Bellwork 927 Put phone up Week 924 928

Bellwork: 9/27 Put phone up Week 9/24 -9/28 1. 2. 3. Explain the difference between an ion and an isotope. Pick an element on the period table, find how many protons, neutrons & electrons that element has. How many valence electrons does your atom have? Is it reactive?

Objective • Students will learn about how biochemistry is an important branch of biology • Today: Students will learn how chemicals bond to form molecules & compounds

Today: 1. Finish fruit loop atoms 2. Whiteboard Practice 3. Unit 2, Pt. 2 Notes

Materials • Fruit Loops Atom Paper • Unit 2 Book

Homework • None

Unit 2: Biochemistry

Fruit Loopy Atoms

3) Make an Ion 1. 2. 3. 4. 5. 6. 7. 8. PICK ONE ION: Li+, Be 2+, or B 2 Use same colors for the same subatomic particles Calculate # of subatomic particles Write names on back of paper plates Use materials to create ion Draw electron shells! Calculate mass of your ion! Begin conclusion questions

Unit 2: Biochemistry

Atomic Structure White Board Practice

Draw the electron configuration & list the # of P, N, & E H

Draw the electron configuration & list the # of P, N, & E S

Draw the electron configuration & list the # of P, N, & E K

Draw the electron configuration & list the # of P, N, & E 2+ K

Draw the electron configuration & list the # of P, N, & E Si

Draw the electron configuration & list the # of P, N, & E 30 Si

Draw the electron configuration & list the # of P, N, & E 17 N

Draw the electron configuration & list the # of P, N, & E 2 He

Draw the electron configuration & list the # of P, N, & E 13 Be

Part II: Chemical Bonding
Chemical Bonding • Definition: Forces that hold atoms together • Two types of chemical bonds: • Ionic Bonds • Covalent Bonds

Ionic Bonds • Definition: Removal of electrons from one atom and transferred to another atom • Commonly occur when atoms in Group IA or IIA react with Group VIIA on Periodic Table • (metal + non-metal) • Ex. Na. Cl

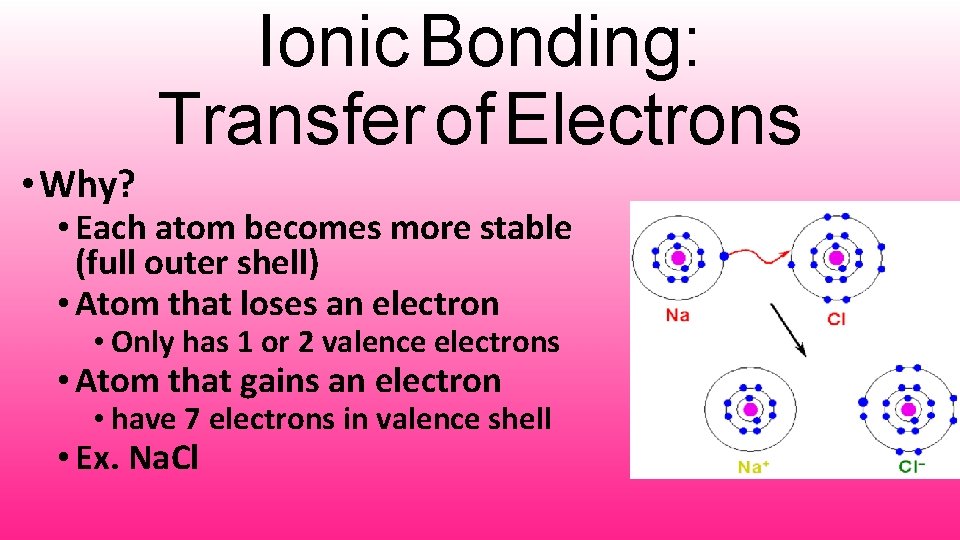
• Why? Ionic Bonding: Transfer of Electrons • Each atom becomes more stable (full outer shell) • Atom that loses an electron • Only has 1 or 2 valence electrons • Atom that gains an electron • have 7 electrons in valence shell • Ex. Na. Cl

Covalent Bonds • Definition: share electrons between two atoms • Commonly, atoms in Groups IIIA-VIA form covalent bonds • (non-metal + nonmetal)

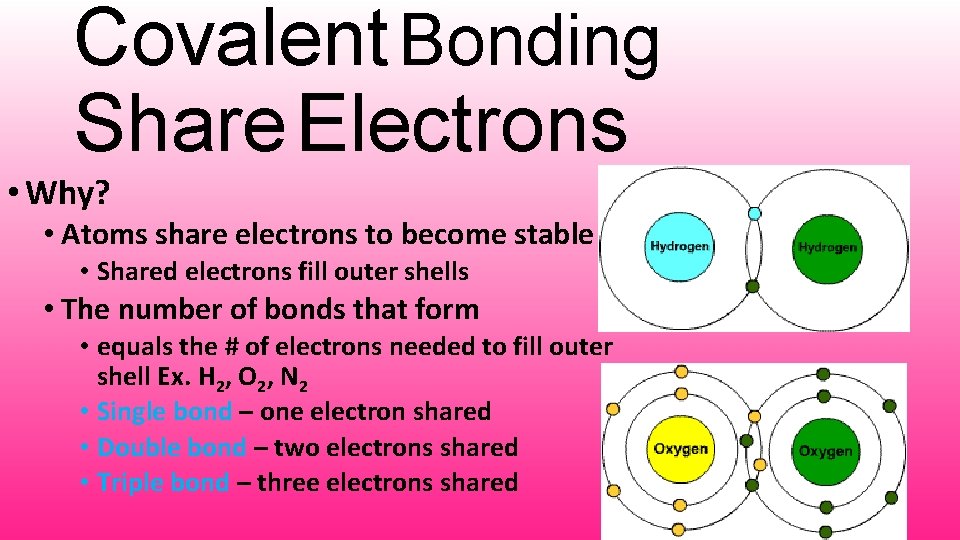
Covalent Bonding Share Electrons • Why? • Atoms share electrons to become stable • Shared electrons fill outer shells • The number of bonds that form • equals the # of electrons needed to fill outer shell Ex. H 2, O 2, N 2 • Single bond – one electron shared • Double bond – two electrons shared • Triple bond – three electrons shared

Note Guide Practice Use a Venn Diagram to compare and contrast ionic bonds and covalent bonds.

Note Guide Practice • Examples we work together: How many covalent bonds are formed in H 2? In N 2? • Now you try it: How many covalent bonds are formed between oxygen atoms in O 2?

Compound Versus • Compound Molecule • Substance formed by the chemical bonding of two or more different elements • All compounds are molecules, but not all molecules are compounds • Molecule • Two or more atoms joined together by chemical bonds • Atoms can be the same element OR different • How is a molecule different than a compound? Write answer on your note guide. • Can Na. Cl be a molecule? Explain why or why not on your note guide. • Is O 2 a compound, molecule or both?

Chemical Reactions • Definition: when atoms combine or change bonding partners 2 H 2 + O 2 2 H 20 reactants products

Chemical Reaction • During chemical reaction: • Atoms rearrange and form new bonds • Results in a chemical change • Changes one set of chemicals into another • Different from a physical change • Physical change does not result in change in chemical composition

Chemical Change vs Physical Change • Examples: • Rust on a Chain : O 2 + Fe 2 O 3 • chemical reaction because bonds break and re-form • Boiling Water : H 2 O(liq) + heat H 2 O(gas) • not a chemical reaction, no rearranged bonds • physical change

? Thumbs up Chemical Change / Thumbs down Physical Change You Cut the Grass You combust gasoline in your car You digest the protein in your hamburger You dissolve sugar in water You dissolve Alka-Seltzer in water
- Slides: 34