BellRinger n n Please put your homework on

Bell-Ringer n n Please put your homework on the left corner of your desk Get a board, marker/chalk, and a wipey thing Take out your Reference Tables and prepare for review Your test will be NEXT CLASS

Review Game Eureeka Atomic Structure

What is similar about a proton and a neutron? They are in the nucleus n They have a similar mass n

History. . What did the Greeks think all things were made of? n Who believed atoms looked like Plum Pudding? n What does the Gold Foil Experiment prove? n Why is the solar system model of an atom not perfect? n

Which of the following elements has the greatest nuclear charge? 1. 2. 3. 4. He N H C


2. Which sub-atomic particle in electrically neutral?

Vocabulary n Define ion- An element with a positive or negative charge n Define isotopen An atom with the same number of protons but a different number of neutrons as another atom

5. Which species has a negative charge? 1. 2. 3. 4. A lithium ion An alpha particle A beta particle An aluminum ion


Which of the below nuclei is an isotope of 10 p 11 n 1. 3. 9 p 11 n 11 p 12 n 2. 4. 11 p 10 n 10 p 9 n

Why are atomic masses not whole numbers? n They are the average of the masses of all the isotopes

What does the outermost principal energy level of Oxygen look like? n Orbital notation please
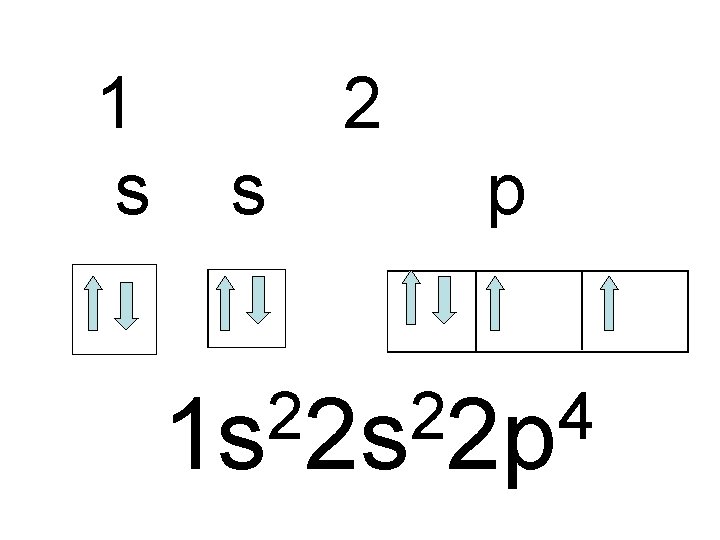
1 s 2 s p 2 2 4 1 s 2 s 2 p

Which has a completely filled 3 rd Principle Energy Level? 1. 2. 3. 4. K Zn Ca Ar


What is the maximum number of electrons a single orbital in the 3 d sublevel may contain? 1. 2. 3. 4. 5 2 3 4


As an S-2 ion loses two electrons and becomes an S 0 atom, the number of protons in the nucleus 1. 2. 3. 4. Increases Remains the same Decreases I don’t know but Mole Day is wonderful

If it’s Sulfur, it will have unchanging protons

Elements over which atomic number on the periodic table are radioactive? *

As an atom in the excited state returns to the ground state, what happens to the energy of the atom?

What is the total number of electrons in a Mg atom? * What is the total number of electrons in a Mg+2 ion? *

A particle is coming out of a radioactive source. What is the radioactive particle?

Draw the electron configuration for fluorine?

Draw an orbital notation for Magnesium n n n How is the Reference Table e-1 configuration related to yours? How many orbitals are completely filled? How many sublevels are filled? What is the valence shell? Could you identify the valence shell without drawing it? Draw the Lewis-Dot diagram

If the two elements below are mixed, what will the emission spectra look like?

That is the end of the game Tally your points and wait in suspense to see who wins n If you want a copy of these questions, pick them up from the chair in front of my room during activity period n
- Slides: 28