Bellringer 6 minutes Which picture below represents a

Bell-ringer: 6 minutes � Which picture below represents a homogeneous mixture and which represents a heterogeneous mixture? �Copy CCSS/Objectives/HW

Mantra - Your Turn! �Today is a great day for science because: �science is power. �We are powerful because: �We always make a difference.

Agenda 1. 2. 3. 4. 5. 6. Lab Safety Contracts – LAST DAY Paper Return/ Grade Sheets/ Leftover Collection Newsletters and Survey Lewis Dots Chemical Changes Summary/ Questions
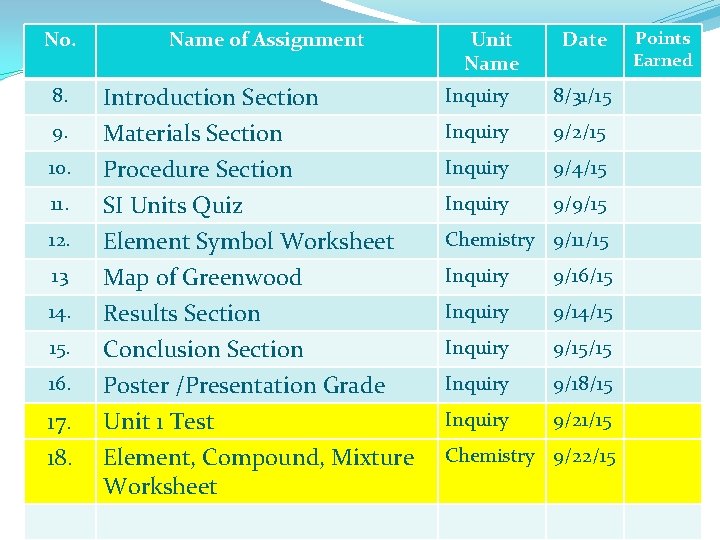
No. Name of Assignment Unit Name Date 8. Introduction Section Inquiry 8/31/15 9. Materials Section Inquiry 9/2/15 10. Procedure Section SI Units Quiz Element Symbol Worksheet Inquiry 9/4/15 Inquiry 9/9/15 Map of Greenwood Results Section Conclusion Section Poster /Presentation Grade Unit 1 Test Element, Compound, Mixture Worksheet Inquiry 9/16/15 Inquiry 9/14/15 Inquiry 9/15/15 Inquiry 9/18/15 Inquiry 9/21/15 11. 12. 13 14. 15. 16. 17. 18. Chemistry 9/11/15 Chemistry 9/22/15 Points Earned

Class Cup Categories of the Day �Quiet Coyote Time �Homework Completion �Cleanliness of Room at the end of class �# of Volunteers �Following Directions the First Time Feedback �Feedback pickup & parent newsletter
Trends in the Table

Worksheet Check �Elements, Compounds, Mixtures �Chemical vs. Physical Changes

�Chemical Change: The change of substances into other substances through a reorganization of the atoms. Copyright © 2010 Ryan P. Murphy
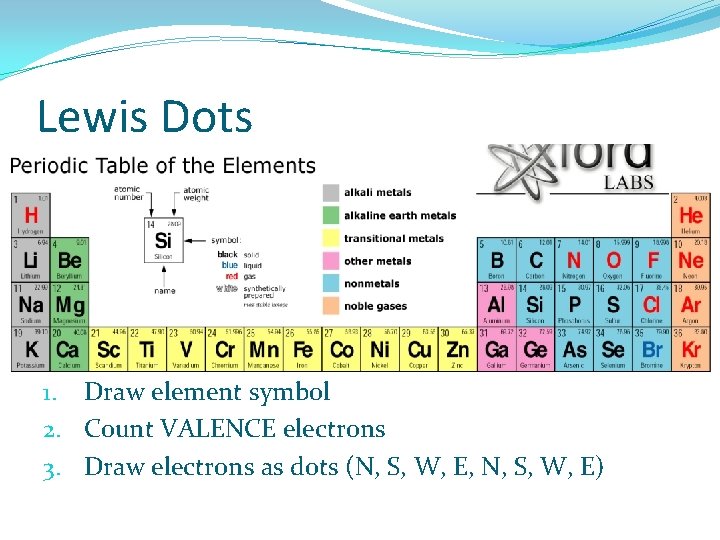
Lewis Dots 1. Draw element symbol 2. Count VALENCE electrons 3. Draw electrons as dots (N, S, W, E, N, S, W, E)

Equations �Practice Worksheet On Website Soon: Balancing equations

Last Items/ Questions �CLOSE-TOED SHOES, HAIR TIED BACK TOMORROW! �Think about your progress and effort in Unit 1 for Reflection/Celebration once grades are back �Extra practice worksheets on website soon �Balancing Equations

�Combustion: When oxygen combines with another compound to form water and carbon dioxide. CH 4(g) + 2 O 2(g) → CO 2(g) + 2 H 2 O(g) Methane oxygen Carbon Dioxide Water Can you name the molecules?

�Answer: Because fire requires oxygen to burn during combustion. �Shutting the windows prevents oxygen from flowing through the building.

�Combustion: A process in which a substance reacts with oxygen to give heat and light. Copyright © 2010 Ryan P. Murphy
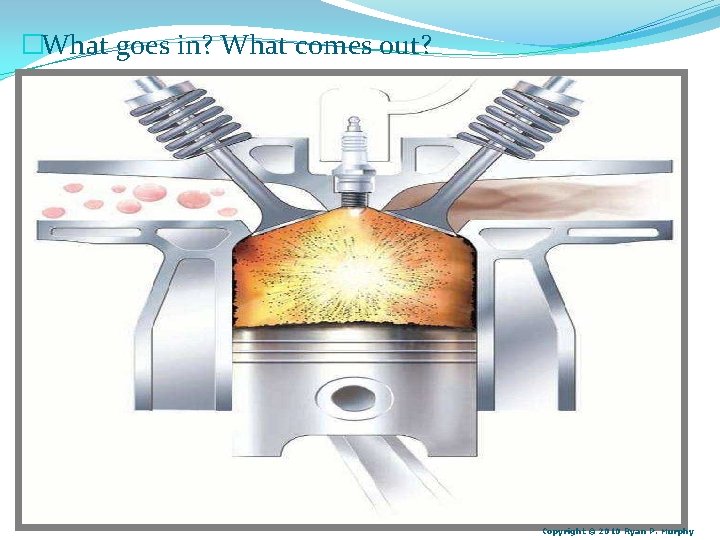
�What goes in? What comes out? Copyright © 2010 Ryan P. Murphy
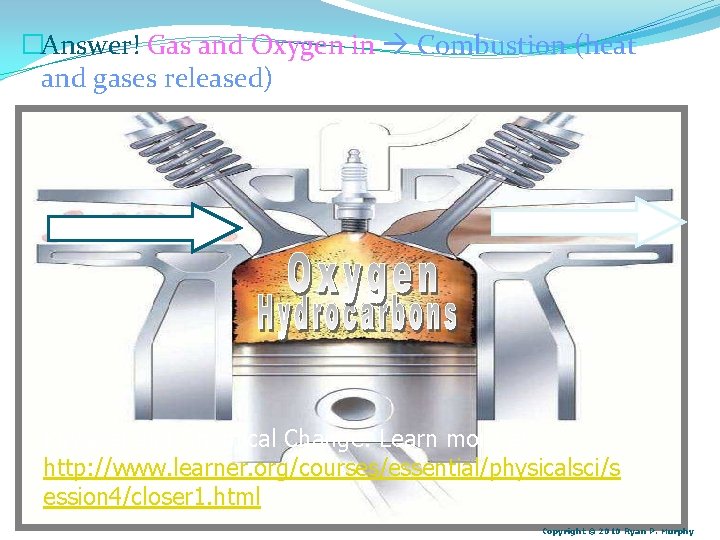
�Answer! Gas and Oxygen in Combustion (heat and gases released) Physical and Chemical Change: Learn more at… http: //www. learner. org/courses/essential/physicalsci/s ession 4/closer 1. html Copyright © 2010 Ryan P. Murphy

�Synthesis Reaction: When two or more simple compounds combine to form a more complicated one. A + B = AB �Zn + 2 HCl → Zn. Cl 2 + H 2 (g) Zn HCl H 2 (g) Zn. Cl 2 Copyright © 2010 Ryan P. Murphy

Rust (oxidation) is a synthesis reaction
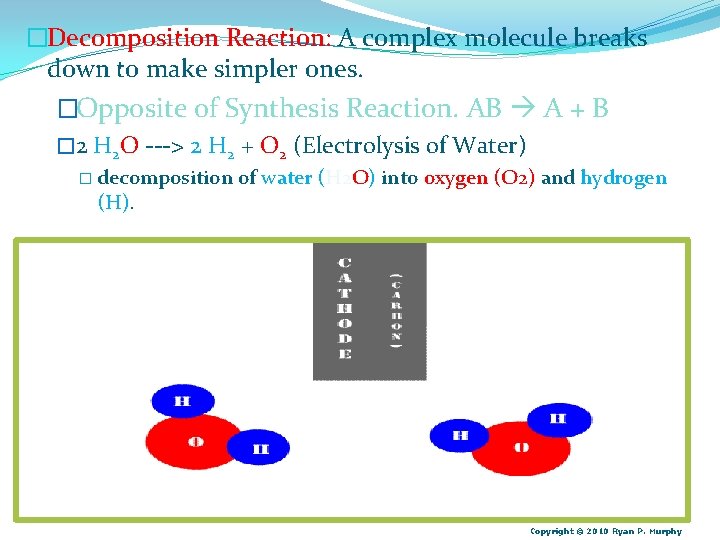
�Decomposition Reaction: A complex molecule breaks down to make simpler ones. �Opposite of Synthesis Reaction. AB A + B � 2 H 2 O ---> 2 H 2 + O 2 (Electrolysis of Water) � decomposition (H). of water (H 2 O) into oxygen (O 2) and hydrogen Copyright © 2010 Ryan P. Murphy

�Video Link. Black Snake Experiment �https: //www. youtube. com/watch? v=8 j. NG 8 w 0 l. J 4 U � 4: 1 Mix of Confectionary Sugar to Baking Soda When baking soda is heated to high temperature carbon dioxide gas is released. The pressure from the release of the gas causes the snake to grow. The snake is black because the sugar is carmelized by the heat. Sugar is a carbohydrate, so when you remove the water from the molecule, you're basically left with elemental carbon. The dehydration reaction is a type of elimination reaction.

Mercury (II) Thiocyanate Reacts with Oxygen)
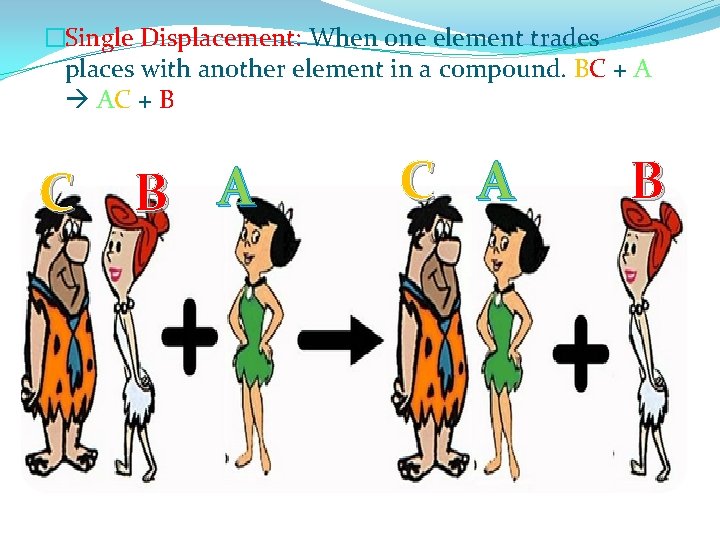
�Single Displacement: When one element trades places with another element in a compound. BC + A AC + B C B A C A B

Iron Reacts with Copper Sulfate

�Double Displacement: When two different molecules switch places, forming two entirely different compounds. �AB + CD AD + CB �Ag. NO 3 + Na. Cl → Ag. Cl + Na. NO 3

�Double Displacement: When two different molecules switch places, forming two entirely different compounds. �AB + CD AD + CB � Ag. NO 3 + Na. Cl → Ag. Cl + Na. NO 3 Month Later Double Date + +
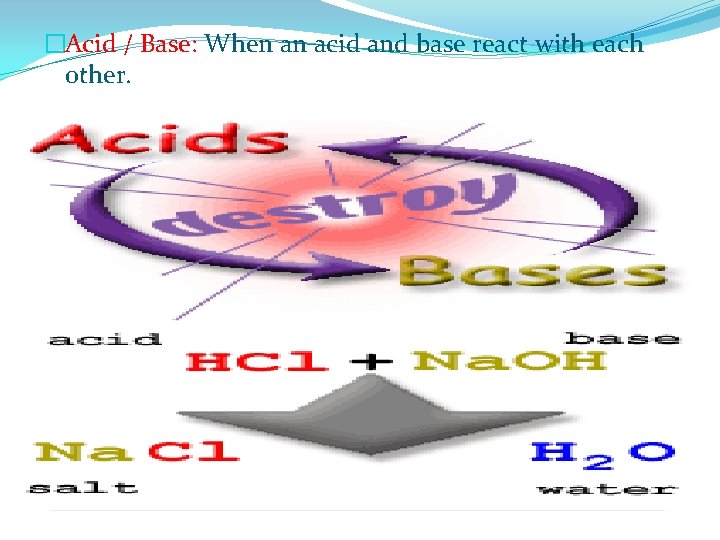
�Acid / Base: When an acid and base react with each other.
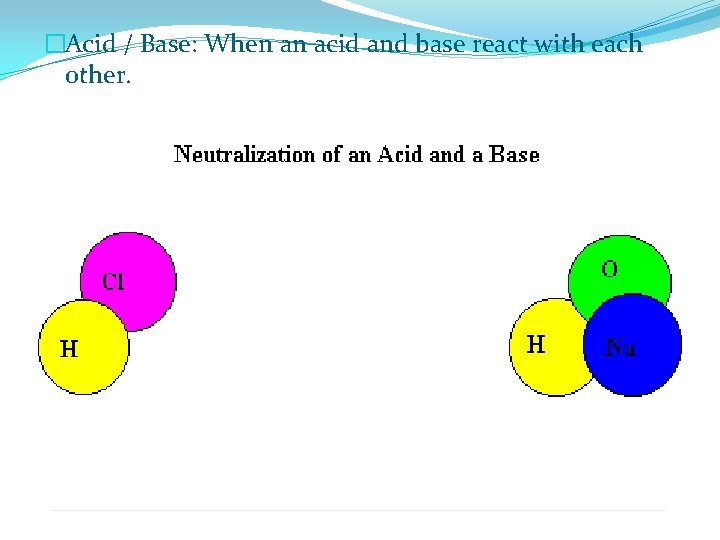
�Acid / Base: When an acid and base react with each other.

�What happens when we mix Red Bull with milk in a clear glass container? �Observe for 5 minutes. �Is it a physical or chemical RXN?

�Answer! Nasty (Chemical RXN) �Red Bull acidic. The acid attaches to one end of the milk protein. This forms salt (a bicarbonate). This alters the structure of the protein causing it to drop out of the suspension into a curdled mass on top.

�Quiz Wiz, Name the type of Chemical Reaction. �Word Bank are the 6 types of chemical reactions. Copyright © 2010 Ryan P. Murphy
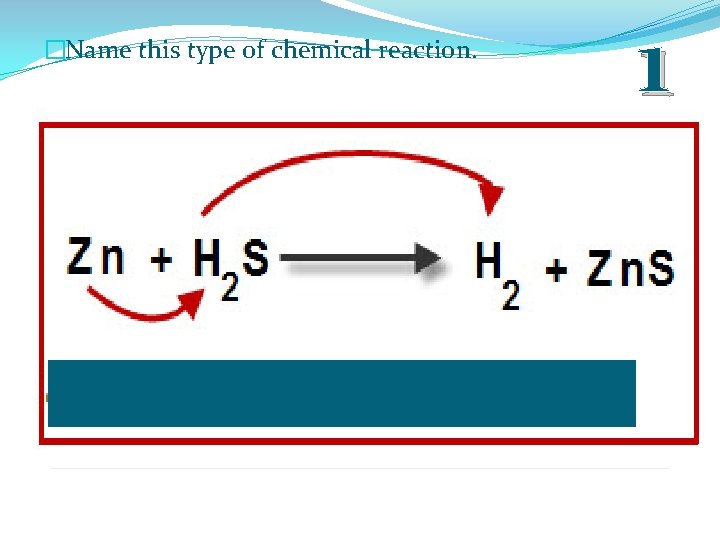
�Name this type of chemical reaction. 1
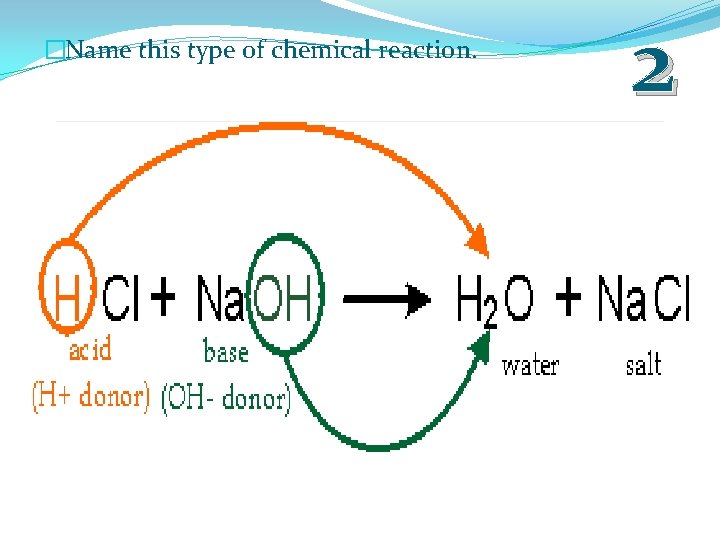
�Name this type of chemical reaction. 2
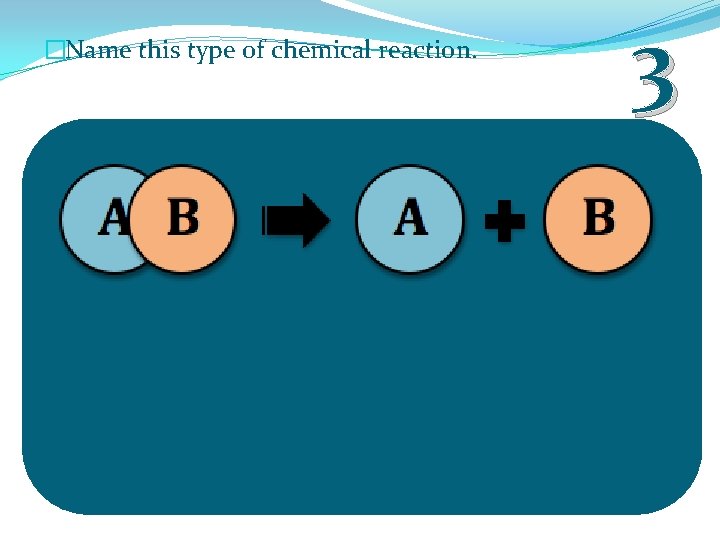
�Name this type of chemical reaction. 3
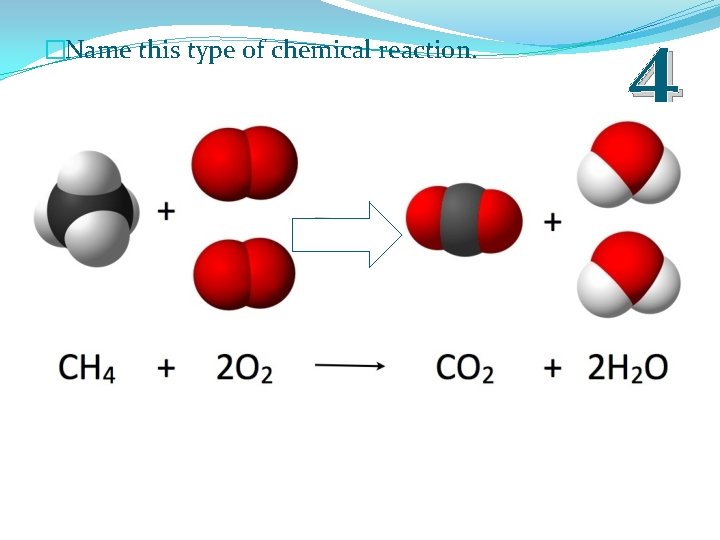
�Name this type of chemical reaction. 4
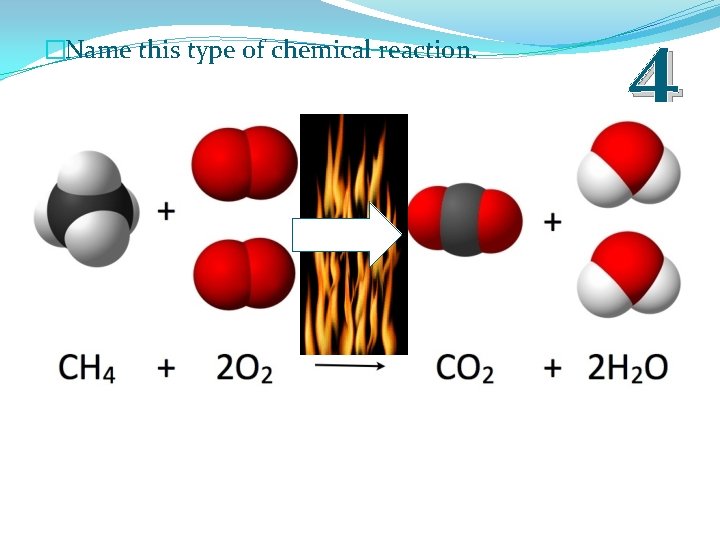
�Name this type of chemical reaction. 4
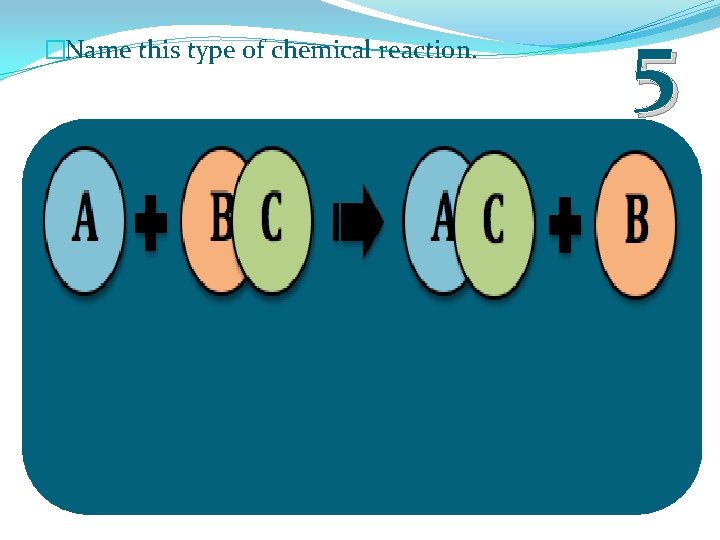
�Name this type of chemical reaction. 5
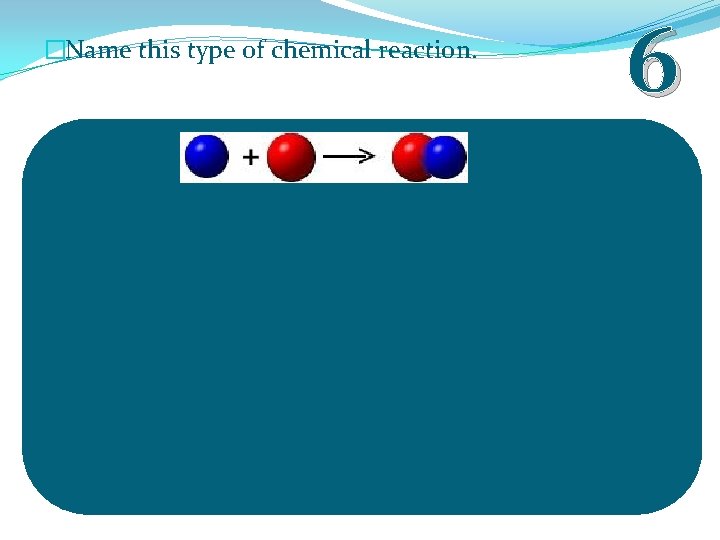
�Name this type of chemical reaction. 6
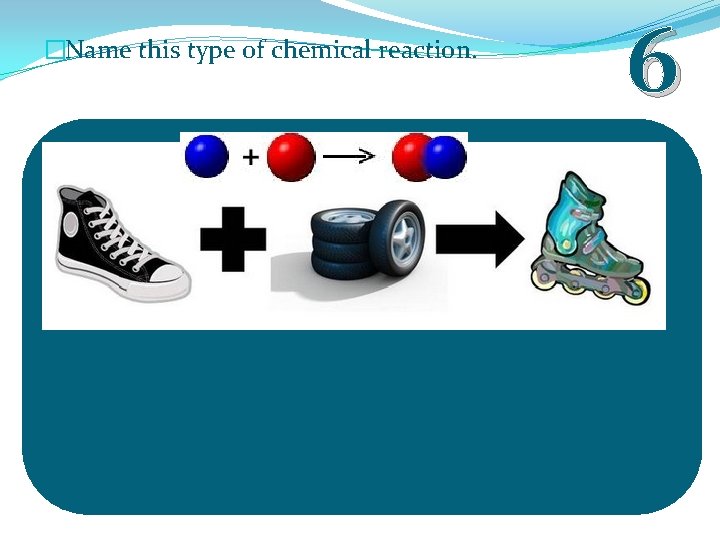
�Name this type of chemical reaction. 6
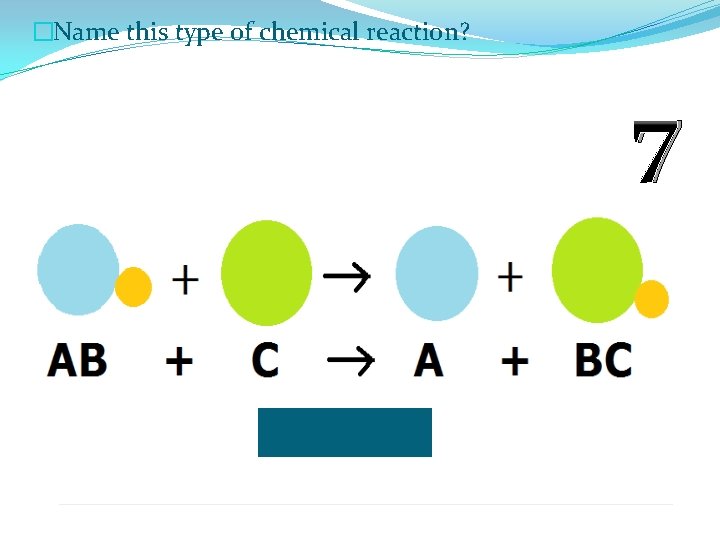
�Name this type of chemical reaction? 7

�Name this chemical reaction? 8
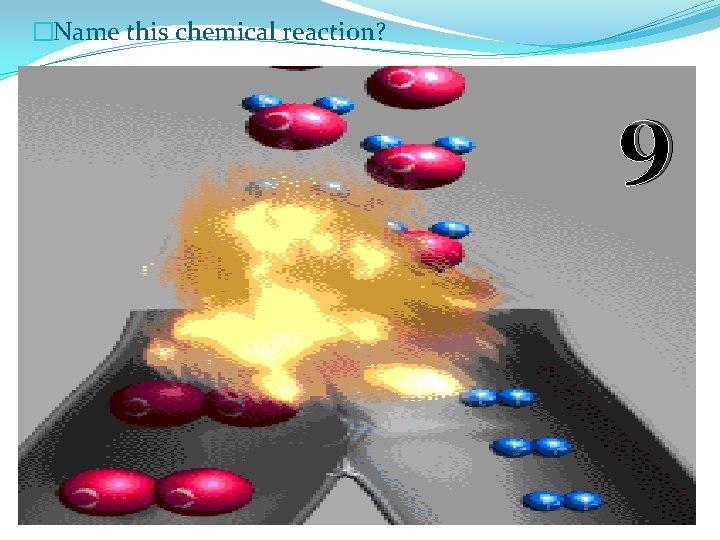
�Name this chemical reaction? 9
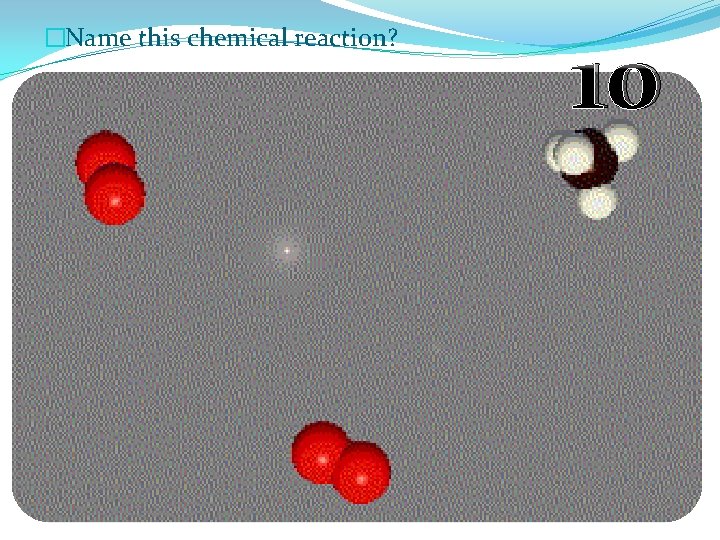
�Name this chemical reaction? 10

�Bonus: Name this split? *11

�Answers, types of chemical reactions.
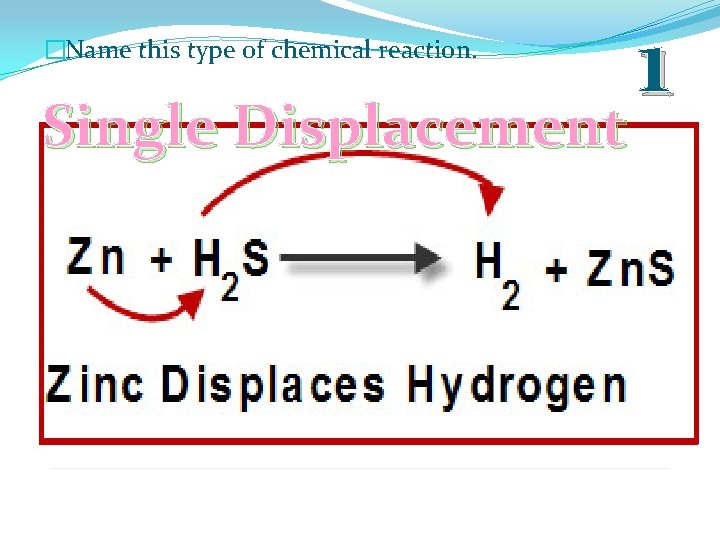
�Name this type of chemical reaction. Single Displacement 1
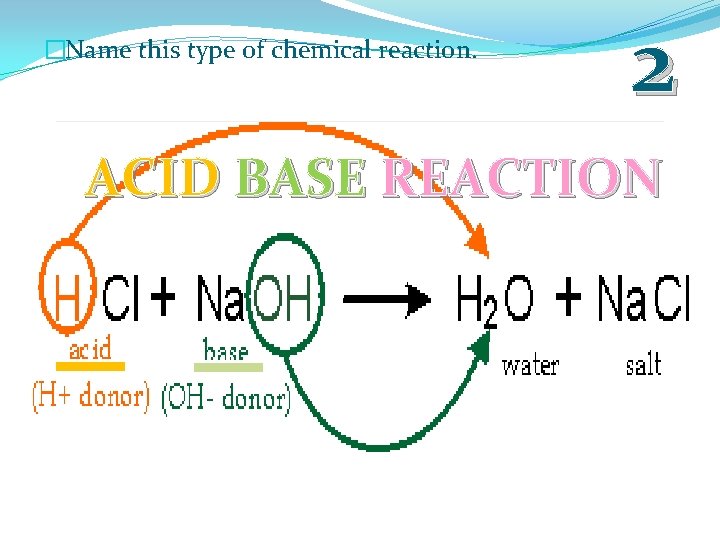
�Name this type of chemical reaction. 2 ACID BASE REACTION
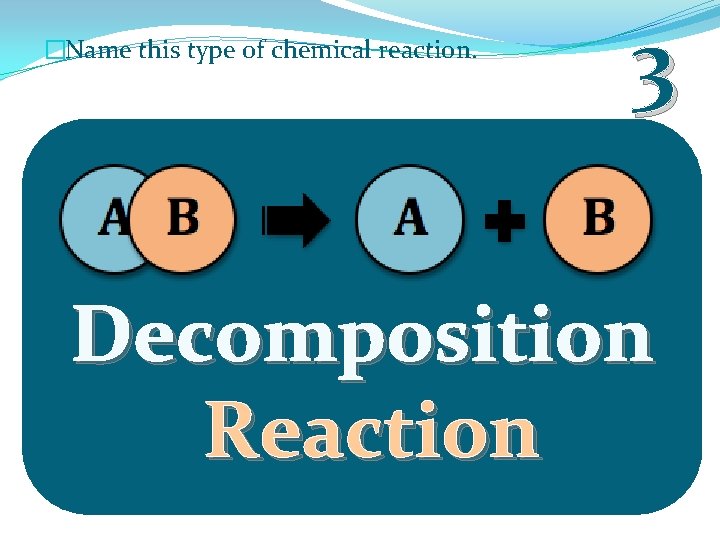
�Name this type of chemical reaction. 3 Decomposition Reaction
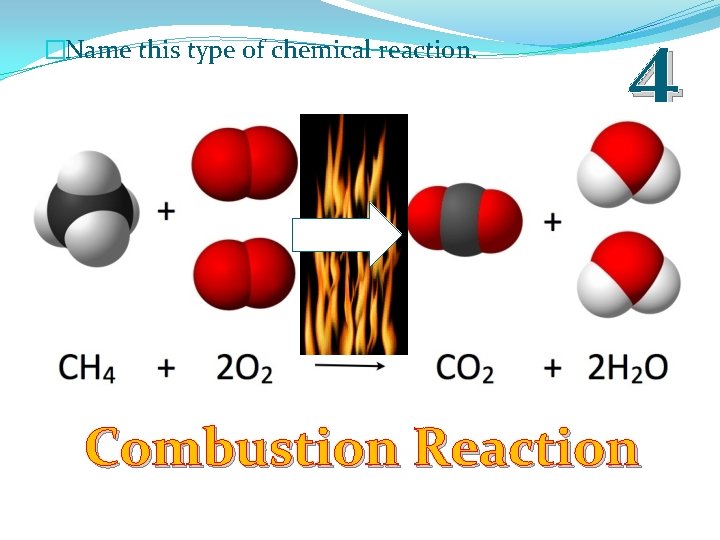
�Name this type of chemical reaction. 4 Combustion Reaction
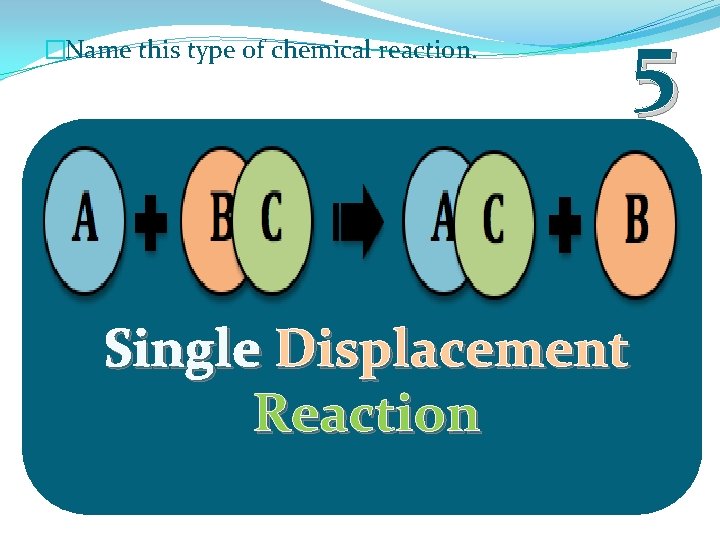
�Name this type of chemical reaction. 5 Single Displacement Reaction
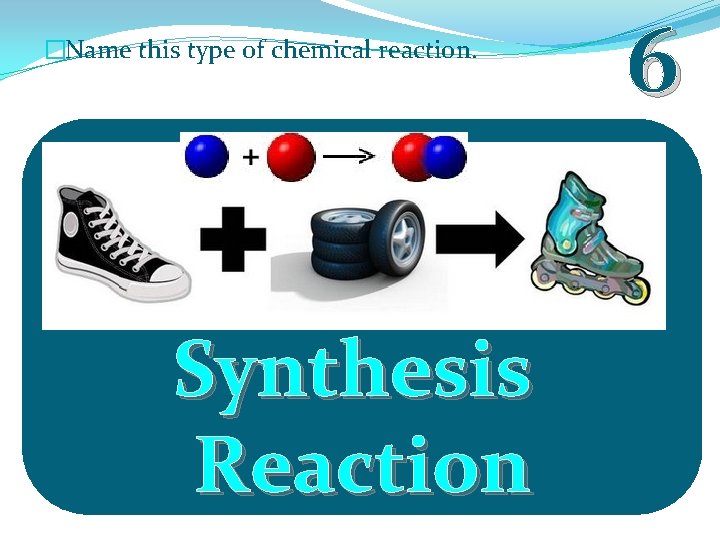
�Name this type of chemical reaction. Synthesis Reaction 6
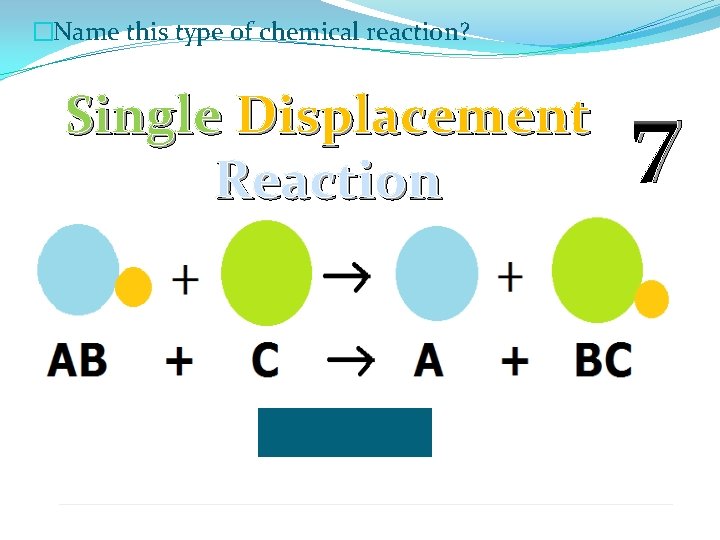
�Name this type of chemical reaction? Single Displacement Reaction 7

�Name this chemical reaction? Double Displacement Reaction 8
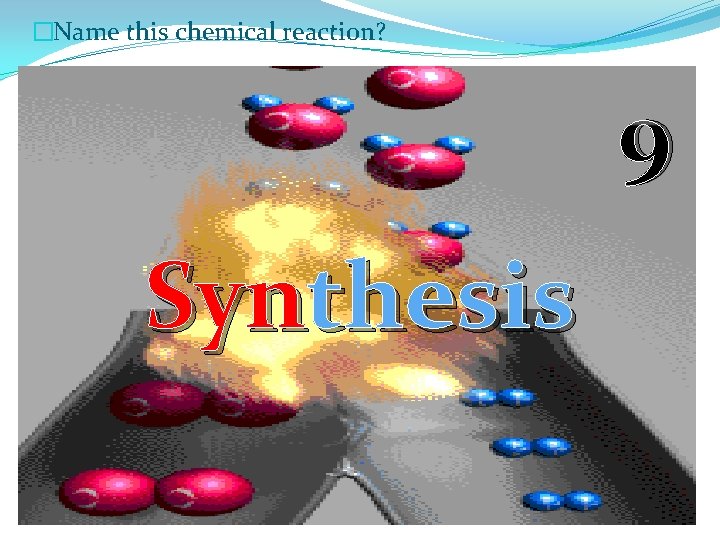
�Name this chemical reaction? 9 Synthesis

�Name this chemical reaction? Combustion Reaction 10

�Bonus: Name this split? Justin Bieber *11 Selena Gomez

�Try and be the first to figure out the hidden picture beneath the boxes. �Raise your hand when you think you know, you only get one guess. Copyright © 2010 Ryan P. Murphy








“I’m changing states. ” “I’m going from a solid to a liquid. ”

“I’m melting. ”

“or am I dissolving which is to become incorporated into a liquid so as to form a solution.

“I don’t know”

�Try and be the first to figure out the hidden picture beneath the boxes. �Raise your hand when you think you know, you only get one guess. Copyright © 2010 Ryan P. Murphy















that hing nd t y n A ass a has m up takes e spac
that hing nd t y n A ass a has m up takes e spac
that hing nd t y n A ass a has m up takes e spac

that hing nd t y n A ass a has m up takes e spac
that hing nd t y n A ass a has m up takes e spac
that hing nd t y n A ass a has m up takes e spac

that hing nd t y n A ass a has m up takes e spac

that hing nd t y n A ass a has m up takes e spac

that hing nd t y n A ass a has m up takes e spac
that hing nd t y n A ass a has m up takes e spac
that hing nd t y n A ass a has m up takes e spac

that hing nd t y n A ass a has m up takes e spac
that hing nd t y n A ass a has m up takes e spac

that hing nd t y n A ass a has m up takes e spac
- Slides: 97