Bell Ringer You need the following items ON

Bell Ringer You need the following items ON YOUR DESK -HW WS on Molar Mass Check your “Compound Formula” with your shoulder partner (2 minutes)
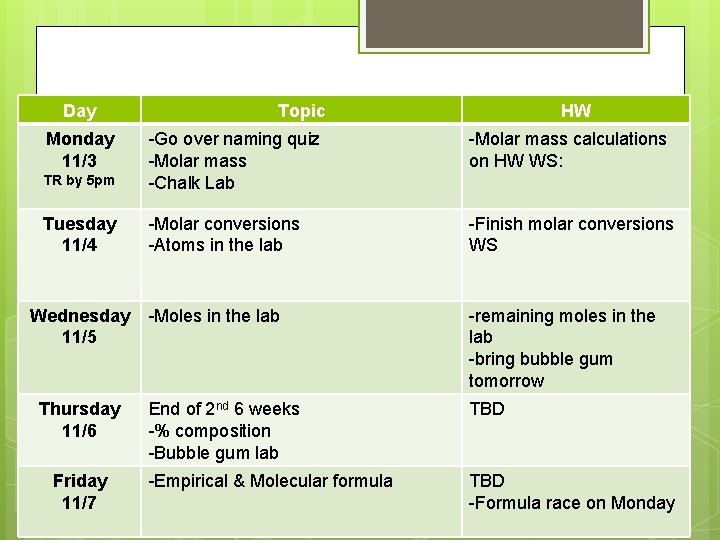
Day Monday 11/3 Topic HW -Molar mass calculations on HW WS: TR by 5 pm -Go over naming quiz -Molar mass -Chalk Lab Tuesday 11/4 -Molar conversions -Atoms in the lab -Finish molar conversions WS -Moles in the lab -remaining moles in the lab -bring bubble gum tomorrow End of 2 nd 6 weeks -% composition -Bubble gum lab TBD -Empirical & Molecular formula TBD -Formula race on Monday Wednesday 11/5 Thursday 11/6 Friday 11/7

Go over naming quiz

What do you remember about the mole?

What is the Mole? • A counting number (like a dozen) • Avogadro’s number (NA) • 1 mol = 6. 02 1023 items A large amount!!!!
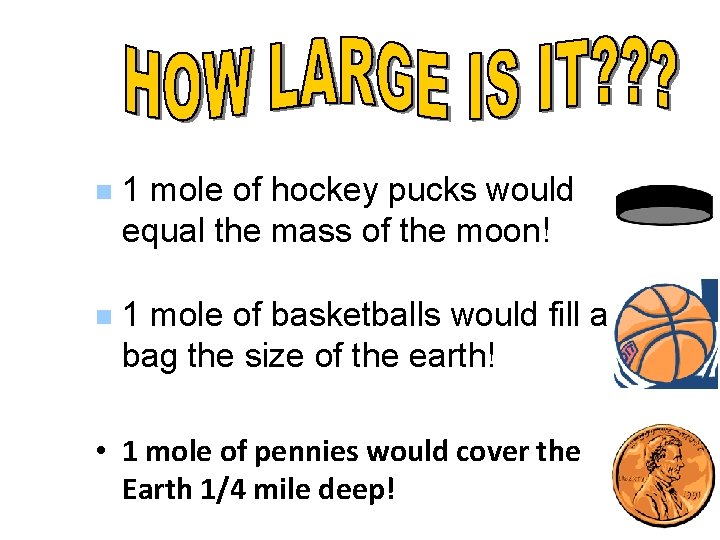
n 1 mole of hockey pucks would equal the mass of the moon! n 1 mole of basketballs would fill a bag the size of the earth! • 1 mole of pennies would cover the Earth 1/4 mile deep!

CORE Concept 6. 022 x 1023 ions/atoms/molecules/formula units = 1 mol = molar mass What do moles have to do with chemistry?

CORE Concept 6. 022 x 1023 ions/atoms/molecules/formula units = 1 mol = molar mass in grams • • 1 mol Fe. Br 2 = 6. 022 x 1023 formula units 1 mol H 2 O = 6. 022 x 1023 molecules 1 mol H 2 O = 6. 022 x 1023 atoms 1 mol Na+1 = 6. 022 x 1023 ions

Molar Mass • Mass of 1 mole of an element or compound. • Atomic mass tells the. . . – atomic mass units per atom (amu) – grams per mole (g/mol) Use all numbers after the decimal place from your periodic table!! Where do you find the molar mass of an element? Carbon-12. 011 g/mol Aluminum-26. 982 g/mol Zinc-65. 39 g/mol

Molar Mass of Compounds Water -H 2 O Ammonium carbonate (NH 4)2 CO 3

Molar Mass of Compounds Water -H 2 O 2(1. 008) + 15. 999 = 18. 015 g/mol 1 mol = 18. 015 g Ammonium carbonate (NH 4)2 CO 3 2(14. 007) + 8(1. 008) + 12. 011 + 3(15. 999) = 96. 09 g/mol 1 mol = 96. 09 g

Let’s go to the mall…

Practice • Calculate the number of moles in 180. 16 grams of glucose (C 6 H 12 O 6). • Looking ahead: Calculate the number of particles of sugar in 180. 16 grams.

Moles of Chalk Lab Mass the piece of chalk before using Mass the piece of chalk after filling a paper with writing/drawing Just to g mol conversions Determine the number of moles used by answering the questions

Reminders Test remediation by TODAY at 5: 00 pm Turn in all assignments with “ 1”s Complete the molar mass column on “Molar Mass WS” for HW tonight – we will be using tomorrow

Exit Ticket Moles of chalk lab g mol Place in folder for tomorrow when completed
- Slides: 16