Bell Ringer you need Periodic Table packet and
Bell Ringer (you need Periodic Table packet and test review out on desk) Turn project in to your period’s lab table. : D
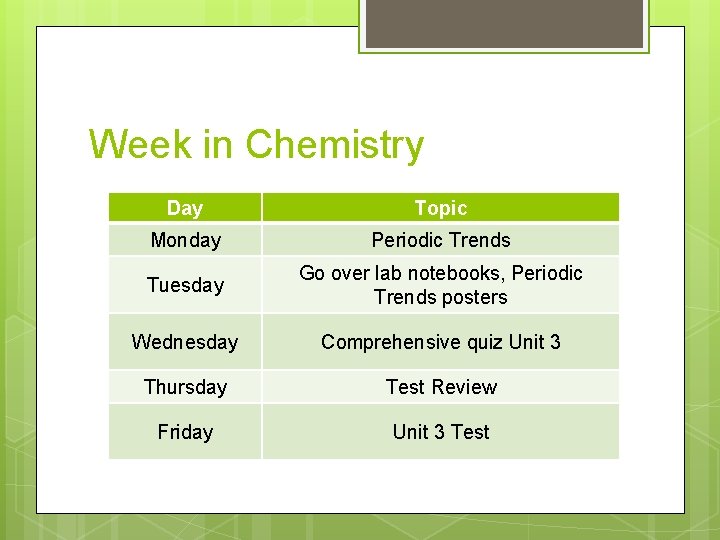
Week in Chemistry Day Topic Monday Periodic Trends Tuesday Go over lab notebooks, Periodic Trends posters Wednesday Comprehensive quiz Unit 3 Thursday Test Review Friday Unit 3 Test

Assignments Passed Back Isotope Quiz (quiz)

Orbital Notation Shows increasing energy levels and the orbitals on those levels. “ 1 s”, “ 2 s”, “ 2 p”, etc – these represent energy levels 1 box – represents one orbital (holds two electrons) Write the orbital notation for Sodium

Periodicity

Periodic Trends Because of the established Periodic Law, scientists began to notice tendencies of certain elemental characteristics to increase or decrease along a row or column of the periodic table of elements. These tendencies are called Periodic Trends.

Coulomb’s Law

4 factors that cause the trends 1. Nuclear Pull (Z) – the number of protons Positive protons pull negative electrons The protons pull on the outer electrons. The more protons, the more pull exerted by the nucleus on the outer electrons.

4 factors that cause the trends 2. Electron repulsion – size of e cloud Electrons are negative and push away from each other The more electrons in an atom’s electron cloud, the more they are pushed away from each other (due to having the same charge), making a bigger cloud.
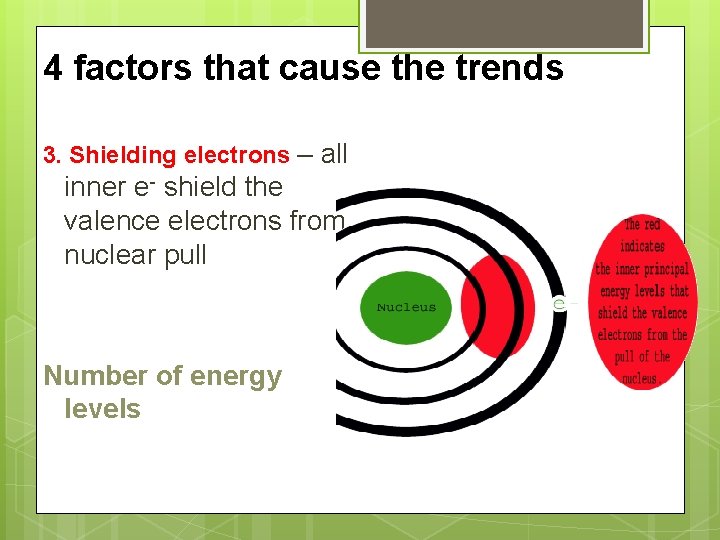
4 factors that cause the trends 3. Shielding electrons – all inner e- shield the valence electrons from nuclear pull Number of energy levels

4 factors that cause the trends 4. Zeff – the “effective” nuclear pull on outer electrons. The nuclear pull taking into account the shielding electrons which are taking most of the force. Zeff= # protons - # non-valence electrons Which element has more effective nuclear pull? Fluorine OR Magnesium

Periodic Trends – Four Corners In your corner, determine what the periodic trend is going across the PT and down the PT. Then explain WHY the trend occurs using one or more of the four factors. Answer the provided questions. Atomic Radius Ionization Energy Electronegativity Ionic Size
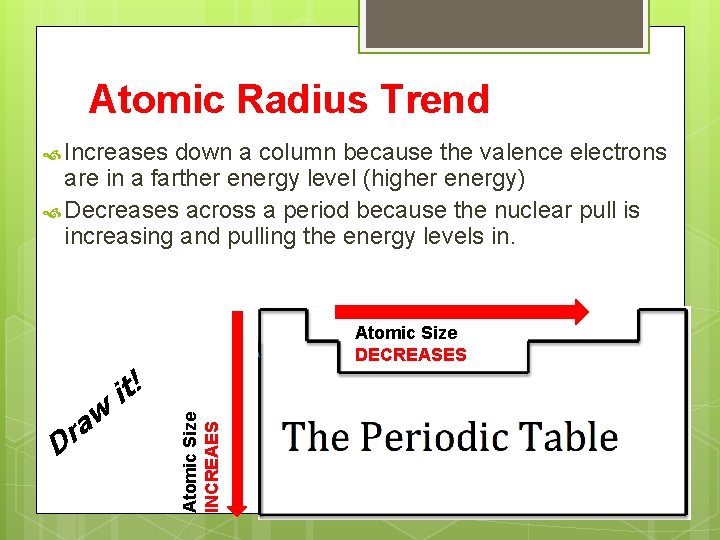
Atomic Radius Trend Increases down a column because the valence electrons are in a farther energy level (higher energy) Decreases across a period because the nuclear pull is increasing and pulling the energy levels in. Atomic Size INCREAES D w a r it! Atomic Size DECREASES
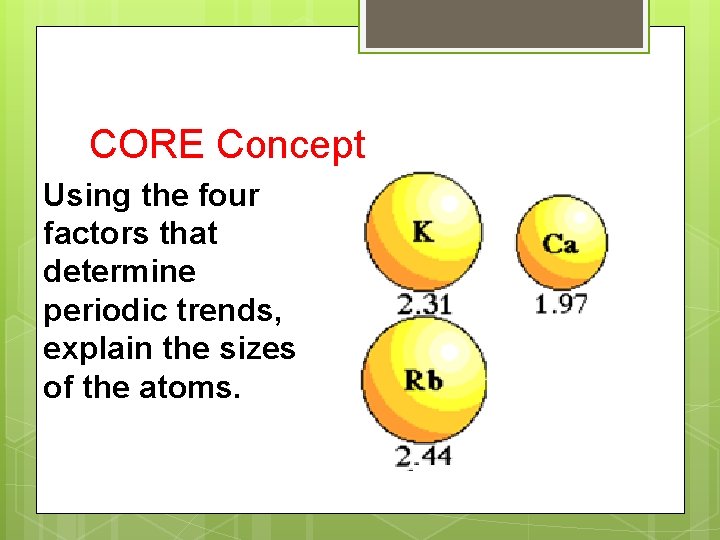
CORE Concept Using the four factors that determine periodic trends, explain the sizes of the atoms.
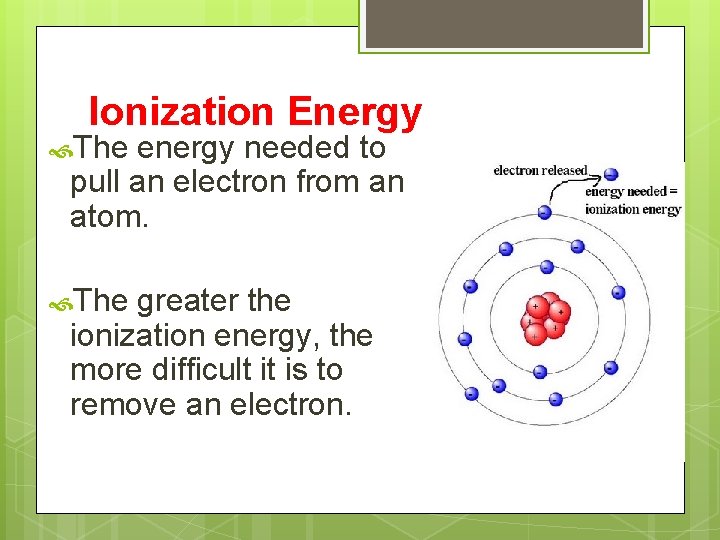
Ionization Energy The energy needed to pull an electron from an atom. The greater the ionization energy, the more difficult it is to remove an electron.
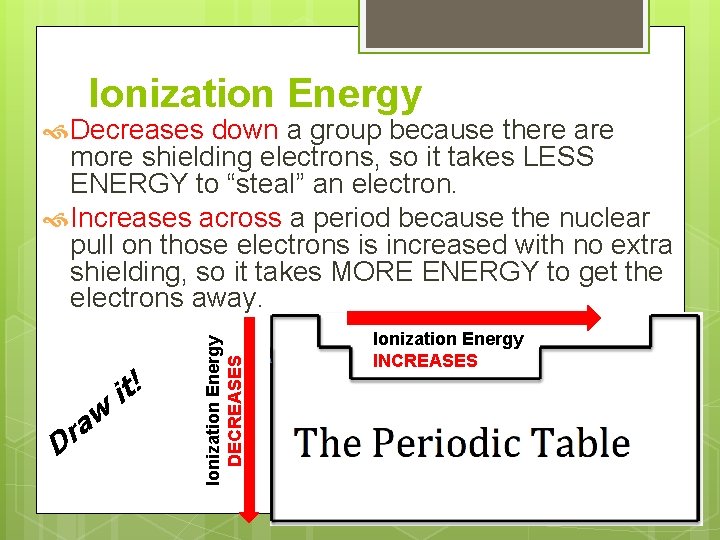
Ionization Energy Decreases down a group because there are D w a r it! Ionization Energy DECREASES more shielding electrons, so it takes LESS ENERGY to “steal” an electron. Increases across a period because the nuclear pull on those electrons is increased with no extra shielding, so it takes MORE ENERGY to get the electrons away. Ionization Energy INCREASES
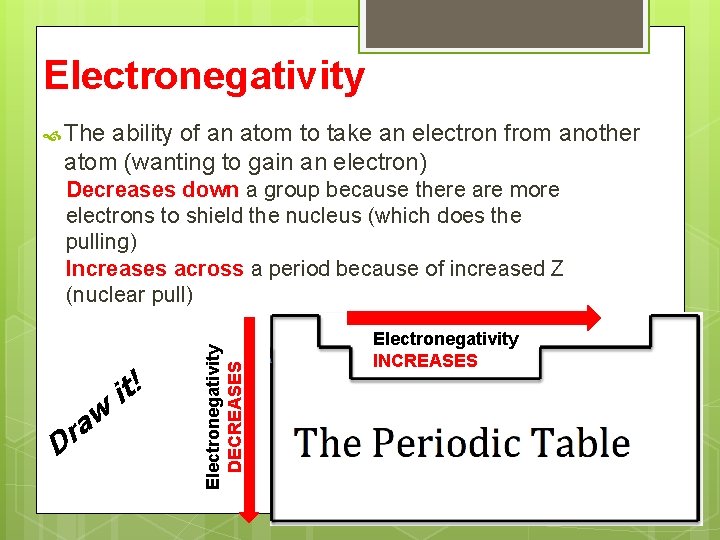
Electronegativity The ability of an atom to take an electron from another atom (wanting to gain an electron) D w a r it! Electronegativity DECREASES Decreases down a group because there are more electrons to shield the nucleus (which does the pulling) Increases across a period because of increased Z (nuclear pull) Electronegativity INCREASES
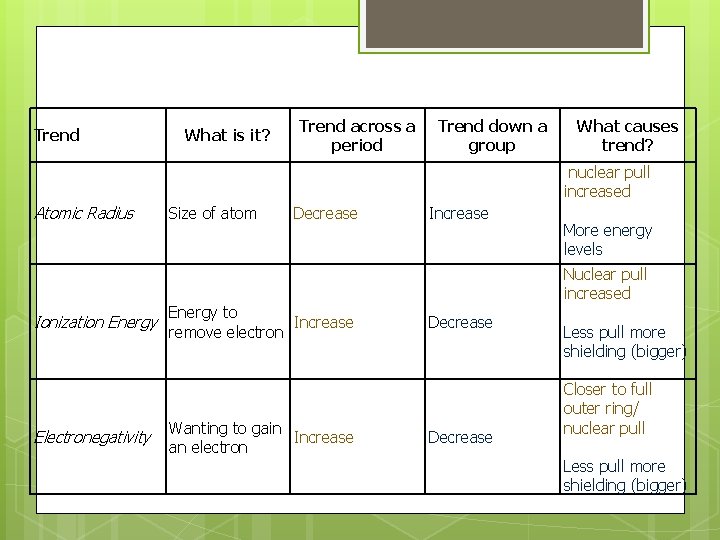
Trend What is it? Trend across a period Trend down a group What causes trend? nuclear pull increased Atomic Radius Ionization Energy Electronegativity Size of atom Decrease Energy to Increase remove electron Wanting to gain Increase an electron Increase More energy levels Nuclear pull increased Decrease Less pull more shielding (bigger) Closer to full outer ring/ nuclear pull Less pull more shielding (bigger)

Ionic Size Metals lose electrons (form cations) less e- repulsion, smaller electron cloud, smaller size Nonmetals gain electrons (form anions) more e- repulsion, larger electron cloud, larger size

Practice 1) Which is the smallest ion? Explain Na+ Cl- P 3 - 2) Which has the highest electronegativity? Explain. As Sn S 3) In the following pairs, which has the larger atomic radius? Explain. Mg or Ba Cu or Cu 2+ S or S 24) In the following pairs, which has the higher ionization energy? Explain. Li or Cs Ca or Br

Reminders Assignments Comprehensive Quiz Test Review Unit 3 Test Due Date Wednesday, 10/1 Friday, 10/3

Exit Ticket Which element has a smallest ionic radius? Which element has a largest ionic radius? Which element requires the least amount of energy to remove a valence electron?
- Slides: 22