Bellringer 1 Copy down homework Finish reasoning Density

Bellringer 1. Copy down homework: Finish reasoning (Density Column & Notes) 2. Take out pride card.

DENSITY COLUMN AND NOTES • Hypothesis: What do you think will happen when we pour syrup, dish soap, water, and oil together in a graduated cylinder? DRAW IT AND LABEL! • Gather Data: Observations (what happened): DRAW IT AND LABEL! Material Mass (g) Volume Density (ml) Syrup 10 Dish Soap 20 Water 10 Oil 15 (g/ml)
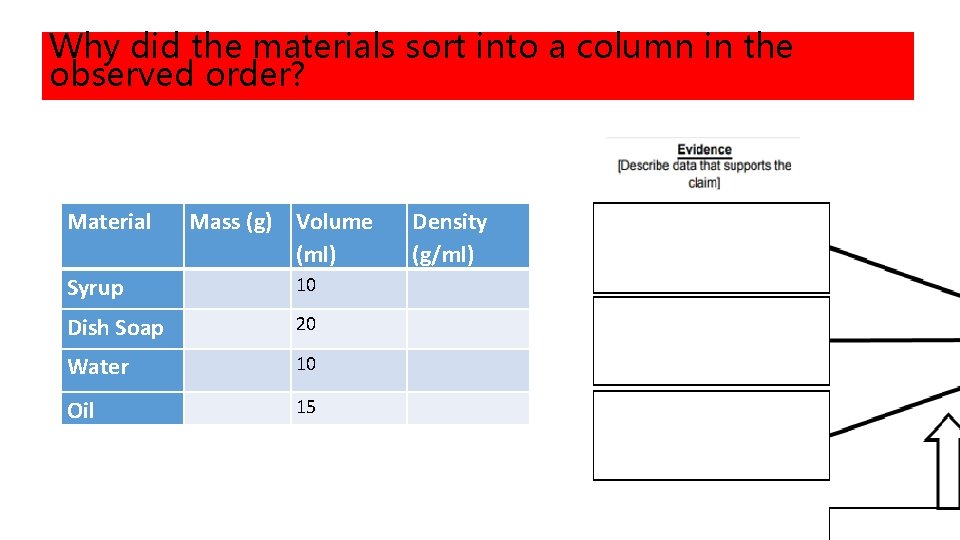
Why did the materials sort into a column in the observed order? Material Mass (g) Volume (ml) Syrup 10 Dish Soap 20 Water 10 Oil 15 Density (g/ml)

Density Notes (brainpop 3: 35) • Mass: The amount of matter in an object • Volume: The amount of space an object takes up. • Density- A measure of how much matter is in a certain volume or space (how packed the molecules are in a space) • Density= Mass/Volume https: //www. youtube. com/watch? v=zlkp. ZZW 29 b 0
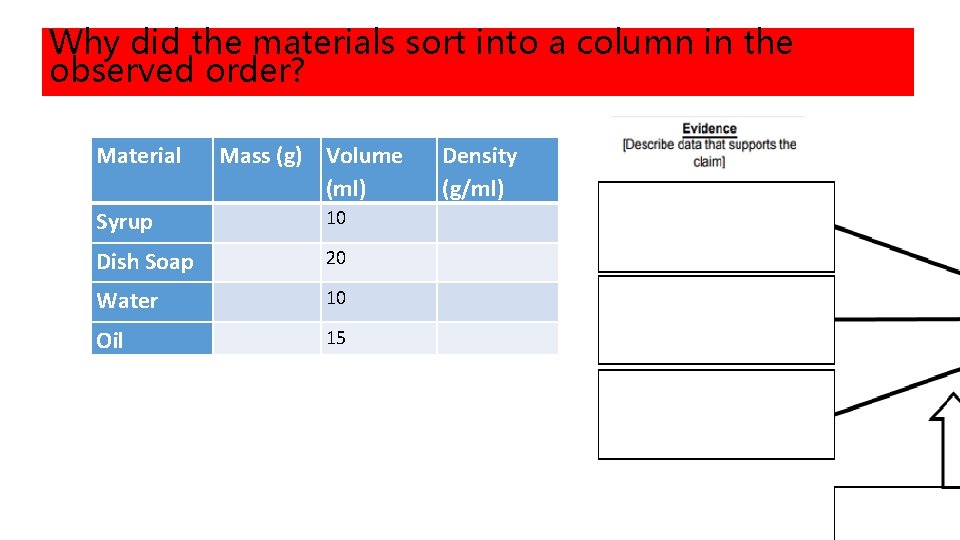
Why did the materials sort into a column in the observed order? Material Mass (g) Volume (ml) Syrup 10 Dish Soap 20 Water 10 Oil 15 Density (g/ml)
Why did the materials sort into a column in the observed order?
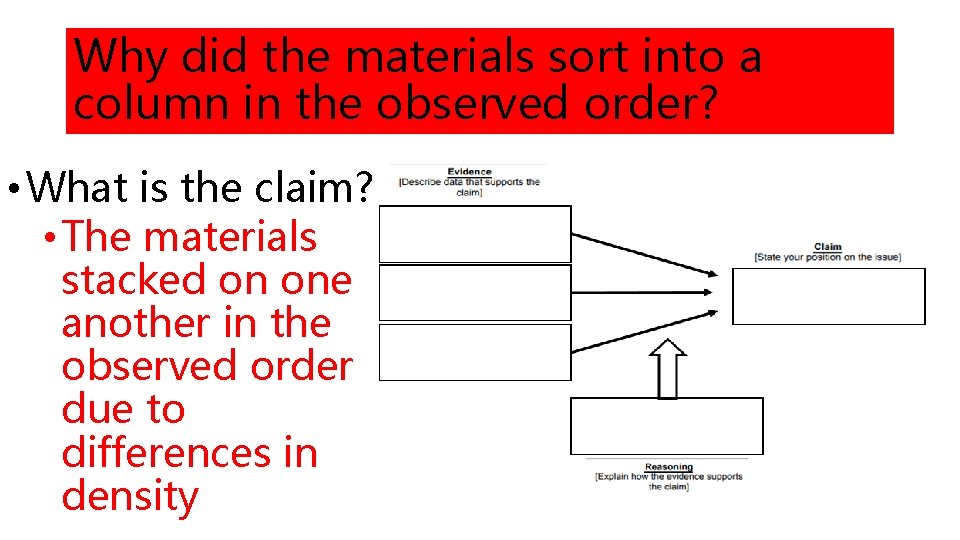
Why did the materials sort into a column in the observed order? • What is the claim? • The materials stacked on one another in the observed order due to differences in density
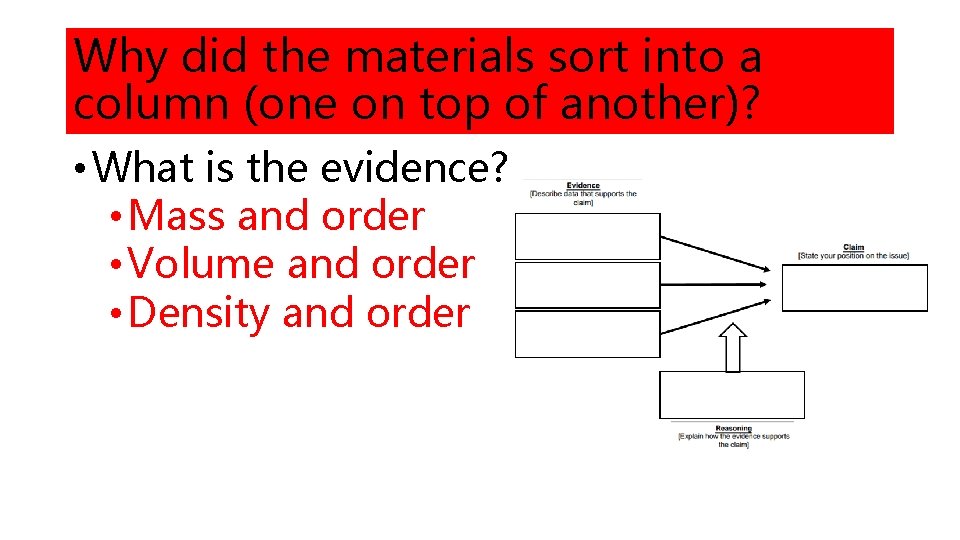
Why did the materials sort into a column (one on top of another)? • What is the evidence? • Mass and order • Volume and order • Density and order

What is the reasoning (how does evidence support claim)? • Why did the materials sort in that order (restate claim)? • Therefore, the differences in density caused the materials to stack on one another and determined the order of the materials. • What is density (Scientific Principle)? • Density is the amount of matter (mass) per space. • Looking at the calculated density and the order of the materials, what is the relationship between the two? • The objects with higher density sank below objects with lower density.

Bellringer 1. Copy down daily learning log & homework • Pg. #4: Coca Cola Vs. Diet Coke Lab 2. Glue in Density Column (Pg. #3) if not glued in. 3. Copy down homework (Finish Coca Cola Lab) 4. Take out pride card

Density (calculations) Ingredients Observations Why does the can of diet coke float on HIGHER IN THE water and the can of coca cola sinks in the water?
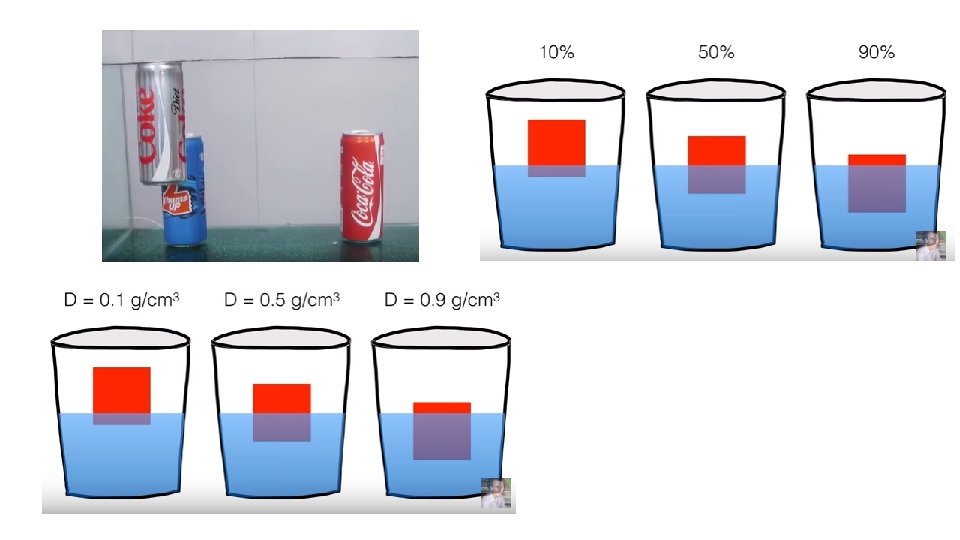

Question: Why does diet coke float HIGHER IN THE on water and coca cola sinks in water? • Determine how you would gather evidence (Facts, data, or observations that support the claim) to support your claim. • Write procedures. 1. Mass: What is mass and how did you find the mass? 2. Volume: What is volume and what is the volume of the cans? (look at the can) 3. Density: How do you calculate density? • Collect Data: Create a table to collect information about the density of each can. Include your units! • Create a claim based on what you know to answer the question above. • Write the reasoning (how does the evidence supports your claim? ). 1. Why does diet coke float HIGHER IN THE on water and coca cola sinks in water? (restate claim) 2. What is density? (Scientific Reasoning). 3. How does your evidence support your claim? (How did the densities compare? Why did the diet coke float higher in the water if the density of water is 1 g/ml? Why were the densities of each soda different? )
Coke vs. Diet Coke • https: //www. youtube. com/watch? v=4 wy. PPlj. Nd. K 8
- Slides: 15