iron K – Potassium Na - Sodium Black = Solid Blue = Liquid Red = Gas
Energy Levels There are up to seven ENERGY LEVELS within the electron cloud on which the electrons orbit the nucleus. The PERIOD (row) on which an element can be found will tell you how many energy levels there are in an atom of that element.

Electron Configurations 1 st energy level holds 2 electrons 2 nd energy level holds 8 electrons 3 rd energy level holds 18 electrons
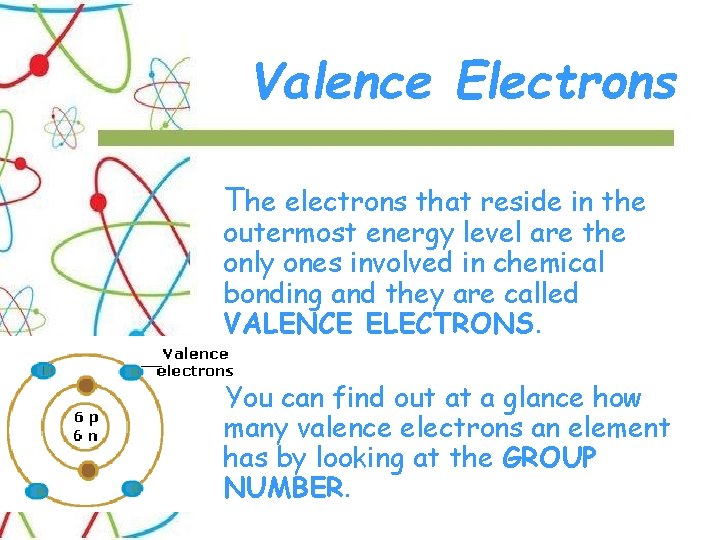
Valence Electrons The electrons that reside in the outermost energy level are the only ones involved in chemical bonding and they are called VALENCE ELECTRONS. You can find out at a glance how many valence electrons an element has by looking at the GROUP NUMBER.

Groups / Families • Elements in the same GROUP / FAMILY (column) have similar properties. • The groups are numbered 1 – 18 from left to right and are typically named after the first element at the top of the group. • Some groups have other names they are known by however:

Group 1 ALKALI METALS – do not occur uncombined in nature and are HIGHLY chemically reactive
Alkali Metals H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Group 2 ALKALINE EARTH METALS – also do not occur uncombined; good conductors
Alkaline Earth Metals H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Groups 3 -12 Transition Metals – also good conductors; form colorful compounds that last a long time since they are not super chemically reactive.
Rare Earth Metals

Lanthanides • • Soft, shiny, malleable metals with high conductivity. Found in a lot of superconductors and laser technology.

Lanthanide Series H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Actinides • • They are radioactive! Many are synthetic meaning the nuclei are unstable (they break apart easily).
Actinide Series H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr

Metalloids Along the stair step line – (7) METALLOIDS that have properties of metals and nonmetals are very useful. Especially for their varying abilities to conduct electricity (i. e. SILICON silicon semiconductors in computer chips).

Non-Metals The NON-METALS are not good conductors but they combine with others readily to form compounds. Many of these elements are crucial in creating and maintaining life (C H N O P S).

Group 17 HALOGENS - meaning “salt forming” Form salts when combined with Alkali Metals They are DIATOMIC, meaning they never exist as a single atom.
Halogens H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr **Notice that this is the only family in which the members come in all normal phases**

Diatomic Elements H 2 He Li Be B C N 2 O 2 F 2 Ne Na Mg Al Si P S Cl 2 Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br 2 Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I 2 Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
Group 18 NOBLE GASES – do not react with other elements. They are noble, they don’t mix with commoners. They are all gases at normal phase (room temperature).
Noble Gases H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr
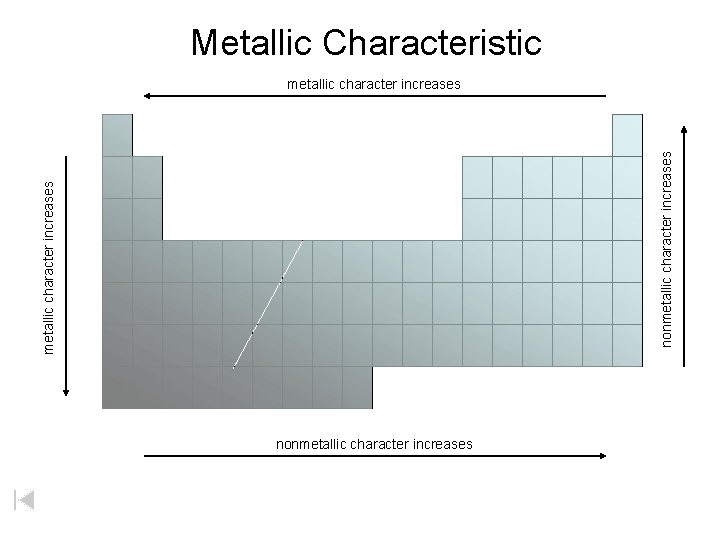
Metallic Characteristic metallic character increases nonmetallic character increases
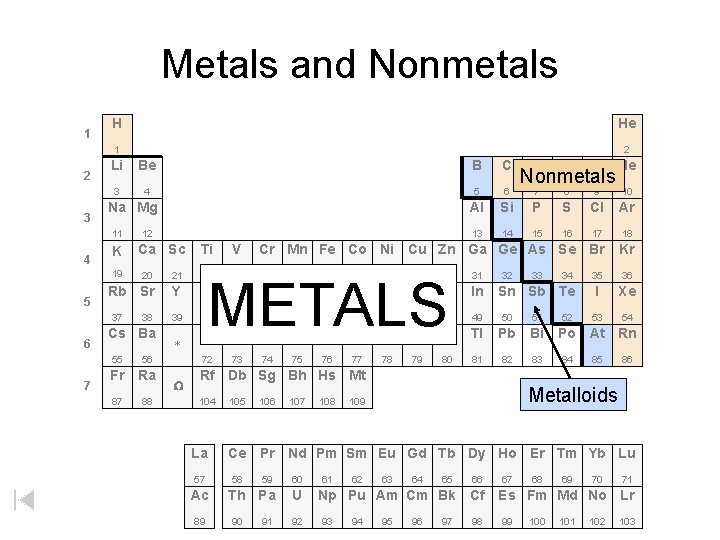
Metals and Nonmetals 1 2 3 H He 1 2 Li Be B C 3 4 5 6 Al 13 Na Mg 11 4 K 19 5 7 Ca Sc O F Ne 7 8 9 10 Si P S Cl Ar 14 15 16 17 18 Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 12 N Nonmetals 38 Cs Ba 55 56 Fr Ra 87 88 * W 25 26 27 28 29 30 METALS 43 44 Re Os 75 76 47 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Po At Rn 84 85 86 Rf Db Sg Bh Hs Mt 104 La 57 Ac 89 105 106 107 108 Metalloids 109 Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 58 59 60 Th Pa U 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103

Properties • A PROPERTY is a characteristic of a substance that can be observed. • A PHYSICAL PROPERTY is one that can be observed without changing the substance. • A CHEMICAL PROPERTY can only be observed while changing the substance.

Physical Properties • Malleability - the ability to be hammered into a thin sheet • Ductility - the ability to be stretched into a wire • Melting/Freezing/Boiling point – temperature at which phase change occurs • Density – the amount of matter in a given volume D=m/v • Solubility – ability to dissolve in another substance • Specific heat - the amount of heat required to heat a substance 1 degree Celsius • Luster - shiny, dull
- Slides: 45