Bell Ringer Tell me the difference between a


Bell Ringer Tell me the difference between a single, double and triple bond. l Give an example of an element that might l l Triple bond? l Double bond l Single bond?

VSEPR theory states electrons pairs shared (in bonds) or unshared (lone pair) will adopt a geometry that maximizes the distance between the bonds or lone pairs. • lone pairs and bonds will be referred to as regions of high electron density • double and triple bonds will count only as 1 region of high electron density

The geometry or shape around the central atom will depend upon: ·The number of bonding electron pairs or shared pairs (multiple bonds count as a single shared pair) on the central atom ·The number of non-bonding pairs or unshared pairs found on the central atom (lone pair)

Polar vs Nonpolar l Polar compounds are asymmetrical and end up with areas with partial charges. This can be due to: l 1. lone pairs on the central atom l 2. different terminal atoms l 3. different types of bonds (one single bond and one triple bond) l Nonpolar compounds are symmetrical with their charges evenly distributed.

A pair of electrons is any one of the following: • a non-bonding pair of electrons • a single bond • a double bond • a triple bond
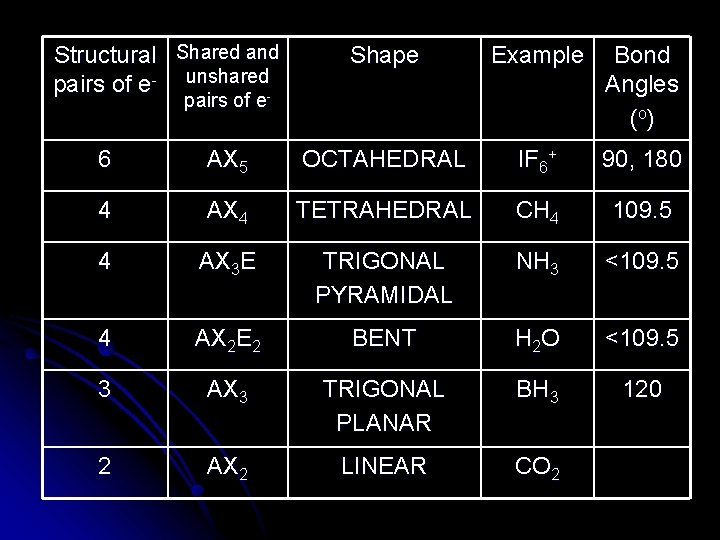
Structural Shared and pairs of e- unshared- Shape Example Bond Angles (o ) pairs of e 6 AX 5 OCTAHEDRAL IF 6+ 90, 180 4 AX 4 TETRAHEDRAL CH 4 109. 5 4 AX 3 E TRIGONAL PYRAMIDAL NH 3 <109. 5 4 AX 2 E 2 BENT H 2 O <109. 5 3 AX 3 TRIGONAL PLANAR BH 3 120 2 AX 2 LINEAR CO 2
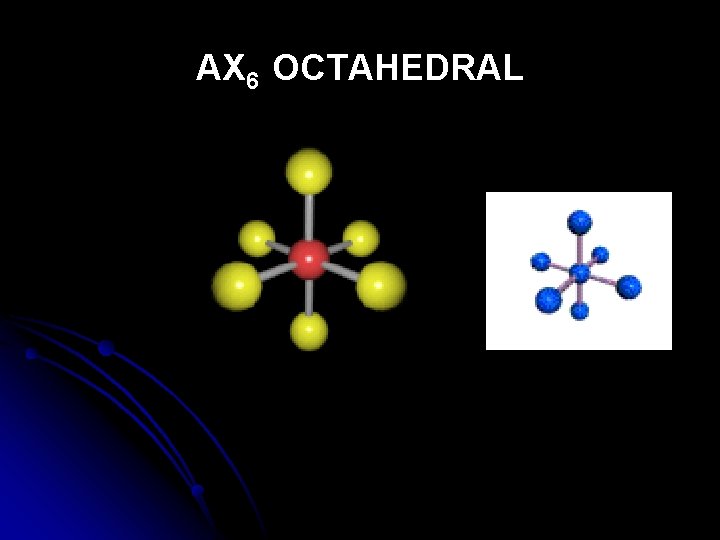
AX 6 OCTAHEDRAL

EXAMPLE Se. F 6 OCTAHEDRAL
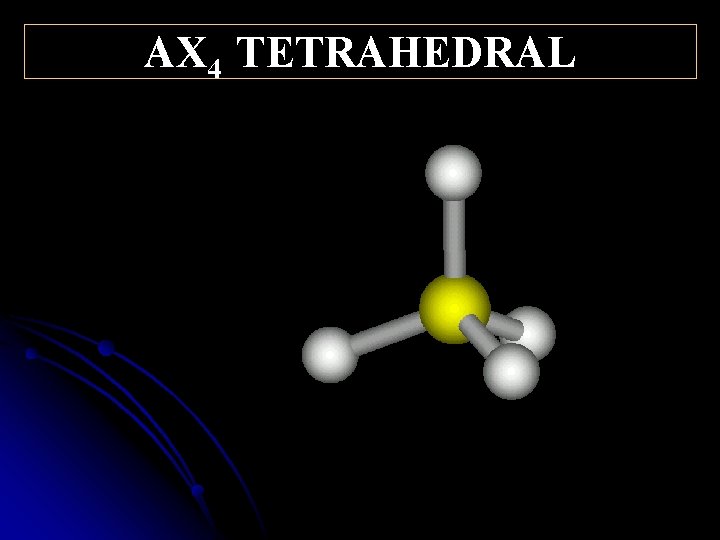
10 9. 5 ° AX 4 TETRAHEDRAL
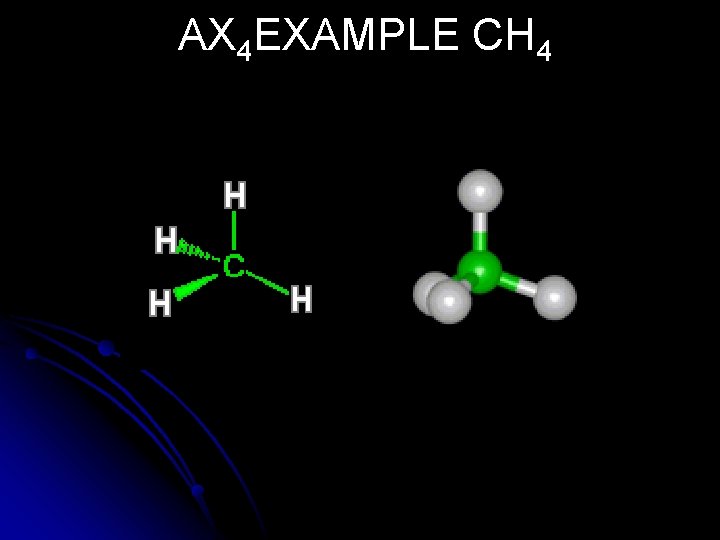
AX 4 EXAMPLE CH 4

AX 3 E EXAMPLE NH 3 TRIGONAL PYRAMIDAL
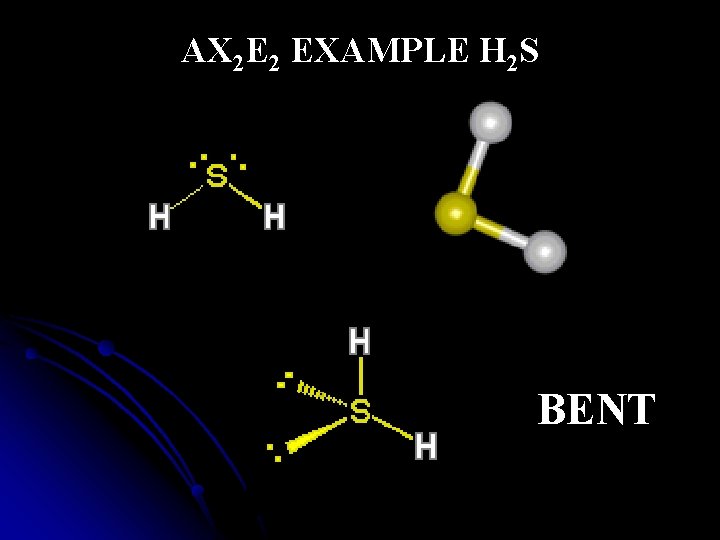
AX 2 E 2 EXAMPLE H 2 S BENT
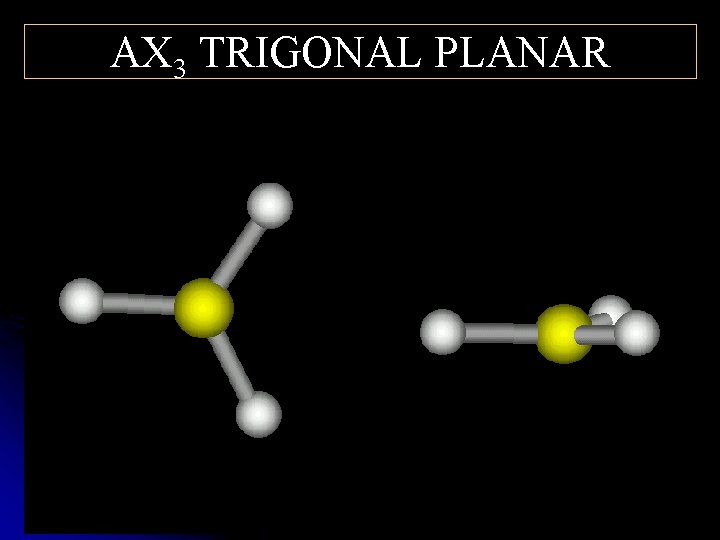
AX 3 TRIGONAL PLANAR ° 0 2 1
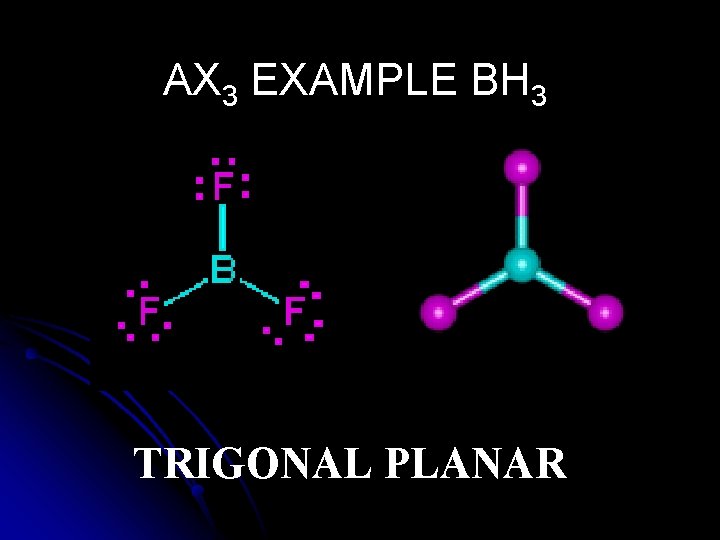
AX 3 EXAMPLE BH 3 TRIGONAL PLANAR

AX 3 WITH DOUBLE BOND H 2 CO TRIGONAL PLANAR

AX 2 E EXAMPLE SO 2 BENT
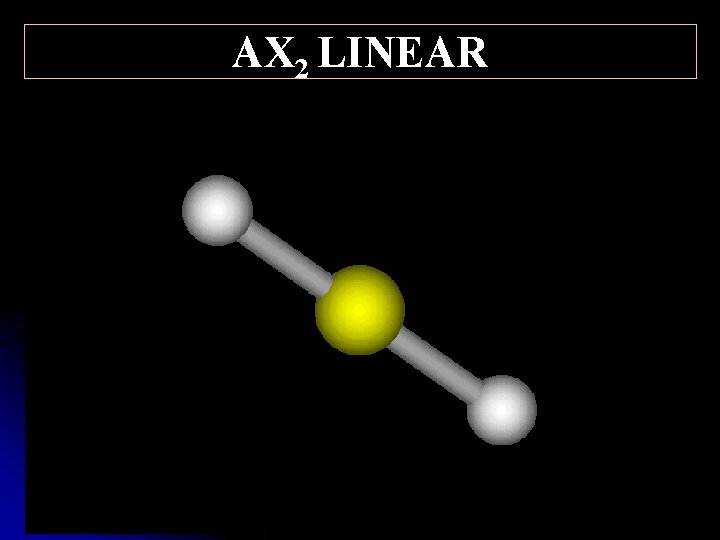
AX 2 LINEAR 180°
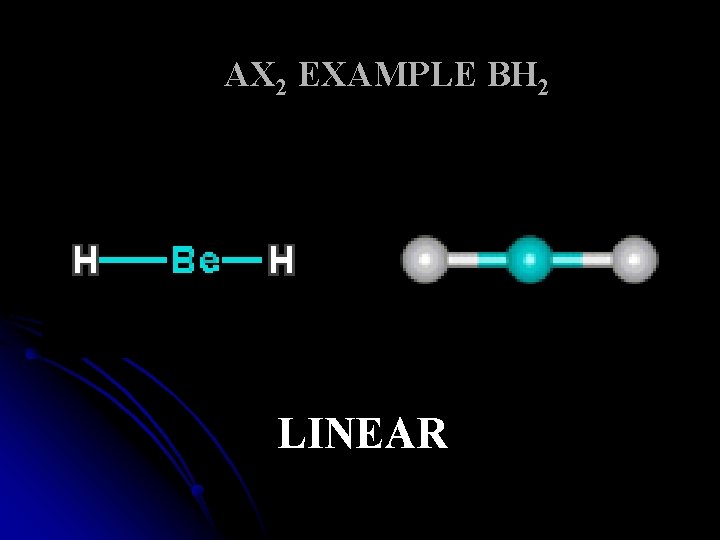
AX 2 EXAMPLE BH 2 LINEAR

AX 2 WITH DOUBLE BOND CO 2
- Slides: 20