Bellringer 492018 1 In your bellringer notebook count

Bellringer: 4/9/2018 1. In your bellringer notebook count how many atoms are in the following compounds: a) H 2 SO 4 b) KCl. O 3 c) NH 3 d) O 2 2. STOTD **QUIZ on Thursday: Balancing Equations & Types of Chemical Reactions **Lab on FRIDAY

Chemical Reactions Chapter 7

Chemical Reactions Chemical Reaction Process that rearranges atoms into different substances Evidence of a Chemical Reaction 1. 2. 3. 4. 5. Temperature change Production of light Color change Production of gas Creation of a precipitate (solid) end

Chemical Equations There are 2 parts in a chemical equation: Reactants To the Left of the arrow 2 H 2(g) + O 2(g) 2 H 2 O(l) Products To the Right of the arrow Yields/Forms/Produces The state of matter is shown with an: (s), (l), (g), (aq) aq- aqueous (dissolved in water) end
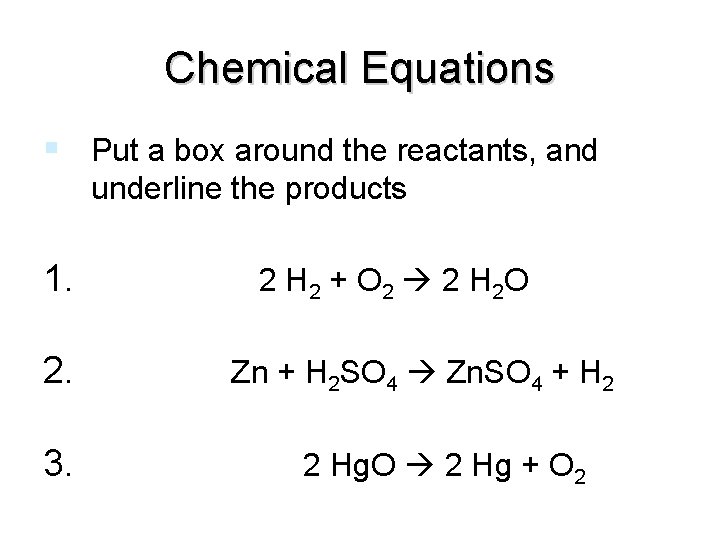
Chemical Equations Put a box around the reactants, and underline the products 1. 2 H 2 + O 2 2 H 2 O 2. Zn + H 2 SO 4 Zn. SO 4 + H 2 3. 2 Hg. O 2 Hg + O 2 end
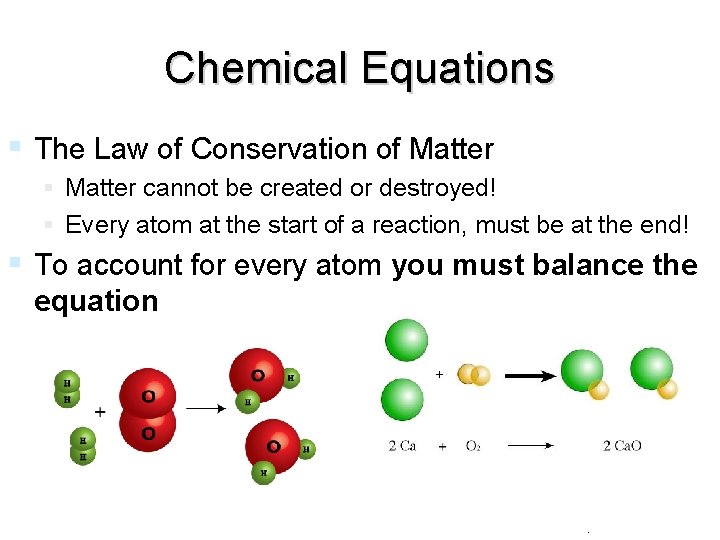
Chemical Equations The Law of Conservation of Matter cannot be created or destroyed! Every atom at the start of a reaction, must be at the end! To account for every atom you must balance the equation end

Chemical Equations Steps to balancing chemical equations: 1. Write the chemical equation 2. List the elements on each side of the equation 3. Place coefficients in front of each formula Distribute (multiply) the element by the coefficient Balance 1 element at a time! 4. If you need part of an atom, multiply everything by 2 5. Double check your work!!! NEVER Change the Subscripts end
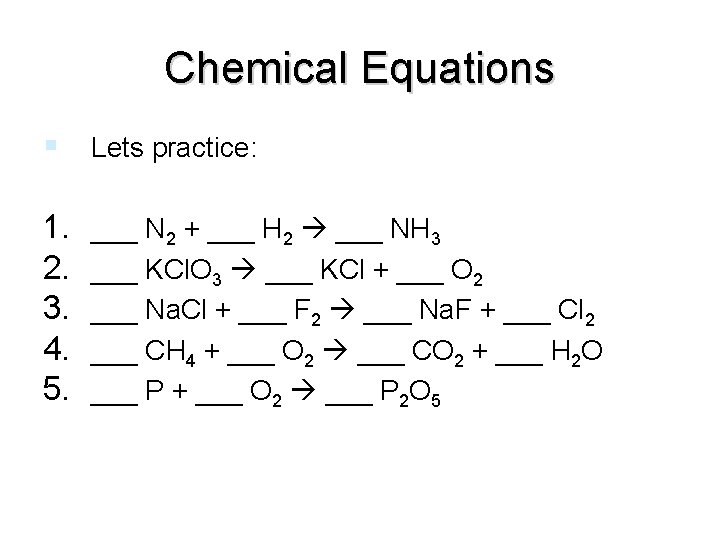
Chemical Equations Lets practice: 1. 2. 3. 4. 5. ___ N 2 + ___ H 2 ___ NH 3 ___ KCl. O 3 ___ KCl + ___ O 2 ___ Na. Cl + ___ F 2 ___ Na. F + ___ Cl 2 ___ CH 4 + ___ O 2 ___ CO 2 + ___ H 2 O ___ P + ___ O 2 ___ P 2 O 5 end
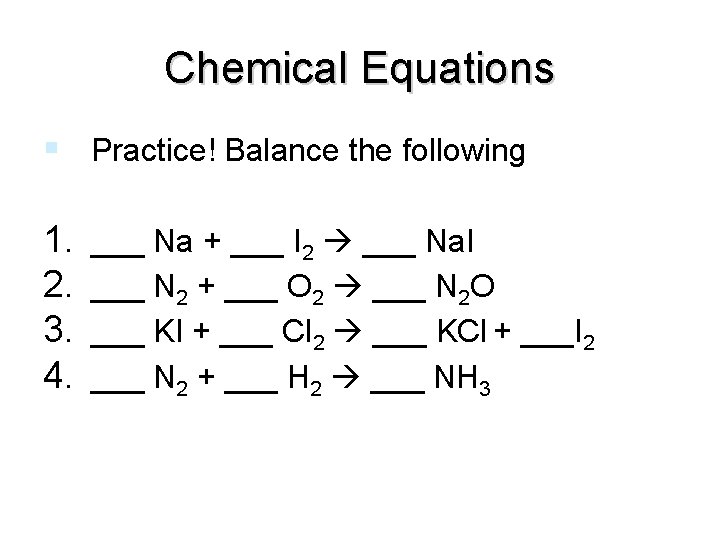
Chemical Equations Practice! Balance the following 1. 2. 3. 4. ___ Na + ___ I 2 ___ Na. I ___ N 2 + ___ O 2 ___ N 2 O ___ KI + ___ Cl 2 ___ KCl + ___I 2 ___ N 2 + ___ H 2 ___ NH 3

Chemical Equations • Word Equations – Names are written out instead of the formulas. – To solve: 1. Box the Reactants and Underline the Products 2. Write the formula of each reactant and product 3. Don’t forget to separate: – Each formula with a “+” – The reactants and products with an “ ” end

Chemical Equations • Write and balance the following equations: 1. Potassium oxide combines with water to make potassium hydroxide 2. Aluminum plus oxygen gas forms aluminum oxide 3. Nitrogen gas plus oxygen gas react and form dinitrogen pentoxide end

Chemical Equations • Practice: 1. Hydrogen plus oxygen yield water 2. Nitrogen plus hydrogen yield ammonium 3. Aluminum bromide plus chlorine yield aluminum chloride and bromine

Bellringer: 4/10/2018 Balance the following chemical equations: 1. ___ Na. Br + ___ Ca(OH)2 ___ Ca. Br 2 + ___Na. OH 2. ___ Na + ___ I 2 ___ Na. I 3. Safety Tip of the Day

Chemical Reactions There are 5 basic chemical reactions: 1. 2. 3. 4. 5. Synthesis Decomposition Single Replacement Double Replacement Combustion end

Chemical Reactions Synthesis 2 or more reactants combine to form 1 product A + B AB 2 Ca + O 2 2 Ca. O end
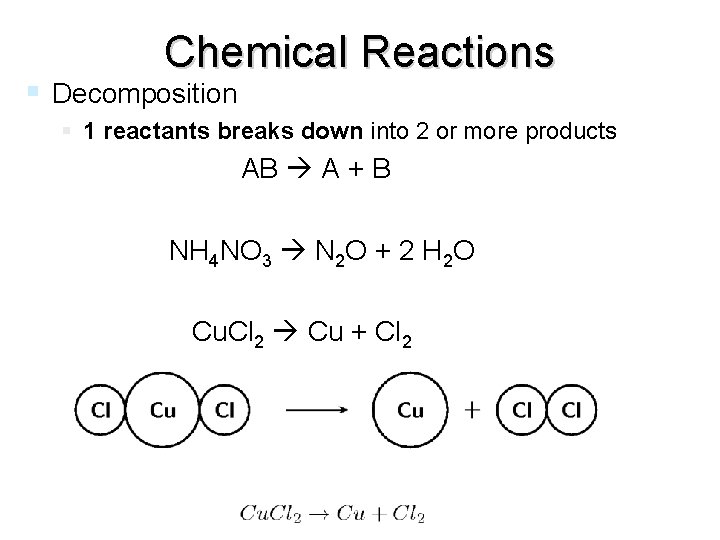
Chemical Reactions Decomposition 1 reactants breaks down into 2 or more products AB A + B NH 4 NO 3 N 2 O + 2 H 2 O Cu. Cl 2 Cu + Cl 2 end
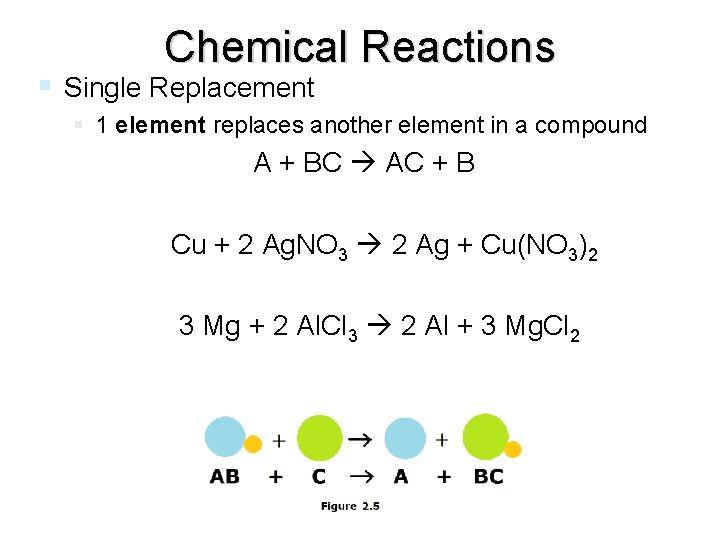
Chemical Reactions Single Replacement 1 element replaces another element in a compound A + BC AC + B Cu + 2 Ag. NO 3 2 Ag + Cu(NO 3)2 3 Mg + 2 Al. Cl 3 2 Al + 3 Mg. Cl 2 end
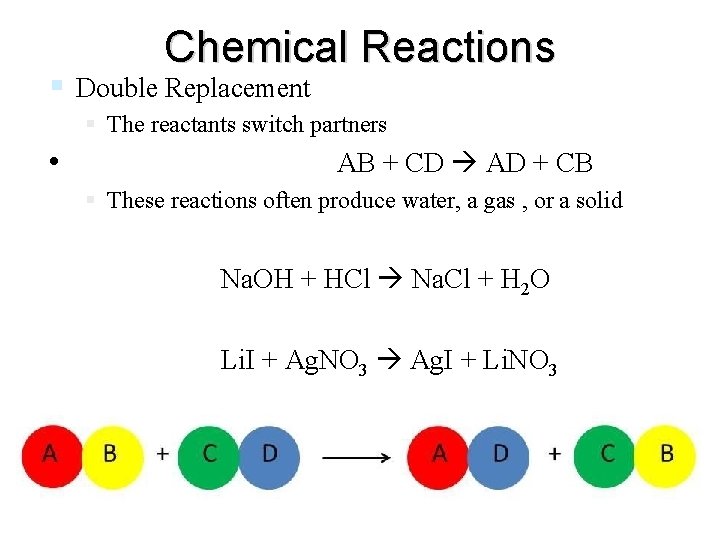
Chemical Reactions Double Replacement The reactants switch partners • AB + CD AD + CB These reactions often produce water, a gas , or a solid Na. OH + HCl Na. Cl + H 2 O Li. I + Ag. NO 3 Ag. I + Li. NO 3 end

Chemical Reactions Combustion Oxygen combines with a compound to give off heat and light Forms CO 2(g) and H 2 O(l) CH 4 + 2 O 2 CO 2 + 2 H 2 O end

Chemical Reaction Classify the following: 1. 2. 3. 4. 5. 2 H 2 + O 2 2 H 2 O Zn + H 2 SO 4 Zn. SO 4 + H 2 2 Hg. O 2 Hg + O 2 Ag. NO 3 + Na. Cl Ag. Cl + Na. NO 3 2 C 2 H 6 + 7 O 2 4 CO 2 + 6 H 2 O end

Bellringer: 4/11/2018 1. What are the 5 basic chemical reactions and their general equations? 2. Classify the following: a) SO 3 + H 2 O H 2 SO 4 b) Ca. Cl 2 + F 2 Ca. F 2 + Cl 2 c) 2 Na. Cl 2 Na + Cl 2 3. Safety Tip of the Day

Bellringer: 1. How do you know if a reaction is endothermic? 2. How do you know if a reaction is exothermic? 3. Provide an example of an exothermic chemical reaction. 4. STOTD

Energy Changes Chemical Energy stored in chemical bonds 2 Types: 1. Exothermic 2. Endothermic end
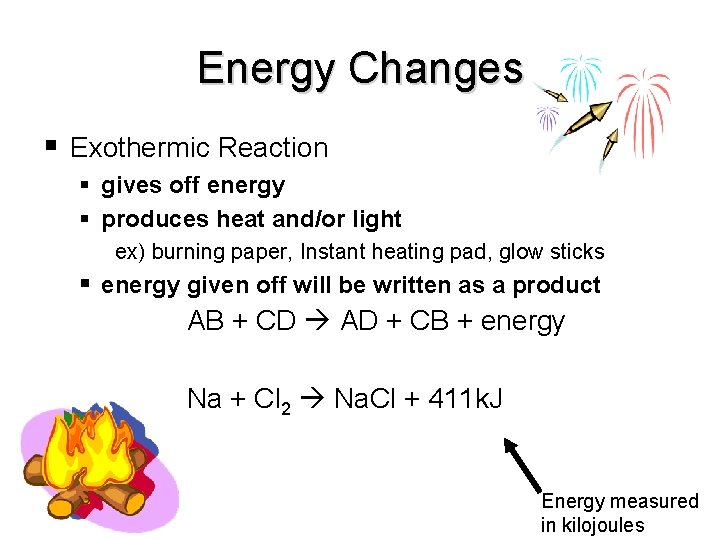
Energy Changes Exothermic Reaction gives off energy produces heat and/or light ex) burning paper, Instant heating pad, glow sticks energy given off will be written as a product AB + CD AD + CB + energy Na + Cl 2 Na. Cl + 411 k. J end Energy measured in kilojoules
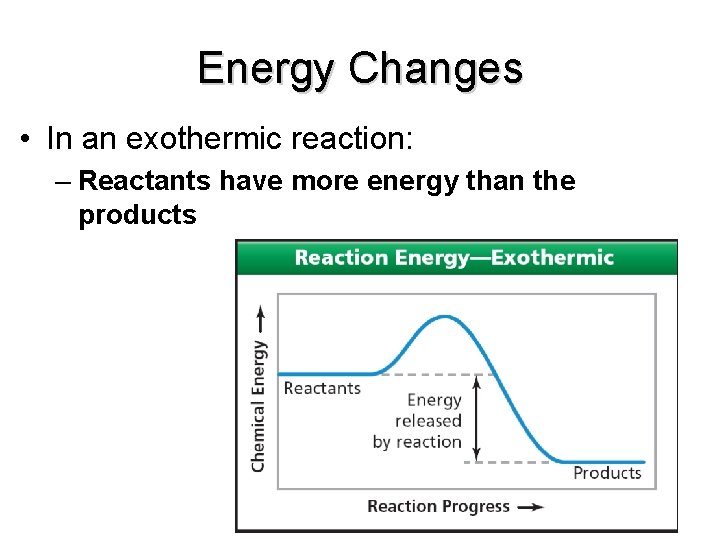
Energy Changes • In an exothermic reaction: – Reactants have more energy than the products end

Energy Changes Endothermic Reaction Absorbs energy Surroundings get colder Ex) Instant icepacks energy taken in will be written as a reactant AB + CD + energy AD + CB 15000 k. J + 6 CO 2 + H 2 O C 6 H 12 O 6 + 6 O 2 Energy measured end in kilojoules
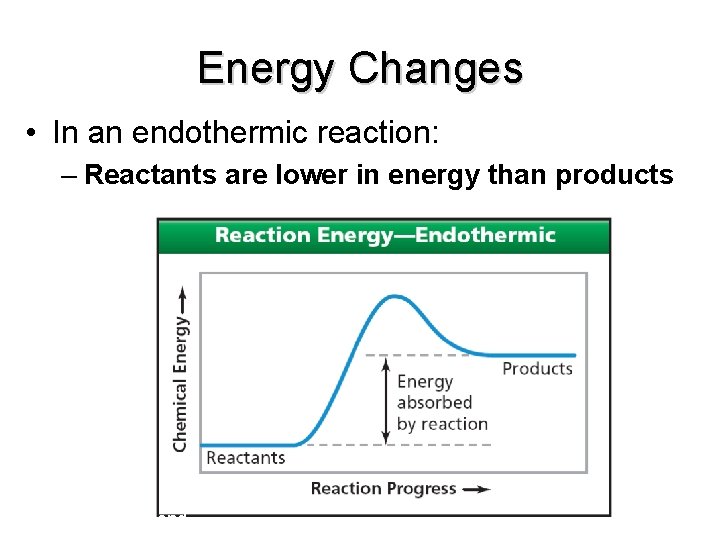
Energy Changes • In an endothermic reaction: – Reactants are lower in energy than products end

Activity With a partner: 1. In your notes write the question, “How can you make a chemical reaction go faster? ” 2. With your partner brainstorm answers to the question and write them down. • Be prepared to share your answers

Reaction Rates Reaction Rate How fast the reactants change to products There are 5 ways to make a reaction go faster: 1. Temperature 2. Surface Area 3. Stirring 4. Concentration 5. Catalysts end

Reaction Rates 1. Temperature Adding heat makes molecules move faster Move Faster = Faster Reaction 2. Surface Area Grind up a substance into a powder More Surface Area = Faster Reaction 3. Stirring makes things move around More Movement = Faster Reaction 4. Concentration Adding more reactants makes it easier for them to combine More Reactants = Faster Reaction end
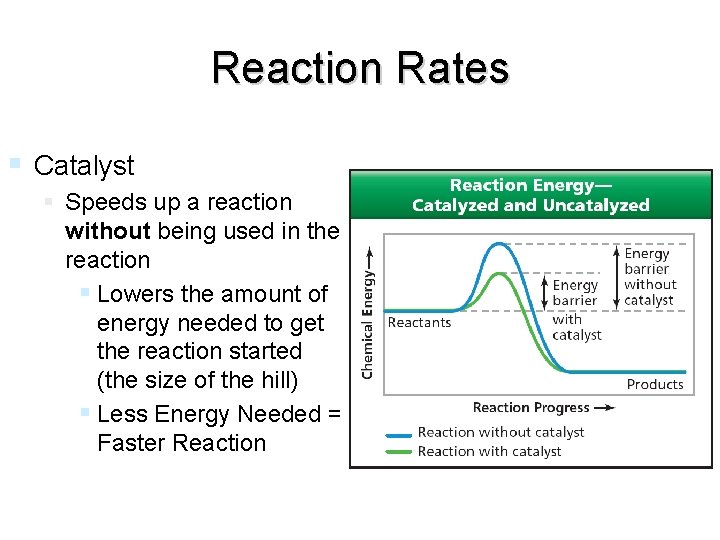
Reaction Rates Catalyst Speeds up a reaction without being used in the reaction Lowers the amount of energy needed to get the reaction started (the size of the hill) Less Energy Needed = Faster Reaction end
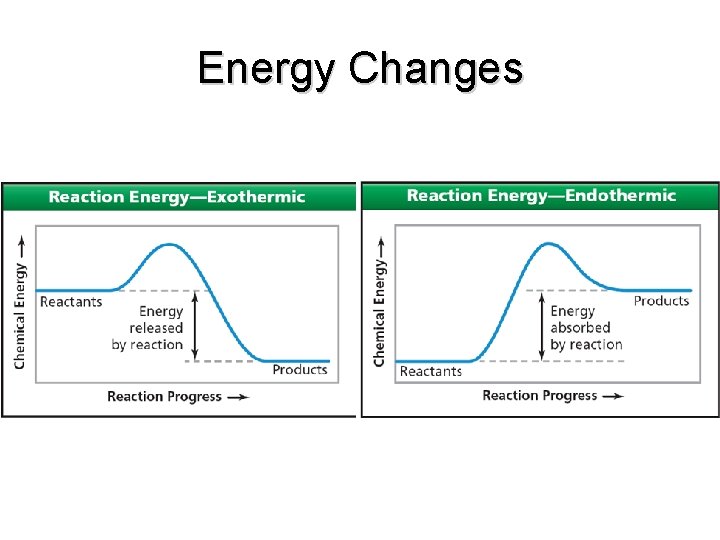
Energy Changes end
- Slides: 32