Bellringers Week 1 BR 1 Write each of

Bellringers Week 1

BR 1 Write each of these 3 TIMES Greek, Latin, and Roman names of elements Argentum = Silver Kalium=Potassium Aurum=Gold Ferrum = Iron Plumbum = Lead Stannum = Tin

BR 2 Write each of these 3 TIMES Greek, Latin, and Roman names of elements Stibium =Antimony Natrium =Sodium Cuprum =Copper Hydrargyrum =Mercury Wolfram =Tungsten

Bellringers Week 2

BR 3 1 H 2 He 3 Li 4 Be 5 B Hydrogen Helium Lithium Beryllium Boron 3 TIMES EACH

BR 4 6 C 7 N 8 O 9 F 10 Ne Carbon Nitrogen Oxygen Fluorine Neon 3 TIMES EACH

BR 5 11 12 13 14 15 Na Sodium Mg Magnesium Al Aluminum Si Silicon P Phosphorus 3 TIMES EACH

Bellringers Week 3

BR 6 16 S Sulfur 17 Cl Chlorine 18 Ar Argon 19 K Potassium 3 TIMES EACH

BR 7 20 Ca Calcium 21 Sc Scandium 22 Ti Titanium 23 V Vanadium 3 TIMES EACH

BR 8 CHEM 24 Cr Chromium 25 Mn Manganese 26 Fe Iron 27 Co Cobalt 3 TIMES EACH

Bellringers Week 4

BR 9 CHEM 28 Ni Nickel 29 Cu Copper 30 Zn Zinc 31 Ga Gallium 32 Ge Germanium 3 TIMES EACH

BR 10 CHEM 33 As Arsenic 34 Se Selenium 35 Br Bromine 36 Kr Krypton 3 TIMES EACH

BR 11 CHEM 37 Rb Rubidium 38 Sr Strontium 39 Y Yttrium 40 Zr Zirconium 3 TIMES EACH

Bellringers Week 5

BR 12 CHEM 41 Nb Niobium 42 Mo Molybdenum 43 Tc Technetium 44 Ru Ruthenium 45 Rh Rhodium 3 TIMES EACH

BR 13 CHEM 46 Pd Palladium 47 Ag Silver 48 Cd Cadmium 49 In Indium 50 Sn Tin 3 TIMES EACH

BR 14 CHEM 51 Sb Antimony 52 Te Tellurium 53 I Iodine 54 Xe Xenon 55 Cs Cesium 3 TIMES EACH

Bellringers Week 6 Prefixes, Suffixes, Root Words QUIZ ON FRIDAY

BR 15 Number Prefix, Suffix, Root Word Definition/Meaning Example 1 a without or not; opposite to asexual, acoelomate 2 ab away from abduct, abnormal 3 ad toward adduct, adrenal 4 aer air, gas aerobic, aerospace 5 amphi on both sides amphibians

BR 16 6 an without anaerobic 7 andr man androgen, android 8 aqua water aquaduct, aquamarine 9 auto self autobiography, automatic two bipolar, biplane 10 bi

BR 17 11 bio life biology, biotic 12 car cancer carcinogen 13 cardi heart cardiology 14 carn meat carnivore 15 ceph toward the head cephalothorax, hydrocephalus

Bellringers Week 7 QUIZ ON FRIDAY

BR 18 16 chiro with the hand chiropractor 17 chloro green chlorophyll, chloroplast 18 chrom color chromosome 19 cide kill matricide 20 cyte cell cytoplasm, amoebocyte

BR 19 21 dent or dont teeth orthodontist, dentures 22 derm skin dermatologist, epidermis 23 eco house ecosystem, economy 24 ecto outer ectoderm 25 endo inner endospores, endodermis

BR 20 26 entomon insect entomology 27 dys disease dysentary 28 epi upon epicardium 29 gamo sexual union gamete, monogamy 30 gastro stomach gastroenterology

Bellringers Week 8 QUIZ ON FRIDAY
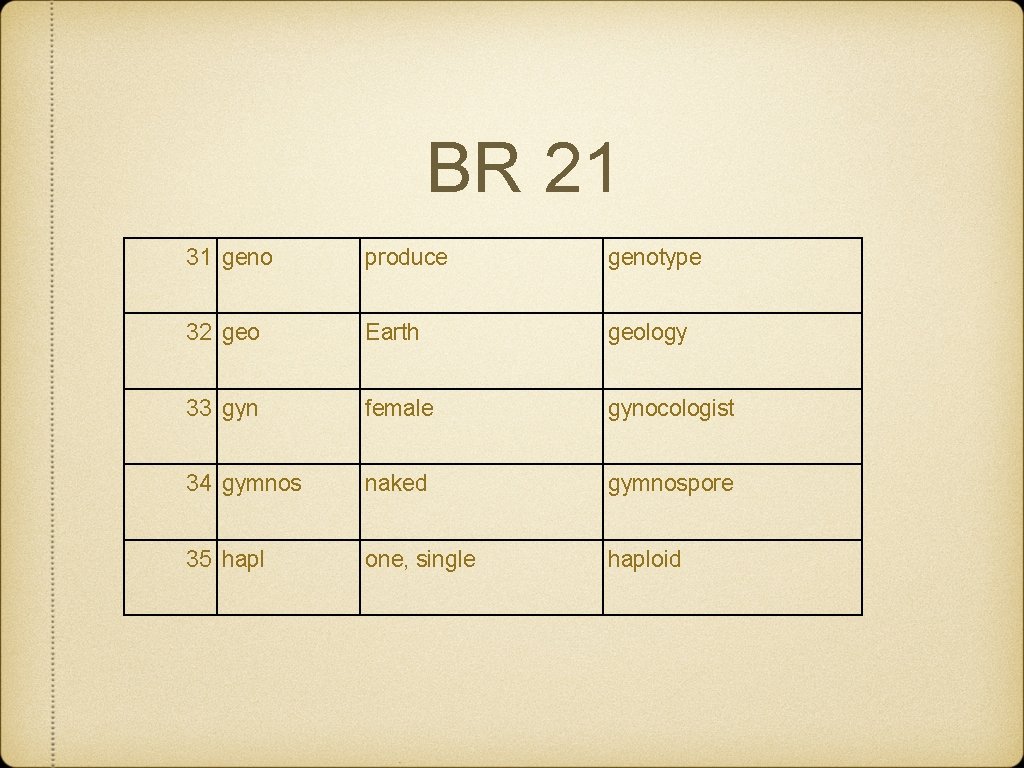
BR 21 31 geno produce genotype 32 geo Earth geology 33 gyn female gynocologist 34 gymnos naked gymnospore 35 hapl one, single haploid
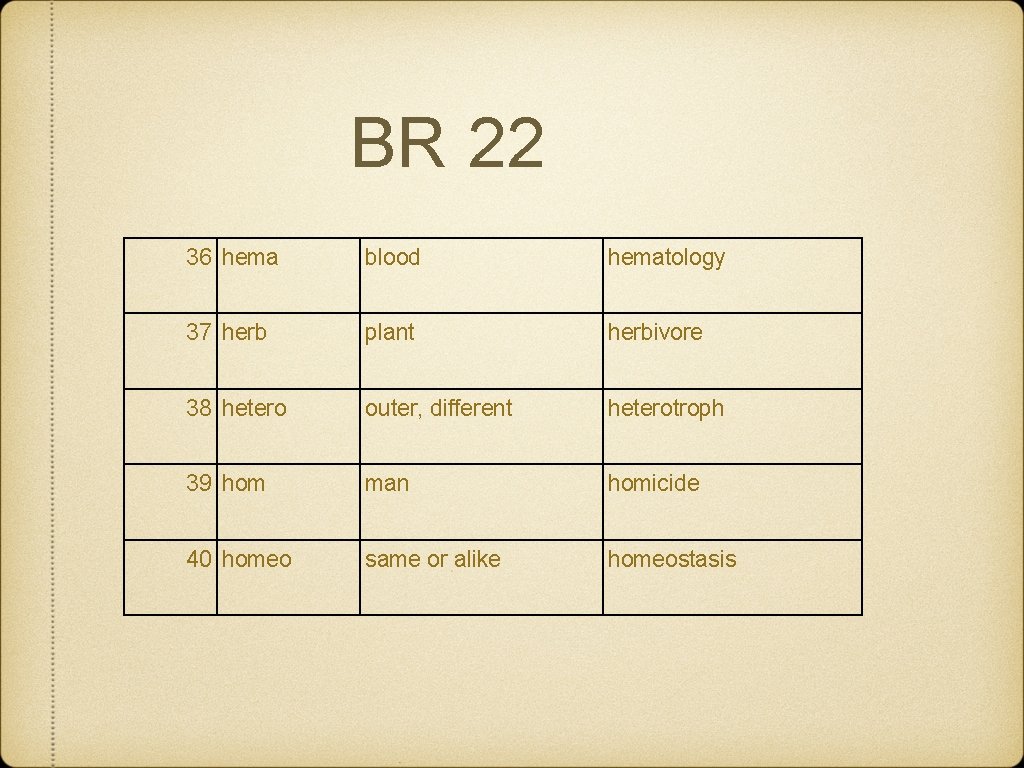
BR 22 36 hema blood hematology 37 herb plant herbivore 38 hetero outer, different heterotroph 39 hom man homicide 40 homeo same or alike homeostasis

BR 23 41. hydro water hydroelectric 42. hyper above hypertonic 43. hypo below hypogastric 44. ichthus fish ichthyology 45. inter between intermedian

Bellringers Week 9 QUIZ ON FRIDAY

BR 24 46. leuco without color leucocytes 47. lyso to loosen or dissolve lysosome 48. macro larger macroorganism 49. mei reduce meiosis 50. meso middle mesoderm

BR 25 51 meta behind metamorphosis 52 micro small microscope 53 mono one monotone 54 morph form morphology 55 myco fungus mycology
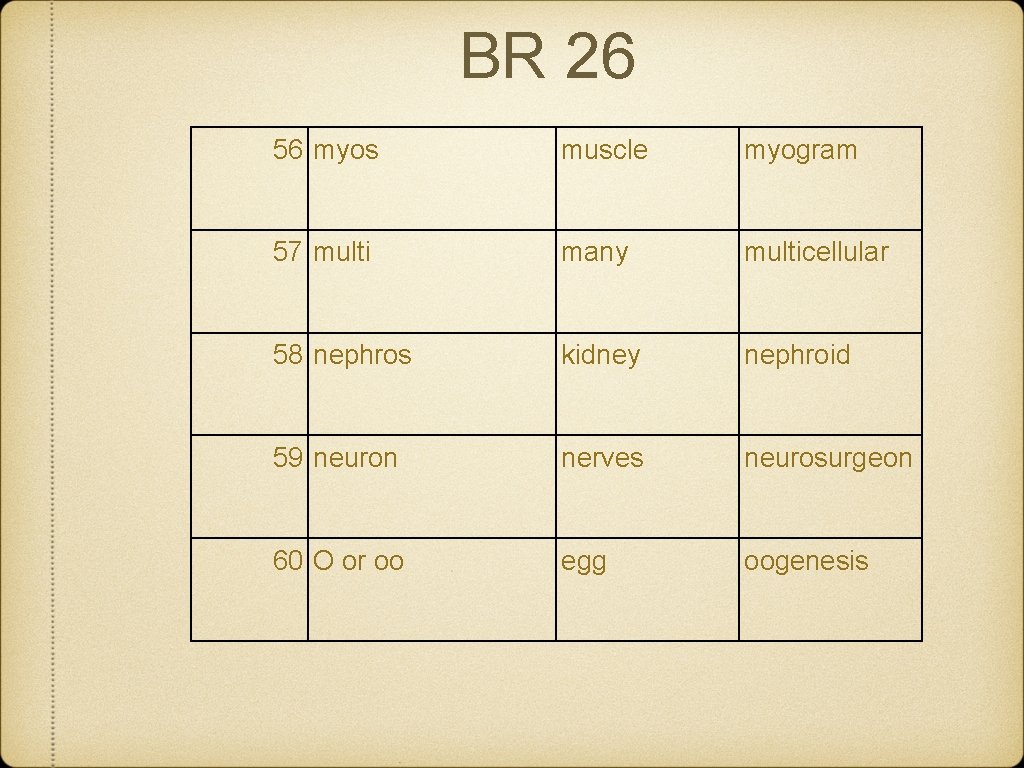
BR 26 56 myos muscle myogram 57 multi many multicellular 58 nephros kidney nephroid 59 neuron nerves neurosurgeon 60 O or oo egg oogenesis

BR 20 CHEM Diatomic Molecules Diatomic molecules are molecules composed of only two atoms, of either the same or different chemical elements. The prefix di- is of Greek origin, meaning "two". If a diatomic molecule consists of two atoms of the same element, such as hydrogen (H 2) or oxygen (O 2), then it is said to be homonuclear. Hydrogen (H 2) Nitrogen (N 2) Oxygen (O 2) Fluorine (F 2) Chlorine (Cl 2) Iodine (I 2) Bromine (Br 2)
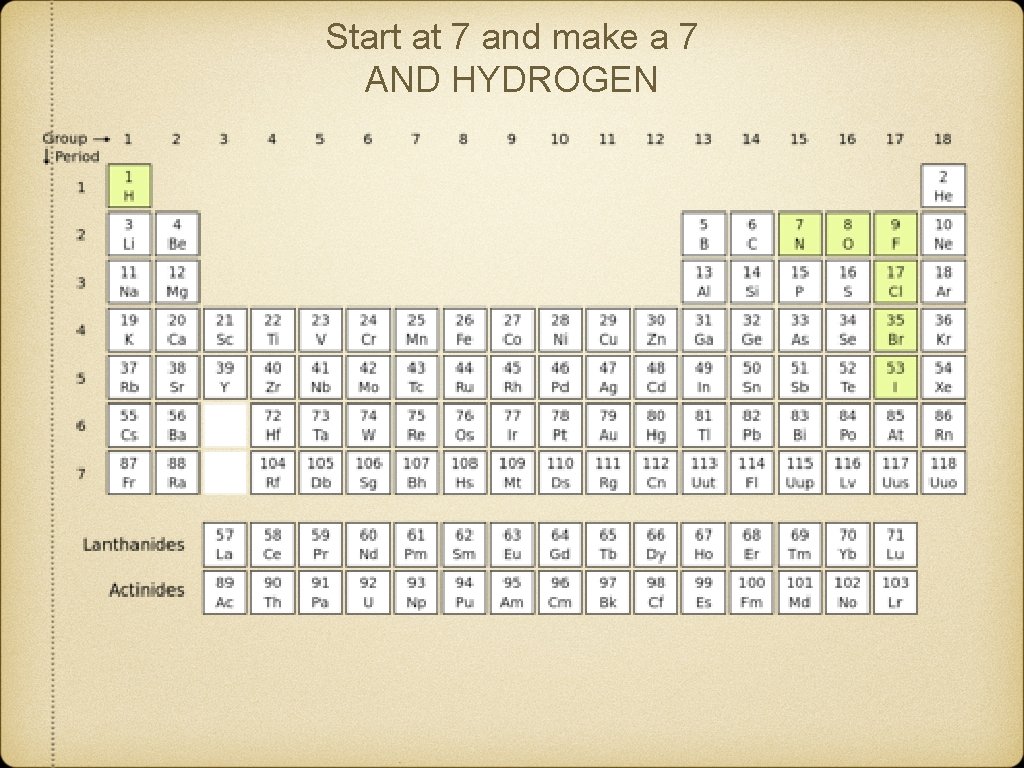
Start at 7 and make a 7 AND HYDROGEN

BR 21 CHEM Natural Elements How many of the elements are man made? Answer: There are 90 elements that occur in nature. Of the first 92 elements on the periodic table, where 1 is hydrogen and 92 is uranium, 90 elements occur naturally. Technetium (atomic number 43) and promethium (atomic number 61) are man-made elements that have no naturally occurring isotopes.
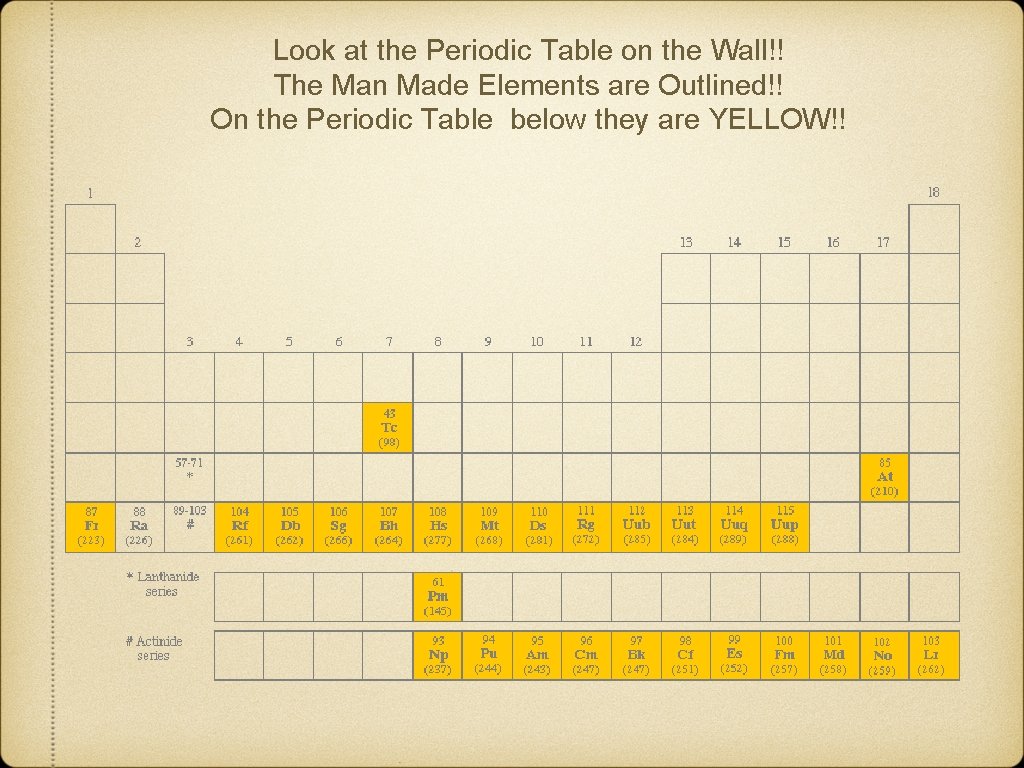
Look at the Periodic Table on the Wall!! The Man Made Elements are Outlined!! On the Periodic Table below they are YELLOW!!

BR 22 CHEM Periodic Table INFO P, N, E To Find the Protons you look at the TOP number To find the Electrons you look at the TOP number To find the NEUTRONS you subtract BOTTOM - TOP (ROUNDED)

BR 20 ON YOUR OWN SHEET OF PAPER Write the first 5 element names and symbols FIVE times each

BR 21 Write the element names 6 -10 and symbols FIVE times each

BR 22 Write the element names 11 -15 and symbols FIVE times each

BR 23 Write the element names 16 -20 and symbols Three times each

BR 8 Write a full paragraph on how the Law of Conservation of Mass and the Law of Conservation of Energy are part of God’s plan for our world. -Include a definition of the Law of Conservation of Mass -Remember the example of the bunny that was on the road -This will be turned in

BR 23 Write a full paragraph on how the Scientific Method is part of God’s plan for our world. -Do you think God meant for us to have a method to reason through things? -Why would God give us the minds/brains to solve problems? -This will be turned in
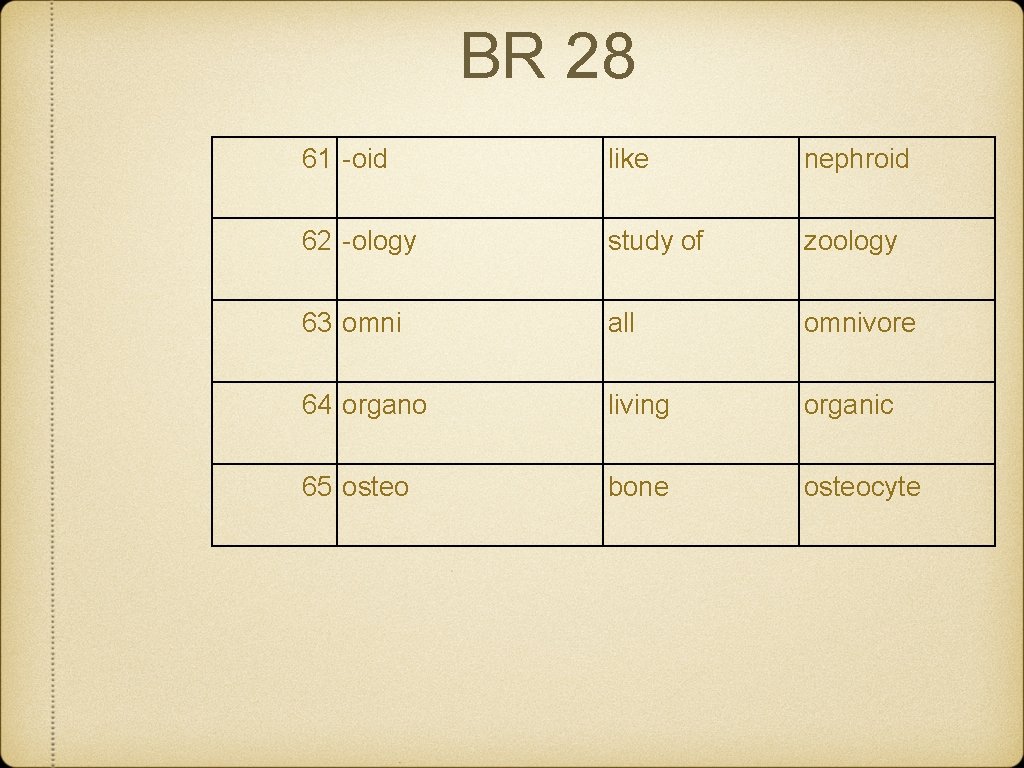
BR 28 61 -oid like nephroid 62 -ology study of zoology 63 omni all omnivore 64 organo living organic 65 osteo bone osteocyte
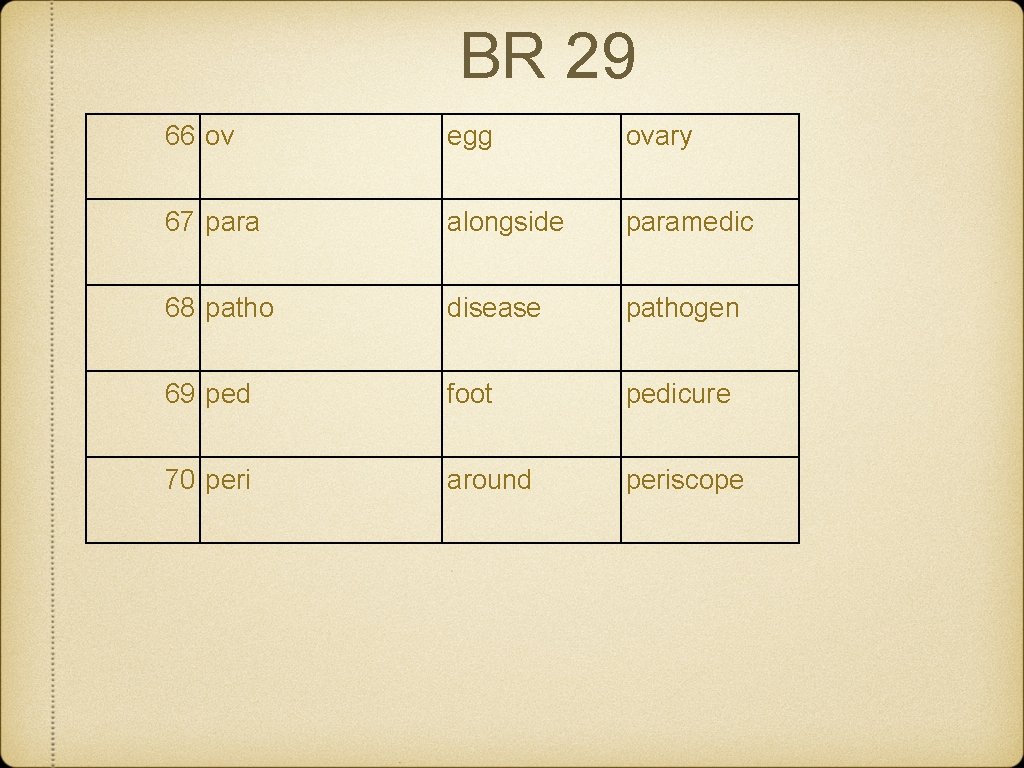
BR 29 66 ov egg ovary 67 para alongside paramedic 68 patho disease pathogen 69 ped foot pedicure 70 peri around periscope
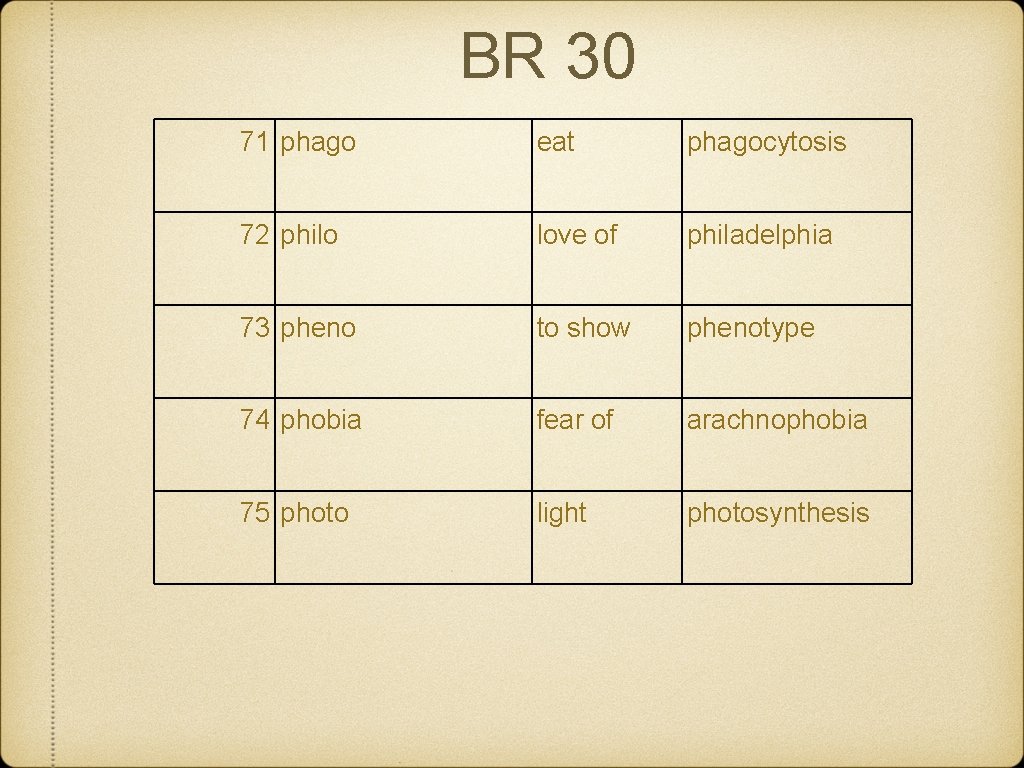
BR 30 71 phago eat phagocytosis 72 philo love of philadelphia 73 pheno to show phenotype 74 phobia fear of arachnophobia 75 photo light photosynthesis
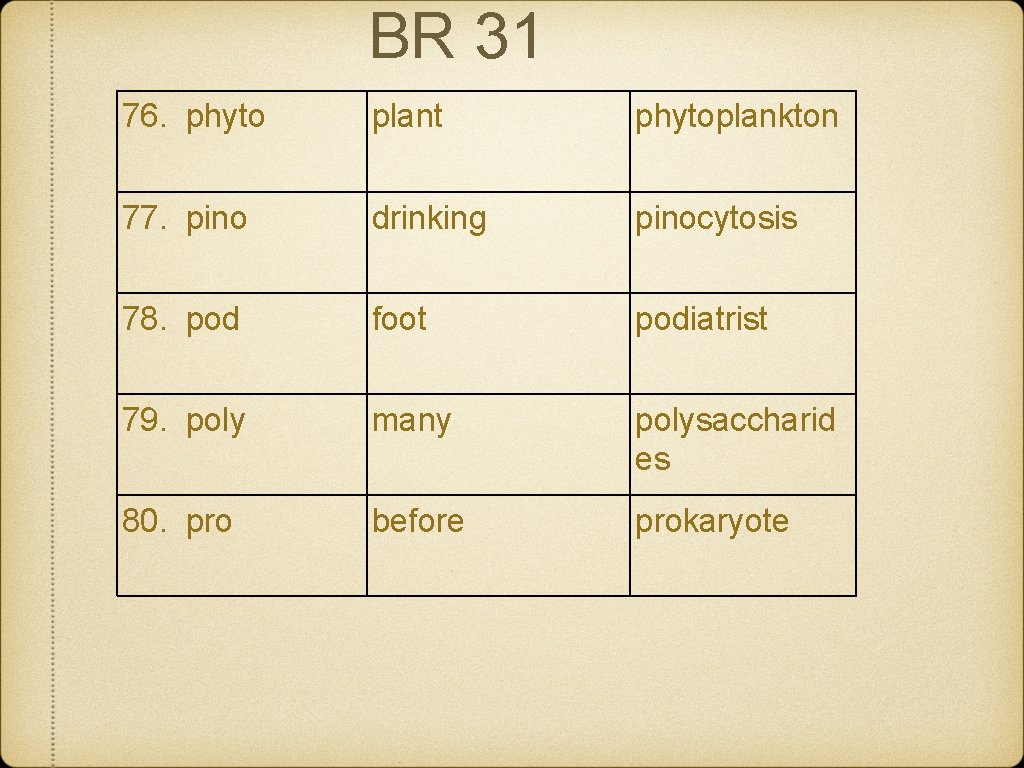
BR 31 76. phyto plant phytoplankton 77. pino drinking pinocytosis 78. pod foot podiatrist 79. poly many polysaccharid es 80. pro before prokaryote

BR 32 81. pseudo FALSE pseudopod 82. pulmo lung pulmonary 83. renos kidney renal 84. stasis to stay homeostasis 85. sub under submarine

BR 33 86. super above superior 87. syn, sym together symbiosis 88. tele distance telepathic 89. telo last, final telophase 90. thermo heat thermometer
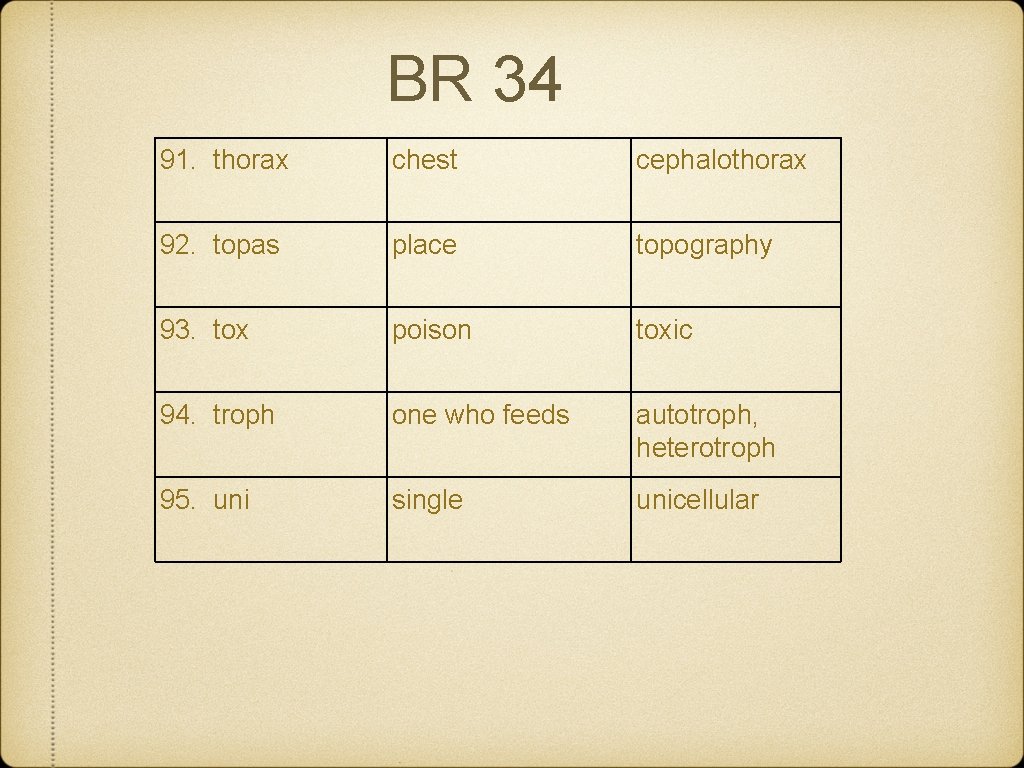
BR 34 91. thorax chest cephalothorax 92. topas place topography 93. tox poison toxic 94. troph one who feeds autotroph, heterotroph 95. uni single unicellular

BR 35 96. vas vessel vascular 97. vena vein intraveneous 98. vivus alive oviparous 99. vore eat carnivore 100. troph one who feeds autotroph

BR 36 101. zoo or zoa animal zoology 102. zygo result of fusion zygote 103. acu sharp acute, acupuncture 104. acy state or quality privacy, infancy 105. age activity shrinkage, courage

BR 37 106. agri pertaining to fields agriculture 107. ambi both ambidextrous 108. bar pressure barometer 109. cad to fall cadaver 110. calor heat calorie
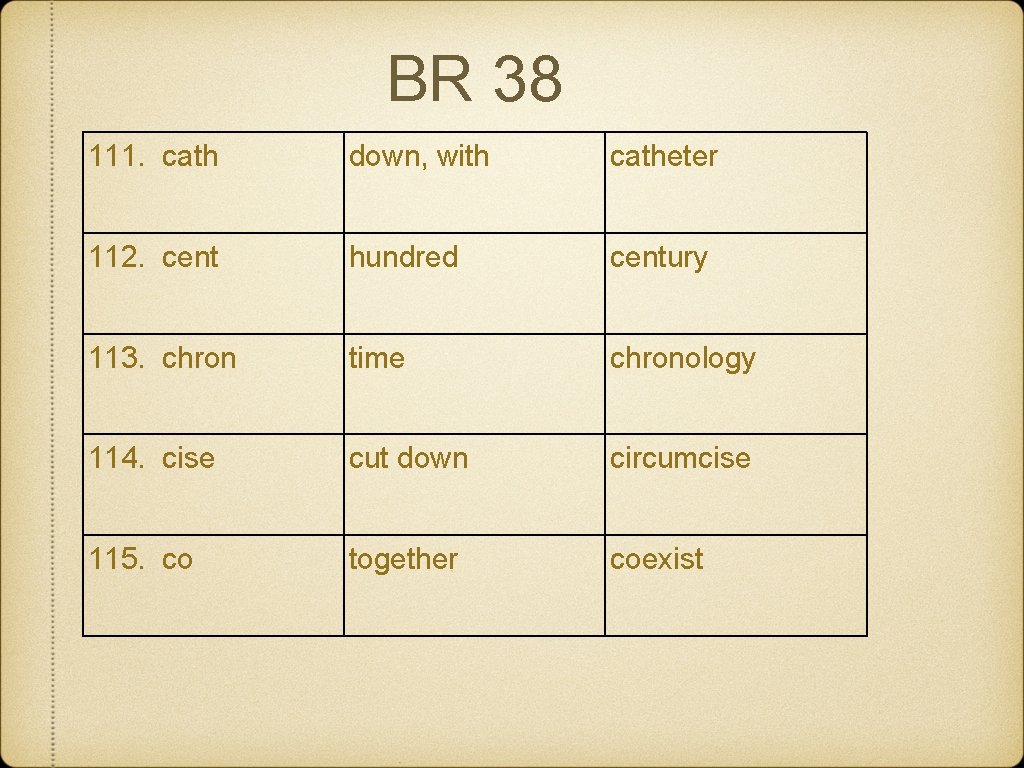
BR 38 111. cath down, with catheter 112. cent hundred century 113. chron time chronology 114. cise cut down circumcise 115. co together coexist
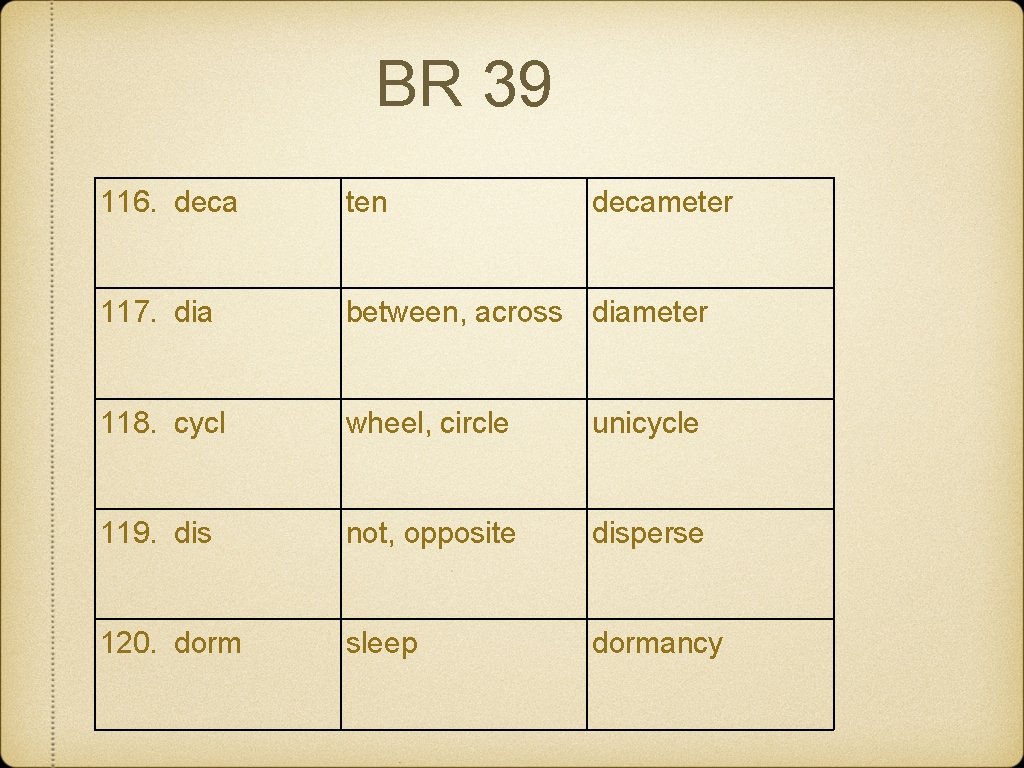
BR 39 116. deca ten decameter 117. dia between, across diameter 118. cycl wheel, circle unicycle 119. dis not, opposite disperse 120. dorm sleep dormancy

BR 40 A monatomic ion is an ion consisting of a single atom. For example, the calcium shown below here is a monatomic ion 2+ Ca THE 2+ or +2 is the CHARGE of the ION It is written as a superscript

BR 41 If an ion contains more than one atom, even if these atoms are of the same element, it is called a polyatomic ion. The polyatomic ion carbonate is shown here CO 32− THE 3 is a subscript (HOW MANY) and the -2 is a superscript (CHARGE)

BR 42 How do you “READ” a polyatomic ion? I will put a polyatomic ion in parentheses for you (OH) Not all resources will have the polyatomic in parentheses The subscript goes on the INSIDE of the parentheses The superscript goes on the outside of the parentheses EXAMPLE: (OH) -1 Is this polyatomic? Yes What is the name? Hydroxide How many O’s and H’s are there? 1 O and 1 H There are no subscripts so you assume the number 1 for Oxygen and Hydrogen What is the charge? -1

BR 43 Dot Structures or Lewis Dot Structures Valence Electron: Outer shell electrons are used in bonding Materials bond in octets which means it wants 8 electrons Use the Periodic Table to find the valence electrons and that is how many dots you use Ex: Ca has 2 valence electrons; so you draw Ca with 2 dots

BR 44 1. Draw the Lewis dot structure for Oxygen. 2. Draw the Lewis dot structure for Potassium.
- Slides: 63