Bellwork 1022 Put phone up Week 1022 1026

Bellwork: 10/22 Put phone up Week 10/22 -10/26 1. Explain how the buffer worked in the lab. 2. What do you think a macromolecule is? 3. Did you have a good weekend?

Objective • Continue to understand the importance and the role of macromolecules in life

Today: 1. Unit 2, pt. 5 & 6 Notes

This Week: • Monday: Unit 2, pt. 5 & 6 notes • Tues/Wed: Unit 2, pt. 7 notes & macromolecules lab • Thursday: Unit 2, pt. 8 notes & Mello Jello Lab • Friday: Mello Jello Lab

Materials • Lab Notebook

Homework • Quiz due tonight @ 11: 59 pm • Read pages 44 -48 • Answer ? s on Google Classroom • Due tonight @ 11: 59 pm

Unit II: Biochemistry
Part V: Macromolecules – Carbohydrates

4 Key Macromolecules: • Carbohydrates • Lipids • Proteins • Nucleic Acids

Macromolecules are made of CARBON! • Carbon • Central element in living things • Presence of carbon – organic chemistry

Carbon = Key Element • Tetravalent – has 4 electrons in valence shell • Can form four covalent bonds • Bonds can be with either other carbon atoms, or hydrogen, oxygen, nitrogen and/or sulfur

Most Macromolecules are: • Polymers – large molecules made of similar building blocks or monomers • Example: • Glucose (monomer) makes up starch (polymer)

Carbohydrates

What Foods Can you Think of that Contain Carbohydrates?
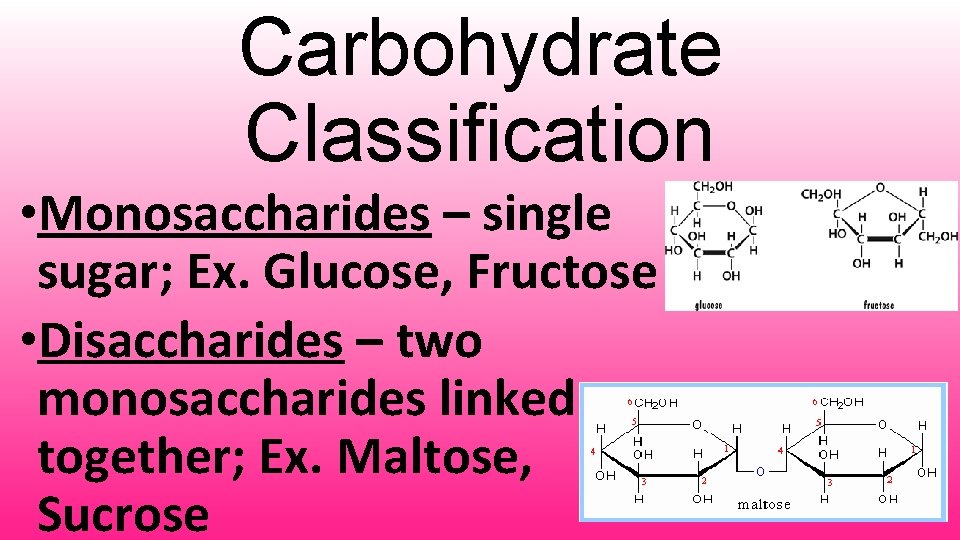
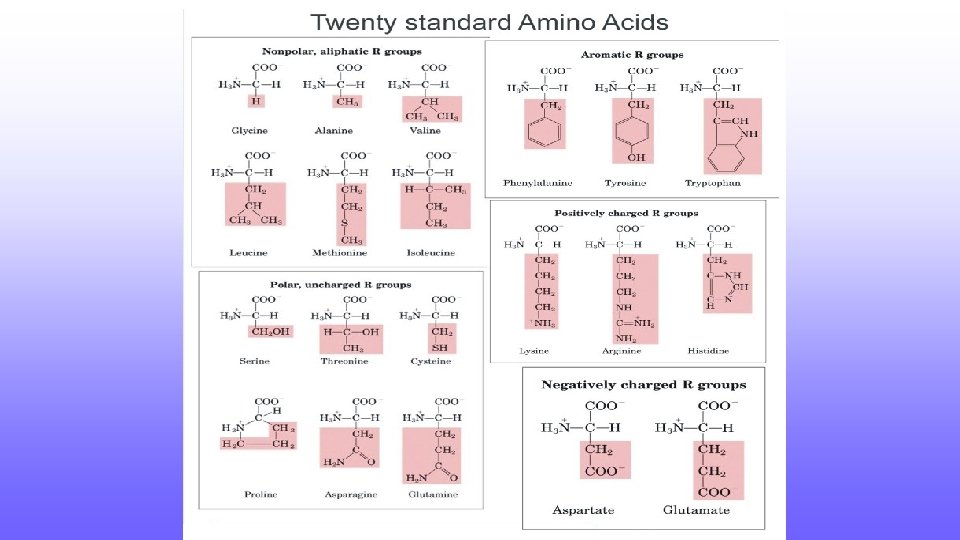
Carbohydrate Classification • Monosaccharides – single sugar; Ex. Glucose, Fructose • Disaccharides – two monosaccharides linked together; Ex. Maltose, Sucrose
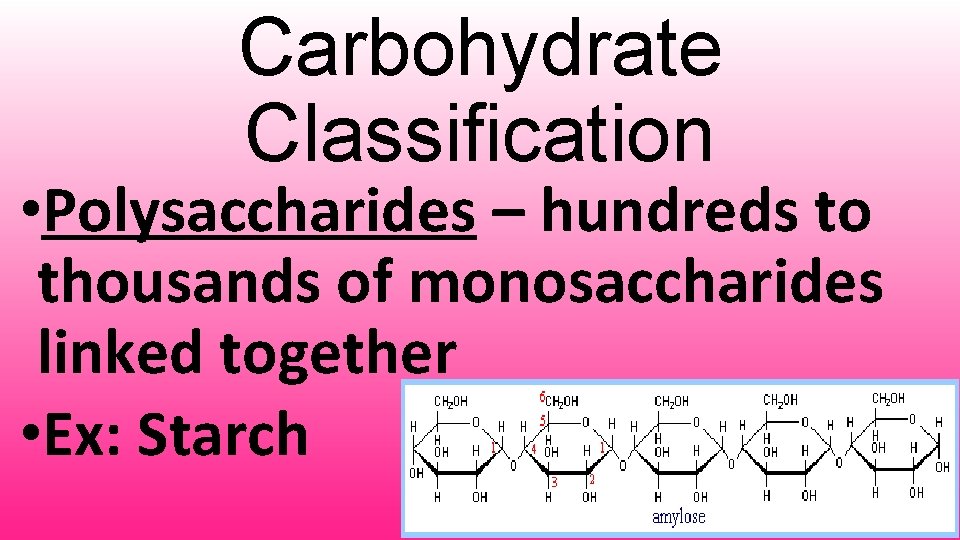
Carbohydrate Classification • Polysaccharides – hundreds to thousands of monosaccharides linked together • Ex: Starch

Functions of Carbohydrates • Storage • Starch in plants • Glycogen – humans, other vertebrates found in liver and muscle cells • Structural – Cannot be broken down • Cellulose – cell wall of plants • Chitin – exoskeleton of arthropods

Part VI: Macromolecules Lipids

Lipids

What Foods Can you Think of that Contain Lipids?

Lipids • Classified because they’re hydrophobic- repel water • These molecules further sub classified as: • Fats and oils • Phospholipids • Steroids
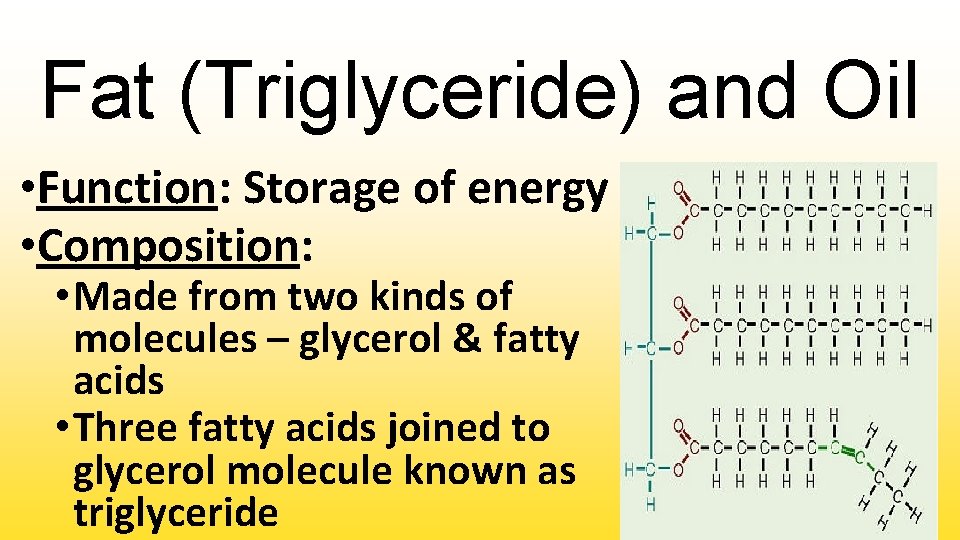
Fat (Triglyceride) and Oil • Function: Storage of energy • Composition: • Made from two kinds of molecules – glycerol & fatty acids • Three fatty acids joined to glycerol molecule known as triglyceride
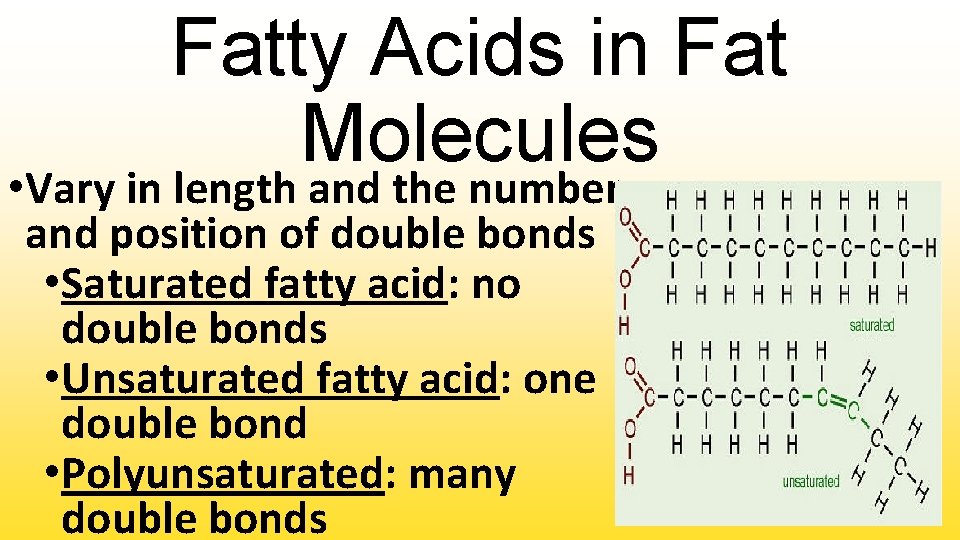
Fatty Acids in Fat Molecules • Vary in length and the number and position of double bonds • Saturated fatty acid: no double bonds • Unsaturated fatty acid: one double bond • Polyunsaturated: many double bonds

Degree of Unsaturation • Greater number of double bonds lowers the melting point • Number of double bonds: • Zero – solid @ room temperature and @ 4∘C – Ex. Butter • Single – liquid @ room temperature, solid @ 4∘C – Ex. Olive Oil • Multiple – liquid @ room temp & 4∘C – Ex. Corn Oil

Biological Significance • Unsaturated fatty acids do not form solids at room temperature • Double bonds in fatty acid chains prevent chains from stacking up and interacting • Result – does not form solid; remains a liquid • Why unsaturated fatty acids healthier? Do not clog arteries and cause heart disease

Activity • What is it about the shape of saturated fats that make them: • Melt at higher temperature? • Clog up your arteries? • Hint: Think of many fat molecules grouped together Be ready to share!

Lipids • Important part of animal diet • Animals primarily use fat for energy storage. Why? • Fat • Stores 9 Kcal/g of energy • Kcal = Cal = 1000 cal • Cal = energy to raise 1 g of water 1 o. C • Carbohydrates & Proteins • Store only 4 k. Cal/g of energy • Fat also used in our bodies to • Cushion vital organs like kidneys • Provide insulation, especially under skin
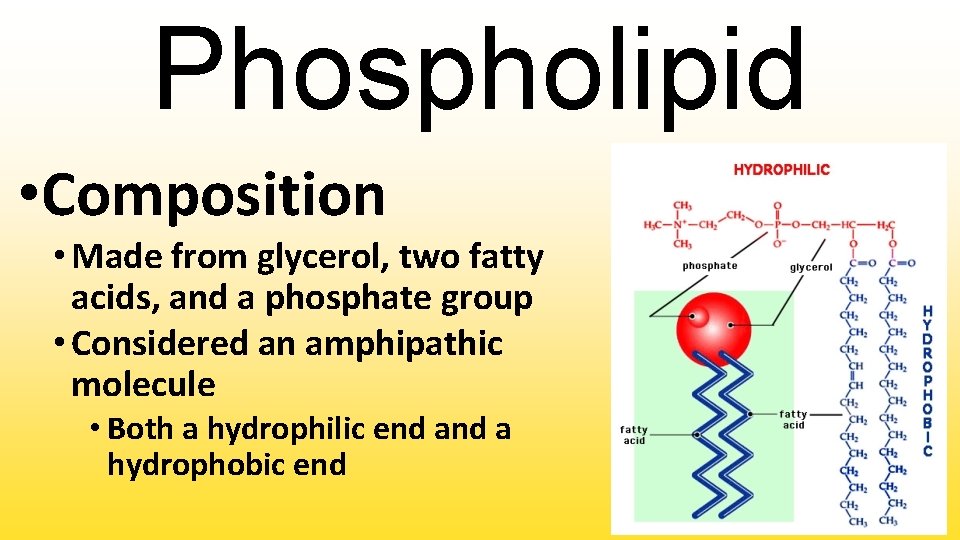
Phospholipid • Composition • Made from glycerol, two fatty acids, and a phosphate group • Considered an amphipathic molecule • Both a hydrophilic end a hydrophobic end
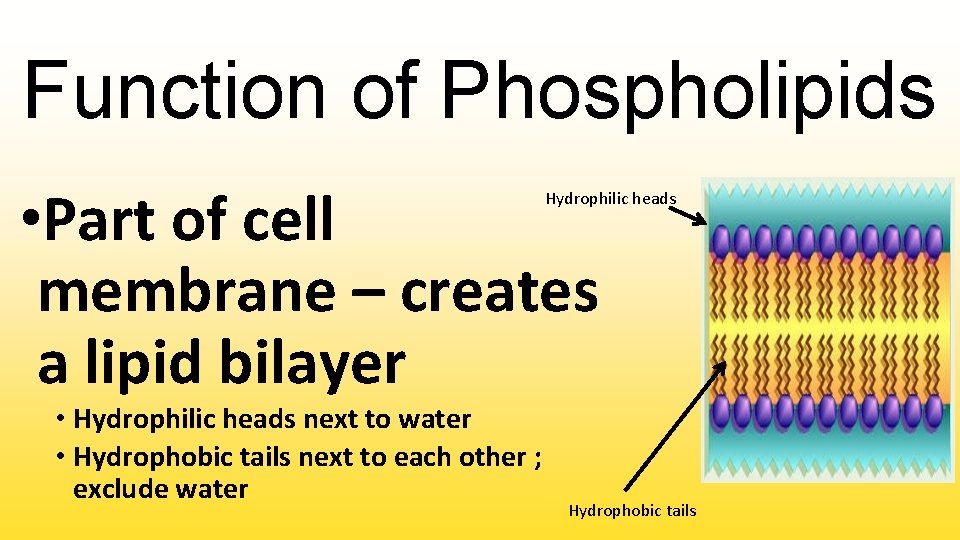
Function of Phospholipids • Part of cell membrane – creates a lipid bilayer Hydrophilic heads • Hydrophilic heads next to water • Hydrophobic tails next to each other ; exclude water Hydrophobic tails
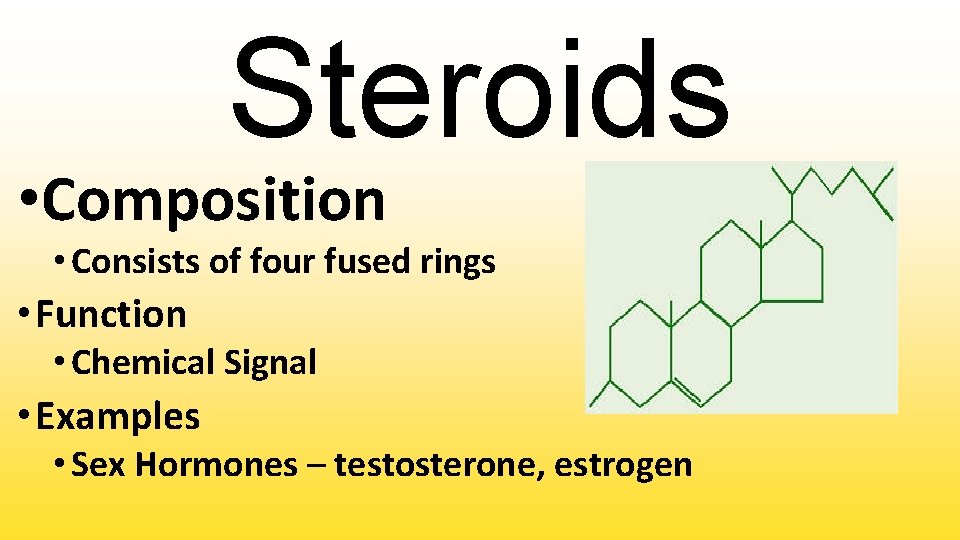
Steroids • Composition • Consists of four fused rings • Function • Chemical Signal • Examples • Sex Hormones – testosterone, estrogen

Answer ? s in Note Guide

Part VII: Macromolecules Proteins

Intro ? • Why does our body need proteins to function?

Proteins • Polymers of amino acids • Have diverse shapes & functions

Diverse Functions • Structural • Provide support & protection • Storage • Energy Storage • Transport • Move ions and small molecules across membranes • Enzymes • Act as catalysts to speed up chemical reactions • Receptors • Bind chemical signals, like hormones

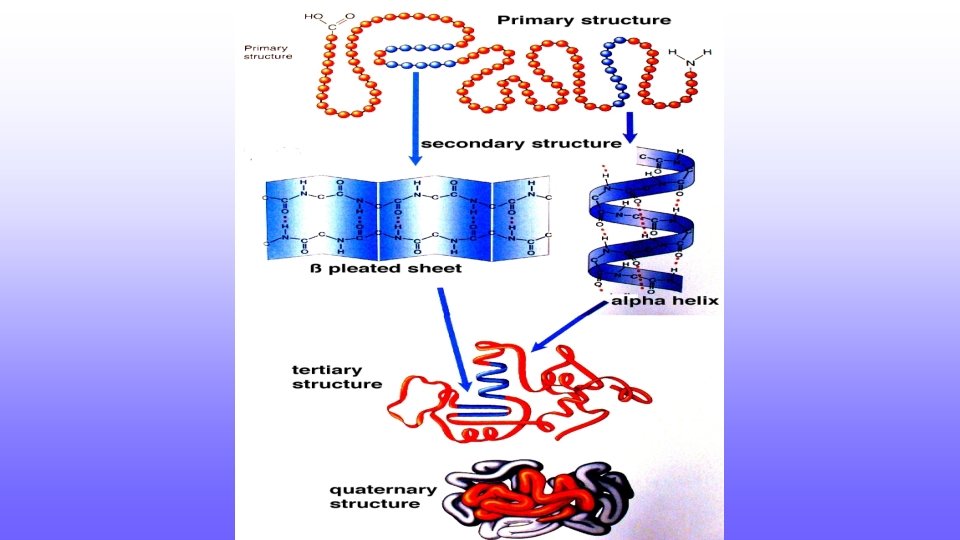
Primary Structure • Polymer composed of a chain of amino acids • Each amino acid has the same basic structure, differing in only the “R group”
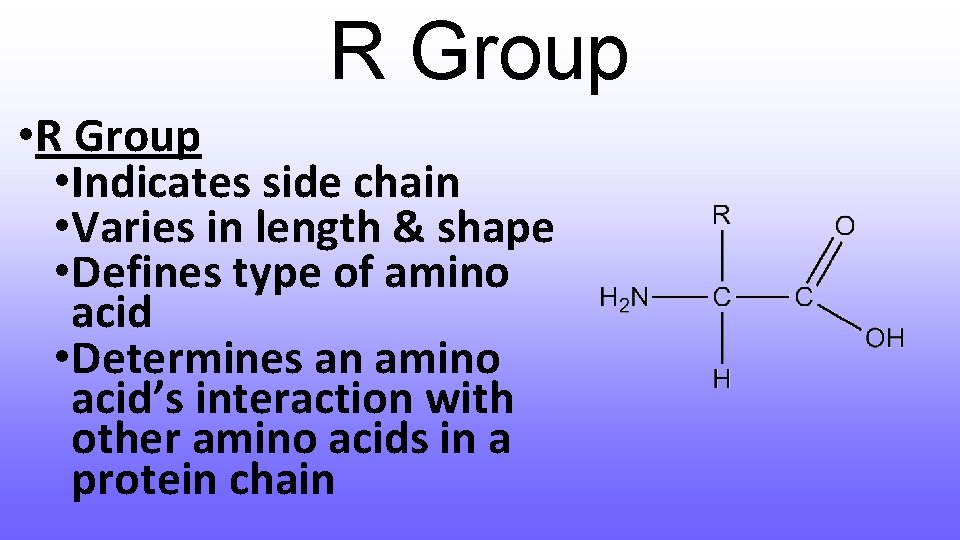
R Group • Indicates side chain • Varies in length & shape • Defines type of amino acid • Determines an amino acid’s interaction with other amino acids in a protein chain
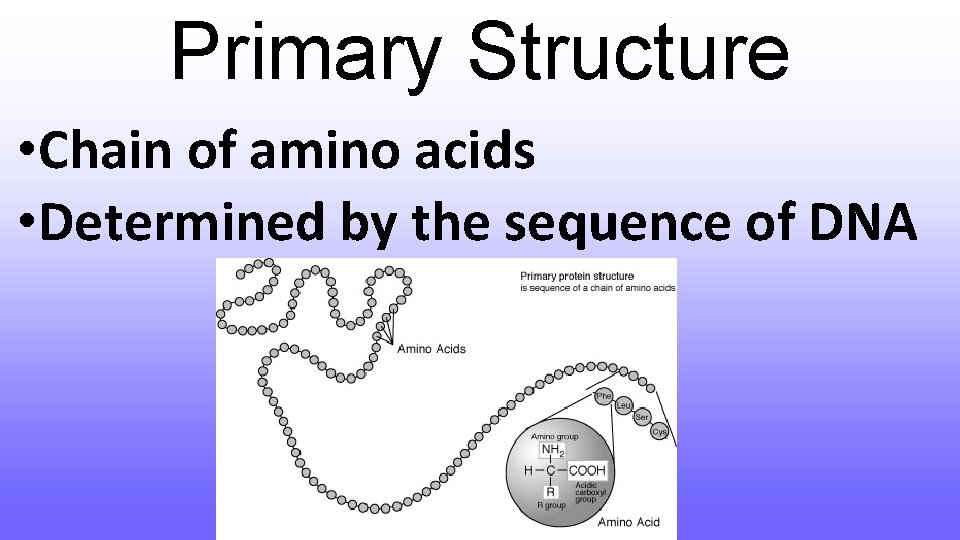
Primary Structure • Chain of amino acids • Determined by the sequence of DNA
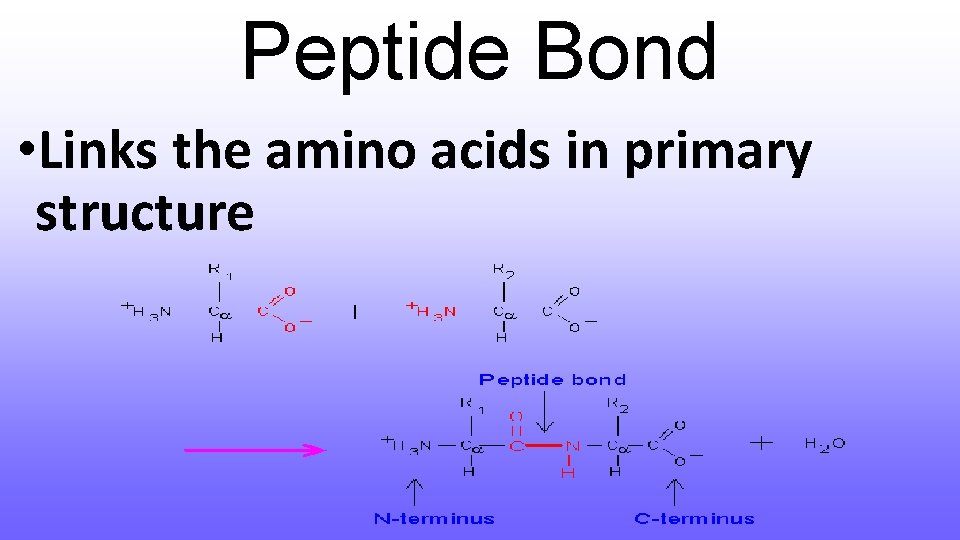
Peptide Bond • Links the amino acids in primary structure
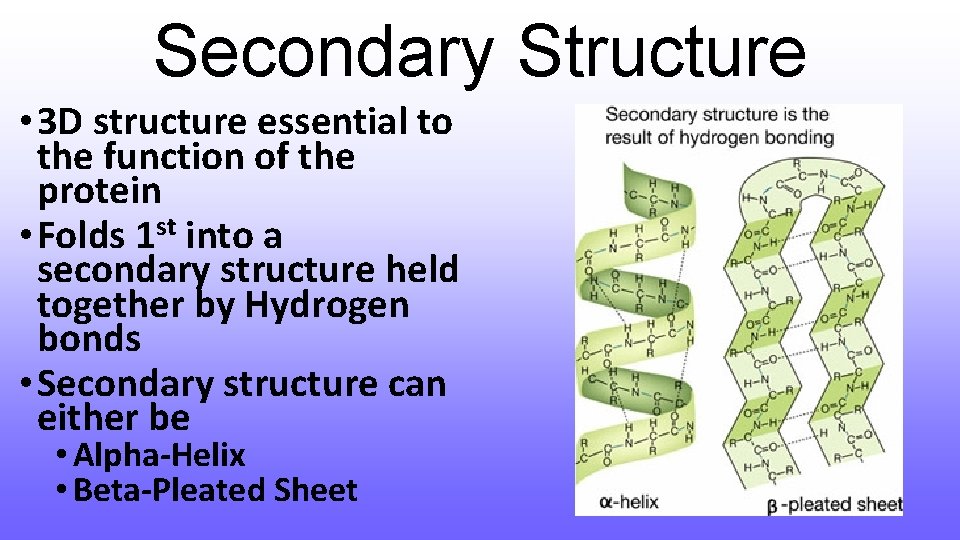
Secondary Structure • 3 D structure essential to the function of the protein • Folds 1 st into a secondary structure held together by Hydrogen bonds • Secondary structure can either be • Alpha-Helix • Beta-Pleated Sheet
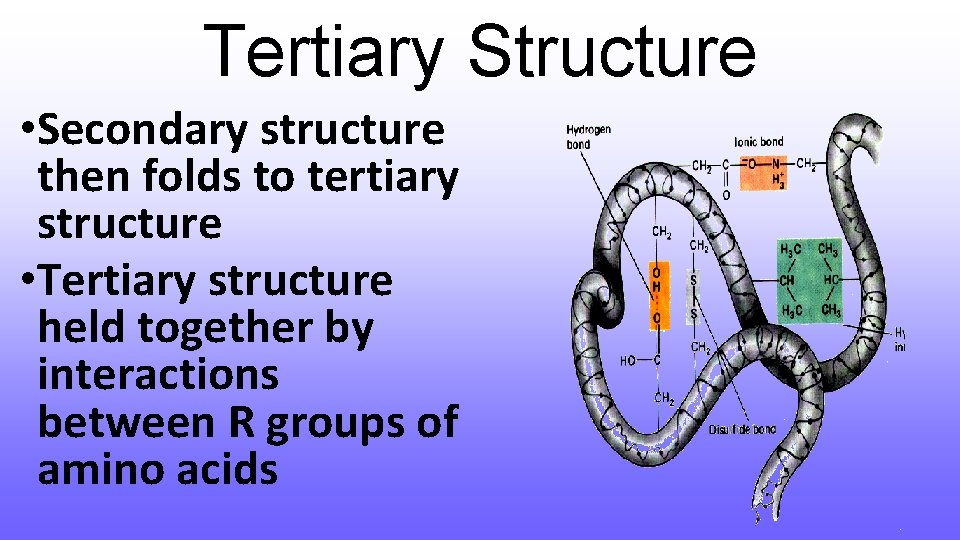
Tertiary Structure • Secondary structure then folds to tertiary structure • Tertiary structure held together by interactions between R groups of amino acids

Quaternary Structure • Proteins that have multiple polypeptides form quaternary structures

Example - Hemoglobin • Function: transport oxygen in blood • Shape: “globular” • With binding sites for 4 oxygen molecules

Hemoglobin • Has quaternary structure • Made of 2 alpha subunits and 2 beta subunits

THINK • Proteins have diverse functions. What is it about proteins that allow them to be so diverse?
- Slides: 47