Bellringer YESTERDAY Using the examples A B C
Bell-ringer: YESTERDAY Using the examples A, B, C, and D on the boards and your knowledge about enzymes, go to https: //forms. gle/w 6 a. VFZLYWfcjo 8 DE 8 (the link is on Moodle and the calendar) for a check for understanding about enzymes. Your participation will be graded. Your quiz is open for you to view your grade. If you did not take your quiz, a late quiz is available. DUE TODAY: Your 2 vocabulary words are due by 11: 59 PM today. I will turn off access to the slides after this time and email those of you that did not complete the assignment. The assignment is on Moodle, under the Biochemistry toggle, 9/29

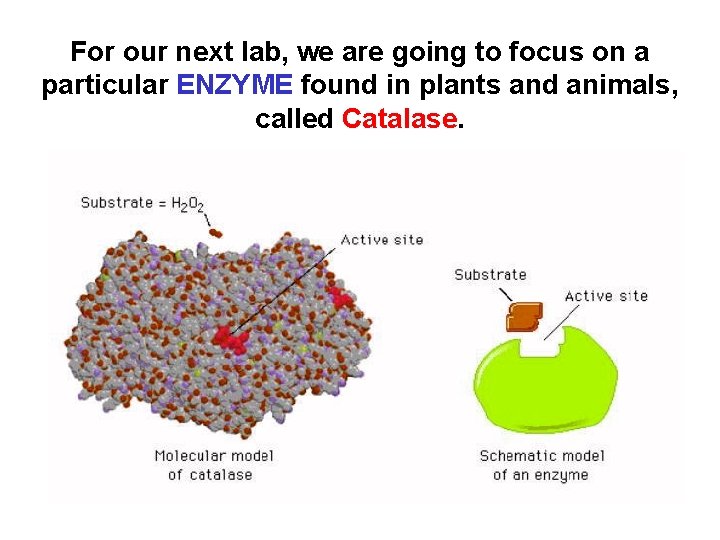
For our next lab, we are going to focus on a particular ENZYME found in plants and animals, called Catalase.

Catalase breaks down H 2 O 2 (which is Hydrogen Peroxide) WAIT…. I HAVE HYDROGEN PEROXIDE IN MY BODY? ? Hydrogen Peroxide is toxic inside your body. H 2 O 2 is naturally produced during cellular respiration when your cells are making energy (as a waste by-product). Catalase quickly arrives on the scene to break down the Hydrogen Peroxide molecule before it can do damage.

Catalase breaks down Hydrogen Peroxide INTO water and oxygen. 2 H 2 O 2 CATALASE 2 H 2 O + O₂ CATALASE Catalase can break down up to 200, 000 molecules of H 2 O 2 in a second. (Under ideal conditions)

We will be using Catalase enzymes found in CHICKEN LIVER We are going to measure the rate at which Catalase breaks down hydrogen peroxide (Dependent Variable) We want to know if the rate at which Catalase breaks down hydrogen peroxide is affected by changes in TEMPERATURE. (Independent Variable)

One of the ways you can measure the rate of this chemical reaction is by measuring the rate of the appearance of oxygen (O 2)

PRE LAB REASEARCH: • A pre-lab sheet is on Moodle/calendar to put in your digital notebook. • Use the background reading to answer the reading questions. • Research the Catalase enzyme • Write your hypothesis.

DATA COLLECTION: • Watch the four tests: - Room temperature catalase reaction - Cool temperature catalase reaction - Warm temperature catalase reaction - Hot temperature catalase reaction • As you watch, you will collect data on the accumulation of OXYGEN every 20 seconds for 5 minutes.

POST LAB ANALYSIS: • You will use the data collected to answer the questions on your Post-Lab Sheet. • Turn in your digital notebook to Google Classroom by Thurs. October 15 th 11: 59 PM. • Summative Grade, if not turned in on time, late points will be deducted and you will not be able to correct a late lab.

RUBRIC: COMPONENT Pre-Lab Slide Data Collection Slide Post-Lab Slide WORTH Exp. Design Questions 3 Background Questions 6 Research Questions 4 Hypothesis 2 Data Collection 12 Rate Calculations 4 Graph 9 Analysis Questions 4 Conclusion 4 Discussion/Reflection 4 TOTAL 52 EARNED FEEDBACK

Upside-Down Graduated Cylinder Lab Procedure: Cylinder spout Water Bath

Reaction Chamber I put 10 m. L of Hydrogen Peroxide (the substrate) at the bottom of the reaction chamber, careful not to get any on the sides. Hydrogen Peroxide (the substrate)

Soaked Disks (Where the enzyme will be) Then I soaked 7 paper disks in the blended chicken liver. With forceps, I stuck all the disks on one side of the reaction chamber, away from the H₂O₂ Hydrogen Peroxide (the substrate)

Room Temp Test: 21. 2°C (70. 16°F) Didn’t do anything ‘special’ to the liver. It had previously been refrigerated, so I let the liver sit out for a few hours before I took the temperature and conducted the test. Cool Temp Test: 11. 3°C (52. 3°F) The blended liver beaker sat in an ice bath for 20 minutes before I took the temperature and conducted the test. Warm Temp Test: 45°C (113°F) The blended liver beaker was put in a heated bath on a Bunsen burner for 20 minutes before I took the temperature and conducted the test. Hot Temp Test: 73. 8°C (164. 8°F) I microwaved the blended liver for 35 seconds. It was gross.

Without mixing the enzyme and substrate, I laid the reaction chamber in the water bath and put the open end of the chamber inside the graduated cylinder.

Oxygen 14 20 0 Time will be the amount of gas already in the graduated cylinder. This number will be subtracted from your 5 minute total m. L.

- Slides: 18