Bellringer 1 What did John Dalton do 2

Bellringer 1. What did John Dalton do? 2. Around what year was the electron discovered? 3. Who discovered that the electrons exist in energy levels? 4. How many energy levels exist in atoms? 5. When was the first atomic theory proposed?

http: //www. youtube. com/watch? v=thn Dx. Fdkz. Zs Crash Course atomic models In notes: Lavoisier Democritus John Dalton J. J. Thomson Ernest Rutherford Niels Bohr

Notes Unit 2 B CH 5 Electron Configurations Or telling electrons where they can go!!
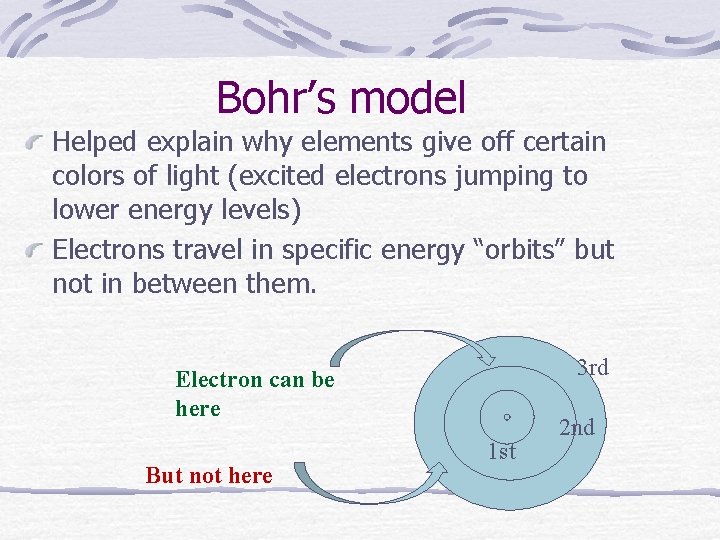
Bohr’s model Helped explain why elements give off certain colors of light (excited electrons jumping to lower energy levels) Electrons travel in specific energy “orbits” but not in between them. 3 rd Electron can be here But not here 1 st 2 nd
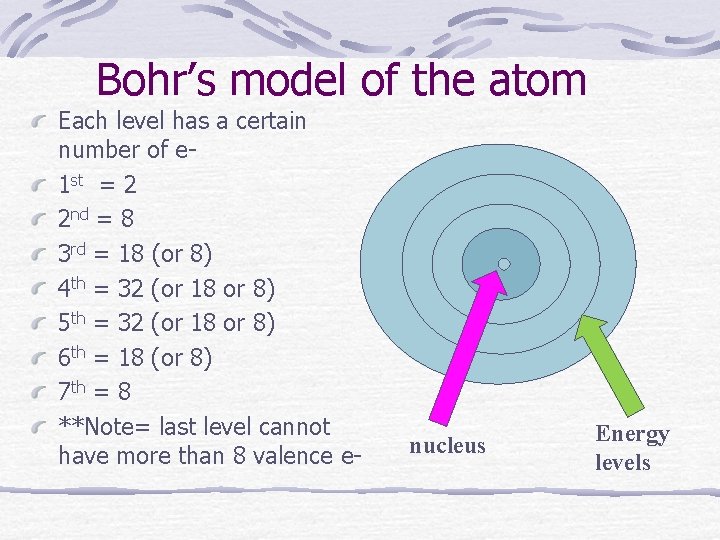
Bohr’s model of the atom Each level has a certain number of e 1 st = 2 2 nd = 8 3 rd = 18 (or 8) 4 th = 32 (or 18 or 8) 5 th = 32 (or 18 or 8) 6 th = 18 (or 8) 7 th = 8 **Note= last level cannot have more than 8 valence e- nucleus Energy levels

Bohr models of various atoms. 1. Draw (or write) the Bohr sequence for Calcium (20) 2. Give the # of valence electrons (last level) 3. Tell if will lose e- (like metals, have less than 4) or gain e- (like nonmetals, have more than 4) or metalloids (either gain or lose) 4. Give the isotope symbol

Bohr models of various atoms. 1. Draw (or write) the Bohr sequence for Bromine 2. Give the # of valence electrons (last level) 3. Tell if will lose e- (like metals, have less than 4) or gain e- (like nonmetals, have more than 4) or metalloids (either gain or lose) 4. Give the isotope symbol

Bohring work For all the following elements: Elements (write the name also) a) 11 d) 23 b) 17 e) 34 c) 20 Draw (or write) the Bohr sequence 1. Give the # of valence electrons 2. Tell if will lose e- or gain e- or either 3. Give the isotope symbol

Niels Bohr’s Atomic model Yes! It’s Bohring! http: //videos. howstuffworks. c om/hsw/5787 -niels-bohrsatomic-model-video. htm
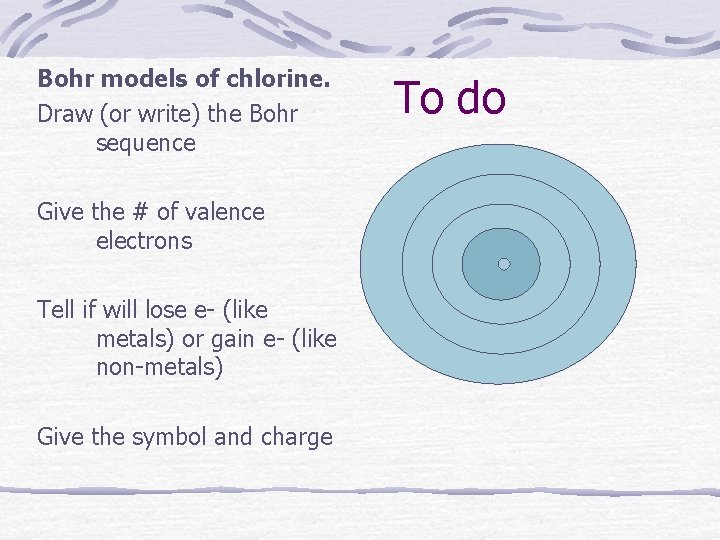
Bohr models of chlorine. Draw (or write) the Bohr sequence Give the # of valence electrons Tell if will lose e- (like metals) or gain e- (like non-metals) Give the symbol and charge To do
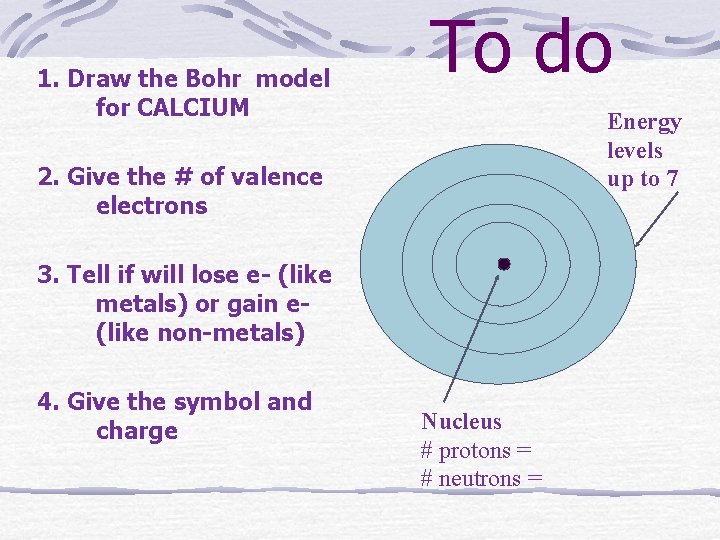
1. Draw the Bohr model for CALCIUM To do Energy levels up to 7 2. Give the # of valence electrons 3. Tell if will lose e- (like metals) or gain e(like non-metals) 4. Give the symbol and charge Nucleus # protons = # neutrons =

Bellringer check notes and PT 1. 2. 3. 4. 5. Give the # of electrons for Cesium. Draw (or write) the Bohr sequence of electrons Give the # of valence electrons (last level) Tell if will lose e- (like metals, have less than 4) or gain e- (like nonmetals, have more than 4) or metalloids (either gain or lose) Give the isotope symbol

Jumping electrons When an electron jumps from its normal or ground state to a higher energy level it absorbs a quantum of energy (heat or electricity). (Quanta is plural) Energy is quantized (absorbs only certain amounts of energy) It is now in the excited state and will give off a photon of light (Electromagnetic radiation) when it returns.

Spectra Each element has a characteristic pattern of light that it emits. This can be used as a “fingerprint” for identification.

Videos http: //www. youtu be. com/watch? v= 1 Jr. O 7 QQ 1 o 1 M UC Berkeley spectra
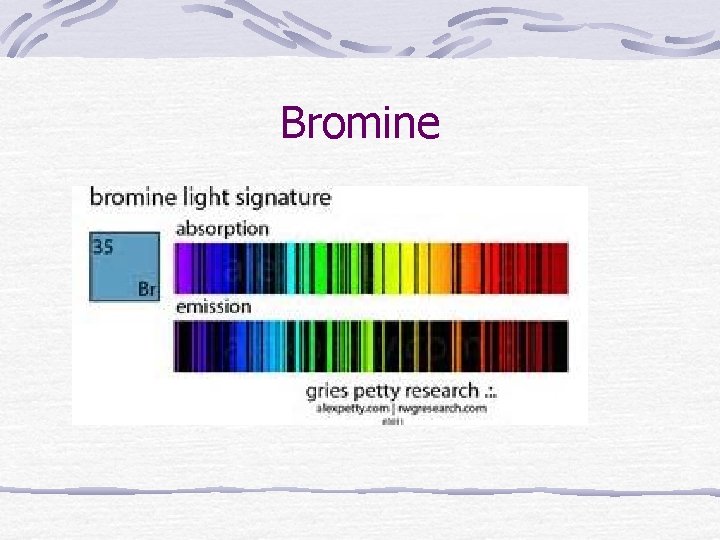
Bromine

Atomic Bookwork HOLT blue book Find these in notes & CH 3 Restate and answer. Tell why Democritus is famous. 2. Tell why Dalton is famous. 3. State 5 of his postulates 4. Draw and label a Cathode ray tube. 5. Describe how it works and how Thomson inferred the existence of the electron. 6. Tell why Millikan is famous. 7. Draw and label the gold foil expt set-up. 8. Explain how it worked and how Rutherford inferred the nucleus of the atom. 9. Tell what Chadwick is famous for. 10. Explain what isotopes are and give the names and isotope symbols for the 3 isotopes of Hydrogen. 1.

Atomic Bookwork HOLT blue book Find these in notes & CH 3 Restate and answer. 1. 2. 3. 4. 5. 6. 7. 8. Tell why Democritus is famous. Tell why Dalton is famous. State 5 of his postulates Draw and label a Cathode ray tube. Describe how it works and how Thomson inferred the existence of the electron. Tell why Millikan is famous. Draw and label the gold foil expt set-up. Explain how it worked and how Rutherford inferred the nucleus of the atom.

Bellringer 1. How many sublevels CAN exist in an 2. 3. 4. 5. 6. atom? Give the letters. Which has a four-leaf-clover shape? Which has a spherical shape? How many (and which) sublevels can exist on level 3? Write the electron configuration for Arsenic, As.

Modern views Electrons can act as particles and sometimes as waves (dual nature) Electrons travel in paths that look like clouds. The size and shape of these depend on how much energy the electrons have. The farther from the nucleus, the more energy. There are 7 energy levels with these max # of e- (2 -8 -1832 -32 -18 -8).

Electron levels Each energy level is divided into different sublevels (s, p, d, f). Each sublevel is divided into orbitals. An orbital is the space where two electrons travel. They have the shape of the sublevel but different orientations. Sublevels: s = 2 e- max = ___ orbitals p = 6 e- max = ___ orbitals d = 10 e- max = ___ orbitals f = 14 e- max = ___ orbitals 1 st = 2 e- max = _______ 2 nd = _8__ e- max = _______ 3 rd = _18__ e- max = _______ 4 th = _32__ e- max = _______ 5 th = __32_ e- max = ______ 6 th = _18__ e- max = _______ 7 th = __8_ e- max = _______
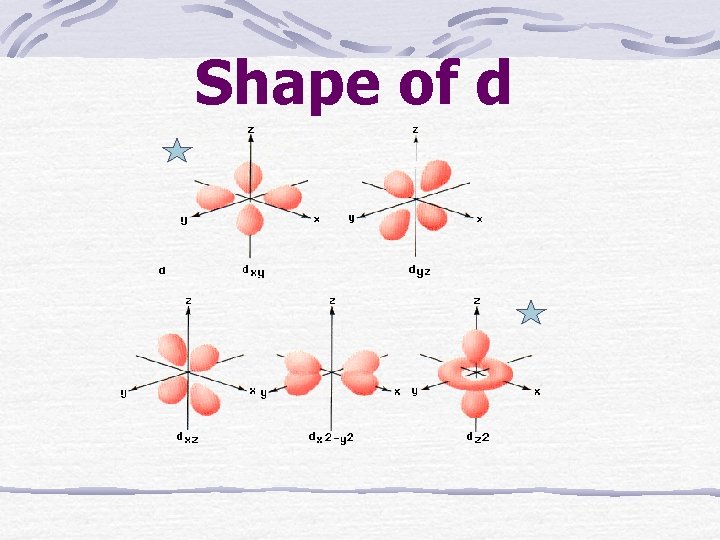
Shape of d
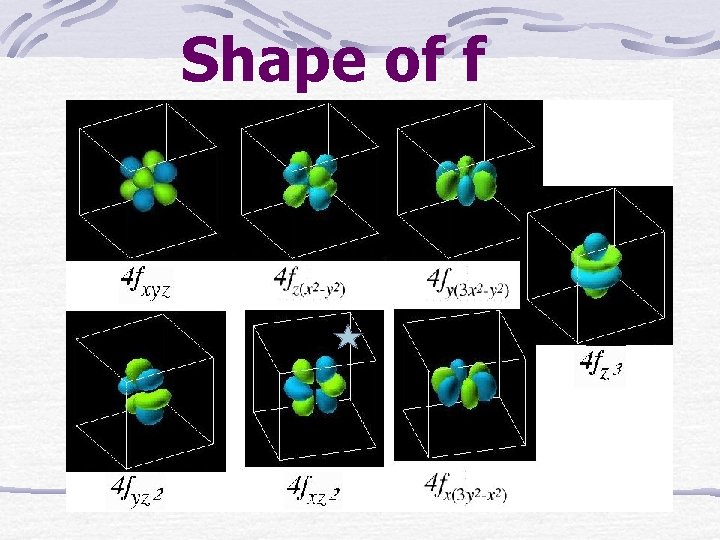
Shape of f

Mind Blowing!! Just imagine an atom with for example 92 electrons like Uranium. All are whizzing around the nucleus at the speed of light in different shaped paths. Just read

Video http: //www. youtube. com/watch? v=Kj. Ngq 16 j. EY&feature=related Thomson etc great 14 min http: //www. youtube. com/watch? v=-FWxd 78 s. OZ 8 CRT Univ Sydney http: //www. youtube. com/watch? v=2 x. KZRp. As. WL 8 CRT http: //www. youtube. com/watch? v=O 9 Goyscbazk

Electron configuration (long form) This gives MORE information about predicting properties. The placement of an elements electrons is written as: These add up to total # of e 2 1 s , 2 2 s , 6 2 p , 2 3 s , 6 3 p
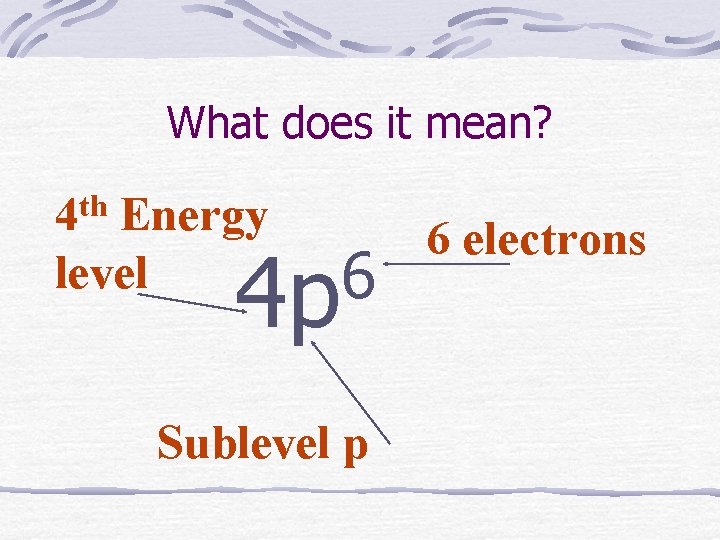
What does it mean? th 4 Energy level 6 4 p Sublevel p 6 electrons

The “Arrow” Diagram pg 160 Electrons arrange themselves in orbitals of increasing energy. s=2 p=6 d= 10 f = 14

Take argon for example. It has an atomic number of 18, so it has 18 electrons. Let’s put these in order. Check it! it should be in group 8 and on period 3. . is it? Valence e-? How many e- lost/gained? Charge of ion? 1 s 2, 2 p 6, 3 s 2, 3 p 6

Bellringer 1) 2) Arsenic has ____ total electrons. Write the electron configuration (long form) for As. 3) And for arsenic’s ion? (Ar 3 -) 1 s 2, 2 p 6, 3 s 2, 3 p 6, 4 s 2, 3 d 10, 4 p 3

Bellringer Write the electron configuration (long form) for Se. 2) Will it lose or gain? What charge will it have? And the E Config for its ion? 1) 3) Write the short form for Indium. 4) Write the short form for Indium ion.

Practice Write the electron configuration (long form) for: Silver ____ electrons And for silver ion? 1 s 2, 2 p 6, 3 s 2, 3 p 6, 4 s 2, 3 d 10, 4 p 6 , 5 s 2 , 4 d 9

Pre AP In notes Practice short form 1 Fe 2 Fe 3+ Hint: remove e from outermost level first 3 Sb 4 Sb 3 - 9 Rubidium ion 5 Cl 10 Magnesium ion 6 Cl 111 Iodide 7 Zn 12 Phosphide 8 Zn 2+

To turn in! Write these for each element: a) symbol and total # of electrons b) short form configuration c) # of valence e d) # e- lost/gained, charge of ion 1. 2. 3. 4. Palladium Indium Scandium Copper 5. Argon 6. Molybdenum 7. Polonium 8. Radium

To turn in! new Write these for each element: a) symbol and total # of electrons b) short form configuration c) # of valence e d) # e- lost/gained, charge of ion 1. 2. 3. 4. Palladium Indium Telliurium Copper 5. Argon 6. Molybdenum 7. Polonium 8. Radium

Homework……in notes (I will check it tomorrow) Write the long and short configuration for: 1. Oxygen 2. Phosphorous 3. Rubidium 4. Krypton 5. Silver

Bellringer A) Write the long-form electron configuration for silver B) tell how many e- it will lose/gain C) give the ionic charge

Electron configuration Shorthand version (Noble gas version) Ex Silver = [1 s 2, 2 p 6, 3 s 2, 3 p 6, 4 s 2, 3 d 10, 4 p 6 ], 5 s 2 , 4 d 9 Take the previous Noble Gas and substitute [Kr], 5 s 2 , 4 d 9

Bellringer 1. How many main energy levels can exist in 2. 3. 4. 5. 6. any atom? What are the 4 sublevels? Non-metals will gain enough electrons to make ___ valence electrons. For 1 s 2, 2 p 6, 3 s 2, 3 p 4 How many valence electrons are there? What will the charge of the ion be?

Videos http: //www. youtube. com/watch? v=rc. Kil E 9 Cda. A Hank electron + config http: //www. youtube. com/watch? v=97 uok_Xvp. M tutorial long/short
![Short-form electron configuration Looks like: [Ar] 4 s 1 ……for potassium One step… You Short-form electron configuration Looks like: [Ar] 4 s 1 ……for potassium One step… You](http://slidetodoc.com/presentation_image_h/2b34563c790cf569fe0b9b6492eebd87/image-42.jpg)
Short-form electron configuration Looks like: [Ar] 4 s 1 ……for potassium One step… You need to know: The 4 sublevel regions of the PT
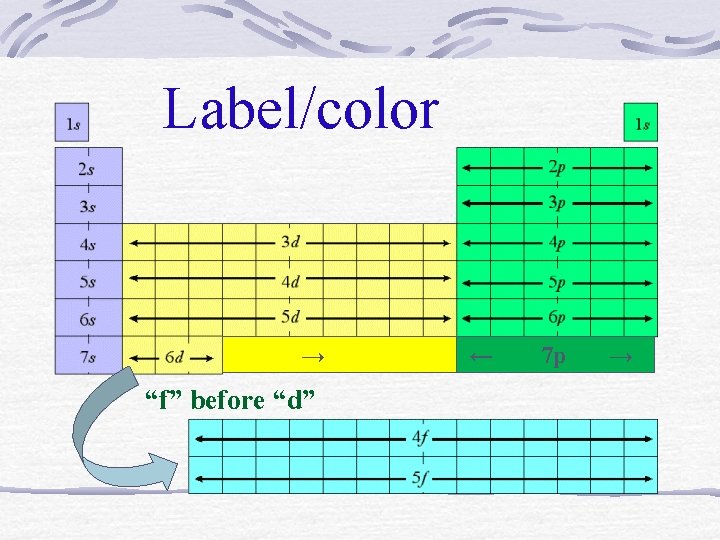
Label/color → “f” before “d” ← 7 p →

One step for short form (using PT) Let’s do the short-form for Fe (26) A) find the PREVIOUS noble gas ………is Argon B) Write it as……. . [Ar] C) Find the period and then the “sublevel blocks”. Answer = [Ar] 4 s 2, 3 d 6 For iron (III) ion (Fe ): [Ar] 3 d 5

More…. 1. Oxygen 2. And O 2 - ? ? 3. Arsenic 4. Aluminum 5. And Al 3+ ? ? 6. Europium 7. Silver 8. Francium 9. Lead (Pb) 10. Iodine (I 1 -)

Exceptions to Aufbau (lowest energy orbital first) Half-filled sublevels are more stable so lower level e- are moved for most transition metals. Ex Cr Cu W etc

Pre AP Bellringer Turn in lab report Write the short form configuration for: a) Silicon b) Tin c) Polonium d) Nickel 2+ion e) Nitride 3 - ion Grade cam quiz tomorrow

Gen Bellringer Write the short form configuration for: a) Silicon b) Tin c) Polonium d) Nickel 2+ion e) Nitride 3 - ion Grade cam quiz tomorrow

Homework short form for 1. Fe 2+ 3. Fe 3+ 4. S 5. S 26. As 7. Silver 8. V 2+ 9. Se 10. Bi

EMR (electromagnetic radiation) All wavelengths = from gamma rays (very short=high energy) to radio and TV (long=low energy). All types of EMR travel at the speed of light Are emitted when excited electrons jump to lower energy levels. A “piece” or quantum of EMR is called a photon.
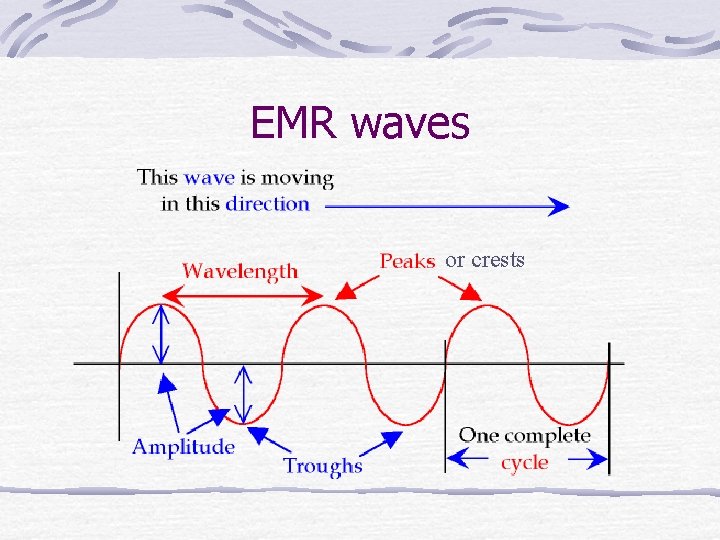
EMR waves or crests
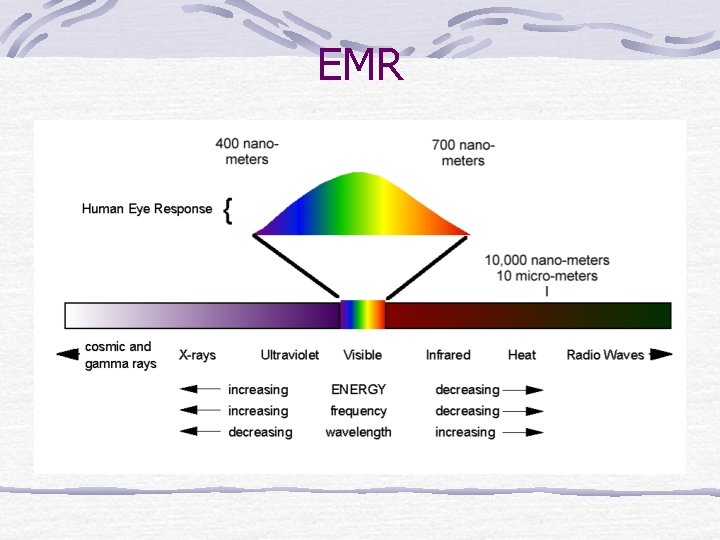
EMR


Wavelength and frequency of light emitted. Formulas: c = λν c = speed of light (300 000 m/s or 3. 00 x 10 λ = wavelength ν = frequency 8 m/s)

Homework To turn in Show formula, substitutions, units, sig figs, etc. . Write the question. Ex. 1 Calculate the frequency of 418. 0 nm light.

Energy of a photon E=hν E = energy of the photon ν = frequency h = Planck’s constant (6. 626 x 10 -34 Joules-s ) Problem: A flame test was done on an unknown compound. The light was seen through a spectroscope and was 456 nm (____ meters) in wavelength. Calculate the frequency of this light. And the energy of one photon of this light.

http: //www. youtube. com/watch? v=lwf. J Pc-r. SXw&list=PL 09 E 558656 CA 5 DF 76 NASA EMR http: //www. youtube. com/watch? v=gtg BHs. Sz. CPE Light/Color Bill Nye

Bellringer 1. Write the long form configuration for K (see page 116 for arrow chart, blue book) 2. Write the short form configuration for Indium 3. Write the short form configuration for Bismuth

Electron orbital diagrams know these! Electrons can behave as a particle but also have wavelike behavior (dual-nature by De. Broglie) Electrons will be in the lowest available energy position first. (Aufbau rule) Electrons will be in separate orbitals (same energy) before pairing up. (Hund’s rule) Electrons will orient themselves to have opposite spins if in same orbital. (Pauli rule) What? ? ? Let’s try applying this…. .
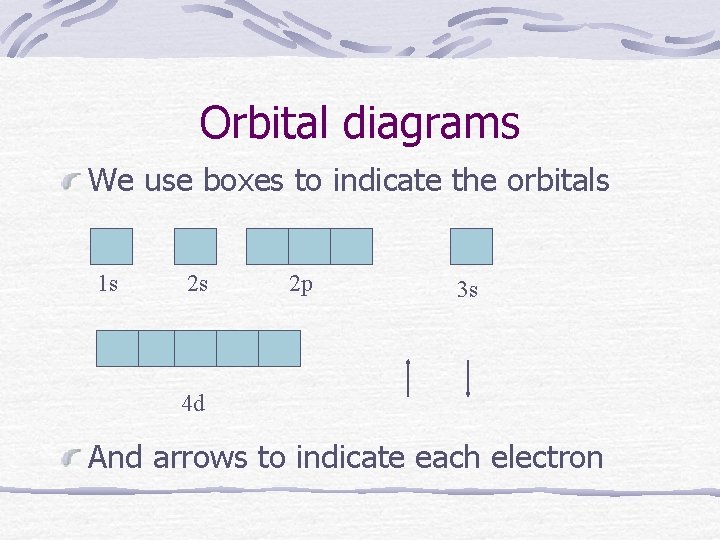
Orbital diagrams We use boxes to indicate the orbitals 1 s 2 s 2 p 3 s 4 d And arrows to indicate each electron

Practice Let’s do oxygen (8 e-) 1 s 2 s 2 p 3 s 3 p So…it has 2 unpaired electrons to use for bonding with other elements. And now aluminum (13 e-) 1 s 2 s 2 p 3 s 3 p
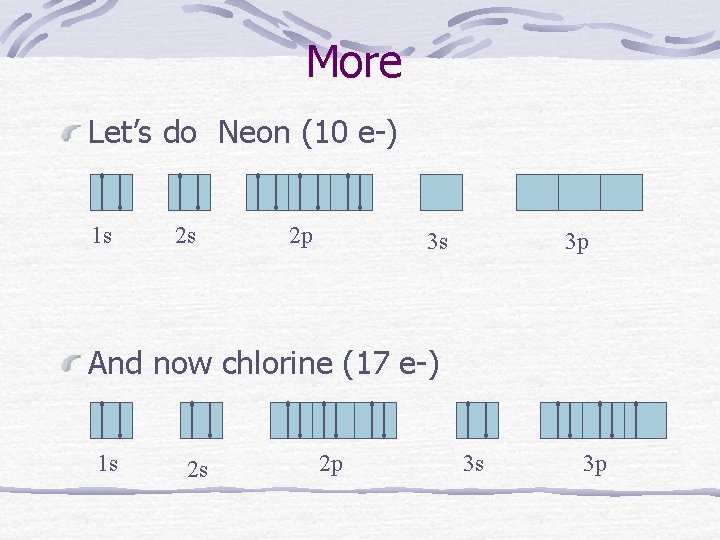
More Let’s do Neon (10 e-) 1 s 2 s 2 p 3 s 3 p And now chlorine (17 e-) 1 s 2 s 2 p 3 s 3 p

Homework… in notes Short form and orbital diagram for V 2+, S 2 -, I, Si and Pd

Today Quiz! Use red book PT Warm-up…. What is the short configuration for Gallium 3+? How do I know it is 3+? Do short form for Cs 1+. And Se 2 -
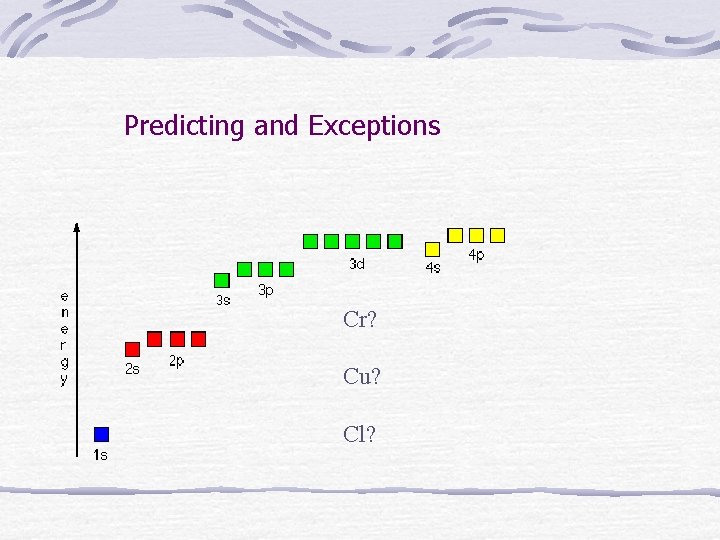
Predicting and Exceptions Cr? Cu? Cl?

Do these for Sn and Ni Predict the charges

And Vanadium Selenium

Bellringer 1. Write the short form configuration for Selenium. 2. Write the orbital diagram for Selenium. 3. Circle the 4 th electron on 4 p

Quantum numbers…where electrons are Tell you the distance an electron is from the nucleus, the shape and position of the orbital, and the spin of the electron. If quantum mechanics has not profoundly shocked you, you haven’t understood it yet. Niels Bohr

Quantum numbers Electron location is defined by the 4 quantum numbers: n = principal QN = energy level (values 1 to 7) l = angular QN = sublevel 0(s) 1(p) 2(d) 3(f) m (or ml) = magnetic QN = orientation of orbital x-y-z (values –l to +l) depends on value of l can be: -3, -2, -1, 0, +1, +2, +3 s (or ms) = spin = to right or left (values + ½ or – ½) s= either ½ (up) or -½ (down) Example: The 4 th electron on 4 p for Selenium

Quantum numbers A) For 4 th electron in 5 d for Mercury Hint: draw the orbital diagram first n = l = m = s= B) For the 2 nd electron in 3 s for Mg n = l = m = s= C) For the last electron written for Iodine n = l = m = s= D) For the 7 th 5 d electron in Gold n = l = m = s= E) For the last electron in Boron n = l = m = s= F) For the 2 nd-to-last electron in Kr n = l = m = s=

Quantum numbers For the 2 s electrons…. n = l = m = s= s = For the 5 th 4 d electron n = l = m = s=

Answers For the 2 s electrons…. n = 2 n = 2 l = 0 m = 0 s= ½ s = -½ For the 5 th 4 d electron n = 4 l = 2 m = (-2, -1, 0, 1, 2) so is 2 s= ½

Bellringer 1. Give QN for the last e- in 3 s 1 n= l= m= s= 2. Give QN for the last e- in 6 p 4 n= l= m= s= 3. Which QN tells the main energy level? 4. Which tells the direction the electron is spinning (on its axis)? 5. Which tells the shape? 6. Which tells the orientation in space? 7. Which tells the sublevel?

Exceptions to the Aufbau rule Elements with full or half full d sublevels are more stable. Electrons can be promoted to d sublevels to gain stability. Ex write the predicted short form and the corrected one for these: Cu Cr Ag Mo

Bellringer An arrow What symbol is used in orbital diagrams to show one electron? 2. What rule says that electrons will occupy the lowest energy orbitals first? Aufbau 3. Who first stated that electrons can behave both as a particle and as a wave? De. Broglie Wavelength 4. What do we call the distance on a wave for a complete cycle? 5. What do we call the height (energy) of a wave? Amplitude 6. What type of light is produced when an electron jumps from the 6 th to the 3 rd level? 7. What color of visible light is produced from the 3 rd to the 2 nd level? 8. Is light produced in this jump? If so, what kind? 3 rd to 6 th 9. Which element is this? …… 3 s 2, 3 p 4 10. Which type of EMR travels the fastest? ( 1.

Bellringer 1. 2. 3. 4. 5. 6. Which of these would have an electron configuration different than what we would predict? A) Mg b) Mn c) Cr d) Hg e) Cl Units of Joules would be for: a) wavelength b) frequency c) speed d) energy Units of Hertz (or 1/sec or sec-1) would be for: a) wavelength b) frequency c) speed d) energy Units of centimeters would be for: a) wavelength b) frequency c) speed d) energy Units of kilometers/sec would be for: a) wavelength b) frequency c) speed d) energy The speed to light is ________ (give number and units)

Wavelength and frequency of light emitted. Formulas: c = λν c = speed of light (300 000 m/s or 3. 00 x 10 λ = wavelength 8 m/s) ν = frequency E=hν E = energy of the photon ν = frequency h = Planck’s constant (6. 626 x 10 -34 Joules ∙ s ) Practice… 1. An element emits light of 1893 nm in wavelength. Calculate the frequency of this light. 2. And its energy. 2) Calculate the wavelength of a certain light with a frequency of 8. 65 x 10 19 1/sec.

Practice whiteboards Formulas: c = λν c = speed of light (300 000 m/s or 3. 00 x 10 8 m/s) Answers: 6. 58 x 10 14 1/s λ = wavelength ν = frequency 8. 27 x 10 -7 m E=hν 7. 05 x 10 16 1/s 2. 97 x 10 -19 J E = energy of the photon ν = frequency 1. 99 x 10 -13 J -34 h = Planck’s constant (6. 626 x 10 Joules ∙ s ) 1. A flame test was done on an unknown compound. The light was seen through a spectroscope and was 456 nm in wavelength. Calculate the frequency of this light. 2. Calculate the wavelength of a certain light with a frequency of 3. 625 x 10 14 1/sec. 3. Determine the frequency of light that is 4. 257 x 10 -7 cm. 4. The light from a burning unknown compound was seen through a spectroscope and was 669. 0 nm in wavelength. And the energy of one photon of this light. 5. Cobalt-60 is an isotope used to treat certain types of cancer. If the wavelength of the gamma rays emitted is 1. 00 x 10 -3 nm, what is the energy?

Bellringer A flame test was done on an unknown compound. The light was seen through a spectroscope and was 2. 187 x 10 -4 cm in wavelength. Test a) Calculate the frequency of this light. will be b) And the energy of one photon of this light. h = Planck’s constant (6. 626 x 10 Joules ∙ s ) on FRIDAY -34
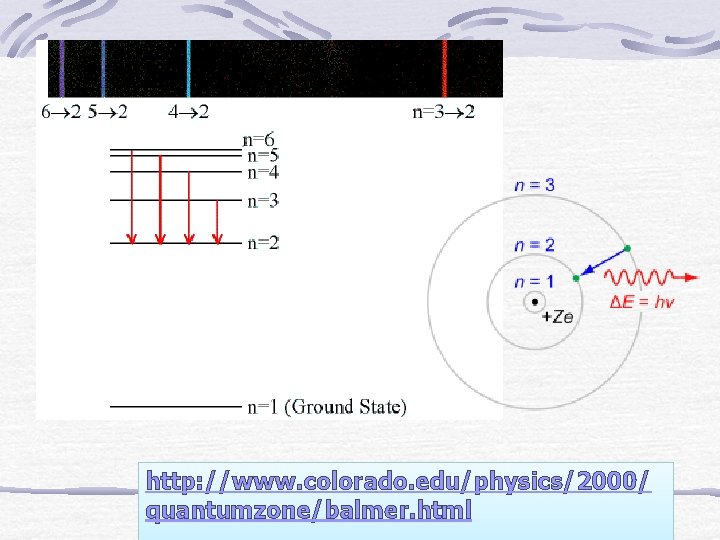
http: //www. colorado. edu/physics/2000/ quantumzone/balmer. html
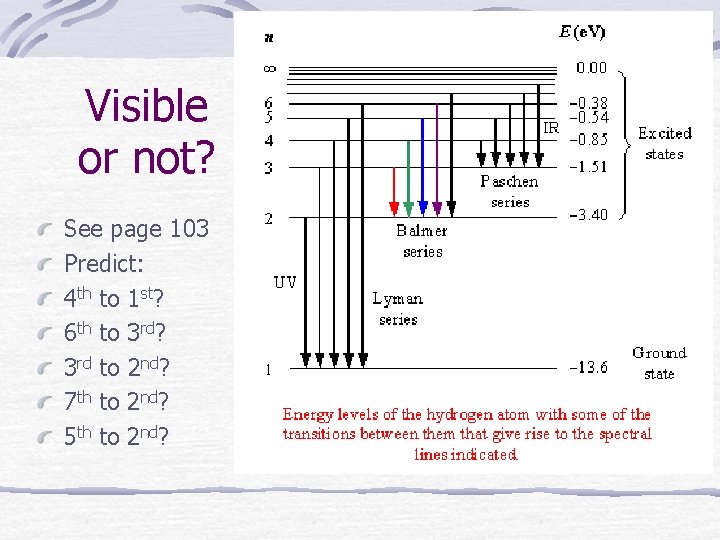
Visible or not? See page 103 Predict: 4 th to 1 st? 6 th to 3 rd? 3 rd to 2 nd? 7 th to 2 nd? 5 th to 2 nd?

More formulas! Rydberg-Bohr is a mathematical formula used to predict the wavelength of light resulting from an electron moving between energy levels of an atom. R = Rydberg constant = 1. 097 x 107 1/m λ = wavelength of the EMR ν = frequency of the EMR h = Planck’s constant = 6. 626 X 10 -34 J s c = speed light = 299, 792, 458 meters per second E = hν c = λν Example: When an electron jumps from the 5 th to the 2 nd level, what is the wavelength, frequency and the energy? Ans = E = x 1018 Joules

Solving MORE Problems Using Hydrogen’s electron in the ground state A) if an electron gains energy and moves from 1 st to 3 rd level, what wavelength and frequency AND energy does the element give off as it jumps back? B) Will this be visible? If so, what color? If not what part of the EMR spectra is it? See page 98 and use the frequency. h = Planck’s constant (6. 626 x 10 -34 Joules ∙ s ) R = Rydberg constant = 1. 097 x 107 1/m You will need 6 checks for full points!

Problem #1 Use Rydberg equation to calculate wavelength when electron returns from n=5 to n=2. 1/λ = 2. 304 x 106 1/m; λ = 4. 341 x 10 -7 m 434 nm = violet Problem #2 What is the wavelength of a photon from a transition of 5 th to 1 st? 1/λ = 2. 304 x 106 1/m; λ = 4. 341 x 10 -7 m 434 nm = violet Problem #3 One electron de-excites from n = 8 to n =2. Another electron deexcites from n = 2 to n = 1. Which emits the greater energy? Explain! 1/λ = 2. 304 x 106 1/m; λ = 4. 341 x 10 -7 m 434 nm = violet

Problem #1 Use Rydberg equation to calculate wavelength when electron returns from n=5 to n=2. Problem #2 For l = 1090, use Rydberg equation to determine initial and final energy levels. Problem #3 One electron de-excites from n = 8 to n =2. Another electron deexcites from n = 2 to n = 1. Which emits the greater energy? Explain!

Problem #1 Use Rydberg equation to calculate wavelength when electron returns from n=5 to n=2. = [1/22 - 1/52] = 0. 25 - 0. 0400 = 0. 21 1/λ = 1. 097 x 107 1/m (0. 21) = 4. 34 x 107 m now convert to nm = 432 nm Blue Problem #2 For l = 10, 900 A, use Rydberg equation to determine initial and final energy levels. 911 A/l = [1/n 12 - 1/n 22] 911 A/10900 A = 0. 0836 = DIF = [1/n 12 - 1/n 22] = 0. 0836 check table for the best fit! The best fit involves n=6 to n=3 [1/32 - 1/62] = 0. 111 - 0. 0278 = 0. 0832 Problem #3 One electron de-excites from n = 8 to n =2. Another electron de-excites from n = 2 to n = 1. Which emits the greater energy? Explain! For n = 8 to n = 2: For n = 2 to n = 1: 911 A/l = [1/22 - 1/82] 911 A/l = [1/12 - 1/22] = 0. 25 - 0. 0156 = 0. 234 = 1. 00 - 0. 25 = 0. 75 = 911 A/0. 234 = 3890 A = 911 A/0. 75 = 1215 A (shorter l higher energy)

Video http: //www. youtube. com/watch? v=x. H 1 k 1 dtgi. VY Tutorial orbital diagrams http: //www. bing. com/videos/search? q= absorption+spectra&FORM=HDRSC 3#vi ew=detail&mid=9 B 6 FDB 6 BABF 5 A 2519 D 60 emission spectra stars

Videos http: //www. youtube. com/watch? v=1 CG zk-n. V 06 g MRI 1 min http: //www. youtube. com/watch? v=t. D 9 7 Vhna-ic attorney explains MRI http: //www. youtube. com/watch? v=_93 TCv. Wad 8 E fun w/MRI

Exit slip For the element Phosphorous (P) and Bromine (Br) Write the: a) short electron configuration. b) # of valence e c) # e- lost/gained d) charge of ion e) orbital diagram

Bellringer 1. Give the 4 quantum numbers for the 2. 3. 4. 5. second-to last highest-energy electron of an actual atom of chromium. Give 2 atoms or ions that are isoelectronic. Who did the gold foil experiment and what did he find? Draw the 2 shapes of a “d” sublevel cloud. Whose rule says that electrons can not have the same spin if they are in the same orbital? TEST on FRIDAY. Review and practice WS today. Study guide online classroom.

Bellringer 1. What is the long form electron configuration for sulfur? 2. How many valence electrons does it have? 3. How many does it need to have a complete valence level? 4. What is the short form for Sc (21)? 5. What is wrong with this? [Ar] 4 s 2, 3 d 6 , 4 p 3 6. Which 2 are isoelectronic? Na 1+, S 2 -, Br 1 -

Bellringer 1. List 3 of Dalton’s postulates and tell if they are still true now. 2. Tell at least 3 ways that the quantum model is different from Bohr’s model. 3. What was the main difference between the models of Democritus and Dalton? 4. Which rule does these disobey? TEST on FRIDAY. Finish practice WS today and work on Study guide, or can do test corrections on last test.

CH 5+6 test Main topics Test CH 5 Main topics (NOT all you should know) Vocab Long + short e config and explain exceptions Orbital diagrams and Valence e 4 quantum numbers Orbital vs sublevel and shapes Hund’s rule, Pauli exclusion principal, Aufbau rule, Heisenberg principle, Bohr model, De. Broglie prinicple Label parts of a wave EMR spectra in general visible = 400 -700 nm Atomic and ionic radii Ionization energy trends Electronegativity trends Calculations E = hν c = λν Rydberg E = -2. 178 x 10 -18 J (1/nf 2 - 1/nf 2 )

Practice Using the Bohr equation, calculate the change in energy when an electron transitions from n = 3 to n= 2 in a hydrogen atom ANS = 3. 01 x 10 – 10 m Hydrogen has a red emission line at 656. 3 nm, what is the energy and frequency of a photon of this light? Note: Planck’s constant = 6. 626 x 10 – 34 J·s, speed of light = 2. 998 x 10 8 m/s ANS = 3. 027 x 10 – 19 J and 4. 568 x 10 14 s– 1 or Hz What are the four quantum numbers for the last electron in each of the following elements? Nitrogen Sulfur Copper ANS = 2 1 +1 + ½ & 3 1 – 1/2 & 3 2 +2 – 1/2 Hydrogen has a red emission line at 656. 3 nm, what is the energy and frequency of a photon of this light? Answer 3. 03 x 10 – 19 J and 4. 59 x 10 14 Hz Much practice! http: //drmadscientist. wordpress. com/2013/06/24/wavelengthpracticeproblems/ And http: //www. chemteam. info/Electrons/calc-wavelength-given-freq. html
- Slides: 96