Bellringer Silently and by yourself Answer these questions
Bellringer Silently and by yourself Answer these questions in complete sentences in your composition book 1. What was your favorite experiment from the lab? 2. The experiment you liked, was there a chemical change, physical change, or both? How did you know 3. What was your least favorite experiment from the lab? Why

Atomic Theory How can we study what we can’t see
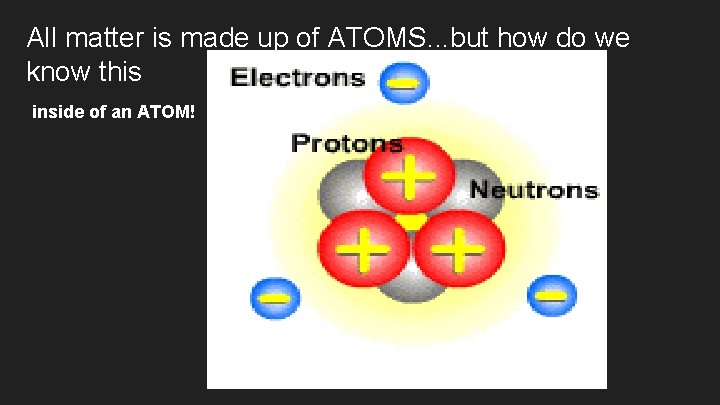
All matter is made up of ATOMS. . . but how do we know this inside of an ATOM!

Lets travel down history

Democritus � Greek philosopher In 430 B. C. proposed the idea that matter was composed of small pieces that could not be cut into smaller parts He used the word atomos meaning “uncuttable”

Dalton � English chemist � Theorized that atoms were like smooth, hard balls that could not be broken into smaller pieces

Dalton’s Theory � All elements consist of atoms that cannot be divided �All atoms of the same element are exactly alike and have the same mass. �Atoms of different elements are different and have different masses �An atom of one element cannot be changed into an atom of a different element. �Compounds are formed when atoms of more than one element combine in a specific ratio.

JJ Thomson � British physicist Discovered that atoms are made of even smaller parts Discovered the negatively charged electron but since atoms have no charge also reasoned that atoms must have positively charged particles. Thomson proposed a model with electrons scattered throughout a ball of positive charge
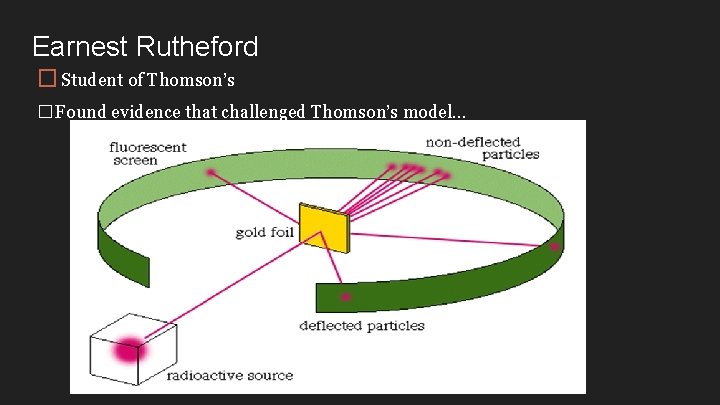
Earnest Rutheford � Student of Thomson’s �Found evidence that challenged Thomson’s model…
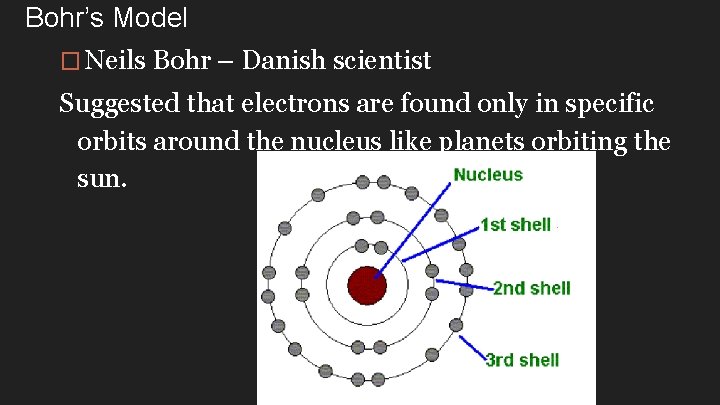
Bohr’s Model � Neils Bohr – Danish scientist Suggested that electrons are found only in specific orbits around the nucleus like planets orbiting the sun.

Cloud Model � In the 1920 s by Austrian physicist Erwin Schrodenger proposed that electrons move rapidly within a cloud-like region around the nucleus.
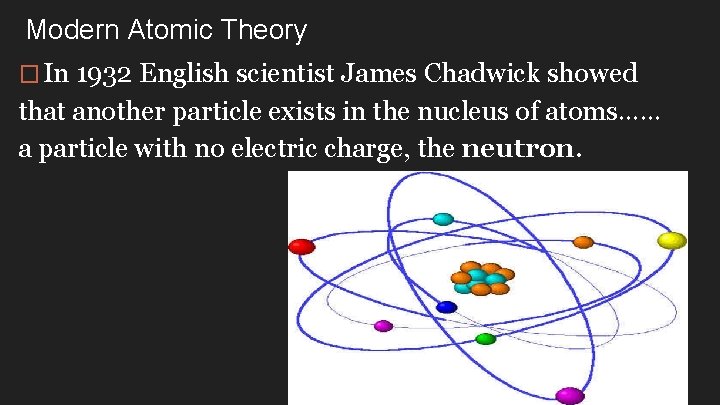
Modern Atomic Theory � In 1932 English scientist James Chadwick showed that another particle exists in the nucleus of atoms…… a particle with no electric charge, the neutron.

Teach Me! � You will write a book about atomic theory � It will have a table of contents and 4 chapters � The content for your book will be the material discussed in class today.
- Slides: 13